-
Give an example of a metal which
(a) is liquid at room temperature. (b) can be easily cut with a knife.
(c) is the best conductor of heat. (d) is a poor conductor of heat.
View Answer play_arrow
-
Explain the meanings of malleable and ductile.
View Answer play_arrow
-
Why is sodium kept immersed in kerosene oil ?
View Answer play_arrow
-
Write the equations for the reactions of
(a) iron with steam (b) calcium and potassium with water
View Answer play_arrow
-
Samples of four metals A, B, C and D were taken and added to the following solutions one by one. The results obtained have been tabulated as follows :
|
Metal
|
Solution to which metal is added
|
|
|
Iron (II) sulphate
|
Copper (II) sulphate
|
Zinc sulphate
|
Silver nitrate
|
|
A
|
No reaction
|
Displacement
|
-
|
-
|
|
B
|
Displacement
|
-
|
No reaction
|
-
|
|
C
|
No reaction
|
No reaction
|
No reaction
|
Displacement
|
|
D
|
No reaction
|
No reaction
|
No reaction
|
No reaction
|
Use the table given above to answer the following questions :
(a) Which is the most reactive metal ?
(b) What would you observe when B is added to solution of copper (II) sulphate?
(c) Arrange the metals. A, B, C and D in order of increasing reactivity.
-
Which gas is produced when a reactive metal reacts with dilute hydrochloric acid? Write the chemical reaction when iron reacts with dilute hydrochloric acid. Iron and dilute
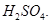
-
What would you observe when zinc is added to a solution of iron (II) sulphate ? Write the chemical reaction that takes place.
View Answer play_arrow
-
(i) Write the electron-dot structures for sodium, oxygen and magnesium.
(ii) Show the formation of
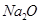
and
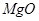
by the transfer of electrons.
(iii) What are the ions present in these compounds?
-
Why do ionic compounds have high melting points?
View Answer play_arrow
-
Define the following terms :
(i) Minerals (ii) Ores (iii) Gangue.
View Answer play_arrow
-
Name two metals which are found in nature in free state.
View Answer play_arrow
-
Which chemical process is used for obtaining a metal from its oxide ?
View Answer play_arrow
-
Metallic oxides of zinc, magnesium and copper were heated with the following metals.
|
Metal
|
Zinc
|
Magnesium
|
Copper
|
|
Zinc Oxide
Magnesium oxide
Copper oxide
|
|
|
|
In which cases, will you find displacement reactions taking place ?
View Answer play_arrow
-
Which metals do not corrode easily ?
View Answer play_arrow
-
What are alloys ?
View Answer play_arrow
-
Which of the following pairs will give displacement reactions ?
(a)
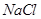
solution and copper metal (b)
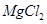
solution and aluminium metal
(c)
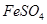
solution and silver metal (d)
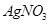
solution and copper metal
-
Which, of the following methods is suitable for preventing' an iron frying pan from rusting?
(a) Applying grease (b) Applying paint
(c) Applying a coating of zinc (d) All of the above
View Answer play_arrow
-
An element reacts with oxygen to give a compound with high melting point. This compound is also water soluble. The element is likely to be :
(a) calcium (b) carbon (c) silicon (d) iron
View Answer play_arrow
-
Food cans are coated with tin and not with zinc because
(a) zinc is costlier than tin (b) zinc has higher melting point than tin
(c) zinc is more reactive than tin (d) zinc is less reactive than tin
View Answer play_arrow
-
You are given a hammer, a battery; a bulb, wires and a switch :
(a) How could you use them to distinguish between samples of metals and non-metals?
(b) Assess the usefulness of these tests in distinguishing between metals and non-metals.
View Answer play_arrow
-
What are amphoteric oxides ? Give two examples of amphoteric oxides.
View Answer play_arrow
-
Name the two metals which will displace hydrogen from dilute acids and two metals which will not.
