question_answer 1) The sound wave produced in a gas is always
A)
longitudinal
done
clear
B)
transverse
done
clear
C)
stationary
done
clear
D)
electromagnetic
done
clear
View Answer play_arrow
question_answer 2) Source of sound and the observer are mutually at rest. If the speed of sound is changed, then the frequency of sound heard by the observer will appear to be
A)
increased
done
clear
B)
decreased
done
clear
C)
unchanged
done
clear
D)
decreasing exponentially
done
clear
View Answer play_arrow
question_answer 3) With what velocity should an observer approach stationary sound source, so that the apparent frequency of sound appear to be double of the initial frequency? (given velocity of sound = v)
A)
\[{{v}_{o}}=\frac{v}{2}\]
done
clear
B)
\[{{v}_{o}}=3v\]
done
clear
C)
\[{{v}_{o}}=2v\]
done
clear
D)
\[{{v}_{o}}=v\]
done
clear
View Answer play_arrow
question_answer 4) A charge q is lying at mid-point of the line joining the two similar charges Q. The system will be in equilibrium, if the value of q is
A)
\[\frac{Q}{2}\]
done
clear
B)
\[-\frac{Q}{2}\]
done
clear
C)
\[\frac{Q}{4}\]
done
clear
D)
\[-\frac{Q}{4}\]
done
clear
View Answer play_arrow
question_answer 5) Charges \[2q,\] \[-q\] and \[-q\] lie at the vertices of a triangle. The value of E and Vat the centroid of equilateral triangle will be
A)
\[E\ne 0\]and \[V\ne 0\]
done
clear
B)
\[E=0\]and \[V=0\]
done
clear
C)
\[E\ne 0\]and \[V=0\]
done
clear
D)
\[E=0\] and \[V\ne 0\]
done
clear
View Answer play_arrow
question_answer 6) Infinite charges of magnitude q each are lying at \[x=1,2,4,8,...\]metre on X-axis. The value of intensity of electric field at point \[x=0\]due to these charges will be
A)
\[12\times {{10}^{9}}q\,N/C\]
done
clear
B)
zero
done
clear
C)
\[6\times {{10}^{9}}q\,N/C\]
done
clear
D)
\[4\times {{10}^{9}}q\,N/C\]
done
clear
View Answer play_arrow
question_answer 7) The capacity of parallel plate capacitor in air and on immersing it into oil is \[50\mu F\] and \[110\mu F\]respectively. The dielectric constant of oil is
A)
\[0.45\]
done
clear
B)
\[0.55\]
done
clear
C)
\[1.10\]
done
clear
D)
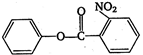
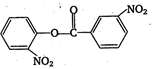
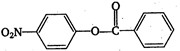
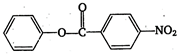
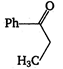
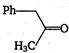
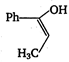
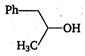
\[2.20\]
done
clear
View Answer play_arrow
question_answer 8) The energy stored in a condenser is in the form of
A)
kinetic energy
done
clear
B)
potential energy
done
clear
C)
elastic energy
done
clear
D)
magnetic energy
done
clear
View Answer play_arrow
question_answer 9) On increasing the plate separation of a charged condenser, the energy
A)
increases
done
clear
B)
decreases
done
clear
C)
remains unchanged
done
clear
D)
becomes zero
done
clear
View Answer play_arrow
question_answer 10) The ratio of electric fields on the axis and at equator of an electric dipole will be
A)
\[1:1\]
done
clear
B)
\[2:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[1:4\]
done
clear
View Answer play_arrow
question_answer 11) Some electric bulbs are connected in series across a \[220\text{ }V\]supply in a room. If one bulb is fused, then remaining bulbs are connected again in series across the same supply. The illumination in the room will be
A)
increase
done
clear
B)
decrease
done
clear
C)
remain the same
done
clear
D)
not continuous
done
clear
View Answer play_arrow
question_answer 12) If one junction of thermocouple is kept at \[{{0}^{o}}C\]and its emf is given by \[e=at+b{{t}^{2}},\]then the neutral temperature will be
A)
\[\frac{a}{b}\]
done
clear
B)
\[-\frac{a}{b}\]
done
clear
C)
\[\frac{a}{2b}\]
done
clear
D)
\[-\frac{a}{2b}\]
done
clear
View Answer play_arrow
question_answer 13) A cube is constructed from 12 identical wires. Current enters one comer of the cube and it leaves the opposite comer. If the resistance of each wire is r, then equivalent resistance will be
A)
\[\frac{6\,r}{5}\]
done
clear
B)
\[\frac{5\,r}{6}\]
done
clear
C)
\[\frac{5\,r}{12}\]
done
clear
D)
\[\frac{12\,r}{5}\]
done
clear
View Answer play_arrow
question_answer 14) A source of emf \[E=15V\] and having negligible internal resistance, is connected to a variable resistance, so that the current in the circuit increases with time as \[I=1.2t+3\]. Then, the total charge that will flow in first \[5\text{ }s\]will be
A)
\[10\text{ }C\]
done
clear
B)
\[20\text{ }C\]
done
clear
C)
\[30\text{ }C\]
done
clear
D)
\[40\text{ }C\]
done
clear
View Answer play_arrow
question_answer 15) The magnetic induction at the centre of a current carrying circular of radius r, is
A)
directly proportional to r
done
clear
B)
inversely proportional to r
done
clear
C)
directly proportional to \[{{r}^{2}}\]
done
clear
D)
inversely proportional to \[{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 16) A current carrying conductor produces
A)
only electric field
done
clear
B)
only magnetic field
done
clear
C)
both electric and magnetic fields
done
clear
D)
neither electric nor magnetic field
done
clear
View Answer play_arrow
question_answer 17) A proton of energy \[8\text{ }eV\]is moving in a circular path in a uniform magnetic field. The energy of an alpha particle moving in the same magnetic field and along the same path will be
A)
\[4\text{ }eV\]
done
clear
B)
\[2\text{ }eV\]
done
clear
C)
\[8\,\,eV\]
done
clear
D)
\[6\,\,eV\]
done
clear
View Answer play_arrow
question_answer 18) A \[1\text{ }m\]long wire is lying at right angles to the magnetic field. A force of 1 kg wt. is acting on it in a magnetic field of \[0.98\text{ }T\]. The current flowing in it will be
A)
\[100\text{ }A\]
done
clear
B)
\[10\text{ }A\]
done
clear
C)
\[1\text{ }A\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 19) The resultant magnetic moment of neon atom will be
