Answer:
Hinsberg's reagent (benzene sulphonylchloride with reacts
with primary, secondary and tertiary amines in a differentmanner. This is used
for the distinction of three types of amines,
(i) Primary amineThe reaction of
benzene sulphonyl chloride withprimary amine proudces N-alkyl benzene
sulphonamide.Hydrogen attached to nitrogen in the product is acidic so it is soluble
in alkali.
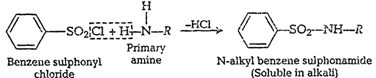 (ii) A secondary amine reacts
with Hinsberg's reagent to fromN,N-dialkyl benzene sulphonamide which
remains insoluble inalkali.
(ii) A secondary amine reacts
with Hinsberg's reagent to fromN,N-dialkyl benzene sulphonamide which
remains insoluble inalkali.
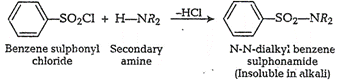 (iii) A tertiary amine does not
react with benzene sulphonyl chloride.
(iii) A tertiary amine does not
react with benzene sulphonyl chloride.
You need to login to perform this action.
You will be redirected in
3 sec