View Answer play_arrow
-
In the electrolytic refining of a metal M, what would you take as the anode, (he cathode and the electrolyte?
View Answer play_arrow
-
Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it, as shown in figure below.
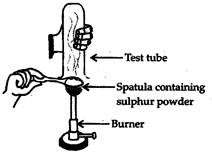
(a) What will be the action of gas on
(i) dry litmus paper? (ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
-
State two ways to prevent the rusting of iron.
View Answer play_arrow
-
What type of oxides are formed when non- metals combine with oxygen ?
View Answer play_arrow
-
Give reasons for the following :
(a) Platinum, gold and silver are used to make jewellery.
(b) Sodium, potassium and lithium are stored under oil.
(c) Aluminium is a highly reactive metal but still used for making cooking utensils.
(d) Carbonate and sulphide ores are usually converted into oxides during the process of extraction.
View Answer play_arrow
-
You must have seen tarnished copper vessels being cleaned with lemon or tamarind juice. Explain why these sour substances are effective in cleaning the vessels.
View Answer play_arrow
-
Differentiate between metals and non-metals on the basis of chemical properties ?.
View Answer play_arrow
-
A man went door to door posing as a goldsmith. He promised to bring back the glitter of old and dull gold ornaments. An unsuspecting lady gave a set of gold bangles to him which he dipped in a particular solution. The bangles sparkled like new but their weight was reduced drastically. The lady got upset and after a futile argument, the man beat a hasty retreat. Can you. predict the nature of the solution used by the imposter ?
View Answer play_arrow
-
Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
View Answer play_arrow
-
question_answer32)
Which of the following property is
generally not shown by metals?
(a) Electrical conduction (b)
Sonorous in nature
(c)
Dullness (d) Ductility
View Answer play_arrow
-
question_answer33)
The ability of metals to be
drawn into thin wire is known as
(a) ductility (b)
malleability (c) sonorousity (d) conductivity
View Answer play_arrow
-
question_answer34)
Aluminium is used for making
cooking utensils. Which of the following properties of aluminium are
responsible for the same?
(i) Good thermal conductivity (ii)
Good electrical conductivity
(iii) Ductility (iv)
High melting point
(a) (i) and (ii) (b)
(i) and (iii) (c) (ii) and (iii) (d) (i)
and (iv)
View Answer play_arrow
-
question_answer35)
Which one of the following metals do not
react with cold as well as hot water?
(a) Na (b)
Ca (c) Mg (d)
Fe
View Answer play_arrow
-
question_answer36)
Which of the following oxide(s) of iron
would be obtained on prolonged reaction of iron with steam?
(a)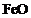 (b)
(b)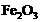 (c)
(c)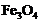 (d)
(d)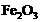 and
and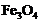
View Answer play_arrow
-
question_answer37)
What happens when calcium is treated
with water?
(i) It does not react with
water.
(ii) It reacts violently with
water.
(iii) If reacts less violently
with water.
(iv) Bubbles of hydrogen gas
formed stick to the surface of calcium.
(a) (i) and (iv) (b)
(ii) and (iii) (c) (i) and (ii) (d)
(iii) and (iv)
View Answer play_arrow
-
question_answer38)
Generally metals react with adds
to give salt and hydrogen gas. Which of the following acids does not give
hydrogen gas on reacting with metals(except Mn and Mg)?
(a)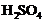 (b)
(b)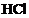 (c)
(c)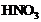 (d)
All of these
(d)
All of these
View Answer play_arrow
-
question_answer39)
The composition of aqua-regia is
(a)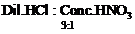 (b)
(b)
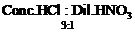
View Answer play_arrow
-
question_answer40)
Which of the following are not
ionic compounds?
(i)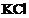 (ii)
(ii)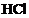 (iii)
(iii)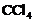 (iv)
(iv)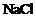 (a) (i) and (ii) (b)
(ii) and (iii) (c) (iii) and (iv) (d) (i)
and (iii)
(a) (i) and (ii) (b)
(ii) and (iii) (c) (iii) and (iv) (d) (i)
and (iii)
View Answer play_arrow
-
question_answer41)
Which one of the following
properties is not generally exhibited by ionic compounds?