A)
infinity
done
clear
B)
\[{{\mu }_{B}}\]
done
clear
C)
zero
done
clear
D)
\[\frac{{{\mu }_{B}}}{2}\]
done
clear
View Answer play_arrow
question_answer 20) The temperature at which ferromagnetic material becomes paramagnetic is called a
A)
neutral temperature
done
clear
B)
Curie temperature
done
clear
C)
inversion temperature
done
clear
D)
critical temperature
done
clear
View Answer play_arrow
question_answer 21) The ultimate individual unit of magnetism in any magnet is called
A)
north pole
done
clear
B)
south pole
done
clear
C)
dipole
done
clear
D)
quadrupole
done
clear
View Answer play_arrow
question_answer 22) The power loss in AC circuit will be minimum when
A)
resistance is high, inductance is high
done
clear
B)
resistance is high, inductance is low
done
clear
C)
resistance is low, inductance is low
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 23) At high frequency, the capacitor offer
A)
more reactance
done
clear
B)
less reactance
done
clear
C)
zero reactance
done
clear
D)
infintie reactance
done
clear
View Answer play_arrow
question_answer 24) The quantity that. remain unchanged in transformer is
A)
voltage
done
clear
B)
current
done
clear
C)
frequency
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 25) A piece of plane glass is placed on a word with letters of different colours. The letters which appear minimum raised are
A)
red
done
clear
B)
green
done
clear
C)
yellow
done
clear
D)
violet
done
clear
View Answer play_arrow
question_answer 26) When light waves suffer reflection at the interface between air and glass, the change of phase of the reflected wave is equal to
A)
zero
done
clear
B)
\[\frac{\pi }{2}\]
done
clear
C)
\[\pi \]
done
clear
D)
\[2\pi \]
done
clear
View Answer play_arrow
question_answer 27) If the wavelength of light is \[4000\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,,\] then the number of waves in 1 mm length will be
A)
\[25\]
done
clear
B)
\[0.25\]
done
clear
C)
\[0.25\times {{10}^{4}}\]
done
clear
D)
\[25\times {{10}^{4}}\]
done
clear
View Answer play_arrow
question_answer 28) The wave theory of light was given by
A)
Maxwell
done
clear
B)
Planck
done
clear
C)
Huygen
done
clear
D)
Young
done
clear
View Answer play_arrow
question_answer 29) In Young's double slit experiment the amplitudes of two sources are \[3a\]and a respectively. The ratio of intensities of bright and dark fringes will be
A)
\[3:1\]
done
clear
B)
\[4:1\]
done
clear
C)
\[2:1\]
done
clear
D)
\[9:1\]
done
clear
View Answer play_arrow
question_answer 30) The diffraction effect can be observed in
A)
only sound waves
done
clear
B)
only light waves
done
clear
C)
only ultrasonic waves
done
clear
D)
sound as well as light waves
done
clear
View Answer play_arrow
question_answer 31) At what distance from a convex lens of focal length 30 cm, an object should be placed, so that the size of the image be \[\frac{1}{2}th\] of the object?
A)
\[30\,cm\]
done
clear
B)
\[60\,cm\]
done
clear
C)
\[15\,cm\]
done
clear
D)
\[90\,cm\]
done
clear
View Answer play_arrow
question_answer 32) Two coherent sources of intensity ratio \[1:4\] produce an interference pattern. The fringe visibility will be
A)
\[1\]
done
clear
B)
\[0.8\]
done
clear
C)
\[0.4\]
done
clear
D)
\[0.6\]
done
clear
View Answer play_arrow
question_answer 33) The specific charge of an electron is
A)
\[1.6\times {{10}^{-19}}C\]
done
clear
B)
\[4.8\times {{10}^{-19}}stat-C\]
done
clear
C)
\[1.76\times {{10}^{-11}}C/kg\]
done
clear
D)
\[1.76\times {{10}^{11}}C/kg\]
done
clear
View Answer play_arrow
question_answer 34) The colour of the second line of Balmer series is:
A)
blue
done
clear
B)
yellow
done
clear
C)
red
done
clear
D)
violet
done
clear
View Answer play_arrow
question_answer 35) If elements with principal quantum number \[n>4\] were not allowed in nature, the number of possible elements would be
A)
\[60\]
done
clear
B)
\[32\]
done
clear
C)
\[4\]
done
clear
D)
\[64\]
done
clear
View Answer play_arrow
question_answer 36) The energy of incident photons corresponding to maximum wavelength of visible light is
A)
\[3.2\text{ }eV\]
done
clear
B)
\[7\,eV\]
done
clear
C)
\[1.55\text{ }eV\]
done
clear
D)
\[1\,eV\]
done
clear
View Answer play_arrow
question_answer 37) If the work function of potassium is \[2\text{ }eV,\]then its photoelectric threshold wavelength is
A)
\[310\text{ }nm\]
done
clear
B)
\[620\text{ }nm\]
done
clear
C)
\[6200\text{ }nm\]
done
clear
D)
\[3100\text{ }nm\]
done
clear
View Answer play_arrow
question_answer 38) Threshold wavelength for a metal\[5200\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]. The photoeletrons will be ejected, if it is irradiated by light from
A)
\[50\text{ }W\]infrared lamp
done
clear
B)
\[\text{1 }W\] infrared lamp
done
clear
C)
\[\text{50 }W\] ultraviolet lamp
done
clear
D)
\[\text{0}\text{.5 }W\] infrared lamp
done
clear
View Answer play_arrow
question_answer 39) An electron and a proton are accelerated through the same potential difference. The ratio of their de-Broglie wavelength will be
A)
\[{{\left( \frac{{{m}_{p}}}{{{m}_{e}}} \right)}^{1/2}}\]
done
clear
B)
\[\frac{{{m}_{e}}}{{{m}_{p}}}\]
done
clear
C)
\[\frac{{{m}_{p}}}{{{m}_{e}}}\]
done
clear
D)
\[\frac{a}{\sqrt{2}}\]
done
clear
View Answer play_arrow
question_answer 40) A particle with rest mass zero is moving with speed c. The de-Broglie wavelength associated with it
A)
zero
done
clear
B)
infinity
done
clear
C)
\[\frac{hv}{c}\]
done
clear
D)
\[\frac{{{m}_{0}}c}{h}\]
done
clear
View Answer play_arrow
question_answer 41) The nearest distance between two atoms in case of a bcc lattice is equal to
A)
\[\frac{a\sqrt{2}}{3}\]
done
clear
B)
\[\frac{a\sqrt{3}}{2}\]
done
clear
C)
\[a\sqrt{3}\]
done
clear
D)
\[\frac{a}{\sqrt{2}}\]
done
clear
View Answer play_arrow
question_answer 42) Which of the following is an amorphous substance?
