question_answer 1) Mass equivalent to energy 931 MeV is
A)
\[6.02\times {{10}^{-27}}\,kg\]
done
clear
B)
\[1.66\times {{10}^{-27}}kg\]
done
clear
C)
\[16.66\,\times {{10}^{-27}}\,kg\]
done
clear
D)
\[6.02\,\times {{10}^{-27}}\,kg\]
done
clear
View Answer play_arrow
question_answer 2) Plutonium-decays with a half-life of 24000 years. If plutonium is stored for 72000 years, then the fraction of plutonium that remains, is
A)
\[\frac{1}{8}\]
done
clear
B)
\[\frac{1}{4}\]
done
clear
C)
\[\frac{1}{3}\]
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 3) If light travels a distance x in \[{{t}_{1}}\,\sec \] in air and 10 x distance in \[{{t}_{2}}\,\sec \] in a medium, the critical angle of the medium will be
A)
\[{{\tan }^{-1}}\left( \frac{{{t}_{1}}}{{{t}_{2}}} \right)\]
done
clear
B)
\[{{\sin }^{-1}}\left( \frac{{{t}_{1}}}{{{t}_{2}}} \right)\]
done
clear
C)
\[{{\sin }^{-1}}\left( \frac{10{{t}_{1}}}{{{t}_{2}}} \right)\]
done
clear
D)
\[{{\tan }^{-1}}\left( \frac{10{{t}_{1}}}{{{t}_{2}}} \right)\]
done
clear
View Answer play_arrow
question_answer 4) In an electromagnetic wave, the electric and magnetic fields are 100 V/m and 0.265 A/m. The maximum energy flow will be
A)
\[79\,W/{{m}^{2}}\]
done
clear
B)
\[13.2\,W/{{m}^{2}}\]
done
clear
C)
\[53.0\,\,W/{{m}^{2}}\]
done
clear
D)
\[26.5\,\,W/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 5) If a current of 10 A flows in one second through a coil and the induced emf is 10V, then the self-inductance of the coil will be
A)
1 H
done
clear
B)
2 H
done
clear
C)
4 H
done
clear
D)
\[\frac{2}{5}H\]
done
clear
View Answer play_arrow
question_answer 6) The frequency of an alternating voltage is 50 cycles/sec and its amplitude is120 V. Then its rms value will be
A)
84.9 V
done
clear
B)
42.4 V
done
clear
C)
56.5 V
done
clear
D)
75.5 V
done
clear
View Answer play_arrow
question_answer 7) Two waves of wavelength 50 cm and 51cm produce 12 beats per sec. The velocity of sound will be
A)
340 m/s
done
clear
B)
332 m/s
done
clear
C)
153 m/s
done
clear
D)
306 m/s
done
clear
View Answer play_arrow
question_answer 8) The wavelength of radiation emitted is\[{{\lambda }_{0}}\] when an electron in hydrogen atom jumps from 3rd to 2nd orbit. If in the hydrogen atom itself, the electron jumps from fourth orbit to second orbit, then wavelength of emtted radiation will be
A)
\[\frac{25}{16}{{\lambda }_{0}}\]
done
clear
B)
\[\frac{27}{20}{{\lambda }_{0}}\]
done
clear
C)
\[\frac{20}{27}{{\lambda }_{0}}\]
done
clear
D)
\[\frac{16}{25}{{\lambda }_{0}}\]
done
clear
View Answer play_arrow
question_answer 9) A thin circular ring of mass M and radius r is rotating about its axis with a constant angular velocity\[\omega \]. If two objects of mass m are attached gently to opposite ends of a diameter of ring, ring will now rotate with an angular velocity given by
A)
\[\frac{2\omega M}{(M-2m)}\]
done
clear
B)
\[\frac{(M-2m)}{M}\]
done
clear
C)
\[\frac{\omega M}{(M+2m)}\]
done
clear
D)
\[\frac{2\omega M}{(M-2m)}\]
done
clear
View Answer play_arrow
question_answer 10) A bomber plane moves horizontally with a speed of 500 m/s and a bomb released from it, strikes the ground in 10 sec. Angle at which it strikes the ground will be \[(g=10\,m/{{s}^{2}})\]
A)
\[{{\tan }^{-1}}\left( \frac{1}{5} \right)\]
done
clear
B)
\[\tan \left( \frac{1}{5} \right)\]
done
clear
C)
\[{{\tan }^{-1}}(1)\]
done
clear
D)
\[{{\tan }^{-1}}(5)\]
done
clear
View Answer play_arrow
question_answer 11) Black hole is
A)
super surface of atmosphere
done
clear
B)
ozone layer
done
clear
C)
super dense planetary material
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 12) The dual nature of light is exhibited by
A)
diffraction and photoelectric effect
done
clear
B)
photoelectric effect
done
clear
C)
refraction and interference
done
clear
D)
diffraction and reflection
done
clear
View Answer play_arrow
question_answer 13) The number of lenses in terrestrial telescope will be
A)
6
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 14) The direction of null points are on the equatorial line of a bar magnet, when the north pole of the magnet is pointing
A)
west
done
clear
B)
east
done
clear
C)
south
done
clear
D)
north
done
clear
View Answer play_arrow
question_answer 15) If the distance between the plates of parallel plate capacitor is halved and the dielectric constant is doubled, then its capacity will
A)
increase by 16 times
done
clear
B)
increase by 4 times
done
clear
C)
increase by 2 times
done
clear
D)
remain the same
done
clear
View Answer play_arrow
question_answer 16) A particle of mass m and charge q is placed at rest in a uniform electric field E and then released, the kinetic energy attained by the particle after moving a distance y, will be
A)
\[{{q}^{2}}Ey\]
done
clear
B)
\[qEy\]
done
clear
C)
\[q{{E}^{2}}y\]
done
clear
D)
\[qE{{y}^{2}}\]
done
clear
View Answer play_arrow
question_answer 17) Time period of a simple pendulum will be double, if we
A)
decrease the length 2 times
done
clear
B)
decrease the length 4 times
done
clear
C)
increase the length 2 times
done
clear
D)
increase the length 4 times
done
clear
View Answer play_arrow
question_answer 18) Two gases A and B having same pressure P, volume V and temperature T are mixed. If mixture has volume and temperature as V and T respectively, then the pressure of the mixture will be
A)
4 P
done
clear
B)
3 P
done
clear
C)
2 P
done
clear
D)
P
done
clear
View Answer play_arrow
question_answer 19) The maximum velocity and maximum acceleration of body moving in a simple harmonic motion are 2 m/s and \[4\,m/{{s}^{2}}\] respectively. Then the angular velocity will be
A)
4 rad/sec
done
clear
B)
3 rad/sec
done
clear
C)
2 rad /sec
done
clear
D)
8 rad/sec
done
clear
View Answer play_arrow
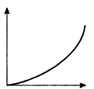
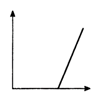
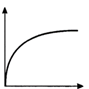
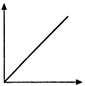
question_answer 20) The velocity v acquired by an electron starting from rest and moving through potential difference V is shown by which of the following the graphs ?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 21) The major carriers of current in a p-type semi-conductor will be
A)
neutrons
done
clear
B)
protons
done
clear
C)
electrons
done
clear
D)
holes
done
clear
View Answer play_arrow
question_answer 22) The longest wavelength in Balmer series of hydrogen spectrum will be
A)
\[6557\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[1216\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[4800\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[5600\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 23) Myopia is corrected by