(a) Solubility in
water (b) Electrical
conductivity in solid state
(c) High melting and boiling
points (d) Electrical conductivity in molten state
View Answer play_arrow
-
question_answer42)
Which of the following metals
exist in their native state in nature?
(i) Cu (ii)
Au
(iii) Zn
(iv) Ag
(a) (i) and (ii) (b)
(ii) and (iii) (c) (ii) and (iv) (d)
(iii) and (iv)
View Answer play_arrow
-
question_answer43)
Metals are refined by using different
methods. Which of the following metals are refined by electrolytic refining?
(i)
Au (ii) Cu
(iii)
Na (iv) K
(a) (i) and (ii) (b)
(i) and (iii) (c) (ii) and (iii) (d)
(ii) and (iv)
View Answer play_arrow
-
question_answer44)
Silver articles become black on
prolonged exposure to air. This is due to the formation of
(a)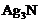 (b)
(b)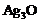 (c)
(c)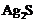 (d)
(d)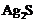 and
and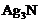
View Answer play_arrow
-
question_answer45)
Galvanization is a method of protecting
iron from rusting by coating it with a thin layer of
(a) gallium (b)
aluminium (c) zinc (d) silver
View Answer play_arrow
-
question_answer46)
Stainless steel is very useful
material for our life. In stainless steel, iron is mixed with
(a) Ni and Cr (b)
Cu and Cr (c) Ni and Cu (d) Cu and Au
View Answer play_arrow
-
question_answer47)
If copper is kept open in air,
it slowly loses its shining brown surface and gains a green coating. It is due
to the formation of
(a)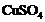 (b)
(b)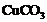 (c)
(c)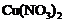 (d)
(d)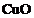
View Answer play_arrow
-
question_answer48)
Generally, metals are solid in nature.
Which one of the following metals is found in liquid state at room temperature?
View Answer play_arrow
-
question_answer49)
Which of the following metals
are obtained by electrolysis of their chlorides in molten state?
(i)
Na (ii) Ca
(iii)
Fe (iv) Cu
(a) (i) and (iv) (b)
(iii) and (iv) (c) (i) and (iii) (d) (i)
and (ii)
View Answer play_arrow
-
question_answer50)
Generally, non-metals are not lustrous.
Which of the following non-metals is lustrous?
(a) Sulphur (b)
Oxygen (c) Nitrogen (d) Iodine
View Answer play_arrow
-
question_answer51)
Which one of the following four
metals would be displaced from the solution of its salts by other three metals?
(a)Mg (b)Ag
(c)Zn (d) Cu
View Answer play_arrow
-
question_answer52)
2 mL each of cone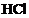 ,
,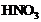 and a
mixture of cone.
and a
mixture of cone.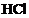 and
cone.
and
cone.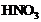 in the
ratio of 3 : 1 were taken in test tubes labeled as A, B and C. A small piece of
metal was put in each test tube. No change occurred in test tubes A and B but
the metal got dissolved in test tube C. The metal could be
(a) Al (b)
Au (c) Cu (d)
Pt
in the
ratio of 3 : 1 were taken in test tubes labeled as A, B and C. A small piece of
metal was put in each test tube. No change occurred in test tubes A and B but
the metal got dissolved in test tube C. The metal could be
(a) Al (b)
Au (c) Cu (d)
Pt
View Answer play_arrow
-
question_answer53)
An alloy is
(a) an element. (b)
a compound
(c) a homogeneous
mixture (d) a heterogeneous mixture
View Answer play_arrow
-
question_answer54)
An electrolytic cell consists of
(i) positively charged
cathode (ii) negatively charged anode
(iii) positively charged
anode (iv) negatively charged cathode
(a) (i) and (ii) (b)
(iii) and (iv) (c) (i) and (iii) (d) (ii)
and (iv)
View Answer play_arrow
-
question_answer55)
During electrolytic refining of
zinc, it gets
(a) deposited on
cathode (b) deposited on anode
(c) deposited on cathode as well
as anode (d) remains in the solution
View Answer play_arrow
-
question_answer56)
An element A is soft and can be cut with
a knife. This is very reactive to air and cannot be kept open in air. It reacts
vigorously with water. Identify the element from the following.
