question_answer 1) The velocity of bullet is reduced from 200 m/s to 100 m/s while travelling through a wooden block of thickness of 10 cm. The retardation assuming to be uniform, will be:
A)
\[15\times {{10}^{4}}m/{{s}^{2}}\]
done
clear
B)
\[13.5\times {{10}^{4}}m/{{s}^{2}}\]
done
clear
C)
\[12\times {{10}^{4}}m/{{s}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 2) A stone tied to one end of spring 80 cm long is whirled in a horizontal circle with a constant speed. If stone makes 25 revolutions in 14 sec, the magnitude of acceleration of stone is:
A)
\[850\text{ }cm/{{s}^{2}}\]
done
clear
B)
\[996\text{ }cm/se{{c}^{2}}\]
done
clear
C)
\[720\text{ }cm/{{s}^{2}}\]
done
clear
D)
\[650\text{ }cm/se{{c}^{2}}\]
done
clear
View Answer play_arrow
question_answer 3) A concave mirror of focal length 15 cm forms an image having twice the linear dimension of the object. The position of the object when the image is virtual will be:
A)
45 cm
done
clear
B)
30 cm
done
clear
C)
7.5 cm
done
clear
D)
22.5 cm
done
clear
View Answer play_arrow
question_answer 4) The thermodynamic co-ordinates of a jar filled with gas A are P, V and T and another jar B filled with another gas are 2P, V/4 and 2T, where the symbols have their usual meaning.. The ratio of the number of molecules of jar A to those of jar B is:
A)
\[4:1\]
done
clear
B)
\[2:1\]
done
clear
C)
\[1:2\]
done
clear
D)
\[1:1\]
done
clear
View Answer play_arrow
question_answer 5) If the pressure of a gas contained in a vessel is increased by 0.4% when heated through\[1{}^\circ C,\]the initial temperature had been:
A)
2500 K
done
clear
B)
250 K
done
clear
C)
\[250{}^\circ C\]
done
clear
D)
\[25{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 6) A fire screen produces sensation of cooling as:
A)
it allows both infrared and visible light but cuts off ultraviolet
done
clear
B)
it allows infrared and cuts off shorter wavelengths
done
clear
C)
it cuts off both visible light and infrared
done
clear
D)
it allows only visible light and cuts off infrared
done
clear
View Answer play_arrow
question_answer 7) For a given angle of projection, if the time of flight of a projectile is doubled, the horizontal range will increase to:
A)
four times
done
clear
B)
thrice
done
clear
C)
once
done
clear
D)
twice
done
clear
View Answer play_arrow
question_answer 8) When the kinetic energy of a body executing S.H.M. is 1/3 of the potential energy. The displacement of the body is\[x\]percent of the amplitude, where\[x\] is:
A)
33
done
clear
B)
87
done
clear
C)
67
done
clear
D)
50
done
clear
View Answer play_arrow
question_answer 9) In the case of horse pulling a cart, the force that causes the horse to move forward is the force that:
A)
the horse exerts on the ground
done
clear
B)
the horse exerts on the cart
done
clear
C)
the ground exerts on the horse
done
clear
D)
the cart exerts on the horse
done
clear
View Answer play_arrow
question_answer 10) If retardation produced by air resistance of projectile is one-tenth of acceleration due to gravity, the time to reach maximum height:
A)
decreases by 11 percent
done
clear
B)
increases by 11 percent
done
clear
C)
decreases by 90 percent
done
clear
D)
increases by 90 percent
done
clear
View Answer play_arrow
question_answer 11) \[y\] component of velocity is 20 and\[x\]component of velocity is 10. The direction of motion of the body with the horizontal at this instant is:
A)
\[{{\tan }^{-1}}(2)\]
done
clear
B)
\[{{\tan }^{-1}}(1/2)\]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[0{}^\circ \]
done
clear
View Answer play_arrow
question_answer 12) The dimension of light year is:
A)
\[[{{L}^{-1}}]\]
done
clear
B)
\[[{{T}^{-1}}]\]
done
clear
C)
\[[L]\]
done
clear
D)
\[[T]\]
done
clear
View Answer play_arrow
question_answer 13) In a good conductor of electricity, the type of bonding that exists is:
A)
ionic
done
clear
B)
van der waal
done
clear
C)
covalent
done
clear
D)
metallic
done
clear
View Answer play_arrow
question_answer 14) Two identical straight wires are stretched so as to produce 6 beats per second when vibrating simultaneously. On changing the tension slightly in one of them, the beat frequency still remains unchanged. Denoting by\[{{T}_{1}}\]and\[{{T}_{2}},\]the higher and the lower initial tensions in the strings, it could be said that while making the above changes in tension:
A)
\[{{T}_{1}}\]was decreased
done
clear
B)
\[{{T}_{1}}\] was increased
done
clear
C)
\[{{T}_{2}}\] was increased
done
clear
D)
\[{{T}_{2}}\] was decreased
done
clear
View Answer play_arrow
question_answer 15) When beats are produced by two progressive waves of the same amplitude and of nearly the same frequency, the ratio of maximum loudness to the loudness of one of the waves will be\[n\]. Where\[n\]is:
A)
3
done
clear
B)
1
done
clear
C)
4
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 16) A body is suspended from a smooth horizontal nail by a string of length 0.25 metre. What minimum horizontal velocity should be given to it in the lowest position so that it may move in a complete vertical circle with the nail at the centre?
A)
\[\sqrt{12.25}\,m{{s}^{-1}}\]
done
clear
B)
\[4.9\,m{{s}^{-1}}\]
done
clear
C)
\[7\sqrt{2}\,m{{s}^{-1}}\]
done
clear
D)
\[\sqrt{9.8}\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 17) Two rain drops falling through air have radii in the ratio\[1:2\]. They will have terminal velocity in the ratio:
A)
\[4:1\]
done
clear
B)
\[1:4\]
done
clear
C)
\[2:1\]
done
clear
D)
\[1:2\]
done
clear
View Answer play_arrow
question_answer 18) A body is acted on by force towards a point. The magnitude of the force is inversely proportional to the square of the distance. The path of body will be:
A)
ellipse
done
clear
B)
hyperbola
done
clear
C)
circle
done
clear
D)
parabola
done
clear
View Answer play_arrow
question_answer 19) An electric immersion heater of 1.08 kW is immersed in water. After the water has reached a temperature of\[100{}^\circ C,\]how much time will be required to produce 100 g of steam?
A)
210 s
done
clear
B)
105 s
done
clear
C)
420 s
done
clear
D)
50 s
done
clear
View Answer play_arrow
question_answer 20) The speed of sound through oxygen at TK is \[\upsilon \text{ }m{{s}^{-1}}\]. As. the temperature becomes 2T and oxygen gas dissociates into atomic oxygen, the speed of sound:
A)
remains the same
done
clear
B)
becomes \[2\upsilon \]
done
clear
C)
becomes \[\sqrt{2\upsilon }\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 21) Illumination of the sun at noon is maximum because:
A)
scattering is reduced at noon
done
clear
B)
refraction of light is minimum at noon
done
clear
C)
rays are incident almost normally
done
clear
D)
the sun is nearer to earth at noon
done
clear
View Answer play_arrow
question_answer 22) Two spheres of radii in the ratio\[1:2\]and densities in the ratio\[2:1\]and of same specific heat, are heated to same temperature and left in the same surrounding. Their rate of cooling will be in the ratio:
A)
\[2:1\]
done
clear
B)
\[1:1\]
done
clear
C)
\[1:2\]
done
clear
D)
\[1:4\]
done
clear
View Answer play_arrow
question_answer 23) The formation of ice is started in a lake with water at \[0{}^\circ C\]. When the atmospheric temperature is\[-10{}^\circ C\]. If time taken for 1 cm of ice to be formed is 7 hours, the time taken for the thickness of ice to increase from 1 cm to 2 cm is:
A)
less than 7 hours
done
clear
B)
7 hours
done
clear
C)
more than 14 hours
done
clear
D)
more than 7 hours but less than 14 hours
done
clear
View Answer play_arrow
question_answer 24) If an ideal flask containing hot coffee is shaken, the temperature of the coffee will:
A)
decrease
done
clear
B)
increase
done
clear
C)
remain same
done
clear
D)
decrease if temperature is below\[4{}^\circ C\]and increase if temperature is equal to or more than\[4{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 25) Water is used to cool the radiators of engines in cars because:
A)
of its low boiling point
done
clear
B)
of its high specific heat
done
clear
C)
of its low density
done
clear
D)
of its easy availability
done
clear
View Answer play_arrow
question_answer 26) Suppose radius of the moons orbit around the earth is doubled. Its period around the earth will become:
A)
1/2 times
done
clear
B)
\[\sqrt{2}\] times
done
clear
C)
\[{{2}^{2/3}}\]times
done
clear
D)
\[{{2}^{3/2}}\] times
done
clear
View Answer play_arrow
question_answer 27) A sphere is accelerated upwards by a cord whose braking strength is four times its weight. The maximum acceleration with which the sphere can move up without breaking the cord is:
A)
\[g\]
done
clear
B)
\[3g\]
done
clear
C)
\[2g\]
done
clear
D)
\[4g\]