A)
cylindrical lens
done
clear
B)
bifocal lens
done
clear
C)
convex lens
done
clear
D)
concave lens
done
clear
View Answer play_arrow
question_answer 24) A moving coil produces an induced emf, this is in accordance with
A)
Amperes law
done
clear
B)
Coulomb law
done
clear
C)
Faradays law
done
clear
D)
Lenzs law
done
clear
View Answer play_arrow
question_answer 25) If a steady current of 4 amp maintained for 40 minutes, deposits 4.5 g of zinc at the cathode, then the chemical equivalent will be
A)
\[51\times {{10}^{-17}}g/C\]
done
clear
B)
\[28\times {{10}^{-6}}g/C\]
done
clear
C)
\[32\times {{10}^{-5}}g/C\]
done
clear
D)
\[47\times {{10}^{-5}}g/C\]
done
clear
View Answer play_arrow
question_answer 26) Two parallel wires of length 9 m each are separated by a distance 0.15 m. If they carry equal currents in the same direction and exerts a total force of \[30\times {{10}^{-7}}N\] on each other, then the value of current must be
A)
2.5 amp
done
clear
B)
3.5 amp
done
clear
C)
1.5 amp
done
clear
D)
0.5 amp
done
clear
View Answer play_arrow
question_answer 27) The frequency of tuning fork is 256 Hz. It will not resonate with a fork of frequency
A)
768 Hz
done
clear
B)
738 Hz
done
clear
C)
512 Hz
done
clear
D)
256 Hz
done
clear
View Answer play_arrow
question_answer 28) Antimony and bismuth are usually used in a thermocouple, because
A)
negative thermal emf is produced
done
clear
B)
constant thermal emf is produced
done
clear
C)
lower thermal emf is produced
done
clear
D)
higher thermal emf is produced
done
clear
View Answer play_arrow
question_answer 29) A person sitting in an open car moving at constant velocity throws a ball vertically into air, the ball falls
A)
exactly in the hand of thrower
done
clear
B)
outside the car
done
clear
C)
in the car ahead the person
done
clear
D)
in the car behind the person
done
clear
View Answer play_arrow
question_answer 30) The force on a rocket moving with a velocity of 300 m/s is 210 N. Then the rate of combustion of the fuel will be
A)
3.5 kg/sec
done
clear
B)
2.1 kg/sec
done
clear
C)
1.4 kg/sec
done
clear
D)
0.7 kg/sec
done
clear
View Answer play_arrow
question_answer 31) Ina p-type semi-conductor, germanium is dopped with
A)
gallium
done
clear
B)
boron
done
clear
C)
aluminum
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 32) The stopping potential, when a metal with work function 0.6 eV is illuminated with light of energy 2 eV will be
A)
1.4 V
done
clear
B)
2.8 eV
done
clear
C)
4.2 eV
done
clear
D)
0.7 V
done
clear
View Answer play_arrow
question_answer 33) Magnetic permeability is maximum for
A)
ferromagnetic substances
done
clear
B)
diamagnetic substances
done
clear
C)
paramagnetic substances
done
clear
D)
all of these
done
clear
View Answer play_arrow
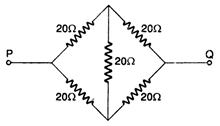
question_answer 34)
The equivalent resistance between P and Q in given figure, will be:
A)
50\[\Omega \]
done
clear
B)
40\[\Omega \]
done
clear
C)
20 \[\Omega \]
done
clear
D)
10\[\Omega \]
done
clear
View Answer play_arrow
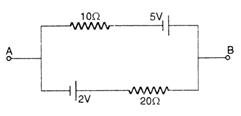
question_answer 35)
The current in the given circuit will be
A)
0.4 A
done
clear
B)
0.3 A
done
clear
C)
0. 2 A
done
clear
D)
0.1 A
done
clear
View Answer play_arrow
question_answer 36) Two charged spheres separated by a distance d exert some force F on each other. If they are immersed in a liquid of dielectric constant 2, then the force exerted by them, if all other conditions are same, is
A)
4 F
done
clear
B)
2 F
done
clear
C)
F
done
clear
D)
F/2
done
clear
View Answer play_arrow
question_answer 37) Two springs of spring constants \[{{k}_{1}}\] and \[{{k}_{2}}\] have equal maximum velocities. When executing simple harmonic motion, the ratio of their amplitudes (masses are equal) will be
A)
\[{{\left( \frac{{{k}_{2}}}{{{k}_{1}}} \right)}^{{1}/{2}\;}}\]
done
clear
B)
\[{{\left( \frac{{{k}_{1}}}{{{k}_{2}}} \right)}^{{1}/{2}\;}}\]
done
clear
C)
\[\frac{{{k}_{2}}}{{{k}_{1}}}\]
done
clear
D)
\[{{k}_{1}}{{k}_{2}}\]
done
clear
View Answer play_arrow
question_answer 38) Colour appears on a thin film of soap and on soap bubble due to:
A)
diffraction
done
clear
B)
interference
done
clear
C)
dispersion
done
clear
D)
refraction
done
clear
View Answer play_arrow
question_answer 39) If the earth loses its gravity, then for a body:
A)
weight becomes zero, but not the mass
done
clear
B)
mass becomes zero but not weight
done
clear
C)
neither mass nor weight is zero
done
clear
D)
both mass and weight are zero
done
clear
View Answer play_arrow
question_answer 40) The critical angle of a medium with respect to air is \[{{45}^{\text{o}}}\]. The refractive index of medium is
A)
1.41
done
clear
B)
1.2
done
clear
C)
1.5
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 41) When an electron is emitted from a nucleus, then effect on its neutron- proton ratio will
A)
remain same
done
clear
B)
increase
done
clear
C)
decrease
done
clear
D)
either (a) or (b)
done
clear
View Answer play_arrow
question_answer 42) The spectrum obtained from a sodium vapour lamp is an example of
A)
absorption spectrum
done
clear
B)
emission spectrum
done
clear
C)
continuous spectrum
done
clear
D)
band spectrum
done
clear
View Answer play_arrow
question_answer 43) Electromagnetic waves travel in a medium which has relative permeability 1.3 and relative permittivity 2.14. Then the speed of the electromagnetic wave in the medium will be
A)
\[13.6\,\times {{10}^{6}}\,m/s\]
done
clear
B)
\[1.8\times {{10}^{2}}\,m/s\]
done
clear
C)
\[3.6\,\times {{10}^{8}}\,m/s\]
done
clear
D)
\[1.8\times {{10}^{8}}\,m/s\]
done
clear
View Answer play_arrow
question_answer 44) A wire of length 0.5 m and carrying the current of 1.2 amp is placed in a uniform magnetic field of induction 2 tesla. If the magnetic field is perpendicular to the length of the wire, then the force on the wire will be
A)
1.2 N
done
clear
B)
4.8 N
done
clear
C)
2.4 N
done
clear
D)
1.8 N
done
clear
View Answer play_arrow
question_answer 45) The resistors of resistances 2\[\Omega \], 4\[\Omega \] 5\[\Omega \] are connected in parallel. The total resistance of the combination will be:
A)
\[\frac{20}{19}\Omega \]
done
clear
B)
\[\frac{19}{20}\Omega \]
done
clear
C)
\[\frac{10}{20}\Omega \]
done
clear
D)
\[\frac{29}{10}\Omega \]
done
clear
View Answer play_arrow
question_answer 46) Assertion: A larger dry cell has higher emf. Reason: The emf of a dry cell is proportional to its size.
