Answer:
(i)
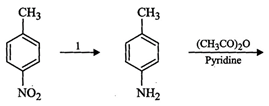
Since the reagent (1) reduces - ![]() to -
to - ![]() , therefore, I is either
Sn/HCl or Fe/HCl.
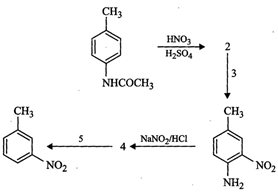
(ii) Since p-methylacetanilide on treatment with a mixture of cone.
, therefore, I is either
Sn/HCl or Fe/HCl.
(ii) Since p-methylacetanilide on treatment with a mixture of cone. ![]() + cone.
+ cone. ![]() gives compound (2), therefore,
compound (2) must be
gives compound (2), therefore,
compound (2) must be
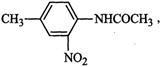 2-nitro-4-methylacetanilide.
(iii) Since reagent (3) brings about hydrolysis of
2-nitro-4-methylacetanilide.
(iii) Since reagent (3) brings about hydrolysis of ![]() to
to ![]() therefore, it must be aqueous
acid, i.e.,
therefore, it must be aqueous
acid, i.e., ![]() .
(iv) Since product (4) is obtained upon treatment of 1°
aromatic amine with
.
(iv) Since product (4) is obtained upon treatment of 1°
aromatic amine with ![]() , therefore, compound
(4) must be a diazonium salt
, therefore, compound
(4) must be a diazonium salt
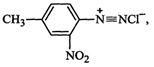 2-Nitro-4= methylbenzenediazonium chloride
(v) Since reagent (5) brings about the substitution of diazo
2-Nitro-4= methylbenzenediazonium chloride
(v) Since reagent (5) brings about the substitution of diazo ![]() group by H or brings about
reduction of diazonium chloride, therefore, it must be aqueous hypophosphorus
acid, i.e.,
group by H or brings about
reduction of diazonium chloride, therefore, it must be aqueous hypophosphorus
acid, i.e., ![]() .
.
You need to login to perform this action.
You will be redirected in
3 sec