A)
Gold
done
clear
B)
Silver
done
clear
C)
Copper
done
clear
D)
Glass
done
clear
View Answer play_arrow
question_answer 43) The maximum efficiency of full wave rectifier is
A)
\[100%\]
done
clear
B)
\[25.20%\]
done
clear
C)
\[40.6%\]
done
clear
D)
\[81.2%\]
done
clear
View Answer play_arrow
question_answer 44) In a npn-transistor, the collector current is\[10\text{ }mA\]. If \[90%\] of the electrons emitted reach the collector, then the emitter current will be
A)
\[9\,\,mA\]
done
clear
B)
\[11\,\,mA\]
done
clear
C)
\[1\,\,mA\]
done
clear
D)
\[0.1\,\,mA\]
done
clear
View Answer play_arrow
question_answer 45)
The given truth table is of A B X 0 0 0 0 1 1 1 0 1 1 1 1
A)
OR gate
done
clear
B)
AND gate
done
clear
C)
NOT gate
done
clear
D)
XOR gate
done
clear
View Answer play_arrow
question_answer 46) If force (F), length (L) and time (T) are assumed to be the fundamental units, then the dimensional formula of the mass will be
A)
\[[F{{L}^{-1}}{{T}^{2}}]\]
done
clear
B)
\[[F{{L}^{-1}}{{T}^{-2}}]\]
done
clear
C)
\[[F{{L}^{-1}}{{T}^{-1}}]\]
done
clear
D)
\[[F{{L}^{2}}{{T}^{2}}]\]
done
clear
View Answer play_arrow
question_answer 47) The horizontal range of a projectile is \[4\sqrt{3}\] times its maximum height. Its angle of projection will be
A)
\[{{45}^{o}}\]
done
clear
B)
\[{{60}^{o}}\]
done
clear
C)
\[{{90}^{o}}\]
done
clear
D)
\[{{30}^{o}}\]
done
clear
View Answer play_arrow
question_answer 48) Two balls of same size but the density of one is greater than that of the other are dropped from the same height, then which ball will reach the earth first (air resistance is negligible)?
A)
Heavy ball
done
clear
B)
Light ball
done
clear
C)
Both simultaneously
done
clear
D)
Will depend upon the density of the balls
done
clear
View Answer play_arrow
question_answer 49) A person moves \[30\text{ }m\]north and then \[20\text{ }m\] towards east and finally 30^2 min south-west direction. The displacement of the person from the origin will be
A)
\[10\text{ }m\]along north
done
clear
B)
\[10\text{ }m\]along south
done
clear
C)
\[10\text{ }m\] along west
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 50) If the velocity of a particle is given by \[v={{(180-16x)}^{1/2}}\,m/s\]. then its acceleration will be
A)
zero
done
clear
B)
\[8\text{ }m/{{s}^{2}}\]
done
clear
C)
\[-8\text{ }m/{{s}^{2}}\]
done
clear
D)
\[4\text{ }m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 51) Neglecting the air resistance, the time of flight of a projectile is determined by
A)
\[{{U}_{vertical}}\]
done
clear
B)
\[{{U}_{horizontal}}\]
done
clear
C)
\[U={{U}_{vertical}}+U_{horizontal}^{2}\]
done
clear
D)
\[U={{(U_{vertical}^{2}+U_{horizontal}^{2})}^{1/2}}\]
done
clear
View Answer play_arrow
question_answer 52) A train is moving towards east and a car is along north, both with same speed. The observed direction of car to the passenger in the train is
A)
east-north direction
done
clear
B)
west-north direction
done
clear
C)
south-east direction
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 53) A body moving with velocity v has momentum and kinetic energy numerically equal. What is the value of v?
A)
\[2\text{ }m/s\]
done
clear
B)
\[\sqrt{2}\text{ }m/s\]
done
clear
C)
\[1\text{ }m/s\]
done
clear
D)
\[0.2\text{ }m/s\]
done
clear
View Answer play_arrow
question_answer 54) If the kinetic energy of a body becomes four times, then its momentum will be
A)
\[{{P}_{new}}=3{{p}_{initial~}}\]
done
clear
B)
\[{{P}_{new}}=4{{p}_{initial~}}\]
done
clear
C)
\[{{P}_{new}}=2{{p}_{initial~}}\]
done
clear
D)
\[{{P}_{new}}={{p}_{initial~}}\]
done
clear
View Answer play_arrow
question_answer 55) A block of mass \[2\text{ }kg\]is lying on an inclined plane, inclined to the horizontal at\[{{30}^{o}}\]. If the coefficient of friction between the block and the plane is \[0.7,\] then magnitude of frictional force acting on the block will be
A)
\[11.9\text{ }N\]
done
clear
B)
\[1.19N\]
done
clear
C)
\[0.19\text{ }N\]
done
clear
D)
\[11.0\text{ }N\]
done
clear
View Answer play_arrow
question_answer 56) A ring of mass m and radius r is melted and then moulded into a sphere. The moment of inertia of the sphere will be
A)
more than that of the ring
done
clear
B)
less than that of the ring
done
clear
C)
equal to that of the ring
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 57) A solid sphere and a hollow sphere of the same material and of a same size can be distinguished without weighing
A)
by determining their moments of inertia about their coaxial axes
done
clear
B)
by rolling them simultaneously on an inclined plane
done
clear
C)
by rotating them about a common axis of rotation
done
clear
D)
by applying equal torque on them
done
clear
View Answer play_arrow
question_answer 58) Point masses \[1,\text{ }2,-3\]and \[4\text{ }kg\]are lying at the point \[(0,0,0)\]\[(2,0,0)\]\[(0,3,0)\] and \[(-2,-2,0)\] respectively. The moment of inertia of this system about x-axis will be
A)
\[43\,kg-{{m}^{2}}\]
done
clear
B)
\[34\,kg-{{m}^{2}}\]
done
clear
C)
\[27\,kg-{{m}^{2}}\]
done
clear
D)
\[72\,kg-{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 59) The radius of gyration of a body about an axis at a distance \[6\text{ }cm\] from its centre of mass is\[10\text{ }cm\]. Then its radius of gyration about a parallel axis through its centre of mass will be
A)
\[80\,\,cm\]
done
clear
B)
\[8\,\,cm\]
done
clear
C)
\[0.8\,\,cm\]
done
clear
D)
\[80\,\,m\]
done
clear
View Answer play_arrow
question_answer 60) Two planets of radii in the ratio \[2:3\]are made from the material of density in the ratio\[3:2\]. Then, the ratio of acceleration due to gravity \[\frac{{{g}_{1}}}{{{g}_{2}}}\] at the surface of the two planets will be
A)
\[1\]
done
clear
B)
\[2.25\]
done
clear
C)
\[4/9\]
done
clear
D)
\[0.12\]
done
clear
View Answer play_arrow
question_answer 61) If the radius of the earth contracts to half of its present day value without change in mass, then the length of the day will be
A)
\[24\text{ }h\]
done
clear
B)
\[48\text{ }h\]