A)
If both assertion and reason are true and reason is the correct explanation of assertion
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion
done
clear
C)
If assertion is true but reason is false
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 47) Assertion: A red object appears dark in the yellow light. Reason: The red colour is scattered less.
A)
If both assertion and reason are true and reason is the correct explanation of assertion
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion
done
clear
C)
If assertion is true but reason is false
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 48) Assertion: In a pressure cooker the water is brought to boil. The cooker is then removed from the stove. Now on removing the lid of the pressure cooker, the water starts boiling again. Reason: The impurities in water bring down its boiling point.
A)
If both assertion and reason are true and reason is the correct explanation of assertion
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion
done
clear
C)
If assertion is true but reason is false
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 49) Assertion: The true geographic north direction is found by using a compass needle. Reason: The magnetic meridian of the earth is along the axis of rotation of the earth.
A)
If both assertion and reason are true and reason is the correct explanation of assertion
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion
done
clear
C)
If assertion is true but reason is false
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 50) Assertion: There are very small sporadic changes in the speed of rotation of the earth. Reason: Shifting of large air masses in the earths atmosphere produce a change in the moment of inertia of the earth causing its speed of rotation to change.
A)
If both assertion and reason are true and reason is the correct explanation of assertion
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion
done
clear
C)
If assertion is true but reason is false
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 51) Fats, on alkaline hydrolysis, gives
A)
oils
done
clear
B)
soaps
done
clear
C)
detergents
done
clear
D)
glycol + acid
done
clear
View Answer play_arrow
question_answer 52) The rate of diffusion of hydrogen gas is
A)
1.4 times to He gas
done
clear
B)
same as He gas
done
clear
C)
5 times to He gas
done
clear
D)
2 times to He gas
done
clear
View Answer play_arrow
question_answer 53) If 0.15 g of a solute, dissolved is 15 g of solvent, is boiled at a temperature higher by \[0.216{{\,}^{\text{o}}}C,\] than that of the pure solvent. The molecular weight of the substances (molal elevation constant for the solvent is \[2.16{}^\circ C\]) is:
A)
100
done
clear
B)
10.1
done
clear
C)
10
done
clear
D)
1.001
done
clear
View Answer play_arrow
question_answer 54) Which of the following is a three dimensional silicate?
A)
Mica
done
clear
B)
Spodumene
done
clear
C)
Zeolite
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 55) Which type of bond is present in \[{{\text{H}}_{\text{2}}}\text{S}\] molecule:
A)
ionic bond
done
clear
B)
covalent bond
done
clear
C)
co-ordinate
done
clear
D)
all of three.
done
clear
View Answer play_arrow
question_answer 56) \[\text{MgC}{{\text{l}}_{\text{2}}}\text{6}{{\text{H}}_{\text{2}}}\text{O}\text{.}\]When heated gives:
A)
magnesium oxide
done
clear
B)
magnesium oxychloride
done
clear
C)
magnesium dichloride
done
clear
D)
magnesium chloride
done
clear
View Answer play_arrow
question_answer 57) A certain mass of gas occupies a volume of 300 c.c. at 27C and 620 mm pressure. The volume of this gas at \[\text{47}{{\,}^{\text{o}}}\text{C}\] and 640 mm pressure will be
A)
400 c.c.
done
clear
B)
510 c.c.
done
clear
C)
310 c.c.
done
clear
D)
350 c.c.
done
clear
View Answer play_arrow
question_answer 58) Which of the following is a polar compound?
A)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
B)
\[CC{{l}_{4}}\]
done
clear
C)
\[HCl\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 59) When sand is heated with hydrofluoric acid and a wet rod is brought in contact with vapours evolving, a white deposit is due to
A)
\[Si{{F}_{4}}\]
done
clear
B)
\[Si{{F}_{2}}\]
done
clear
C)
\[{{H}_{4}}Si{{O}_{4}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 60) Which of the following process is concerned with the formation of ethers?
A)
Wurtz synthesis
done
clear
B)
Williamson synthesis
done
clear
C)
Kolbe synthesis
done
clear
D)
Hofmann synthesis
done
clear
View Answer play_arrow
question_answer 61) Which of the following gives methane \[[C{{H}_{4}}]\]on hydrolysis?
A)
\[F{{e}_{3}}{{O}_{4}}\]
done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
C)
\[Ca{{C}_{2}}\]
done
clear
D)
\[A{{l}_{4}}{{C}_{3}}\]
done
clear
View Answer play_arrow
question_answer 62) The number of isomers in \[{{\text{C}}_{\text{4}}}{{\text{H}}_{\text{10}}}\text{O}\] are:
A)
7
done
clear
B)
8
done
clear
C)
6
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 63) In which of the following, the bond length between hybridized carbon atom and other carbon atom is minimum?