done
clear
View Answer play_arrow
question_answer 28) The potential due to dipole at a point is proportional to:
A)
\[{{r}^{-2}}\]
done
clear
B)
\[{{r}^{3}}\]
done
clear
C)
\[{{r}^{2}}\]
done
clear
D)
\[r\]
done
clear
View Answer play_arrow
question_answer 29) The capacitance of a spherical conductor with radius 1 m is:
A)
\[9\times {{10}^{-9}}F\]
done
clear
B)
\[1\,\mu F\]
done
clear
C)
\[2.5\times {{10}^{-10}}F\]
done
clear
D)
\[1\times {{10}^{-6}}F\]
done
clear
View Answer play_arrow
question_answer 30) A body moves for a total of nine second starting from rest with uniform acceleration and then -with uniform retardation, which is twice the value of acceleration and then stops. The duration of uniform acceleration is:
A)
3 s
done
clear
B)
4.5 s
done
clear
C)
5 s
done
clear
D)
6 s
done
clear
View Answer play_arrow
question_answer 31) For achromatic combination of lenses, if we use two lenses of focal length and\[f\] respectively, dispersive powers\[\omega \]and\[\omega ,\]then:
A)
\[\omega ={{\omega }_{0}},\omega =2{{\omega }_{0}},f=2f\]
done
clear
B)
\[\omega ={{\omega }_{0}},\omega =2{{\omega }_{0}},f=-2f\]
done
clear
C)
\[\omega ={{\omega }_{0}},\omega =2{{\omega }_{0}},f=f/2\]
done
clear
D)
\[\omega ={{\omega }_{0}},\omega =2{{\omega }_{0}},f=-f/2\]
done
clear
View Answer play_arrow
question_answer 32) A convex lens makes a real image of 4 cm long on a screen. When the lens is shifted to a new position without disturbing the object or the screen, we get a real image on the screen, which is 16 cm long. The length of the object is:
A)
1/4 cm
done
clear
B)
8 cm
done
clear
C)
20 cm
done
clear
D)
2 cm
done
clear
View Answer play_arrow
question_answer 33) In a sinusoidal wave, the time required for a particular point to move from maximum displacement is 0.17 sec. Then the frequency of the wave is:
A)
0.38 Hz
done
clear
B)
0.78 Hz
done
clear
C)
2.9 Hz
done
clear
D)
1.56 Hz
done
clear
View Answer play_arrow
question_answer 34) Bernoullis principle is based on the law of conservation of:
A)
angular momentum
done
clear
B)
linear momentum
done
clear
C)
mass
done
clear
D)
energy
done
clear
View Answer play_arrow
question_answer 35) The colour of a star indicates its:
A)
temperature
done
clear
B)
distance
done
clear
C)
velocity
done
clear
D)
size
done
clear
View Answer play_arrow
question_answer 36) Efficiency of a HW rectifier is nearly:
A)
80%
done
clear
B)
60%
done
clear
C)
40%
done
clear
D)
20%
done
clear
View Answer play_arrow
question_answer 37) The current in a diode is related to the voltage V by the equation:
A)
\[I\propto {{V}^{1/2}}\]
done
clear
B)
\[I\propto {{V}^{3/2}}\]
done
clear
C)
\[I\propto {{V}^{2}}\]
done
clear
D)
\[I\propto V\]
done
clear
View Answer play_arrow
question_answer 38) An X-ray photon has a wavelength\[0.01\overset{o}{\mathop{\text{A}}}\,\]. Its momentum (in\[kg\text{ }m{{s}^{-1}}\]) is:
A)
\[6.66\times {{10}^{-22}}\]
done
clear
B)
\[3.3\times {{10}^{-32}}\]
done
clear
C)
\[6.6\times {{10}^{-22}}\]
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 39) The equation \[{{4}_{1}}{{H}^{1}}{{\xrightarrow[{}]{{}}}_{2}}H{{e}^{4++}}+2{{e}^{+}}+26\,MeV\] represents:
A)
\[\gamma -\]decay
done
clear
B)
\[\beta -\]decay
done
clear
C)
fission
done
clear
D)
fusion
done
clear
View Answer play_arrow
question_answer 40)
An infinitely long straight conductor is bent into the shape as shown in figure. It carries a current\[i\]A and radius of the circular loop is r m. The magnetic field at the centre of the loop is:
A)
\[{{\mu }_{0}}i(\pi -1)/2\pi r\]
done
clear
B)
\[{{\mu }_{0}}i(\pi +1)/2\pi r\]
done
clear
C)
\[{{\mu }_{0}}/2\pi r\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 41) The dimensional formula for thermal resistance is:
A)
\[{{M}^{-1}}{{L}^{-2}}{{T}^{3}}\theta \]
done
clear
B)
\[{{M}^{-1}}{{L}^{2}}{{T}^{-3}}\theta \]
done
clear
C)
\[M{{L}^{2}}{{T}^{-2}}\theta \]
done
clear
D)
\[M{{L}^{2}}{{T}^{-2}}{{\theta }^{-1}}\]
done
clear
View Answer play_arrow
question_answer 42) A truck travelling due north at\[20\text{ }m{{s}^{-1}}\]turns west and travels with same speed. What is the change in velocity?
A)
\[20\sqrt{2}\,m{{s}^{-1}}\]south-west
done
clear
B)
\[40\text{ }m{{s}^{-1}}\]south-west
done
clear
C)
\[\text{20}\sqrt{2}\text{ }m{{s}^{-1}}\]north-west
done
clear
D)
\[\text{40 }m{{s}^{-1}}\]north-west
done
clear
View Answer play_arrow
question_answer 43) An iron bar of length\[I\]and having a cross-section A is heated from\[0{}^\circ \]to\[100{}^\circ \]. If this bar is so held that it is not permitted to expand or bend, the force that is developed, is:
A)
inversely proportional to the cross-sectional area of the bar
done
clear
B)
independent of the length of the bar
done
clear
C)
inversely proportional to the length of the bar
done
clear
D)
directly proportional to the length of the bar
done
clear
View Answer play_arrow
question_answer 44) A body of uniform cross-sectional area floats in a liquid of density thrice its value. The fraction of exposed height will be:
A)
2/3
done
clear
B)
5/6
done
clear
C)
1/6
done
clear
D)
1/3
done
clear
View Answer play_arrow
question_answer 45) The restoring force of S.H.M. is maximum when particle:
A)
displacement is maximum
done
clear
B)
half way between them
done
clear
C)
when crossing mean position
done
clear
D)
at rest
done
clear
View Answer play_arrow
question_answer 46) Two forces of 12N and 8N act upon a body. The resultant force on the body has a maximum value of:
A)
4 N
done
clear
B)
0 N
done
clear
C)
20 N
done
clear
D)
8 N
done
clear
View Answer play_arrow
question_answer 47) Which of following pairs does not have similar dimensions?
A)
Plancks constant and angular momentum
done
clear
B)
Tension and surface tension
done
clear
C)
Angle and strain
done
clear
D)
Stress and pressure
done
clear
View Answer play_arrow
question_answer 48) Four lenses are made from the same type of glass. The radius of curvature of each face is given below. Which will have the greatest positive power?
A)
10 cm convex and 15 cancave
done
clear
B)
5 cm convex and 10 cm concave
done
clear
C)
15 cm convex and plane
done
clear
D)
20 cm convex and 30 cm concave
done
clear
View Answer play_arrow
question_answer 49) The slope of a graph drawn between threshold frequency and stopping potential is:
A)
\[e\]
done
clear
B)
\[h\]
done
clear
C)
\[\frac{h}{e}\]
done
clear
D)
\[he\]
done
clear
View Answer play_arrow
question_answer 50)
The truth table given below belongs for which gate?
A
B
C
0
0
1
0
1
1
1
0
1
1
1
0
A)
OR
done
clear
B)
XOR
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
question_answer 51) If the rotational velocity of a dynamo armature is doubled, then induced emf will be:
A)
unchanged
done
clear
B)
four times
done
clear
C)
two times
done
clear
D)
half
done
clear
View Answer play_arrow
question_answer 52) There is 10 units of charge at the centre of circle of radius 1 m. The work done in moving 1 unit of charge once around the circle is:
A)
150 unit
done
clear
B)
100 unit
done
clear
C)
10 unit
done
clear
D)
zero
done
clear
View Answer play_arrow
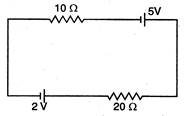
question_answer 53)
The current in the given circuit is:
A)
0.4 A
done
clear
B)
0.3 A
done
clear
C)
0.2 A
done
clear
D)
0.1 A
done
clear
View Answer play_arrow
question_answer 54) If the radius of soap bubble is four times that of another, then the ratio of their pressure will be:
A)
\[1:4\]
done
clear
B)
\[4:1\]
done
clear
C)
\[16:1\]
done
clear
D)
\[1:16\]
done
clear
View Answer play_arrow
question_answer 55) Coefficient of superficial expansion of solid is\[2\times {{10}^{-5}}/{}^\circ C\]. Its coefficient of linear expansion is:
A)
\[4\times {{10}^{-5}}/{}^\circ C\]
done
clear
B)
\[3\times {{10}^{-5}}/{}^\circ C\]
done
clear
C)
\[2\times {{10}^{-5}}/{}^\circ C\]
done
clear
D)
\[1\times {{10}^{-5}}/{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 56) Energy radiated from a body is directly proportional to:
A)
\[{{T}^{4}}\]
done
clear
B)
\[{{T}^{3}}\]
done
clear
C)
\[{{T}^{2}}\]
done
clear
D)
T
done
clear
View Answer play_arrow
question_answer 57) From the top of a tower, a particle is thrown vertically downwards with a velocity of 10 m/s. The ratio of the distances covered by it in the 3rd and 2nd seconds of the motion is (Here \[g=10\text{ }m/{{s}^{2}}\]):
A)
\[6:3\]
done
clear
B)
\[3:6\]
done
clear
C)
\[7:5\]
done
clear
D)
\[5:7\]
done
clear
View Answer play_arrow
question_answer 58) A body whose momentum is constant, must have constant:
A)
acceleration
done
clear
B)
velocity
done
clear
C)
force
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 59) Which of the following is used as a moderator in nuclear reactor?