(a) Mg (b)
Na (c) P (d)
Ca
View Answer play_arrow
-
question_answer57)
Alloys are homogeneous mixtures of a metal
with a metal or non-metal. Which among the following alloys contain non-metal
as one of its constituents?
(a) Brass (b)
Bronze (c) Amalgam (d) Steel
View Answer play_arrow
-
question_answer58)
Which among the following statements is
incorrect for magnesium metal?
(a) It burns in oxygen with a
dazzling white flame
(b) It reacts with cold water to
form magnesium oxide and evolves hydrogen gas
(b) It reacts with hot water to
form magnesium hydroxide and evolves hydrogen gas
(d) It reacts with steam to form
magnesium hydroxide and evolves hydrogen gas
View Answer play_arrow
-
question_answer59)
Which among the following alloys
contain mercury as one of its constituents?
(a) Stainless steel (b)Alnico
(c) Solder (d) Zinc amalgam
View Answer play_arrow
-
question_answer60)
Reaction between X and Y, forms
compound Z. X loses electron and Y gains electron. Which of the following
properties is not shown by Z?
(a) Has high melting
point (b) Has low melting point
(c) Conducts electricity in
molten state (d) Occurs as solid
View Answer play_arrow
-
question_answer61)
The electronic configurations of three
elements X, Y and Z are X ? 2, 8; Y ? 2, 8, 7 and Z ? 2, 8, 2. Which of the
following is correct?
(a) X is a
metal (b) Y
is a metal
(c) Z is a
non-metal (d) Visa
non-metal and Z is a metal
View Answer play_arrow
-
question_answer62)
Although metals form basic oxides, which
of the following metals form an amphoteric oxide?
(a) Na (b)
Ca (c) Al (d) Cu
View Answer play_arrow
-
question_answer63)
Generally, non-metals are not conductors
of electricity. Which of the following is a good conductor of electricity?
(a) Diamond (b)
Graphite (c) Sulphur (d)
Fullerene
View Answer play_arrow
-
question_answer64)
Electrical wires have a coating of an
insulating material. The material, generally used is
(a) sulphur (b)
graphite (c) PVC (d) All
can be used
View Answer play_arrow
-
question_answer65)
Which of the following
non-metals is a liquid?
(a) Carbon (b)
Bromine (c) Phosphorus (d) Sulphur
View Answer play_arrow
-
question_answer66)
Which of the following can undergo a
chemical reaction?
(a)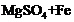 (b)
(b)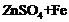 (c)
(c)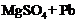 (d)
(d)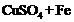
View Answer play_arrow
-
question_answer67)
Which one of the following
figures correctly describes the process of electrolytic refining?
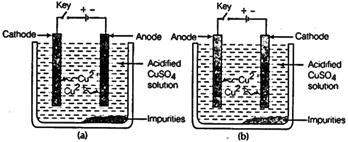
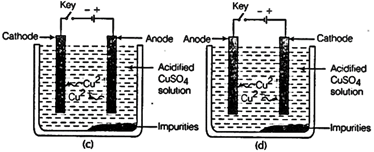
View Answer play_arrow
-
question_answer68)
Iqbal treated a lustrous,
divalent element M with sodium hydroxide. He observed the formation of bubbles
in reaction mixture. He made the same observations when this element was
treated with hydrochloric acid. Suggest how can be identify the produced gas.
Write chemical equations for both the reactions.