done
clear
C)
\[\text{6 }h\]
done
clear
D)
\[\text{12 }h\]
done
clear
View Answer play_arrow
question_answer 62) A person will get more quantity of matter in kg-wt at
A)
poles
done
clear
B)
at latitude of \[{{60}^{o}}\]
done
clear
C)
equator
done
clear
D)
satellite
done
clear
View Answer play_arrow
question_answer 63) The unit of the coefficient of viscosity in SI system is
A)
\[m/kg-s\]
done
clear
B)
\[~m-s/k{{g}^{2}}\]
done
clear
C)
\[kg/m-{{s}^{2}}\]
done
clear
D)
\[kg/m-s\]
done
clear
View Answer play_arrow
question_answer 64) If the excess pressure inside a soap bubble is balanced by oil column of height \[2\text{ }mm,\]then the surface tension of soap solution will be ( \[r=1\text{ }cm\]and density \[d=0.8\text{ }g/\text{ }cc\])
A)
\[3.9\text{ }N/m\]
done
clear
B)
\[3.9\times {{10}^{-1}}N/m\]
done
clear
C)
\[3.9\times {{10}^{-2}}N/m\]
done
clear
D)
\[3.9\,dyne/m\]
done
clear
View Answer play_arrow
question_answer 65) A vessel, whose bottom has round holes with diameter of \[1\text{ }mm\]is filled with water. Assuming that surface tension acts only at holes. Then, the maximum height to which the water can be filled in vessel without leakage is (surface tension of water is \[75\times {{10}^{-3}}\text{ }N/m\]and \[g=10\text{ }m/{{s}^{2}}\])
A)
\[3\,cm\]
done
clear
B)
\[0.3\,cm\]
done
clear
C)
\[3\text{ }mm\]
done
clear
D)
\[~3\text{ }m\]
done
clear
View Answer play_arrow
question_answer 66) For which of the two pairs, the angle of contact is same?
A)
Water and glass, glass and mercury
done
clear
B)
Pure water and glass, glass and alcohol
done
clear
C)
Silver and water, mercury and glass
done
clear
D)
Silver and chromium, water and chromium
done
clear
View Answer play_arrow
question_answer 67) At what temperature the mis velocity of helium molecules will be equal to that of hydrogen moekules at NTP?
A)
\[844\text{ }K\]
done
clear
B)
\[64\text{ }K\]
done
clear
C)
\[{{273}^{o}}C\]
done
clear
D)
\[273\text{ }K\]
done
clear
View Answer play_arrow
question_answer 68) Which of the following is unique function of initial and final states?
A)
\[dQ\]
done
clear
B)
\[dW\]
done
clear
C)
\[dU\]
done
clear
D)
\[\Delta Q\,\,and\,\,\Delta W\]
done
clear
View Answer play_arrow
question_answer 69) If the initial temperatures of metallic sphere and disc of same radius and nature are equal, then the ratio of their rate of cooling will be
A)
\[1:4\]
done
clear
B)
\[4:1\]
done
clear
C)
\[1:2\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
question_answer 70) What will be the ratio of temperatures of sun and moon, if the wavelengths of their maximum emission radiations rates are \[140\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]and \[4200\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]respectively?
A)
\[1:30\]
done
clear
B)
\[30:1\]
done
clear
C)
\[42:14\]
done
clear
D)
\[14:42\]
done
clear
View Answer play_arrow
question_answer 71) A bar magnet is oscillating in the earth's magnetic field with time period T. If its mass is increased four times, then its time period will be
A)
\[4T\]
done
clear
B)
\[2T\]
done
clear
C)
\[T\]
done
clear
D)
\[r\]
done
clear
View Answer play_arrow
question_answer 72) The time period of a simple pendulum, when it is made to oscillate on the surface of moon
A)
increases
done
clear
B)
decreases
done
clear
C)
remains unchanged
done
clear
D)
becomes infinite
done
clear
View Answer play_arrow
question_answer 73) A condenser of capacity \[20\mu F\] is first charged and then discharged through a \[10\text{ }mH\] inductance. Neglecting the resistance of the coil, the frequency of the resulting vibrations will be
A)
\[356\,cycle/h\]
done
clear
B)
\[356\,cycle/s\]
done
clear
C)
\[356\times {{10}^{3}}\,cycle/s\]
done
clear
D)
\[3.56\,cycle/s\]
done
clear
View Answer play_arrow
question_answer 74) Infinite springs with force constants k, 2k, 4k and 8 k... respectively are connected in series. The effective force constant of the spring will be
A)
\[2k\]
done
clear
B)
\[k\]
done
clear
C)
\[\frac{k}{2}\]
done
clear
D)
\[2048\]
done
clear
View Answer play_arrow
question_answer 75) The intensity of sound gets reduced by 10% on passing through a slab. The reduction in intensity on passage through three consecutive slabs is
A)
\[30%\]
done
clear
B)
\[27.1%\]
done
clear
C)
\[20%\]
done
clear
D)
\[36%\]
done
clear
View Answer play_arrow
question_answer 76) The hydrogen electrode is dipped in a solution of \[pH=3\]at \[25{{\,}^{o}}C.\] The potential of the cell would be (the value of.2.303 RT/F is 0.059 V)
A)
\[~0.177\,V\]
done
clear
B)
\[~0.087\,V\]
done
clear
C)
\[~-0.177\,V\]
done
clear
D)
\[0.059\,V\]
done
clear
View Answer play_arrow
question_answer 77) Specific conductivity of a solution
A)
increases with dilution
done
clear
B)
decreases with dilution
done
clear
C)
remains unchanged with dilution
done
clear
D)
depends on mass of electrolyte
done
clear
View Answer play_arrow
question_answer 78) \[\text{1 mol}\]of \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\]is mixed with 2 moles of \[\text{NaOH}\text{.}\]The heat evolved will be
A)
\[57.3\,kJ\]
done
clear
B)
\[2\times 57.3\,kJ\]
done
clear
C)
\[57.3/2\,kJ\]
done
clear
D)
cannot be predicted
done
clear
View Answer play_arrow
question_answer 79) In a reversible process, \[\Delta {{S}_{system}}+\Delta {{S}_{\text{surrounding}}}\] is
A)
\[~>0\]
done
clear
B)
\[<0\]
done
clear
C)
\[~>0\]
done
clear
D)
\[=0\]
done
clear
View Answer play_arrow
question_answer 80) For the reaction, \[{{N}_{2}}+3{{H}_{2}}2NH;\Delta H=?\]
A)
\[\Delta E=+\,2RT\]
done
clear
B)
\[\Delta E-\,2RT\]
done
clear
C)
\[\Delta E+\,RT\]
done
clear
D)
\[\Delta E-\,RT\]
done
clear
View Answer play_arrow
question_answer 81) One mole of a perfect gas expands isothermally to ten times of its original volume. The change in entropy is
A)
0.1 R
done
clear
B)
2.303 R
done
clear
C)
10.0 R
done
clear
D)
100.0 R
done
clear
View Answer play_arrow
question_answer 82) Which of the following solutions will have the highest boiling point?