A)
Propyne
done
clear
B)
Propene
done
clear
C)
Butane
done
clear
D)
Propane
done
clear
View Answer play_arrow
question_answer 64) The amount of silver deposited by passing 241.25 coulomb of current through silver nitrate solution is
A)
2.7g
done
clear
B)
2.7mg
done
clear
C)
0.27g
done
clear
D)
0.54g
done
clear
View Answer play_arrow
question_answer 65) The shape of \[\text{I}{{\text{F}}_{7}}\]molecule is
A)
pentagonal bipyramidal
done
clear
B)
trigonal bipyramidal
done
clear
C)
tetrahedral
done
clear
D)
octahedral
done
clear
View Answer play_arrow
question_answer 66) Oxidation state of iron in \[\text{F}{{\text{e}}_{\text{3}}}{{\text{O}}_{\text{4}}}\]is
A)
\[\frac{8}{3}\]
done
clear
B)
\[\frac{3}{4}\]
done
clear
C)
\[\frac{3}{2}\]
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 67) Reduction of nitrobenzene, in the presence of Zn and KOH, gives
A)
hydrazobenzene
done
clear
B)
nitrobenzene
done
clear
C)
azobenzene
done
clear
D)
azoxybenzene
done
clear
View Answer play_arrow
question_answer 68) An orbital in which \[n=4\]and \[l=2\]is expressed by
A)
\[4\,s\]
done
clear
B)
\[4\,p\]
done
clear
C)
\[4d\]
done
clear
D)
\[sp\]
done
clear
View Answer play_arrow
question_answer 69) Hydrogen peroxide is prepared in the laboratory by
A)
adding \[\text{Mn}{{\text{O}}_{\text{2}}}\]to dil \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\]
done
clear
B)
by passing \[\text{C}{{\text{O}}_{2}}\]into \[\text{Ba}{{\text{O}}_{\text{2}}}\]
done
clear
C)
by adding \[\text{N}{{\text{a}}_{\text{2}}}{{\text{O}}_{\text{2}}}\]to cold water
done
clear
D)
by adding \[\text{Pb}{{\text{O}}_{\text{2}}}\]into \[\text{KMn}{{\text{O}}_{\text{4}}}\]
done
clear
View Answer play_arrow
question_answer 70) The correct order of reactivity of halogens is
A)
\[F>Cl>Br>I\]
done
clear
B)
\[F<Cl>Br<I\]
done
clear
C)
\[F<Cl<Br<I\]
done
clear
D)
\[F<Cl<Br>I\]
done
clear
View Answer play_arrow
question_answer 71) Cyanohydrin of which of the following forms lactic acid?
A)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[HCHO\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 72) Which of the following compound reacts with chloroform and a base to form phenyl isocyanide?
A)
Phenol
done
clear
B)
Aniline
done
clear
C)
Benzene
done
clear
D)
Nitro benzene
done
clear
View Answer play_arrow
question_answer 73) If gas, at constant temperature and pressure expands then its
A)
entropy increases and then decreases
done
clear
B)
internal energy increases
done
clear
C)
internal energy remains the same
done
clear
D)
internal energy decreases
done
clear
View Answer play_arrow
question_answer 74) In a polar molecule, the ionic charge is \[4.8\times {{10}^{-10}}\] e. s. u. If the inter ionic distance is one \[\overset{\text{o}}{\mathop{\text{A}}}\,\]unit, then the dipole moment is
A)
41.8 debye
done
clear
B)
4.18 debye
done
clear
C)
4.8 debye
done
clear
D)
0.48 debye
done
clear
View Answer play_arrow
question_answer 75) The e. m. f. of a galvanic cell with electrode potentials of \[Z{{n}^{2+}}/Zn=-0.76\,V.\]and that of \[C{{u}^{2+}}/Cu=+\,0.34V,\]is
A)
+ 0.34V
done
clear
B)
+ 0.76V
done
clear
C)
-1.1 V
done
clear
D)
+1.1 V
done
clear
View Answer play_arrow
question_answer 76) Identify the transition element
A)
\[1{{s}^{2}},\text{ }2{{s}^{2}}\text{ }2{{p}^{6}},\text{ }3{{s}^{2}},\text{ }3{{p}^{6}},\text{ }4{{s}^{2}}\]
done
clear
B)
\[1{{s}^{2}},\text{ }2{{s}^{2}}\text{ }2{{p}^{6}},\text{ }3{{s}^{2}},\text{ }3{{p}^{6}}\text{ }3{{d}^{2}},\text{ }4{{s}^{2}}\]
done
clear
C)
\[1{{s}^{2}},\text{ }2{{s}^{2}}\text{ }2{{p}^{6}},\text{ }3{{s}^{2}},\text{ }3{{p}^{6}}\text{ }3{{d}^{10}},\text{ }4{{s}^{2}}\text{ }4{{p}^{2}}\]
done
clear
D)
\[1{{s}^{2}},\text{ }2{{s}^{2}}\text{ }2{{p}^{6}},\text{ }3{{s}^{2}}\text{ }3{{p}^{6}}\text{ }3{{d}^{10}},\text{ }4{{s}^{2}}\text{ }4{{p}^{1}}\]
done
clear
View Answer play_arrow
question_answer 77) Which of the following is more soluble in ammonia?
A)
\[\text{AgCl}\]
done
clear
B)
\[\text{AgBr}\]
done
clear
C)
\[\text{Agl}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 78) An important product in the ozone depletion by chloroflurocarbons is :
A)
\[\text{C}{{\text{l}}_{\text{2}}}\]
done
clear
B)
\[\text{OCl}\]
done
clear
C)
\[\text{O}{{\text{F}}_{2}}\]
done
clear
D)
\[{{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}\]
done
clear
View Answer play_arrow
question_answer 79) Which of the following is heavy water?
A)
\[{{H}_{2}}{{O}^{18}}\]
done
clear
B)
\[{{H}_{2}}{{O}^{16}}\]
done
clear
C)
\[{{H}_{2}}{{O}_{3}}\]
done
clear
D)
\[{{D}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 80) Propylene on hydrolysis -with sulphuric acid forms
A)
\[n-\]propyl alcohol
done
clear
B)
isopropyl alcohol
done
clear
C)
ethyl alcohol
done
clear
D)
butyl alcohol
done
clear
View Answer play_arrow
question_answer 81) The volume of oxygen liberated from 15ml of 20 volume \[{{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}\]is
A)
250 ml
done
clear
B)
300 ml
done
clear
C)
150 ml
done
clear
D)
200 ml
done
clear
View Answer play_arrow
question_answer 82) Which of the following is purest form of iron?