View Answer play_arrow
-
question_answer69)
During extraction of metals,
electrolytic refining is used to obtain pure metals, (a)Which material will be
used as anode and cathode for refining of silver metal by this process? (b)
Suggest a suitable electrolyte also. (c) In this electrolytic cell, where do we
get pure silver after passing electric current?
View Answer play_arrow
-
question_answer70)
Why should the metal sulphides
and carbonates be converted to metal oxides in the process of extraction of
metal from them?
View Answer play_arrow
-
question_answer71)
Generally, when metals are
treated with mineral acids, hydrogen gas is liberated but when metals (except
Mn and Mg) are treated with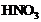 ,hydrogen
is not liberated, why?
,hydrogen
is not liberated, why?
View Answer play_arrow
-
question_answer72)
Compound X and aluminium are
used to join railway tracks, (a) Identify the compound X (b) Name the reaction
(c) Write down its reaction.
View Answer play_arrow
-
question_answer73)
When a metal X is treated with
cold water, it gives a basic salt Y with molecular formula XOH (molecular mass
=40) and liberates a gas Z which easily catches fire. Identify X, Y and Z and
also write the reaction involved.
View Answer play_arrow
-
question_answer74)
A non-metal X exists in two
different forms Y and Z. Y is hardest natural substance, whereas Z is a good
conductor of electricity. Identify X, Y and Z.
View Answer play_arrow
-
question_answer75)
The following reaction takes
place when aluminium powder is heated with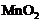
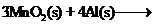
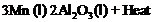 (a) Is aluminium getting
reduced?
(b) Is
(a) Is aluminium getting
reduced?
(b) Is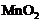 getting oxidized?
getting oxidized?
View Answer play_arrow
-
question_answer76)
What are the constituents of
solder alloy? Which property of solder makes it suitable for welding electrical
wires?
View Answer play_arrow
-
question_answer77)
A metal A, which is used in thermite
process, when heated with oxygen gives an oxide B, which is amphoteric in
nature. Identify A and B. Write down the reactions of oxide B with HCl and NaOH.
View Answer play_arrow
-
question_answer78)
A metal that exists as a liquid at room
temperature is obtained by heating its sulphide in the presence of air.
Identify the metal and its ore and give the reactions involved.
View Answer play_arrow
-
question_answer79)
Give the formulae of the stable binary
compounds that would be formed by the combination of following pairs of
elements.
(a) Mg and N2 (b)
Li and O2 (c) Al and Cl2 (d)
K and O2
View Answer play_arrow
-
question_answer80)
What happens when
(a) 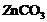 is heated
in the absence of oxygen?
(b) a mixture of
is heated
in the absence of oxygen?
(b) a mixture of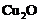 and
and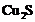 is heated?
is heated?
View Answer play_arrow
-
question_answer81)
A non-metal A is an important
constituent of our food and forms two oxides B and C. Oxide B is toxic whereas
C causes global warming.
(a) Identify A, B and C.
(b) To which group of periodic
table does A belong?
View Answer play_arrow
-
question_answer82)
Give two examples each of the
metals that are good conductors and comparatively poor conductors of heat
respectively.
View Answer play_arrow
-
question_answer83)
Name one metal and one non-metal
that exist in liquid state at room temperature. Also name two metals having
melting point less than 310 K(37°C).
View Answer play_arrow
-
question_answer84)
An element A reacts with water
to form a compound B which is used in white washing. The compound B on heating
forms an oxide C which on treatment with water gives back B. Identify A, B and
C and give the reactions involved.
View Answer play_arrow
-
question_answer85)
An alkali metal A gives a
compound B (molecular mass = 40) on reacting with water. The compound B gives a
soluble compound C on treatment with aluminium oxide. Identify A, B and C and
give the reactions involved.
View Answer play_arrow
-
question_answer86)
Give the reaction involved
during extraction of zinc from its ore by roasting of zinc ore. (b) calcination
of zinc ore.