A)
Heavy water
done
clear
B)
Uranium
done
clear
C)
Plutonium
done
clear
D)
Cadmium
done
clear
View Answer play_arrow
question_answer 60) In Bohrs theory, relation between main quantum number\[n\]and radius of orbit r is:
A)
\[r\propto {{n}^{2}}\]
done
clear
B)
\[r\propto \frac{1}{{{n}^{2}}}\]
done
clear
C)
\[r\propto \frac{1}{n}\]
done
clear
D)
\[r\propto n\]
done
clear
View Answer play_arrow
question_answer 61) Action of nitrous acid on ethyl amine gives:
A)
nitro methane
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow
question_answer 62) Which one of- the following is least likely to act as allewis base?
A)
\[{{I}^{+}}\]
done
clear
B)
\[I\]
done
clear
C)
\[SC{{l}_{2}}\]
done
clear
D)
\[PC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 63) The molecule which has T-shaped structure is:
A)
\[PC{{l}_{3}}\]
done
clear
B)
\[Cl{{F}_{3}}\]
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
\[BC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 64) The conjugate acid of\[N{{H}_{2}}\]is:
A)
\[{{N}_{2}}{{H}_{2}}\]
done
clear
B)
\[NH_{4}^{+}\]
done
clear
C)
\[N{{H}_{2}}OH\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 65) \[E{}^\circ\] for \[F{{e}^{2+}}+2{{e}^{-}}\xrightarrow[{}]{{}}Fe\] is \[-0.44\] volt and \[E{}^\circ \] for \[Z{{n}^{2+}}+2{{e}^{-}}\xrightarrow[{}]{{}}Zn\] is \[-0.76\]volt thus:
A)
\[Zn\] is more electropositive than\[Fe\]
done
clear
B)
\[Zn\] is more electronegative than \[Fe\]
done
clear
C)
\[Fe\]is more electropositive than \[Zn\]
done
clear
D)
none.
done
clear
View Answer play_arrow
question_answer 66) Which isotope is likely to be most stable?
A)
\[_{30}Z{{n}^{66}}\]
done
clear
B)
\[_{30}Z{{n}^{64}}\]
done
clear
C)
\[_{30}Z{{n}^{71}}\]
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 67) The molecular formula of Phosphorus is:
A)
\[{{P}_{5}}\]
done
clear
B)
\[{{P}_{2}}\]
done
clear
C)
\[{{P}_{4}}\]
done
clear
D)
\[{{P}_{1}}\]
done
clear
View Answer play_arrow
question_answer 68) Polyethylene is a resin obtained by polymerization of:
A)
styrene
done
clear
B)
isoprene
done
clear
C)
ethylene
done
clear
D)
butadiene
done
clear
View Answer play_arrow
question_answer 69) The term\[\frac{-dc}{dt}\]in a rate equation refers to:
A)
the decrease in concentration of the reactant with time
done
clear
B)
the concentration of a reactant
done
clear
C)
the change in concentration of the reactant
done
clear
D)
the velocity constant of the reaction
done
clear
View Answer play_arrow
question_answer 70) \[{{H}_{3}}P{{O}_{3}}\]is:
A)
A dibasic acid
done
clear
B)
A tribasic acid
done
clear
C)
monobasic
done
clear
D)
neutral
done
clear
View Answer play_arrow
question_answer 71) Cassiterite is an ore of:
A)
iron
done
clear
B)
lead
done
clear
C)
mercury
done
clear
D)
tin
done
clear
View Answer play_arrow
question_answer 72) The substance used as pigment in paint is:
A)
borax
done
clear
B)
alumina
done
clear
C)
lithopone
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 73) A certain mass of gas occupies a volume of 2 litre at STP keeping the pressure constant. At what temperature would the gas occupy a volume of 4 litre?
A)
\[50{}^\circ C\]
done
clear
B)
\[100{}^\circ C\]
done
clear
C)
\[273{}^\circ C\]
done
clear
D)
\[546{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 74) Benzoic acid dissolved in benzene shows a molecular weight of:
A)
61
done
clear
B)
244
done
clear
C)
366
done
clear
D)
122
done
clear
View Answer play_arrow
question_answer 75) In the compounds\[KMn{{O}_{4}}\]and\[{{K}_{2}}C{{r}_{2}}{{O}_{7}},\]the highest oxidation state is of the element:
A)
\[Mn\]
done
clear
B)
\[K\]
done
clear
C)
\[O\]
done
clear
D)
\[Cr\]
done
clear
View Answer play_arrow
question_answer 76) Among\[HF,C{{H}_{4}},C{{H}_{3}}OH\]and\[{{N}_{2}}{{O}_{4}},\]inter molecular hydrogen bond is expected:
A)
in two
done
clear
B)
in all
done
clear
C)
in all leaving one
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 77) Which gas has the higher partial pressure in atmosphere?
A)
\[{{N}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 78) What bond order does\[L{{i}_{2}}\]have?
A)
3
done
clear
B)
1
done
clear
C)
2
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 79) The element present in gun-metal is:
A)
\[Co\]
done
clear
B)
\[Cu\]
done
clear
C)
\[Sc\]
done
clear
D)
\[Ti\]
done
clear
View Answer play_arrow
question_answer 80) The weakest acid is:
A)
\[HI\]
done
clear
B)
\[HBr\]
done
clear
C)
\[HCl\]
done
clear
D)
\[HF\]
done
clear
View Answer play_arrow
question_answer 81) Carborundum is the commercial name of:
A)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
B)
\[SiC\]
done
clear
C)
\[BN\]
done
clear
D)
\[Si{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 82) Which conversion is an oxidation?
A)
\[SO_{4}^{2-}.....SO_{3}^{2-}\]
done
clear
B)
\[C{{u}^{2+}}....Cu\]
done
clear
C)
\[{{H}^{+}}....H\]
done
clear
D)
\[{{H}^{-}}....H\]
done
clear
View Answer play_arrow
question_answer 83) Ionic hydrides react with water to give:
A)
hydride ions
done
clear
B)
acidic solutions
done
clear
C)
protons
done
clear
D)
basic solutions
done
clear
View Answer play_arrow
question_answer 84) For the cell, \[TI|T{{I}^{+}}(0.001\,M)||C{{u}^{2+}}(0.1)M|Cu\] \[{{E}_{cell}}\] at\[25{}^\circ C\]is\[0.83\text{ }V.\text{ }{{E}_{cell}}\]can be increased:
A)
by decreasing\[[C{{u}^{2+}}]\]
done
clear
B)
by increasing\[[C{{u}^{2+}}]\]
done
clear
C)
by increasing\[[T{{i}^{+}}]\]
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 85) Electrolytes, when dissolved in water, dissociate into their constituent ions. The degree of dissocation of a weak electrolyte increases with:
A)
the presence of a substance yielding common ion
done
clear
B)
decreasing temperature
done
clear
C)
decreasing concentration of the electrolyte
done
clear
D)
increasing concentration of the electrolyte
done
clear
View Answer play_arrow
question_answer 86) The number of p-electrons in bromine atom is:
A)
12
done
clear
B)
15
done
clear
C)
7
done
clear
D)
17
done
clear
View Answer play_arrow
question_answer 87) If the ionization potential for hydrogen atom is 13.6 eV, then the ionization potential for\[H{{e}^{+}}\]ion should be:
A)
13.6 eV
done
clear
B)
6.8 eV
done
clear
C)
54.4 eV
done
clear
D)
72.2 Ev
done
clear
View Answer play_arrow
question_answer 88) Pinacol is:
A)
3-methyl butan-2-ol
done
clear
B)
2, 3-dimethyl-2, 3-butandiol
done
clear
C)
2, 3-dimethyl-2-propanone
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 89) In glycine, the basic group is:
A)
\[-CO{{O}^{-}}\]
done
clear
B)
\[-COOH\]
done
clear
C)
\[-N{{H}_{2}}\]
done
clear
D)
\[-NH_{3}^{+}\]
done
clear
View Answer play_arrow
question_answer 90) Which alkyl halide is preferentially hydrolysed by SN mechanism?
