Charged Particles
Category : 7th Class
Electron, Proton and Neutron are the three fundamental particles, which an atom is made up of. Out of the three, electron and proton are charged particles, whereas neutron is the neutral particle. Electron is negatively charged and proton is positively charged. Magnitude of charge on a proton is equal to that of an electron, but nature of the charge is opposite.
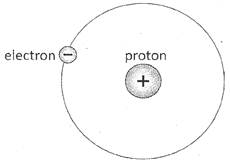
In an atom, number of protons is equal to the number of electrons. Therefore, atoms are neutral. But transfer of electrons from one atom to other atom causes the atoms to be charged. The atom, which looses the electrons gets positively charged because number of positively charged particle (protons) in the atom gets greater than that of negatively charged particles (electrons). Therefore, magnitude of total positive charge becomes greater than that of negative charge. And the atom, which receives the electrons gets negatively charged, because number of negatively charged particle (electrons) in the atom gets greater than that of positively charged particles (protons). Hence, total magnitude of positive charge becomes greater than that of negative charge.
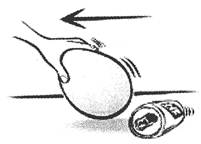
![]() Charge Produced Due to Direction
Charge Produced Due to Direction
Proton and neutron are present in the center of an atom, which is called nucleus. The electrons are revolving around the nucleus. The electrons close to the nucleus are tightly held by force of attraction produced by protons. And the electrons, which are far from the nucleus are loosely held; because force of attraction becomes weaker as the distance increases.
When we rub two substances with each other, some electrons, which are loosely held by nucleus, are transferred from one substance to other. As a result, the is substance that looses electrons, gets positively charged and the substance that gains electrons, gets negatively charged.
![]() Force Around a Charged Particles
Force Around a Charged Particles
Charged particles have a force around them. This force can be experienced by bringing another charged particle close to them. Nature of the force is attraction or repulsion depending on the nature of the charged particles. If the charged particles are alike (both positively charged or both negatively charged), the force of repulsion works between them. And if charged particles are unlike (one positively charged and other negatively charged), the force of attraction works between them. This is why between two protons or two electrons there is a force of repulsion and between an electron and a proton there is a force of attraction.
![]() Movement of charged particles
Movement of charged particles
Moving of charged particles has a number of applications for us, as this movement produces electricity. We already know that protons and electrons are the charged particles and when transfer of electrons occurs between two atoms, both the atoms get charged. Electricity is produced when any charged body move, like electrons, protons or charged atoms. Protons are tightly held in the nucleus and it cannot move from atom to atom like electron. Therefore, the electricity that we get is produced by moving electrons.

![]() Which one of the following kind of energy is produced when a charged particle moves?
Which one of the following kind of energy is produced when a charged particle moves?
(a) Electrical energy
(b) Chemical energy
(c) Light energy
(d) Sound energy
(e) None of these
Answer: (a)
![]() An atom gets charged because of ______.
An atom gets charged because of ______.
(a) Transfer of proton
(b) Transfer of neutron
(c) Transfer of electron
(d) Transfer of nucleus
(e) None of these
Answer: (c)
You need to login to perform this action.
You will be redirected in
3 sec