A)
Steel
done
clear
B)
Wrought iron
done
clear
C)
Grey cast iron
done
clear
D)
White cast iron
done
clear
View Answer play_arrow
question_answer 83) Carborundum is
A)
\[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
B)
\[A{{l}_{2}}{{O}_{3}}.2{{H}_{2}}O\]
done
clear
C)
\[AlC{{l}_{3}}\]
done
clear
D)
\[\text{SiC}\]
done
clear
View Answer play_arrow
question_answer 84) The hydrolysis product of \[C{{H}_{3}}COC{{H}_{3}}+C{{H}_{3}}MgBr\]is
A)
\[n-\] butyl alcohol
done
clear
B)
tertiary butyl alcohol
done
clear
C)
secondary butyl alcohol
done
clear
D)
isopropyl alcohol
done
clear
View Answer play_arrow
question_answer 85) Molecular formula of chloropicrin is
A)
\[CHC{{l}_{3}}N{{O}_{2}}\]
done
clear
B)
\[CC{{l}_{3}}N{{O}_{3}}\]
done
clear
C)
\[CC{{l}_{2}}N{{O}_{2}}\]
done
clear
D)
\[CC{{l}_{3}}N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 86) Which of the following does not give a borax bead test?
A)
Ferrous salt
done
clear
B)
Chromium
done
clear
C)
Cobalt
done
clear
D)
Sodium
done
clear
View Answer play_arrow
question_answer 87) The inert gas abundantly found in atmosphere is
A)
Ae
done
clear
B)
Kr
done
clear
C)
He
done
clear
D)
Ar.
done
clear
View Answer play_arrow
question_answer 88) Which of the following electronic configuration is not possible?
A)
\[1{{s}^{2}},2{{s}^{2}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}}\]
done
clear
C)
\[[Ar]\,3{{d}^{10}},4{{s}^{2}}4{{p}^{2}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{2}},3{{s}^{1}}\]
done
clear
View Answer play_arrow
question_answer 89) Chloroform, on warming with Ag power, gives
A)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 90) The shape of a molecule of \[\text{N}{{\text{H}}_{\text{3}}}\text{,}\]in which central atoms contains lone pair of electron, is
A)
tetrahedral
done
clear
B)
planar trigonal
done
clear
C)
square planar
done
clear
D)
pyramidal
done
clear
View Answer play_arrow
question_answer 91) Which is correct about ionization potential?
A)
It is independent of atomic radii
done
clear
B)
It increases with increase in atomic radii
done
clear
C)
It remains constant with increase in atomic radii
done
clear
D)
It decreases with increase in atomic radii
done
clear
View Answer play_arrow
question_answer 92) In the reaction \[HN{{O}_{3}}+{{P}_{4}}{{O}_{10}}\xrightarrow{{}}4HP{{O}_{3}}+x\] the product \[x\] is
A)
\[{{N}_{2}}{{O}_{3}}\]
done
clear
B)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 93) When lead nitrate is heated, it gives
A)
\[\text{N}{{\text{O}}_{\text{2}}}\]
done
clear
B)
\[\text{NO}\]
done
clear
C)
\[{{\text{N}}_{\text{2}}}{{\text{O}}_{\text{5}}}\]
done
clear
D)
\[{{\text{N}}_{\text{2}}}\text{O}\]
done
clear
View Answer play_arrow
question_answer 94) Equivalant weight of a bivalent metal is 37.2. The molecular weight of its chloride is
A)
412.2
done
clear
B)
216
done
clear
C)
145.4
done
clear
D)
108.2
done
clear
View Answer play_arrow
question_answer 95) Vinegar contains how much % of acetic acid?
A)
15-20%
done
clear
B)
90-100%
done
clear
C)
7-8%
done
clear
D)
10-12%
done
clear
View Answer play_arrow
question_answer 96) Assertion: \[\text{HCl}{{\text{O}}_{\text{4}}}\]is a stronger acid than\[\text{HCl}{{\text{O}}_{3}}.\] Reason: Oxidation state of \[\text{Cl}\] in \[\text{HCl}{{\text{O}}_{4}}\]is + 8 and in \[\text{HCl}{{\text{O}}_{\text{3}}}\text{+}\,\text{5}\text{.}\]
A)
If both assertion and reason are true and reason is not the correct explanation of assertion.
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion.
done
clear
C)
If assertion is true but reason is false.
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 97) Assertion: The free gaseous Cr atom has six unpaired electrons. Reason: Half-filled s orbital has greater stability.
A)
If both assertion and reason are true and reason is not the correct explanation of assertion.
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion.
done
clear
C)
If assertion is true but reason is false.
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 98) Assertion: The \[[Ni{{(en)}_{3}}C{{l}_{2}}]C{{l}_{2}}\] (en= ethylene diamine) has lower stability than \[[Ni{{(N{{H}_{3}})}_{6}}C{{l}_{2}}]\]. Reason: In \[[Ni{{(en)}_{3}}C{{l}_{2}}]\] the geometry of Ni is trigonal bipyramidal.
A)
If both assertion and reason are true and reason is not the correct explanation of assertion.
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion.
done
clear
C)
If assertion is true but reason is false.
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 99) Assertion: Sb (III) is not precipitated as sulphide when in its alkaline solution \[{{H}_{2}}S\] is passed. Reason: The concentration of \[{{S}^{2-}}\] ion in alkaline medium is inadequate for precipitation.
A)
If both assertion and reason are true and reason is not the correct explanation of assertion.
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion.
done
clear
C)
If assertion is true but reason is false.
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 100) Assertion: Nuclear binding energy per nucleon is in the order \[-_{4}^{9}Be>_{3}^{7}Li>_{2}^{4}He\]. Reason: Binding energy per nucleon increases linearly with difference in number of neutrons and protons.
A)
If both assertion and reason are true and reason is not the correct explanation of assertion.
done
clear
B)
If both assertion and reason are true but reason is not the correct explanation of assertion.
done
clear
C)
If assertion is true but reason is false.
done
clear
D)
If both assertion and reason are false
done
clear
View Answer play_arrow
question_answer 101) Pond is an example of which ecosystem?
