Atoms
Category : 4th Class
All the things that we can see around us, whether living or non-living, are matter if it takes up space and has weight. It can be dog, cat, bat, ball, air, water, etc.

Matters
Matter is made up of tiny particles. These tiny particles are too small to be seen. Millions and millions of these tiny particles put together to form a matter. These tiny particles are known as molecules. A molecule is the smaller unit of any substance that has all the properties of that particular substance. A molecule of aluminum has all the properties of aluminium. The molecule of different substances are different from each other. For example molecule of aluminium is different from the molecule of iron. The amount of space that a matter takes up is called its volume.
.
![]() Atoms
Atoms
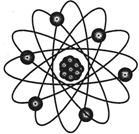
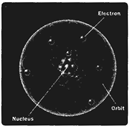
Atoms are made up of fundamental particles electrons, protons, and neutrons. Protons and neutrons lie in the center of the atom and electrons are moving around the nucleus in certain orbit.
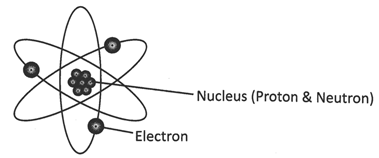
Diagrammatic representation of Atom
All materials of the universe are made up of minute particles called atoms. Structure of atoms of some elements has been given below
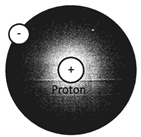
Atom of hydrogen Atom of carbon Atom of nitrogen
Atom of lithium Atom of oxygen
Atoms are the basic building blocks of matter. Atoms can join together to form molecules, which in turn form the objects around us. It is not possible to breakdown the atom further retaining the properties of the element. Atoms are not visible to the naked eye. For example the atoms of the element carbon cannot be further broken down and each part has the properties of carbon.

![]() Consider the statements given bellow and select the right answer.
Consider the statements given bellow and select the right answer.
Statement A: Molecules are made up of atoms.
Statement B: At the center of an atom is the nucleus.
(a) Statement: A is True
(b) Statement: B is True
(c) Both Statement: A and B are True
(d) Both Statement: A and B are false
(e) None of these
Answer: (c)
![]() Which of the following instrument helps us to see the tiny atoms?
Which of the following instrument helps us to see the tiny atoms?
(a) 
(b) 
(c) 
(d) All of these
(e) None of these
Answer: (c)
You need to login to perform this action.
You will be redirected in
3 sec