View Answer play_arrow
-
question_answer87)
A metal M does not liberate
hydrogen from adds but reacts with oxygen to give a black colour product.
Identify M and black coloured product and also explain the reaction of M with
oxygen.
View Answer play_arrow
-
question_answer88)
An element forms an oxide A2O3
which is acidic in nature. Identify A as a metal or non-metal.
View Answer play_arrow
-
question_answer89)
A solution of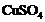 was kept
in an iron pot. After few days the iron pot was found to have a number of holes
in it. Explain the reason in terms of reactivity. Write the equation of the
reaction involved.
was kept
in an iron pot. After few days the iron pot was found to have a number of holes
in it. Explain the reason in terms of reactivity. Write the equation of the
reaction involved.
View Answer play_arrow
-
question_answer90)
A non-metal A which is the largest
constituent of air, when heated with H2 in 1: 3 ratio in the
presence of catalyst (Fe) gives a gas B. On heating with O2 it gives
an oxide C. If this oxide is passed into water in the presence of air, it gives
an acid D which acts as a strong oxidizing agent.
(a) Identify A, B, C and D.
(b) To which group of the
periodic table does this non-metal belongs?
View Answer play_arrow
-
question_answer91)
Give the steps involved in the
extraction of metals of low and medium reactivity from their respective
sulphide ores.
View Answer play_arrow
-
question_answer92)
Explain the following
(a) Reactivity of Al decreases
if it is dipped in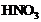 .
(b) Carbon cannot reduce the
oxides of Na or Mg.
(c) NaCI is not a conductor of
electricity in solid state whereas it does conduct electricity in aqueous
solution as well as in molten state.
(d) Iron articles are galvanized.
(e) Metals like Na, K, Ca and Mg
are never found in their free state in nature.
.
(b) Carbon cannot reduce the
oxides of Na or Mg.
(c) NaCI is not a conductor of
electricity in solid state whereas it does conduct electricity in aqueous
solution as well as in molten state.
(d) Iron articles are galvanized.
(e) Metals like Na, K, Ca and Mg
are never found in their free state in nature.
View Answer play_arrow
-
question_answer93)
(a) Given below are the steps
for extraction of copper from its ore. Write the reaction involved.
(i) Roasting of copper (I)
sulphide.
(ii) Reduction of copper (I)
oxide with copper (I) sulphide.
(iii) Electrolytic refining
(b) Draw a neat and well labeled
diagram for electrolytic refining of copper.
View Answer play_arrow
-
question_answer94)
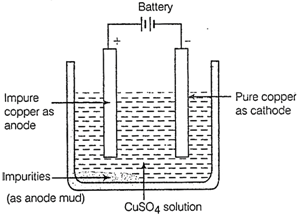 Of the three metals X, Y and Z, X reacts
with cold water, Y with hot water and Z with steam only. Identify X, Y and Z
and also arrange the min order of increasing reactivity.
Of the three metals X, Y and Z, X reacts
with cold water, Y with hot water and Z with steam only. Identify X, Y and Z
and also arrange the min order of increasing reactivity.
View Answer play_arrow
-
question_answer95)
An element A burns with golden flame in
air. It reacts with another element B, atomic number 17 to give a product C. An
aqueous solution of product C on electrolysis gives a compound D and liberates
hydrogen. Identify A, B, C and D. Also write down the equations for the reactions
involved.
View Answer play_arrow
-
question_answer96)
Two ores A and B were taken. On heating,
ore A gives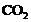 whereas,
ore B gives
whereas,
ore B gives 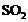 . What
steps will you take to convert them into metals?
. What
steps will you take to convert them into metals?
View Answer play_arrow
 (a) What will be the action of gas on
(i) dry litmus paper? (ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
(a) What will be the action of gas on
(i) dry litmus paper? (ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
![]() (b)
(b)
![]()
![]() (b)
(b)![]() (c)
(c)![]() (d)
(d)![]()