A)
Natural
done
clear
B)
Artificial
done
clear
C)
Forest
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 102) The ecological niche of population is a
A)
geographical area that it covers
done
clear
B)
place where it lives
done
clear
C)
set of conditions and resource it uses
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 103) Mendels dihybrid cross ratio is
A)
1 : 2 : 1
done
clear
B)
9 : 3 : 3 : 1
done
clear
C)
9 : 3 : 4
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 104) Parachute mechanism of seed dispersal occurs in
A)
sunflower
done
clear
B)
Calotropis
done
clear
C)
mango
done
clear
D)
apple
done
clear
View Answer play_arrow
question_answer 105) Hand of man, wing of bat and flipper of seal represents
A)
vestigeal organs
done
clear
B)
analogous organs
done
clear
C)
evolutionary organs
done
clear
D)
homologous organs
done
clear
View Answer play_arrow
question_answer 106) If an organism is crossed with homozygous recessive individual, it is called
A)
back cross
done
clear
B)
test cross
done
clear
C)
simple cross
done
clear
D)
monohybrid cross
done
clear
View Answer play_arrow
question_answer 107) Which is correct for the structure of cell wall of bacteria and fungi?
A)
Both are made up of cellulose
done
clear
B)
Both have mucopeptide
done
clear
C)
Both are made up of N-acetylglucosamine
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 108) Photorespiration takes place only in
A)
lysosomes of plant cell
done
clear
B)
green parts of the plant
done
clear
C)
mitochondria of plant cell
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 109) Nitrogen fixation means:
A)
\[{{N}_{2}}\] changes in \[NO_{3}^{-}\]
done
clear
B)
\[{{N}_{2}}\] changes in \[N{{H}_{3}}\]
done
clear
C)
\[NO_{2}^{-}\] changes into nitrates
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 110) In a pond ecosystem, benthos means
A)
primary consumers in the depth of a pond
done
clear
B)
virus
done
clear
C)
zooplankton on the water surface
done
clear
D)
bacteria
done
clear
View Answer play_arrow
question_answer 111) There are special proteins that help to open up DNA double helix in front of the replication fork. These protein are
A)
DNA ligase
done
clear
B)
DNA gyrase
done
clear
C)
DNA polymerase I
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 112) Peripatus is known as a connecting link, because it has the characters of both
A)
Aves and Fishes
done
clear
B)
Reptiles and Birds
done
clear
C)
Fishes and Amphibians
done
clear
D)
Arthropoda and Annelids
done
clear
View Answer play_arrow
question_answer 113) In which segment the clitellum is present in earthworm?
A)
16 segments
done
clear
B)
17-19 segments
done
clear
C)
14-16 segments
done
clear
D)
5-6 segments
done
clear
View Answer play_arrow
question_answer 114) Which of the following is absent in the mouth part of housefly?
A)
Labrum
done
clear
B)
Epipharynx
done
clear
C)
Mandibles
done
clear
D)
Maxillary palps
done
clear
View Answer play_arrow
question_answer 115) The colour of the body in earthworm is brown due to
A)
porphyrin
done
clear
B)
haemoglobin
done
clear
C)
blood
done
clear
D)
haemocyanin
done
clear
View Answer play_arrow
question_answer 116) The egg found in monotremata is
A)
microlecithal
done
clear
B)
mesolecithal
done
clear
C)
macrolecithal
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 117) Which hormone causes stunted growth in pea ?
A)
Gibberellic acid
done
clear
B)
Auxin
done
clear
C)
Cytokinin
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
question_answer 118) In five kingdom classification of Whittaker, eukaryotes were assigned to
A)
3 of 5 kingdoms
done
clear
B)
2 of 5 kingdoms
done
clear
C)
4 of 5 kingdoms
done
clear
D)
all the 5 kingdoms
done
clear
View Answer play_arrow
question_answer 119) Which is responsible for recycling of material?
A)
Bacteria
done
clear
B)
Algae
done
clear
C)
Fungi
done
clear
D)
Virus
done
clear
View Answer play_arrow
question_answer 120) Which is following group of plant can grow in \[{{N}_{2}}\] deficient soil?
A)
Bryophytes
done
clear
B)
Algae
done
clear
C)
Insectivorous
done
clear
D)
Pteridophytes
done
clear
View Answer play_arrow
question_answer 121) Which region is responsible for origin of rhizoids in Funaria?
A)
Lateral region
done
clear
B)
Dorsal region
done
clear
C)
Ventral region
done
clear
D)
Basal region
done
clear
View Answer play_arrow
question_answer 122) Powdery mildews of crops are caused by
A)
basidiomycetes
done
clear
B)
phycomycetes
done
clear
C)
ascomycetes
done
clear
D)
eumycocetes
done
clear
View Answer play_arrow
question_answer 123) The codons, causing chain termination are
A)
AGT, TAG, UGA
done
clear
B)
UAG, UGA, UAA
done
clear
C)
TAG, TAA, TGA
done
clear
D)
GAT, AAT, AGT
done
clear
View Answer play_arrow
question_answer 124) Biological concept of species is mainly based on
A)
methods of reproduction only
done
clear
B)
reproductive isolation
done
clear
C)
morphological features only
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 125) The organism which carries a disease from one organism to another is called?
A)
Parasite
done
clear
B)
Host
done
clear
C)
Vector
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 126) The morphine obtained from opium is a
A)
tannin
done
clear
B)
alkaloids
done
clear
C)
gums
done
clear
D)
latex
done
clear
View Answer play_arrow
question_answer 127) Hybrid vigour is mostly due to
A)
superiority of all the genes
done
clear
B)
homozygosity of pure characters
done
clear
C)
heterozygosity
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 128) Flowers and lateral branches arise from the
A)
lateral buds
done
clear
B)
lentices
done
clear
C)
stomata
done
clear
D)
cuticle
done
clear
View Answer play_arrow
question_answer 129) Chiasma shows the sites of
A)
spindle formation
done
clear
B)
synapsis
done
clear
C)
crossing over
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 130) The process of cytokinesis refers to the division of
A)
nucleus
done
clear
B)
chromosomes
done
clear
C)
cytoplasm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 131) Which of the following organelle does not contain RNA?
A)
Chromosome
done
clear
B)
Plasmalemma
done
clear
C)
Nucleolus
done
clear
D)
Ribosome
done
clear
View Answer play_arrow
question_answer 132) Arachidonic acid is
A)
non-essential fatty acid
done
clear
B)
essential fatty acid
done
clear
C)
olyunsaturated fatty acid
done
clear
D)
both b and c
done
clear
View Answer play_arrow
question_answer 133) Which of the following is polymerized to form proteins ?
A)
Protein
done
clear
B)
Carbohydrates
done
clear
C)
Amino acid
done
clear
D)
Muramic acid
done
clear
View Answer play_arrow
question_answer 134) All enzymes are basically
A)
carbohydrates
done
clear
B)
steroids
done
clear
C)
proteins
done
clear
D)
lipo-proteins
done
clear
View Answer play_arrow
question_answer 135) The nucleus is separated from surrounding cytoplasm by a nuclear membrane, which is
A)
single layered without pores
done
clear
B)
double layered with pores
done
clear
C)
single layered with pores
done
clear
D)
double layered without pores
done
clear
View Answer play_arrow
question_answer 136) Which organ has a single unit membrane?