A)
\[\text{0}\text{.1 M FeC}{{\text{l}}_{\text{3}}}\]
done
clear
B)
\[\text{0}\text{.1 M BaC}{{\text{l}}_{2}}\]
done
clear
C)
\[\text{0}\text{.1 M NaCl}\]
done
clear
D)
\[\text{0}\text{.1}\,\text{M}\,\text{urea}\]
done
clear
View Answer play_arrow
question_answer 83) Maximum freezing point falls in
A)
camphor
done
clear
B)
naphthalene
done
clear
C)
benzene
done
clear
D)
water
done
clear
View Answer play_arrow
question_answer 84) Azeotropic mixture of \[\text{HCl}\]and water has
A)
\[\text{48 }\!\!%\!\!\text{ HCl}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ 22}\text{.2 }\!\!%\!\!\text{ HCl}\]
done
clear
C)
\[\text{36 }\!\!%\!\!\text{ HCl}\]
done
clear
D)
\[\text{ }\!\!~\!\!\text{ 20}\text{.2 }\!\!%\!\!\text{ HCl}\]
done
clear
View Answer play_arrow
question_answer 85) Vapour pressure of dilute aqueous solution of glucose is 750 mm of mercury at 373 K. The mole fraction of solute is
A)
\[\frac{1}{76}\]
done
clear
B)
\[\frac{1}{7.6}\]
done
clear
C)
\[\frac{1}{38}\]
done
clear
D)
\[\frac{1}{10}\]
done
clear
View Answer play_arrow
question_answer 86) Volume of \[\text{0}\text{.1}\,\text{M}\,{{\text{K}}_{\text{2}}}\text{C}{{\text{r}}_{\text{2}}}{{\text{O}}_{\text{7}}}\] required to oxidize 35 mL of \[\text{0}\text{.5 M FeS}{{\text{O}}_{\text{4}}}\]solution is
A)
29.2 mL
done
clear
B)
17.5 mL
done
clear
C)
175 mL
done
clear
D)
145 Ml
done
clear
View Answer play_arrow
question_answer 87) \[100\,cc\]of \[\text{0}\text{.6}\,\text{N}\,{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\]and \[\text{200 cc}\]of \[\text{0}\text{.3}\,\text{N}\,\text{HCl}\]were mixed together. The normality of the solution will be
A)
0.2 N
done
clear
B)
0.4 N
done
clear
C)
0.8 N
done
clear
D)
0.6 N
done
clear
View Answer play_arrow
question_answer 88) The rate of diffusion of a gas is proportional to
A)
\[\frac{p}{\sqrt{d}}\]
done
clear
B)
\[\sqrt{\frac{p}{d}}\]
done
clear
C)
\[\frac{p}{d}\]
done
clear
D)
\[\frac{\sqrt{p}}{d}\]
done
clear
View Answer play_arrow
question_answer 89) Molar volume of \[\text{C}{{\text{O}}_{\text{2}}}\]is maximum at
A)
NTP
done
clear
B)
\[\text{0}{{\,}^{o}}\text{C}\]and 2.0 atm
done
clear
C)
\[127{{\,}^{o}}C\] and\[\text{ }\!\!~\!\!\text{ 1 atm}\]
done
clear
D)
\[~273{{\,}^{o}}C\] and \[2.0\,\text{atm}\]
done
clear
View Answer play_arrow
question_answer 90) Number of atoms of oxygen present in 10.6 g of \[\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\]will be
A)
\[6.02\times {{10}^{23}}\]
done
clear
B)
\[12.04\times {{10}^{22}}\]
done
clear
C)
\[1.806\times {{10}^{23}}\]
done
clear
D)
\[31.80\times {{10}^{2}}\]
done
clear
View Answer play_arrow
question_answer 91) The equilibrium \[{{P}_{4}}(s)+6C{{l}_{2}}(g)\rightleftharpoons 4PC{{l}_{3}}(g)\]is attained by mixing equal moles of\[{{\text{P}}_{\text{4}}}\]and\[\text{C}{{\text{l}}_{\text{2}}}\]in an evacuated vessel. Then, at equilibrium
A)
\[[C{{l}_{2}}]>[PC{{l}_{3}}]\]
done
clear
B)
\[[C{{l}_{2}}]>[{{P}_{4}}]\]
done
clear
C)
\[[{{P}_{4}}]>[C{{l}_{2}}]\]
done
clear
D)
\[[PC{{l}_{3}}]>[{{P}_{4}}]\]
done
clear
View Answer play_arrow
question_answer 92) The activation energy for most of the reactions is approximately \[50\,\text{kJ}\,\text{mo}{{\text{l}}^{-1}}.\] The value of temperature coefficient for such reactions is
A)
\[~>2\]
done
clear
B)
\[~>3\]
done
clear
C)
\[~<\,\,1\]
done
clear
D)
\[~>4\]
done
clear
View Answer play_arrow
question_answer 93) If the mass defect of \[_{\text{4}}^{\text{9}}\text{X}\]is\[\text{0}\text{.090 u,}\] then binding energy per nucleon is \[\text{(1}\,\text{u}\,\text{=}\,\text{931}\text{.5}\,\text{MeV)}\]
A)
\[\text{9}\text{.315 MeV}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ 931}\text{.5 MeV}\]
done
clear
C)
\[\text{ }\!\!~\!\!\text{ 83}\text{.0 MeV}\]
done
clear
D)
\[\text{ }\!\!~\!\!\text{ 8}\text{.38 MeV}\]
done
clear
View Answer play_arrow
question_answer 94) \[\text{50 mL}\]of\[\text{0}\text{.1}\,\text{M}\,\text{HCl}\] and 50 mL of 0.2 M \[\text{NaOH}\] are mixed. The pH of the resulting solution is
A)
1.30
done
clear
B)
4.2
done
clear
C)
12.70
done
clear
D)
11.70
done
clear
View Answer play_arrow
question_answer 95) A substance \[{{A}_{x}}{{B}_{y}}\]crystallises in a face centred cubic lattice in which A atom occupies each comer of cube and atom B occupies the centres of each face of the cube. Identify the correct composition of the substance \[{{A}_{x}}{{B}_{y}}.\]
A)
\[A{{B}_{3}}\]
done
clear
B)
\[{{A}_{4}}{{B}_{3}}\]
done
clear
C)
\[{{A}_{3}}B\]
done
clear
D)
composition cannot be specified
done
clear
View Answer play_arrow
question_answer 96) In coagulating the colloidal solution of \[\text{A}{{\text{s}}_{\text{2}}}{{\text{S}}_{\text{3}}}\]which has the maximum coagulating value?
