Answer:
Being
an unsymmetrical ketone, oxidation occurs on either side of the C = O group
giving a mixture of 2- methyl propanoic acid, 3-methylbutanoic acid and
propan-2-one. Propan-2-one on further oxidation gives a mixture of ethanoic
acid and methanoic acid. Subsequent oxidation of methanoic acid gives ![]() and
and ![]()
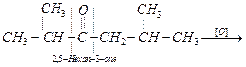
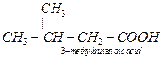
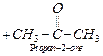
You need to login to perform this action.
You will be redirected in
3 sec