A)
\[{{(C{{H}_{3}})}_{3}}C.Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
D)
\[C{{H}_{3}}Cl\]
done
clear
View Answer play_arrow
question_answer 91) The number of\[\pi -\]bonds present in propyne is:
A)
4
done
clear
B)
1
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 92) When metals react with non-metals, the metal atoms tend to?
A)
share electrons
done
clear
B)
lose electrons
done
clear
C)
gain electrons
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 93) 1 kg of\[NaOH\]solution contains 4g of\[NaOH\]. The approximate concentration of the solution is:
A)
about 0.1 N
done
clear
B)
decinormol
done
clear
C)
0.1 molal
done
clear
D)
0.1 molar
done
clear
View Answer play_arrow
question_answer 94) How much of\[0.1\text{ }M\text{ }{{H}_{2}}S{{O}_{4}}\]solution is required to neutralize 50 ml of\[0.2\text{ }M\text{ }NaOH\]solution:
A)
50 ml
done
clear
B)
5.0 ml
done
clear
C)
0.50ml
done
clear
D)
100 ml
done
clear
View Answer play_arrow
question_answer 95) The rate for the reaction\[RCl+NaOH(aq)\xrightarrow[{}]{{}}ROH+NaCl\]is given by rate\[=k[RCl],\]the freezing point of the, reaction is:
A)
unaffacted by increasing the temp. of the reaction
done
clear
B)
decreased on increasing the temp. of the reaction
done
clear
C)
halved on reducing the concentration of \[RCl\]to half
done
clear
D)
doubled on doubling the concentration of\[NaOH\]
done
clear
View Answer play_arrow
question_answer 96) The sub atomic particle, not found in the nucleus, is:
A)
proton
done
clear
B)
neutron
done
clear
C)
electron
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 97) Emission of a beta particle by an atom of an element results in the formation of:
A)
isomer
done
clear
B)
isotope
done
clear
C)
isobar
done
clear
D)
isomorph
done
clear
View Answer play_arrow
question_answer 98) Which type of isomerism is most common among ethers?
A)
Metamerism
done
clear
B)
Functional
done
clear
C)
Chain
done
clear
D)
Position
done
clear
View Answer play_arrow
question_answer 99) Glucose is hydrolysed by zymase into:
A)
amino acids
done
clear
B)
alcohol
done
clear
C)
aromatic acids
done
clear
D)
dicarboxylic acid
done
clear
View Answer play_arrow
question_answer 100) For the equilibrium, \[PC{{l}_{5}}PC{{l}_{3}}+C{{l}_{2}},{{K}_{c}}=\frac{{{\alpha }^{2}}}{(1-\alpha )V}\] temperature remaining constant:
A)
\[{{K}_{c}}\]may increase or decrease with the change in volume depending upon its numerical value
done
clear
B)
\[{{K}_{c}}\]will increase with the increase in volume
done
clear
C)
\[{{K}_{c}}\]will increase with the decrease in volume
done
clear
D)
\[{{K}_{c}}\]will not change with the change in volume
done
clear
View Answer play_arrow
question_answer 101) 20g of a binary electrolyte (mol. Wt = 100) are dissolved in 500 g of water. The depression in freezing point of the solution is\[0.74{}^\circ C\](\[{{K}_{f}}=\]\[1.86\text{ }K\text{ }molalit{{y}^{-1}}\]) The degree of ionization of the electrolyte is:
A)
0 %
done
clear
B)
100 %
done
clear
C)
75%
done
clear
D)
50 %
done
clear
View Answer play_arrow
question_answer 102) A spontaneous change is one in which the system suffers:
A)
a lowering of free energy
done
clear
B)
a lowering of energy
done
clear
C)
an increase in internal energy
done
clear
D)
no energy change
done
clear
View Answer play_arrow
question_answer 103) According to equation, \[{{C}_{6}}{{H}_{6}}(l)+\frac{15}{2}{{O}_{2}}(g)\xrightarrow[{}]{{}}2{{H}_{2}}O(l)+6C{{O}_{2}}(g)\] \[\Delta H=-3264.4\text{ }kJ/mol\]. The energy evolved when 7.8 g of benzene is burnt in air will be:
A)
3.264 kJ/mol
done
clear
B)
32.64 kJ/mol
done
clear
C)
326.4 kJ/mol
done
clear
D)
163.22 kJ/mol
done
clear
View Answer play_arrow
question_answer 104) When\[PC{{l}_{5}}\]reacts with sulphuric acid, sulphuryl chloride\[(S{{O}_{2}}C{{l}_{2}})\]is formed as the final product. This shows that sulphuric acid:
A)
has two hydroxyl groups in its structure
done
clear
B)
is a derivative of sulphur dioxide
done
clear
C)
is a dibasic acid
done
clear
D)
has greater affinity for water
done
clear
View Answer play_arrow
question_answer 105) In which reaction addition takes place according to Markownikoffs rule?
A)
\[C{{H}_{3}}CH=CHC{{H}_{3}}+B{{r}_{2}}\xrightarrow[{}]{{}}\]
done
clear
B)
\[C{{H}_{2}}=C{{H}_{2}}+HBr\xrightarrow[{}]{{}}\]
done
clear
C)
\[C{{H}_{3}}CH=C{{H}_{2}}+HBr\xrightarrow[{}]{{}}\]
done
clear
D)
\[C{{H}_{3}}CH=C{{H}_{2}}+B{{r}_{2}}\xrightarrow[{}]{{}}\]
done
clear
View Answer play_arrow
question_answer 106) Which statement is false? (Assume complete dissociation in each case):
A)
If 2.0 litre of a solution of\[{{H}_{2}}S{{O}_{4}}\]contains 0.1 mole, then pH of the solution is 2
done
clear
B)
The concentration of\[O{{H}^{-}}\]in 0.005 M\[HN{{O}_{3}}\]is \[2.0\times {{10}^{-12}}mol/1\]
done
clear
C)
The pH of 0.01 M KOH is 12
done
clear
D)
In a 0.001 M solution of\[NaOH\]the concentration of\[{{H}^{+}}\]is\[{{10}^{-3}}\text{ }mol/litre\]
done
clear
View Answer play_arrow
question_answer 107) The hydrolysis constant of a salt of weak acid and weak base is inversely proportional to:
A)
ionic product of water
done
clear
B)
dissocation constant of both weak acid and weak base
done
clear
C)
dissociation constant of weak acid
done
clear
D)
dissociation constant of weak base
done
clear
View Answer play_arrow
question_answer 108) Acetamide and ethyl amine are distinguished by reacting with:
A)
\[B{{r}_{2}}\]water
done
clear
B)
acidic\[KMn{{O}_{4}}\]
done
clear
C)
aq.\[NaOH\]and heat
done
clear
D)
aq.\[HCl\]and heat
done
clear
View Answer play_arrow
question_answer 109) Which shows the easier electrophilic substitution in ring?
A)
N-acetyl aniline
done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{3}}Cl\]
done
clear
C)
Aniline
done
clear
D)
Nitrobenzene
done
clear
View Answer play_arrow
question_answer 110) 300 ml of a gas at\[27{}^\circ C\]is cooled to\[-3{}^\circ C\]at constant pressure. The final volume is:
A)
\[350\text{ }l\]
done
clear
B)
\[270\text{ }ml\]
done
clear
C)
\[540\text{ }ml\]
done
clear
D)
\[135\text{ }ml\]
done
clear
View Answer play_arrow
question_answer 111) Oxygen and the oxide ion have the:
A)
same proton number
done
clear
B)
same electronic configuration
done
clear
C)
same electron number
done
clear
D)
same size
done
clear
View Answer play_arrow
question_answer 112) Which compound shows hydrogen bonding?
A)
\[RC{{H}_{2}}NHC{{H}_{3}}\]
done
clear
B)
\[RC{{H}_{2}}CHO\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[HCl\]
done
clear
View Answer play_arrow
question_answer 113) The temperature of 20 litre of nitrogen was increased from 10 K to 30 K at a constant pressure. Change in volume will be:
A)
20 litre
done
clear
B)
40 litre
done
clear
C)
60 litre
done
clear
D)
80 litre
done
clear
View Answer play_arrow
question_answer 114) The activity of alkaline earth metals as reducing agents:
A)
decreases from Be to Ba
done
clear
B)
increases from Be to Ba
done
clear
C)
increases from Be to\[Ca\]and decreases from \[Ca\]to Ba
done
clear
D)
decreases from Be to\[Ca\]and increases from\[Ca\]to Ba
done
clear
View Answer play_arrow
question_answer 115) A radioactive isotope has a half life of 10 days. If today there are 125 g of it left what was its original weight 40 days earlier?
A)
2000 g
done
clear
B)
1250 g
done
clear
C)
1000g
done
clear
D)
600g
done
clear
View Answer play_arrow
question_answer 116) Oxidation state of oxygen in\[{{H}_{2}}{{O}_{2}}\]is:
A)
\[-2\]
done
clear
B)
\[-1\]
done
clear
C)
\[+1\]
done
clear
D)
\[+2\]
done
clear
View Answer play_arrow
question_answer 117) Which is the property of non-metal?