A)
\[\text{NaCl}\]
done
clear
B)
\[KCl\]
done
clear
C)
\[BaC{{l}_{2}}\]
done
clear
D)
\[AlC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 97) Which of the following is the strongest oxidising agent?
A)
\[HOCl\]
done
clear
B)
\[HCl{{O}_{2}}\]
done
clear
C)
\[HCl{{O}_{3}}\]
done
clear
D)
\[HCl{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 98) In the equation \[4M+8C{{N}^{-}}+2{{H}_{2}}O+{{O}_{2}}\xrightarrow{{}}4{{[M{{(CN)}_{2}}]}^{-}}\] \[+\,4\,O{{H}^{-}}\] Identify the metal M.
A)
copper
done
clear
B)
iron
done
clear
C)
silver
done
clear
D)
zinc
done
clear
View Answer play_arrow
question_answer 99) The formula of azurite is
A)
\[CuC{{O}_{3}}.Cu{{(OH)}_{2}}\]
done
clear
B)
\[2CuC{{O}_{3}}.Cu{{(OH)}_{2}}\]
done
clear
C)
\[CuC{{O}_{3}}.2Cu{{(OH)}_{2}}\]
done
clear
D)
\[CuS{{O}_{4}}.Cu{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 100) The decreasing order of bond angle is
A)
\[N{{O}_{2}}>NO_{2}^{+}>NO_{2}^{-}\]
done
clear
B)
\[NO_{2}^{-}>N{{O}_{2}}>NO_{2}^{+}\]
done
clear
C)
\[NO_{2}^{+}>N{{O}_{2}}>NKO_{2}^{-}\]
done
clear
D)
\[NO_{2}^{+}>NO_{2}^{-}>N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 101) The fresh precipitate can be transformed in colloidal state by
A)
peptization
done
clear
B)
coagulation
done
clear
C)
diffusion
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 102) Milk is
A)
fat disperse4 in water
done
clear
B)
fat dispersed in milk
done
clear
C)
fat dispersed in fat
done
clear
D)
water dispersed in milk
done
clear
View Answer play_arrow
question_answer 103) Purest form of iron is
A)
cast iron
done
clear
B)
pig iron
done
clear
C)
wrought iron
done
clear
D)
steel
done
clear
View Answer play_arrow
question_answer 104) Most unstable hydride is
A)
\[~N{{H}_{3}}\]
done
clear
B)
\[~P{{H}_{3}}\]
done
clear
C)
\[~As{{H}_{3}}\]
done
clear
D)
\[~Bi{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 105) Out of the following metals that cannot be obtained by electrolysis of the aqueous solution of its salts is
A)
Ag
done
clear
B)
Cr
done
clear
C)
Cu
done
clear
D)
Mg
done
clear
View Answer play_arrow
question_answer 106) \[\text{KI}\]and \[\text{CuS}{{\text{O}}_{\text{4}}}\]solution when mixed gives
A)
\[Cu{{l}_{2}}+{{K}_{2}}S{{O}_{4}}\]
done
clear
B)
\[C{{u}_{2}}{{I}_{2}}+{{K}_{2}}S{{O}_{4}}\]
done
clear
C)
\[{{K}_{2}}S{{O}_{4}}+C{{u}_{2}}{{I}_{2}}+{{I}_{2}}\]
done
clear
D)
\[{{K}_{2}}S{{O}_{4}}+Cu{{I}_{2}}+{{I}_{2}}\]
done
clear
View Answer play_arrow
question_answer 107) The strongest reducing agent among the following is
A)
\[{{F}^{-}}\]
done
clear
B)
\[C{{l}^{-}}\]
done
clear
C)
\[B{{r}^{-}}\]
done
clear
D)
\[{{I}^{-}}\]
done
clear
View Answer play_arrow
question_answer 108) \[\text{Xe}{{\text{F}}_{\text{6}}}\]on complete hydrolysis gives
A)
\[\text{Xe}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ Xe}{{\text{O}}_{\text{2}}}\]
done
clear
C)
\[Xe{{O}_{3}}\]
done
clear
D)
\[\text{ }\!\!~\!\!\text{ Xe}{{\text{O}}_{\text{4}}}\]
done
clear
View Answer play_arrow
question_answer 109) The correct name of the compound\[[Cu{{(N{{H}_{3}})}_{4}}]{{(N{{O}_{3}})}_{2}},\]according to IUPAC system is
A)
cuprammonium nitrate
done
clear
B)
tetrammine copper(II) dinitrate
done
clear
C)
tetrammine copper(II) nitrate
done
clear
D)
tetrammine copper(II) dinitrite
done
clear
View Answer play_arrow
question_answer 110) Which of the following complex species does not involve inner orbital hybridisation?
