A) straight line
B) increasing curve
C) straight line with slope
D) None of the above
Correct Answer: D
Solution :
According to Boyle's law, at constant temperature the volume of a given mass of a gas is inversely proportional to its pressure \[p\propto \frac{1}{V}\] \[pV=\]constant At constant temperature, on plotting a graph between p and V, a hyperbola curve is obtained.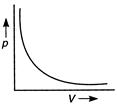
You need to login to perform this action.
You will be redirected in
3 sec