A)
electronegative
done
clear
B)
basic nature of oxide
done
clear
C)
reducing property
done
clear
D)
low ionization potential
done
clear
View Answer play_arrow
question_answer 118) Hofmann bromide reaction is used to prepare:
A)
\[1{}^\circ \]amine
done
clear
B)
\[2{}^\circ \]amine
done
clear
C)
\[3{}^\circ \]amine
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 119) The main component of oil of winter green is:
A)
salicylic acid
done
clear
B)
methyl salicylate
done
clear
C)
acetyl salicylic acid
done
clear
D)
salicyladehyde
done
clear
View Answer play_arrow
question_answer 120) Which pair is isoelectronic?
A)
\[Ar\] and \[Cl\]
done
clear
B)
\[N{{a}^{+}}\]and\[Ne\]
done
clear
C)
\[N{{a}^{+}}\]and \[Mg\]
done
clear
D)
\[Mg\]and \[Ne\]
done
clear
View Answer play_arrow
question_answer 121) Pasteur and Koch are related to:
A)
discovery of nucleic acids (DNA and RNA)
done
clear
B)
discovery of ultracentrifuge
done
clear
C)
germ theory of disease
done
clear
D)
gene splicing
done
clear
View Answer play_arrow
question_answer 122) Upon amniocentesis of a pregnant woman, it is found that the embryo contains both, Barr body and F-body. The syndrome likely to be associated with the embryo is:
A)
Edwards syndrome
done
clear
B)
Downs syndrome
done
clear
C)
Klinefelters syndrome
done
clear
D)
Pataus syndrome
done
clear
View Answer play_arrow
question_answer 123) The retina of nocturnal birds contain:
A)
cones only
done
clear
B)
rods only
done
clear
C)
both a and b
done
clear
D)
either a or b
done
clear
View Answer play_arrow
question_answer 124) Among the following which one is longest phase in prophase of meosis?
A)
Leptotene
done
clear
B)
Zygotene
done
clear
C)
Pachytene
done
clear
D)
Diplotene
done
clear
View Answer play_arrow
question_answer 125) The cavity in the region of diencephalon in the brain of rabbit is called:
A)
lateral ventricle
done
clear
B)
third ventricle
done
clear
C)
foramen of monro
done
clear
D)
iter
done
clear
View Answer play_arrow
question_answer 126) Graafian follicle contains:
A)
oogonial cells
done
clear
B)
corpus luteum
done
clear
C)
theca extema and theca interna
done
clear
D)
corpus albicans
done
clear
View Answer play_arrow
question_answer 127) Excretion means:
A)
formation of those substances which have some role in the body
done
clear
B)
removal of such substances which have never been part of the body
done
clear
C)
removal of useless substances and substances present in excess
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 128) Arthropoda are characterised by:
A)
jointed appendages
done
clear
B)
open blood vascular system
done
clear
C)
triploblastic
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 129) A red blood cell (RBC) was kept in a certain solution for few minutes and it got burst. The said solution was:
A)
isotonic
done
clear
B)
concentrated sugar solution
done
clear
C)
hypertonic
done
clear
D)
hypotonic
done
clear
View Answer play_arrow
question_answer 130) The alternate host of Taenia saginata is:
A)
cow
done
clear
B)
pig
done
clear
C)
dog
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 131) Mammary glands are modified:
A)
sweat glands
done
clear
B)
ceruminous glands
done
clear
C)
glands of Zeis
done
clear
D)
inguinal glands
done
clear
View Answer play_arrow
question_answer 132) End of long bones are covered with:
A)
muscles
done
clear
B)
cartilage
done
clear
C)
adipose tissue
done
clear
D)
bone marrow
done
clear
View Answer play_arrow
question_answer 133) Which of the following is correct?
A)
Mesoderm - brain
done
clear
B)
Ectoderm - liver
done
clear
C)
Mesoderm - skeleton
done
clear
D)
Endodermis - epidermis
done
clear
View Answer play_arrow
question_answer 134) The generic name of tusk shell is:
A)
Dentalium
done
clear
B)
Chaetodenna
done
clear
C)
Chiton
done
clear
D)
Neopima
done
clear
View Answer play_arrow
question_answer 135) From Ascaris egg, first larva hatches out in the:
A)
intestine of host
done
clear
B)
stomach of host
done
clear
C)
outside the body
done
clear
D)
uterus of female Ascaris
done
clear
View Answer play_arrow
question_answer 136) Which of the following presumably possesses a cranial capacity larger than modern man?
A)
Neanderthal man
done
clear
B)
Peeking man
done
clear
C)
Australopitheciis
done
clear
D)
Cro-magnon man
done
clear
View Answer play_arrow
question_answer 137) Spontaneous generation theory was given by:
A)
F. Reddi
done
clear
B)
L. Splanznii
done
clear
C)
Louis Pasteur
done
clear
D)
Aristotle
done
clear
View Answer play_arrow
question_answer 138) Electro cardiograph was developed by:
A)
Hans Berger
done
clear
B)
Willem Kolff
done
clear
C)
Willen Einthoven
done
clear
D)
Wiehelm Roentgen
done
clear
View Answer play_arrow
question_answer 139) Various enzymes and hormones can be produced on large scale by employing the recombinant:
A)
DNA technique
done
clear
B)
RNA technique
done
clear
C)
viral technique
done
clear
D)
bacterial technique
done
clear
View Answer play_arrow
question_answer 140) Which structure is not related to respiration in frog?
A)
Diaphragm
done
clear
B)
Skin
done
clear
C)
Buccal cavity
done
clear
D)
Lungs
done
clear
View Answer play_arrow
question_answer 141) Increase of blood sugar level is known as:
A)
diabetes insipidus
done
clear
B)
diabetes mellitus
done
clear
C)
hypoglycemia
done
clear
D)
both a and b
done
clear
View Answer play_arrow
question_answer 142) Mosaic disease in tobacco is due to:
A)
bacteria
done
clear
B)
virus
done
clear
C)
mycoplasma
done
clear
D)
algae
done
clear
View Answer play_arrow
question_answer 143) Pulse beat is measured from:
A)
arteries
done
clear
B)
veins
done
clear
C)
capillaries
done
clear
D)
nerves
done
clear
View Answer play_arrow
question_answer 144) Which of the following are examples of vestigial structures in man?
A)
Wisdom tooth, head, nails
done
clear
B)
Wisdom tooth, vermiform appendix, hair
done
clear
C)
Wisdom tooth, vermiform appendix, coccyx
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 145) Immunoglobulins are made up of:
A)
two polypeptide chains
done
clear
B)
four polypeptide chains
done
clear
C)
three ammo acids only
done
clear
D)
five nucleotides
done
clear
View Answer play_arrow
question_answer 146) Which of the following disorders can be regarded as madness?
A)
Psychosis
done
clear
B)
Epilepsy
done
clear
C)
Insomnia
done
clear
D)
Parkinsonism
done
clear
View Answer play_arrow
question_answer 147) Vitamin D is produced in human body by:
A)
muscles
done
clear
B)
skin
done
clear
C)
nerves
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 148) Common characteristics of mosquito, housefly and cockroaches are:
A)
one pair each of wings and halters
done
clear
B)
three pair of legs and one pair of developed wings
done
clear
C)
two pair of legs and two compound eyes
done
clear
D)
compound and simple eyes
done
clear
View Answer play_arrow
question_answer 149) Most recent insecticides in India are:
A)
chlorinated hydrocarbons
done
clear
B)
organo phosphorus compounds
done
clear
C)
carbamides
done
clear
D)
pyrethroids
done
clear
View Answer play_arrow
question_answer 150) Tube feet are locomotory organs in:
A)
Annelida
done
clear
B)
Mollusca
done
clear
C)
Echinodermata
done
clear
D)
Arthropoda
done
clear
View Answer play_arrow
question_answer 151) Ozone depletion in stratosphere shall result in:
A)
forest fires
done
clear
B)
green house effect
done
clear
C)
global warming
done
clear
D)
increased incidence of skin cancer
done
clear
View Answer play_arrow
question_answer 152) Acetabulum is the cavity of:
A)
pelvic girdle of frog
done
clear
B)
sternum of frog
done
clear
C)
pectoral girdle of frog
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 153) Which of the following is a pair of analogous organs?