A)
\[{{[Co{{F}_{6}}]}^{3-}}\]
done
clear
B)
\[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
C)
\[{{[Fe{{(CN)}_{6}}]}^{3-}}\]
done
clear
D)
\[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 111) \[{{\,}_{\text{27}}}\text{C}{{\text{o}}^{\text{60}}}\]is radioactive because
A)
its atomic number is high
done
clear
B)
it has high \[\frac{p}{n}\]ratio
done
clear
C)
it has high\[\frac{n}{p}\]ratio
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 112) The correct order of solubility of the sulphates of alkaline earth metals in water is
A)
\[~Be>Ca>Mg>Ba>Sr\]
done
clear
B)
\[~Mg>Be>Ba>Ca>Sr\]
done
clear
C)
\[Be>Mg>Ca>Sr>Ba\]
done
clear
D)
\[Mg>Ca>Ba>Be>Sr\]
done
clear
View Answer play_arrow
question_answer 113) Correct order of radii is
A)
\[N<Be<B\]
done
clear
B)
\[{{F}^{-}}<{{O}^{2-}}<{{N}^{3-}}\]
done
clear
C)
\[Na<Li<K\]
done
clear
D)
\[F{{e}^{3+}}<F{{e}^{2+}}<F{{e}^{4+}}\]
done
clear
View Answer play_arrow
question_answer 114) A sudden large jump between the values of first and second ionisation energies of elements would be associated with which of the following electronic configurations?
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{1}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{1}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{1}}3{{p}^{2}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 115) Which one shows most pronounced inert pair effect?
A)
\[\text{Si}\]
done
clear
B)
\[Sn\]
done
clear
C)
\[Pb\]
done
clear
D)
C
done
clear
View Answer play_arrow
question_answer 116) Which of the following will form a colourless complex?
A)
\[N{{i}^{2+}}\]
done
clear
B)
\[C{{u}^{+}}\]
done
clear
C)
\[T{{i}^{2+}}\]
done
clear
D)
\[F{{e}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 117) Silver containing lead as impurity is purified by
A)
poling
done
clear
B)
cupellation
done
clear
C)
lavigation
done
clear
D)
distillation
done
clear
View Answer play_arrow
question_answer 118) The metal extracted by cyanide process is
A)
silver
done
clear
B)
copper
done
clear
C)
iron
done
clear
D)
sodium
done
clear
View Answer play_arrow
question_answer 119) On the extraction of iron, the slag produced is
A)
\[CO\]
done
clear
B)
\[FeSi{{O}_{3}}\]
done
clear
C)
\[MgSi{{O}_{3}}\]
done
clear
D)
\[Casi{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 120) Complex forming tendency is more for
A)
\[N{{a}^{+}}\]
done
clear
B)
\[{{K}^{+}}\]
done
clear
C)
\[L{{i}^{+}}\]
done
clear
D)
\[R{{b}^{+}}\]
done
clear
View Answer play_arrow
question_answer 121)
In the reaction,
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 122)
The IUPAC name of the following compound is
A)
propionic anhydride
done
clear
B)
dipropanoic anhydride
done
clear
C)
ethoxy propanoic acid
done
clear
D)
propanoic anhydride
done
clear
View Answer play_arrow
question_answer 123) Which of the following compounds is not aromatic?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 124) Which of the following is the most stable cation?
A)
\[{{F}_{3}}C-CH_{2}^{\oplus }\]
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C{{H}^{\oplus }}\]
done
clear
C)
\[CH_{3}^{\oplus }\]
done
clear
D)
\[CF_{3}^{\oplus }\]
done
clear
View Answer play_arrow
question_answer 125)
The product A is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 126) Tautomerism is not exhibited by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 127)
In the compound
A)
\[S,S\]
done
clear
B)
\[R,S\]
done
clear
C)
\[S,R\]
done
clear
D)
\[R,R\]
done
clear
View Answer play_arrow
question_answer 128)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 129) Hinsberg reagent is
A)
\[{{C}_{6}}{{H}_{S}}S{{O}_{3}}H\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}NO\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}S{{O}_{2}}Cl\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}{{N}_{2}}Cl\]
done
clear
View Answer play_arrow
question_answer 130) Acetaldehyde cannot show
A)
lodoform test
done
clear
B)
Lucas test
done
clear
C)
Benedict's test
done
clear
D)
Toilers test
done
clear
View Answer play_arrow
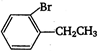
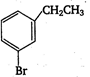
question_answer 131) Ethylbenzene with bromine in presence of \[\text{FeB}{{\text{r}}_{\text{3}}}\text{,}\]predominantly gives
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 132) Which of the following will be most readily dehydrated under acidic conditions?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 133) Which of the following cannot reduce Fehling solution?
A)
\[\text{HCOOH}\]
done
clear
B)
\[\text{ }\!\!~\!\!\text{ }{{\text{H}}_{\text{3}}}\text{CCOOH}\]
done
clear
C)
\[\text{HCHO}\]
done
clear
D)
\[~{{H}_{3}}CCHO\]
done
clear
View Answer play_arrow
question_answer 134) Absolute alcohol is prepared by
A)
vacuum distillation
done
clear
B)
azeotropic distillation
done
clear
C)
steam distillation
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 135) Which of the following compounds is resistant to nucleophilic attack by hydroxyl ion?
A)
Methylacetate
done
clear
B)
Acetonitrile
done
clear
C)
Acetamide
done
clear
D)
Diethyl ether
done
clear
View Answer play_arrow
question_answer 136) Hydrogenation of benzoyl chloride in presence of Pd on\[\text{BaS}{{\text{O}}_{\text{4}}}\]gives
A)
benzyl alcohol
done
clear
B)
benzaldehyde
done
clear
C)
benzoicacid
done
clear
D)
phenol
done
clear
View Answer play_arrow
question_answer 137) \[Ph-C\equiv C-C{{H}_{3}}\xrightarrow{H{{g}^{2+}}/{{H}^{+}}}A\]The product A is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 138) Ethyl amine reacts with nitrous acid to form
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH,{{N}_{2}},{{H}_{2}}O\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}N_{2}^{+}C{{l}^{-}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}NHOH,N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 139) Rice is deficient in
A)
lysine
done
clear
B)
alanine
done
clear
C)
glycine
done
clear
D)
leucine
done
clear
View Answer play_arrow
question_answer 140) Mutarotation does not occur in
A)
sucrose
done
clear
B)
D-glucose
done
clear
C)
L-glucose
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 141) Aldehyde which is formed during photo synthesis of plants is
A)
methanol
done
clear
B)
acetaldehyde
done
clear
C)
propanal
done
clear
D)
phenylmethanal
done
clear
View Answer play_arrow
question_answer 142)
Coupling of diazonium salts of following takes place in the order
A)
\[IV<II<III<I\]
done
clear
B)
\[IV>III<II<I\]
done
clear
C)
\[II<IV<I<III\]
done
clear
D)
\[I<II<III<IV\]
done
clear
View Answer play_arrow
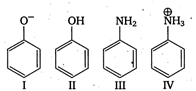
question_answer 143) Which is decreasing order of strength of bases? \[\bar{O}H,\bar{N}{{H}_{2}},HC\equiv {{C}^{-}}\]and \[C{{H}_{3}}CH_{2}^{-}\]
A)
\[{{H}_{3}}CCH_{2}^{-}>NH_{2}^{-}>HC\equiv {{C}^{-}}>O{{H}^{-}}\]
done
clear
B)
\[HC\equiv {{C}^{-}}>C{{H}_{3}}CH_{2}^{-}>NH_{2}^{-}>O{{H}^{-}}\]
done
clear
C)
\[O{{H}^{-}}>NH_{2}^{-}>C{{H}^{-}}>{{H}_{3}}CCH_{2}^{-}\]
done
clear
D)
\[NH_{2}^{-}>HC\equiv {{C}^{-}}>O{{H}^{-}}>{{H}_{3}}CCH_{2}^{-}\]
done
clear
View Answer play_arrow
question_answer 144) The reagent that reacts with nitromethane to form methyl hydroxylamine is
A)
\[Zn/HCl\]
done
clear
B)
\[Zn/N{{H}_{4}}Cl\]
done
clear
C)
\[Zn/NaOH\]
done
clear
D)
\[Sn/HCl\]
done
clear
View Answer play_arrow
question_answer 145) Which is most basic?