A)
Lysosome
done
clear
B)
Mitochondria
done
clear
C)
Golgi body
done
clear
D)
Nucleolus
done
clear
View Answer play_arrow
question_answer 137) Which of the following animal has a notochord throughout its life?
A)
Fish
done
clear
B)
Bird
done
clear
C)
Snake
done
clear
D)
Amphioxus
done
clear
View Answer play_arrow
question_answer 138) Piercing and sucking mouth parts are found in
A)
mosquitoes
done
clear
B)
housefly
done
clear
C)
butterfly
done
clear
D)
ticks
done
clear
View Answer play_arrow
question_answer 139) When the number of organisms at successive levels are plotted they assume the shape of a pyramid. This is called the pyramid of
A)
Biomass
done
clear
B)
Number
done
clear
C)
Energy
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 140) Effect of pollution is on
A)
crossing over
done
clear
B)
ecological balance
done
clear
C)
linkage
done
clear
D)
mutation
done
clear
View Answer play_arrow
question_answer 141) Deforestation causes
A)
thermal pollution
done
clear
B)
noise pollution
done
clear
C)
soil erosion
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 142) A thin film of water is held by the soil particles under the influence of internal attractive force. It is called
A)
hygroscopic water
done
clear
B)
gravitational water
done
clear
C)
combined water
done
clear
D)
capillary water
done
clear
View Answer play_arrow
question_answer 143) Pneumatophores are usually present in
A)
Murrya
done
clear
B)
Eichhornia
done
clear
C)
Avicinnea
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 144) Balloon like outgrowth of parenchyma into the lumen of vessels, is known as
A)
tyloses
done
clear
B)
tunica
done
clear
C)
corpous
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 145) In some plants the style is shorter. But in some others, it is longer than the stamens This condition is called
A)
homogamy
done
clear
B)
homostyly
done
clear
C)
heterostyly
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 146) Interferon is
A)
bacteria
done
clear
B)
anti-viral
done
clear
C)
anti-algal
done
clear
D)
anti-bacterial
done
clear
View Answer play_arrow
question_answer 147) Bacterium have flagella with all over body is known as
A)
peritrichous
done
clear
B)
amphitrichous
done
clear
C)
monotrichous
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 148) Helotism is a term used for the symbiosis of
A)
algae and bacteria
done
clear
B)
algae and bryophytes
done
clear
C)
algae and pteridophytes
done
clear
D)
algae and fungi
done
clear
View Answer play_arrow
question_answer 149) In Dryopteris the opening mechanism of sporangium is effectively operated by
A)
stalk
done
clear
B)
stomium
done
clear
C)
annulus
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 150) In DNA, when AGCT occur, their association is as per which of the following pair ?
A)
AG-CT
done
clear
B)
AC-GT
done
clear
C)
AT-GC
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 151) Anus is absent in
A)
Unio
done
clear
B)
Pheretima
done
clear
C)
Periplaneta
done
clear
D)
Fasciolci
done
clear
View Answer play_arrow
question_answer 152) The vegetation of Rajasthan is
A)
xerophytic
done
clear
B)
deciduous
done
clear
C)
hydrophytic
done
clear
D)
arctic
done
clear
View Answer play_arrow
question_answer 153) In food chain, lion is a
A)
tertiary consumer
done
clear
B)
secondary consumer
done
clear
C)
primary consumer
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 154) Agricultural chemicals include
A)
promotors
done
clear
B)
pesticides
done
clear
C)
fertilizers
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 155) Biological equilibrium is found among the
A)
producers, consumers and decomposers
done
clear
B)
producers and consumers
done
clear
C)
producers and decomposers
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 156) Light energy in photosynthesis is utilized in
A)
\[{{H}_{2}}O\] converted into \[{{H}_{2}}\]
done
clear
B)
ADP converted into ATP
done
clear
C)
ATP converted into ADP
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 157) Nitrates are converted into \[{{N}_{2}}\] by
A)
\[{{N}_{2}}\] fixing bacteria
done
clear
B)
sulphur fixing bacteria
done
clear
C)
denitrifying bacteria
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 158) Genetic dwarfness can be overcome by treating with
A)
cytokinin
done
clear
B)
auxins
done
clear
C)
gibberellins
done
clear
D)
ethylene
done
clear
View Answer play_arrow
question_answer 159) The nephridia, in earthworm, are analogous to
A)
gills of prawn
done
clear
B)
trachea of insects
done
clear
C)
nematoblasts of Hydra
done
clear
D)
flame cells of Planaria
done
clear
View Answer play_arrow
question_answer 160) Which special feature present in slime moulds ?
A)
Capsule
done
clear
B)
Capillitium
done
clear
C)
Claters
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 161) Aggregate fruit develops from
A)
multicarpellary, apocarpous ovary
done
clear
B)
multicarpellary ovary
done
clear
C)
multicarpellary, syncarpous ovary
done
clear
D)
monocarpellary ovary
done
clear
View Answer play_arrow
question_answer 162) Respiratory quotient (R.Q.) during early stages of germination of castor seed is
A)
one
done
clear
B)
zero
done
clear
C)
more than one
done
clear
D)
less than one
done
clear
View Answer play_arrow
question_answer 163) In DNA helix, cytosine is paired with guanine by
A)
three hydrogen bonds
done
clear
B)
two hydrogen bonds
done
clear
C)
single hydrogen bond
done
clear
D)
covalent bond
done
clear
View Answer play_arrow
question_answer 164) High content of lysine is present in
A)
wheat
done
clear
B)
apple
done
clear
C)
maize
done
clear
D)
banana
done
clear
View Answer play_arrow
question_answer 165) In a pond ecosystem the pyramid of biomass is always
A)
irregular
done
clear
B)
inverted
done
clear
C)
upright
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 166) Members of phylum Porifera are
A)
exclusively marine animals
done
clear
B)
exclusively fresh water animals
done
clear
C)
mostly fresh water animals but few are marine animals
done
clear
D)
mostly marine animals but few are fresh water animals
done
clear
View Answer play_arrow
question_answer 167) Hydra recognises its prey by
A)
nematocyst
done
clear
B)
some special organs
done
clear
C)
chemical stimulus of prey
done
clear
D)
mechanical stimulus of prey
done
clear
View Answer play_arrow
question_answer 168) Shell of mollusc is produced by its
A)
radula
done
clear
B)
thorax
done
clear
C)
mantle
done
clear
D)
abdomen
done
clear
View Answer play_arrow
question_answer 169) Asymmetry in gastropoda is due to
A)
torsion
done
clear
B)
coiling
done
clear
C)
twisting
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 170) The joint of femur, with pelvic girdle, is
A)
hinge joint
done
clear
B)
pivot joint
done
clear
C)
non-movable joint
done
clear
D)
ball and socket joint
done
clear
View Answer play_arrow
question_answer 171) Which of the following is the part of mid brain of rabbit?