A)
Contractile vacuole in Amoeba and uriniferous tubule in frog
done
clear
B)
Paddle of whale and front legs of horse
done
clear
C)
Mouth parts in insects
done
clear
D)
Fore limbs in lizard and wings in birds
done
clear
View Answer play_arrow
question_answer 154) Role of typhlosole in earthworm is to:
A)
control blood flow
done
clear
B)
increase absorptive surface area
done
clear
C)
produce digestive enzymes
done
clear
D)
crush food
done
clear
View Answer play_arrow
question_answer 155) National wild life protection act was formulated during:
A)
1972
done
clear
B)
1973
done
clear
C)
1952
done
clear
D)
1992
done
clear
View Answer play_arrow
question_answer 156) Glucagon is secreted by:
A)
adrenal medulla
done
clear
B)
\[\beta \]cells of islets of Langerhans
done
clear
C)
\[\alpha \]cells of islet of Langerhans
done
clear
D)
adrenal cortex
done
clear
View Answer play_arrow
question_answer 157) Scala tympani is connected to scala vestibuli by:
A)
stapes
done
clear
B)
space of nuel
done
clear
C)
helicoterma
done
clear
D)
basilar membrane
done
clear
View Answer play_arrow
question_answer 158) Eurythermal animals and plants are those:
A)
which can tolerate only a small variation in temperature
done
clear
B)
which can tolerate large variation in temperature
done
clear
C)
which cannot tolerate any change in temperature
done
clear
D)
which are affected by temperature
done
clear
View Answer play_arrow
question_answer 159) Ammoniogenesis is process of:
A)
production of\[N{{H}_{3}}\]in liver
done
clear
B)
excretion of ammonia by tadpole of frog
done
clear
C)
exchange of\[N{{a}^{+}}\]for\[N{{H}_{3}}\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 160) Mollusc which does not have ink gland is:
A)
Pila
done
clear
B)
Loligo
done
clear
C)
Sepia
done
clear
D)
Octopus
done
clear
View Answer play_arrow
question_answer 161) If number of chromosome in foot of fern embryo is 8, what should be the number in its spores?
A)
4
done
clear
B)
8
done
clear
C)
23
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 162) Casparian strips are characteristic of:
A)
endodermis
done
clear
B)
epidermis
done
clear
C)
epiblema
done
clear
D)
xylem vessels
done
clear
View Answer play_arrow
question_answer 163) Which is true for meristematic tissue?
A)
Their cells have dense cytoplasm and prominent nuclei
done
clear
B)
Their cells are dead with large intercellular spaces
done
clear
C)
These are usually modified for storage
done
clear
D)
Their cells are most primitive, living and without nuclei
done
clear
View Answer play_arrow
question_answer 164) When funicle, chalaza and micropyle lie in one straight line or lie in same longitudinal axis the ovule is called:
A)
Anatropous
done
clear
B)
Orthotropous
done
clear
C)
Amphitropous
done
clear
D)
Campylotropous
done
clear
View Answer play_arrow
question_answer 165) In Funaria dispersal of spores is achieved with the help of:
A)
columella
done
clear
B)
calyptra
done
clear
C)
peristome
done
clear
D)
elaters
done
clear
View Answer play_arrow
question_answer 166) In Pinus the endosperm is:
A)
haploid
done
clear
B)
diploid
done
clear
C)
triploid
done
clear
D)
teteraploid
done
clear
View Answer play_arrow
question_answer 167) Chipko Movement is related to:
A)
plant conservation
done
clear
B)
project tiger
done
clear
C)
animal breeding
done
clear
D)
operation flood
done
clear
View Answer play_arrow
question_answer 168) In the following there is no difference:
A)
trophic level 1st and herbivores
done
clear
B)
primary consumers and herbivores
done
clear
C)
primary carnivore and trophic level IInd
done
clear
D)
secondary consumer and herbivores
done
clear
View Answer play_arrow
question_answer 169) Simondosia chinensis is commonly known as:
A)
amla
done
clear
B)
poppy
done
clear
C)
teak wood
done
clear
D)
jojoba
done
clear
View Answer play_arrow
question_answer 170) Tuisi belongs to family:
A)
Asclepiadaceae
done
clear
B)
Labiatae
done
clear
C)
Umbelliferae
done
clear
D)
Rubiaceae
done
clear
View Answer play_arrow
question_answer 171) Plasmid is:
A)
bacteriophage
done
clear
B)
DNA molecule incorporailed in bacterial chromosome
done
clear
C)
DNA molecule present in mitochondria
done
clear
D)
extra chromosomal genetic element of bacteria
done
clear
View Answer play_arrow
question_answer 172) Plant and animal cells, both have in common:
A)
cell membrane and nucleolus
done
clear
B)
cell membrane and cell wall
done
clear
C)
nucleolus and chloroplast
done
clear
D)
nucleus and cell wall
done
clear
View Answer play_arrow
question_answer 173) Gibberellins cause:
A)
shortening of genetically tall plants
done
clear
B)
elongation of genetically dwarf plants
done
clear
C)
promotion of rooting
done
clear
D)
yellowing of young leaves
done
clear
View Answer play_arrow
question_answer 174) Which of the following is associated with synthesis of enzymes?
A)
Translation
done
clear
B)
Replication
done
clear
C)
Transduction
done
clear
D)
Transformation
done
clear
View Answer play_arrow
question_answer 175) During anaerobic respiration in yeast:
A)
\[{{H}_{2}}O\] and\[C{{O}_{2}}\]are end products
done
clear
B)
\[C{{O}_{2}},\]ethanol and energy are end products
done
clear
C)
\[C{{O}_{2}}\]and\[{{H}_{2}}O\]are end products
done
clear
D)
\[C{{O}_{2}},\]acetic acid and energy are end products
done
clear
View Answer play_arrow
question_answer 176) Hatch and Slack cycle is found in:
A)
\[{{C}_{4}}\]plants
done
clear
B)
\[{{C}_{3}}\]plants
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 177) The site of protein synthesis is:
A)
smooth E.R
done
clear
B)
rough E.R
done
clear
C)
plasmalemma
done
clear
D)
nucleus
done
clear
View Answer play_arrow
question_answer 178) The modem concept of gene is:
A)
a segment of DNA, capable of crossing over
done
clear
B)
functional unit of DNA
done
clear
C)
a segment of RNA
done
clear
D)
a segment of chromosome
done
clear
View Answer play_arrow
question_answer 179) The disease Oriental sore is caused by:
A)
bacteria
done
clear
B)
virus
done
clear
C)
protozoa
done
clear
D)
fungus
done
clear
View Answer play_arrow
question_answer 180) Viroids differ from viruses in having:
A)
naked RNA molecules only
done
clear
B)
naked DNA molecules only
done
clear
C)
naked DNA packed with viral genome
done
clear
D)
satellite RNA packed with viral genome
done
clear
View Answer play_arrow
question_answer 181) Quantum yield of photosynthesis is:
A)
33%
done
clear
B)
9%
done
clear
C)
12%
done
clear
D)
8%
done
clear
View Answer play_arrow
question_answer 182) Guttation is manifestation of:
A)
osmosis
done
clear
B)
transpiration
done
clear
C)
root pressure
done
clear
D)
photosynthesis
done
clear
View Answer play_arrow
question_answer 183) Water rises in the stem due to:
A)
cohesion and transpirational pull
done
clear
B)
turgor pressure
done
clear
C)
osmotic pressure
done
clear
D)
root absorption
done
clear
View Answer play_arrow
question_answer 184) Fensom and Jones suggested which of the following method for translocation of solute?
A)
Osmosis
done
clear
B)
Plasmolysis
done
clear
C)
Diffusion
done
clear
D)
Electro-osmosis
done
clear
View Answer play_arrow
question_answer 185) Glycolysis is a part of:
A)
anaerobic respiration only
done
clear
B)
aerobic respiration only
done
clear
C)
both a and b
done
clear
D)
Krebs cycle
done
clear
View Answer play_arrow
question_answer 186) Which of the following movements in plants is not related to change in auxin level?
A)
Nyctinastic leaf movement
done
clear
B)
Movement of root towards soil
done
clear
C)
Movement of sunflower, tracking the direction of sun
done
clear
D)
Movement of shoot towards light
done
clear
View Answer play_arrow
question_answer 187) The waxy substance associated with cell wall of cork cells is:
A)
suberin
done
clear
B)
cutin
done
clear
C)
hemicellulose
done
clear
D)
lignin
done
clear
View Answer play_arrow
question_answer 188) In which of the following there is no differentiation of bark, sap wood and heart wood?
A)
Ashok
done
clear
B)
Neem
done
clear
C)
Mango
done
clear
D)
Date palm
done
clear
View Answer play_arrow
question_answer 189) An auxotroph is:
A)
a plant that responds by vending towards sun
done
clear
B)
an organism that depends on another organism for meeting its nutritional requirements
done
clear
C)
a mutant which has lost its ability to synthesize one or more essential compounds
done
clear
D)
a plant that is able to synthesize its own carbohydrate
done
clear
View Answer play_arrow
question_answer 190) In plants vacuole contains:
A)
soil
done
clear
B)
water and dissolved substance
done
clear
C)
cytoplasm
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 191) The number of base pairs per helical turn in Z-DNA is:
A)
10
done
clear
B)
11
done
clear
C)
12
done
clear
D)
13
done
clear
View Answer play_arrow
question_answer 192) In mushroom gills are meant for:
A)
respiration
done
clear
B)
nutrition
done
clear
C)
bears spores which help in reproduction
done
clear
D)
enhancing buoyancy
done
clear
View Answer play_arrow
question_answer 193) Which of the following cause disease in human beings?
A)
Rhizopus
done
clear
B)
Pnccinia
done
clear
C)
Aspergilhis
done
clear
D)
Cystoptis
done
clear
View Answer play_arrow
question_answer 194) Heterosporous pteridophytes always produce:
A)
monoecious gametophytes
done
clear
B)
dioecious gametophytes
done
clear
C)
homothallic gametophyte
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 195) Term meiosis was proposed by:
A)
Farmer and Moore
done
clear
B)
Flemming
done
clear
C)
Strasburger
done
clear
D)
Darlington
done
clear
View Answer play_arrow
question_answer 196) In somatic cells DNA synthesis takes place during:
A)
S-phase
done
clear
B)
\[{{G}_{1}}\]phase
done
clear
C)
\[{{G}_{2}}\]phase
done
clear
D)
Prophase of mitosis
done
clear
View Answer play_arrow
question_answer 197) Edible portion of mango is:
A)
endocarp
done
clear
B)
mesocarp
done
clear
C)
cotyledons
done
clear
D)
endosperm
done
clear
View Answer play_arrow
question_answer 198) Pigment that absorbs red and far red light is:
A)
phytochrome
done
clear
B)
cytochrome
done
clear
C)
carotene
done
clear
D)
xanthophyll
done
clear
View Answer play_arrow
question_answer 199) Inheritance of characters not located in the gene but the young one resembling only the female part is due to:
A)
cytoplasmic inheritance
done
clear
B)
chromosomal inheritance
done
clear
C)
plastid inheritance
done
clear
D)
epigenesist
done
clear
View Answer play_arrow
question_answer 200) Okazaki fragments are produced during the synthesis of:
A)
m-RNA
done
clear
B)
Protein
done
clear
C)
t-RNA
done
clear
D)
DNA
done
clear
View Answer play_arrow
question_answer 201) The second meotic division leads to:
A)
separation of sex chromosomes
done
clear
B)
fresh DNA synthesis
done
clear
C)
separation of chromatids and centromere
done
clear
D)
separation of homologous chromosomes
done
clear
View Answer play_arrow
question_answer 202) The true stomach in ruminants where most of digestion take place is:
A)
rumen
done
clear
B)
omasum
done
clear
C)
reticulum
done
clear
D)
abomasum
done
clear
View Answer play_arrow
question_answer 203) Which of the following cells are related with inflammatory reactions and immediate hypersensitivity?
A)
Mast cells
done
clear
B)
Plasma cells
done
clear
C)
Tebroblast cells
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 204) In which pneumatophores are found?
A)
Tinospom
done
clear
B)
Finns
done
clear
C)
Rhizophom
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 205) Glyoxylate cycle occurs in:
A)
lysosomes
done
clear
B)
ribosomes
done
clear
C)
glyoxysomes
done
clear
D)
peroxisomes
done
clear
View Answer play_arrow
question_answer 206) Axolotal is name given to larva of:
A)
Roundworm
done
clear
B)
Amphioxris
done
clear
C)
Ambylostoma
done
clear
D)
Silkworm
done
clear
View Answer play_arrow
question_answer 207) Polygomim type of embryosac is:
A)
8 celled 8 nucleate
done
clear
B)
7 celled 8 nucleate
done
clear
C)
7 nucleate 8 celled
done
clear
D)
A female gamete
done
clear
View Answer play_arrow
question_answer 208) Photo respiration could easily by detected in:
A)
\[{{C}_{3}}\]plants
done
clear
B)
\[{{C}_{4}}\]plants
done
clear
C)
both a and b
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 209) Quiescent centre theory was proposed by:
A)
Nagelli
done
clear
B)
Schmidt
done
clear
C)
Hanstein
done
clear
D)
Clowes
done
clear
View Answer play_arrow
question_answer 210) In which of the following tissue preparations signet ring appearence is obtained?
A)
Apithelial tissue
done
clear
B)
Dense connective tissue
done
clear
C)
Adipose tissue
done
clear
D)
Reticular tissue
done
clear
View Answer play_arrow
question_answer 211) Scion is term related to:
A)
Embryology
done
clear
B)
Emasculation
done
clear
C)
Grafting
done
clear
D)
Palaeobotany
done
clear
View Answer play_arrow
question_answer 212) Which gland in female is a counter part of Cowper gland in male?
A)
Bartholin gland
done
clear
B)
Clatoris
done
clear
C)
Perineal gland
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 213) Correct sequence of electron acceptor of ATP syn diesis is:
A)
\[Cyt\text{ }a,\text{ }{{a}_{3}},\text{ }b,\text{ }c\]
done
clear
B)
\[Cyt\text{ }b,\text{ }c,\text{ }a,\text{ }{{a}_{3}}\]
done
clear
C)
\[Cyt\text{ }b,\text{ }c,\text{ }{{a}_{3}},\text{ }a\]
done
clear
D)
\[Cyt\text{ }c,\text{ }b,\text{ }a,\text{ }{{a}_{3}}\]
done
clear
View Answer play_arrow
question_answer 214) Regulation to restrict the movement of diseased plant material from one place to another are called:
A)
plant regulations
done
clear
B)
plant quarantine
done
clear
C)
plant protection
done
clear
D)
crop rotation
done
clear
View Answer play_arrow
question_answer 215) The name of first cloned sheep is:
A)
Dolly
done
clear
B)
Polly
done
clear
C)
Molley
done
clear
D)
Holly
done
clear
View Answer play_arrow
question_answer 216) Which of the following are symtoms of radiation desease?
A)
Red and ulcered skin
done
clear
B)
Nausea and loss of hair
done
clear
C)
Nausea and anaemia
done
clear
D)
Ulcered skin, anaemia, nausea and loss of hair
done
clear
View Answer play_arrow
question_answer 217) What is the generic name of Portugese man of war.
A)
Aurelia
done
clear
B)
Obelia
done
clear
C)
Physalia
done
clear
D)
Velella
done
clear
View Answer play_arrow
question_answer 218) Endemic plants are those which are:
A)
cosmopolition in distribution
done
clear
B)
restricted to grow over certain areas
done
clear
C)
found in artic region
done
clear
D)
gregarious in habit
done
clear
View Answer play_arrow
question_answer 219) Sigma factor is related to:
A)
RNA polymerase
done
clear
B)
DNA polymerase
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 220) Double fertilization is a characteristic of :
A)
angiosperms
done
clear
B)
gymnosperms
done
clear
C)
thallophytes
done
clear
D)
all plants
done
clear
View Answer play_arrow
question_answer 221) He is always .......... to his uncle because he consider him a ..... fellow.
A)
disrespectful; venerable
done
clear
B)
respectful; dihonourable
done
clear
C)
respectful; venerable
done
clear
D)
impudent, respectable
done
clear
View Answer play_arrow
question_answer 222) If misery is effect of ill fortune, it ought to be pitied, if it is of ........ it ought to be
A)
virtue; criticized
done
clear
B)
calamity; reverenced
done
clear
C)
virtue; protected
done
clear
D)
virtue; reverenced
done
clear
View Answer play_arrow
question_answer 223) All the performances of human art, at which we look with praise or wonder, are ......... of the restless ..... of perseverance.
A)
manifestations, pronouncement
done
clear
B)
projections, component
done
clear
C)
instances, force
done
clear
D)
proofs, humanity
done
clear
View Answer play_arrow
question_answer 224) Due to .......... rainfall this year, there will be .......... cut in water supply.
A)
enough, substantial
done
clear
B)
meagre, least
done
clear
C)
sufficient, no
done
clear
D)
surplus/abundant
done
clear
View Answer play_arrow
question_answer 225) The ........ imposed for non-payment was too ........ for it to bring in improvement in collection.
A)
fine, severe
done
clear
B)
toll, simple
done
clear
C)
penalty, low
done
clear
D)
damage, cruel
done
clear
View Answer play_arrow
question_answer 226) Only with executive ........ can the organisation concentrate its energies on ............ competitive advantage over time.
A)
position, embarking
done
clear
B)
deployment, directing
done
clear
C)
contingent/fabricating
done
clear
D)
commitment, custoning
done
clear
View Answer play_arrow
question_answer 227) The human mind is never ......... it advances or it ......... .
A)
absolute, diminishes
done
clear
B)
dynamic, stops
done
clear
C)
perfect, disintegrates
done
clear
D)
stationary, retrogrades
done
clear
View Answer play_arrow
question_answer 228) I am not...... to sell you my house unless you offer a more ...... price.
A)
conform, true
done
clear
B)
prepared, realistic
done
clear
C)
ready, correct
done
clear
D)
having, actual
done
clear
View Answer play_arrow
question_answer 229) Vision is usually ........ most effectively when many different ....... are used.
A)
developed, manifestations
done
clear
B)
adapted, organisations
done
clear
C)
communicated, vehicles
done
clear
D)
exhibited, forms
done
clear
View Answer play_arrow
question_answer 230) The ........ thinkers are not interested in propagating....... ideas among the masses.
A)
practical, idea
done
clear
B)
original, hackneyed
done
clear
C)
religious, spiritual
done
clear
D)
secular, political
done
clear
View Answer play_arrow
question_answer 231) Directions: Read each sentence to find out whether there is any error in it. The error, if any will in one part of the sentence. The number of that part is the answer.
A)
The girl said
done
clear
B)
that she preferred
done
clear
C)
the blue gown
done
clear
D)
than the black one
done
clear
E)
No error
done
clear
View Answer play_arrow
question_answer 232) Directions: Read each sentence to find out whether there is any error in it. The error, if any will in one part of the sentence. The number of that part is the answer.