A)
Diencephalon
done
clear
B)
Cerebrum
done
clear
C)
Corpora quadrigemina
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 172) Which of the following is a motor nerve?
A)
Auditory
done
clear
B)
Abducens
done
clear
C)
Optic nerve
done
clear
D)
Trigeminal nerve
done
clear
View Answer play_arrow
question_answer 173) Which of the following is called emergency gland of the body?
A)
Testis
done
clear
B)
Thymus
done
clear
C)
Adrenal
done
clear
D)
Pituitary
done
clear
View Answer play_arrow
question_answer 174) Which of the following is both exocrine and endocrine gland
A)
Liver
done
clear
B)
Pancreas
done
clear
C)
Thyroid
done
clear
D)
Adrenal
done
clear
View Answer play_arrow
question_answer 175) Corpus luteum is developed from
A)
oocyte
done
clear
B)
nephrostome
done
clear
C)
graffian follicle
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 176) In most mammals, the testes are located in scrotal sac for
A)
spermatogenesis
done
clear
B)
sex differentiation
done
clear
C)
more space to visceral organs
done
clear
D)
independent functioning of kidney
done
clear
View Answer play_arrow
question_answer 177) Third cleavage of frogs development is
A)
equatorial
done
clear
B)
vertical
done
clear
C)
latitudinal
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 178) Which of the following is a transparent tissue?
A)
Tendon
done
clear
B)
Hyaline cartilage
done
clear
C)
Fibrous cartilage
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 179) Which of the following are not true cells in the blood
A)
Platelets
done
clear
B)
Monocytes
done
clear
C)
Neutrophils
done
clear
D)
Basophils
done
clear
View Answer play_arrow
question_answer 180) Vitamin D is synthesized in skin, by the action of sunlight on
A)
cholesterol
done
clear
B)
seven-hydroxy-cholesterol
done
clear
C)
cephanocholesterol
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 181) In which animal, diaphragm has no role in respiration?
A)
Frog
done
clear
B)
Rat
done
clear
C)
Camel
done
clear
D)
Rabbit
done
clear
View Answer play_arrow
question_answer 182) The area of inner surface of bronchiole is
A)
\[1\,{{m}^{2}}\]
done
clear
B)
\[10\,\,{{m}^{2}}\]
done
clear
C)
\[100\,\,{{m}^{2}}\]
done
clear
D)
\[1000\,\,{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 183) During the process of blood coagulation, vitamin K helps in
A)
the formation of prothrombin
done
clear
B)
the formation of thromboplastin
done
clear
C)
the conversion of fibrinogen into fibrin
done
clear
D)
the conversion of prothrombin into thrombin
done
clear
View Answer play_arrow
question_answer 184) The heart muscles are
A)
striated and involuntary
done
clear
B)
striated and voluntary
done
clear
C)
smooth and involuntary
done
clear
D)
non-striated and involuntary
done
clear
View Answer play_arrow
question_answer 185) Which of the following amino acids is present in ornithine cycle?
A)
Valine and systine
done
clear
B)
Arginin and citrulline
done
clear
C)
(c) Glycine and methionine
done
clear
D)
(b) None of these
done
clear
View Answer play_arrow
question_answer 186) The joint of radio-ulna with the upper arm is
A)
hinge joint
done
clear
B)
pivot joint
done
clear
C)
socket joint
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 187) Haversian canals are present in
A)
bone marrow
done
clear
B)
hyaline cartilage
done
clear
C)
bone matrix
done
clear
D)
calcified cartilage
done
clear
View Answer play_arrow
question_answer 188) Law of segregation of gametes was proposed by
A)
Mendel
done
clear
B)
de Vries
done
clear
C)
Robert Hook
done
clear
D)
Huxley
done
clear
View Answer play_arrow
question_answer 189) Which of the following evidences does not favour the Lamarckian concept of inheritance of acquired characters?
A)
Absence of limbs in snakes
done
clear
B)
Presence of webbed toes in aquatic birds
done
clear
C)
Melanization in peppered moth
done
clear
D)
Lack of pigment in cave-dwelling animals
done
clear
View Answer play_arrow
question_answer 190) The most important example of point mutation is found in a disease called
A)
thalassemia
done
clear
B)
night blindness
done
clear
C)
Downs syndrome
done
clear
D)
sickle cell anaemia
done
clear
View Answer play_arrow
question_answer 191) Assertion: Agricultural output increased several times after introduction of DDT. Reason: DDT was the first insecticide used on a wide scale.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 192) Assertion: Diabetes insipidus is marked by excessive urination and too much thirst of water. Reason: Anti-diuretic hormone (ADH) is secreted by the posterior lobe of pituitary gland.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 193) Assertion: Coacervates are believed to be the precursors of life. Reason: Coacervates were self-duplicating aggregates of proteins surrounded by lipid molecules.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 194) Assertion: Tapeworm roundworm and pinworm are endoparasites of human intestine. Reason: Improperly cooked food is the source of intestinal infections.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 195) Assertion: Fish meal is a rich source of protein for cattle and poultry. Reason: Fish meal is produced from non-edible parts of fishes like fins, tail etc.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 196) Assertion: Animals adopt different strategies to survive in hostile environment. Reason: Praying mantis is green in colour which merges with plant foliage.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 197) Assertion: Among the primates, chimpanzee is the closest relative of the present day humans. Reason: The banding pattern in the autosome numbers 3 and 6 of man and chimpanzee is remarkably similar.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 198) Assertion: Dope test is used to estimate the level of blood alcohol by analyzing the breath of persons drinking alcohol. Reason: A drunken person usually feels tense and less talkative.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 199) Assertion: The honey bee queen copulates only ones in her life time. Reason: The honey bee queen can lay fertilized as well as unfertilized eggs.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow
question_answer 200) Assertion: From evolutionary point of view, human gestation period is believed to be shortening. Reason: One major evolutionary trend in humans has been the larger head undergoing relatively faster growth rate in the foetal stage.
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
E)
E
done
clear
View Answer play_arrow