A)
There is
done
clear
B)
no other way
done
clear
C)
to end war
done
clear
D)
except diasarmament
done
clear
E)
No error
done
clear
View Answer play_arrow
question_answer 233) Directions: Read each sentence to find out whether there is any error in it. The error, if any will in one part of the sentence. The number of that part is the answer.
A)
I was pretty sure that
done
clear
B)
he would support my views
done
clear
C)
for changing the old age
done
clear
D)
and static structure of our organisation
done
clear
E)
No error
done
clear
View Answer play_arrow
question_answer 234) Directions: Read each sentence to find out whether there is any error in it. The error, if any will in one part of the sentence. The number of that part is the answer.
A)
If you had
done
clear
B)
work hard
done
clear
C)
you should have
done
clear
D)
certainly got the scholarship
done
clear
E)
No error
done
clear
View Answer play_arrow
question_answer 235) Directions: Read each sentence to find out whether there is any error in it. The error, if any will in one part of the sentence. The number of that part is the answer.
A)
Entering
done
clear
B)
the crowded store
done
clear
C)
I saw two
done
clear
D)
vaguely familiar faces
done
clear
E)
No error
done
clear
View Answer play_arrow
question_answer 236)
Directions: In each of the questions below four sentences are given winch are denoted by P, Q, R, S. By using all the four sentences you have to frame a meaningful para. The correct order of the sentences is your answer. Choose from the four alternatives the one having the correct order of the sentences. The exhibition committee: (P) attractive and useful (Q) to make the exhibition (R) making effort (S) has been
A)
SRPQ
done
clear
B)
QPSR
done
clear
C)
QPRS
done
clear
D)
SRQP
done
clear
View Answer play_arrow
question_answer 237)
Directions: In each of the questions below four sentences are given winch are denoted by P, Q, R, S. By using all the four sentences you have to frame a meaningful para. The correct order of the sentences is your answer. Choose from the four alternatives the one having the correct order of the sentences. You must have a note: (P) otherwise she will be terribly worried (Q) for your mother (R) at your usual hour (S) if you are not back at home
A)
QPSR
done
clear
B)
QPRS
done
clear
C)
SRQP
done
clear
D)
SRPQ
done
clear
View Answer play_arrow
question_answer 238)
Directions: In each of the questions below four sentences are given winch are denoted by P, Q, R, S. By using all the four sentences you have to frame a meaningful para. The correct order of the sentences is your answer. Choose from the four alternatives the one having the correct order of the sentences. Though he was only a child: (P) he often much work harder (Q) than was either necessary (R) in order to please his parents (S) or good for him
A)
PRSQ
done
clear
B)
RPQS
done
clear
C)
RQSP
done
clear
D)
PRSQ
done
clear
View Answer play_arrow
question_answer 239)
Directions: In each of the questions below four sentences are given winch are denoted by P, Q, R, S. By using all the four sentences you have to frame a meaningful para. The correct order of the sentences is your answer. Choose from the four alternatives the one having the correct order of the sentences. They tell us: (P) in some places (Q) there must have been (R) that when the great glaciers of the ice age melted (S) vast steppes or wide treeless plains
A)
RQPS
done
clear
B)
RQSP
done
clear
C)
QRPS
done
clear
D)
QRSP
done
clear
View Answer play_arrow
question_answer 240)
Directions: In each of the questions below four sentences are given winch are denoted by P, Q, R, S. By using all the four sentences you have to frame a meaningful para. The correct order of the sentences is your answer. Choose from the four alternatives the one having the correct order of the sentences. There are people: (P) to be able to say (Q) not because they enjoy the book (R) who read a book (S) but because they want
A)
PSQR
done
clear
B)
SQPR
done
clear
C)
RSPQ
done
clear
D)
RQSP
done
clear
View Answer play_arrow
question_answer 241) Which one of the following Indian state has got its own separate state level constitution?
A)
Assam
done
clear
B)
Jammu and Kashmir
done
clear
C)
Arunachal Pradesh
done
clear
D)
Sikkim
done
clear
View Answer play_arrow
question_answer 242) Which Indian state is most densely populated?
A)
Rajasthan
done
clear
B)
Andhara Pradesh
done
clear
C)
Bihar
done
clear
D)
W. Bengal
done
clear
View Answer play_arrow
question_answer 243) Which of the following pairs is not corrrectly matched?
A)
Bharat Natyam: Tamil Nadu
done
clear
B)
Mohiniattam: Orissa
done
clear
C)
Kuchipudi: Andhra Pradesh
done
clear
D)
Bhangra: Punjab
done
clear
View Answer play_arrow
question_answer 244) Who was the author of the book Utopia?
A)
Dante
done
clear
B)
Eramus
done
clear
C)
Mochi Avelli
done
clear
D)
Thomas Moore
done
clear
View Answer play_arrow
question_answer 245) Which of the following is the most multilingual city in the wbrld?
A)
New York
done
clear
B)
Landon
done
clear
C)
Paris
done
clear
D)
Hongkong
done
clear
View Answer play_arrow
question_answer 246) Which state is the largest producer of rubber in India?
A)
Assam
done
clear
B)
Tamil Nadu
done
clear
C)
Kerala
done
clear
D)
Kamataka
done
clear
View Answer play_arrow
question_answer 247) Which of the following places in India withnesses the maximum difference between the length of days and night at various times of the year?
A)
Kanya Kumari
done
clear
B)
Mirzapur
done
clear
C)
Allahabad
done
clear
D)
Zanskar
done
clear
View Answer play_arrow
question_answer 248) Which Indian port handles the maximum Cargo traffic?
A)
Cochin
done
clear
B)
Kandia
done
clear
C)
Mumbai
done
clear
D)
Vishakhapatnam
done
clear
View Answer play_arrow
question_answer 249) Which term is used for the part of solar radiation that is reflected back from the earth without heating it?
A)
Albedo
done
clear
B)
Zero
done
clear
C)
Absorption
done
clear
D)
Einino
done
clear
View Answer play_arrow
question_answer 250) Which of the following states has the highest percentage of tank irrigation in the total irrigated area?
A)
Andhra Pradesh
done
clear
B)
Kamataka
done
clear
C)
Orrisa
done
clear
D)
Tamil Nadu
done
clear
View Answer play_arrow
question_answer 251) In a family, the father took\[\frac{1}{4}\]of the cake and he had three times as much of others had. The total number of family members is:
A)
9
done
clear
B)
12
done
clear
C)
3
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 252) If two mixers and one T. V. costs Rs. 7000. While two TV. and one mixer costs Rs. 9800. The value of TV is:
A)
Rs. 8400
done
clear
B)
Rs. 4200
done
clear
C)
Rs. 2800
done
clear
D)
Rs. 2100
done
clear
View Answer play_arrow
question_answer 253) A copper sphere of diameter 18 cm. is drawn into a wire of diameter 4 mm. Length of the wire is:
A)
214 m
done
clear
B)
242 m
done
clear
C)
243m
done
clear
D)
244m
done
clear
View Answer play_arrow
question_answer 254) Choose the correct alternative as Music : Guitar : Performer. :
A)
Trick : Roper : Acrobat
done
clear
B)
Dance : Tune : Instrument
done
clear
C)
Food : Recipe : Cook
done
clear
D)
Patient : Medicine : Docter
done
clear
View Answer play_arrow
question_answer 255)
In this question, the symbols are used as follows: A B means Add B to A A B means Subtract B from A A @ means Divide A by B A* B means Multiply A by B
The total air fare is calculated by adding 15 % of basic fare as fuel surcharge, 2% of the basic fare as IAIA charges and by 200 as airport tan to the basic fare. If the basic fare of a sector is B, which of the following will represent the total fare?
A)
\[B\left( B*15 \right)100\left( B*2 \right)100\text{ }200\]
done
clear
B)
\[B\left( B*15 \right)100\left( B*2 \right)100\text{ }200\]
done
clear
C)
\[B(B*5)100(B*2)100\,\,200\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 256) In 10 years, A will be twice as old as B was 10 years ago. If at present A is of 9 years older than B, the present age of B is:
A)
19 year
done
clear
B)
29 year
done
clear
C)
39 year
done
clear
D)
49 year
done
clear
View Answer play_arrow
question_answer 257) In a certain code INSTITUTION is written as NOITUTITSNI. How is PERFECTION written in that code?
A)
NOICTEFRED
done
clear
B)
NOITCEFERP
done
clear
C)
NOITCEFREP
done
clear
D)
NOITCFFREP
done
clear
View Answer play_arrow
question_answer 258) If PALAM could be given code number 43, what code number can be given to SANTACRUZ?
A)
120
done
clear
B)
123
done
clear
C)
85
done
clear
D)
75
done
clear
View Answer play_arrow
question_answer 259) Find out missing number: \[2,9,28,65\]
A)
121
done
clear
B)
195
done
clear
C)
126
done
clear
D)
103
done
clear
View Answer play_arrow
question_answer 260) Page is related to Book as Leaf is related to:
A)
Root
done
clear
B)
Green
done
clear
C)
Tree
done
clear
D)
Forest
done
clear
View Answer play_arrow