A)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
B)
\[({{C}_{6}}{{H}_{5}}N{{H}_{2}})\]
done
clear
C)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}}NH\]
done
clear
View Answer play_arrow
question_answer 146) For d-electron, the orbital angular momentum is
A)
\[\frac{\sqrt{6}h}{2\pi }\]
done
clear
B)
\[\frac{\sqrt{2}h}{2\pi }\]
done
clear
C)
\[\frac{h}{2\pi }\]
done
clear
D)
\[\frac{2h}{\pi }\]
done
clear
View Answer play_arrow
question_answer 147) Two nodal planes are present in
A)
\[{{\pi }^{*}}2{{p}_{x}}\]
done
clear
B)
\[\sigma 2{{p}_{z}}\]
done
clear
C)
\[\pi 2{{p}_{x}}\]
done
clear
D)
\[\pi 2{{p}_{y}}\]
done
clear
View Answer play_arrow
question_answer 148) One gram mole of a gas at NTP occupies 22.4 L. This fact was derived from
A)
law of gaseous volumes
done
clear
B)
Avogadro's hypothesis
done
clear
C)
Berzelius hypothesis
done
clear
D)
Dalton's atomic theory
done
clear
View Answer play_arrow
question_answer 149) At\[\text{90}{{\,}^{\text{o}}}\text{C,}\] pure water has\[[{{H}_{3}}{{O}^{+}}]={{10}^{-6}}\,mol/L.\] The value of \[{{K}_{w}}\]at \[\text{90}{{\,}^{\text{o}}}\text{C}\]is
A)
\[{{10}^{-6}}\]
done
clear
B)
\[{{10}^{-8}}\]
done
clear
C)
\[{{10}^{-12}}\]
done
clear
D)
\[{{10}^{-14}}\]
done
clear
View Answer play_arrow
question_answer 150) The pH of a buffer solution of \[\text{0}\text{.1}\,\text{M}\,\text{N}{{\text{H}}_{\text{4}}}\text{OH}\]\[\text{ }\!\![\!\!\text{ p}{{\text{K}}_{b}}\text{=5}\text{.0 }\!\!]\!\!\text{ }\]and \[\text{0}\text{.01}\,\text{M}\,\text{N}{{\text{H}}_{\text{4}}}\text{Cl}\]is
A)
1
done
clear
B)
4
done
clear
C)
10
done
clear
D)
13
done
clear
View Answer play_arrow
question_answer 151) If a point \[(a{{t}^{2}},\,\,2at)\] be the extremity of a focal Chord of parabola \[{{y}^{2}}=4ax,\] then the length of the focal chord is
A)
\[a{{\left( t+\frac{1}{t} \right)}^{2}}\]
done
clear
B)
\[a{{\left( t+\frac{2}{t} \right)}^{3}}\]
done
clear
C)
\[a{{\left( t+\frac{1}{t} \right)}^{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 152) In a test, an examines either guesses or copies or knows the answer to a multiple choice questions with four choices. The probability that he makes a guess; is \[\frac{1}{3}\] and the probability that he copies the answer is \[\frac{1}{6}\].The probability that his answer is correct given that he copied it is\[\frac{1}{8}\]. The probability that his answer is correct, given that he guessed it is\[\frac{1}{4}\]. The probability that they knew the answer to the questions given that he correctly answered is
A)
\[\frac{24}{31}\]
done
clear
B)
\[\frac{31}{24}\]
done
clear
C)
\[\frac{24}{29}\]
done
clear
D)
\[\frac{29}{24}\]
done
clear
View Answer play_arrow
question_answer 153) Three of the six vertices of a regular hexagon are chosen at random. The probability that the triangle with three vertices is equilateral is equal to
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{1}{5}\]
done
clear
C)
\[\frac{1}{10}\]
done
clear
D)
\[\frac{1}{20}\]
done
clear
View Answer play_arrow
question_answer 154) The value of \[\underset{n\to \infty }{\mathop{\lim }}\,\,\cos \left( \frac{x}{2} \right)\cos \left( \frac{x}{4} \right)\,\cos \left( \frac{x}{8} \right).....\,\cos \left( \frac{x}{{{2}^{n}}} \right)\]Is
A)
\[\frac{x}{\sin \,x}\]
done
clear
B)
\[\frac{x}{\cos \,x}\]
done
clear
C)
\[\frac{(\sin x)}{x}\]
done
clear
D)
\[\frac{(cosx)}{x}\]
done
clear
View Answer play_arrow
question_answer 155) The order and degree of the differential equation \[y=\frac{dy}{dx}x+\sqrt{{{a}^{2}}{{\left( \frac{dy}{dx} \right)}^{2}}+{{b}^{2}}}\] is
A)
\[3,1\]
done
clear
B)
\[1,2\]
done
clear
C)
\[2,1\]
done
clear
D)
\[1,3\]
done
clear
View Answer play_arrow
question_answer 156) \[y=a{{e}^{mx}}+b{{e}^{-mx}}\] satisfies which of the following differential equations
A)
\[\frac{dy}{dx}-my=0\]
done
clear
B)
\[\frac{dy}{dx}+my=0\]
done
clear
C)
\[\frac{{{d}^{2}}y}{d{{x}^{2}}}-{{m}^{2}}y=0\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 157) If \[f(x)={{\left( \frac{x}{1-|x|} \right)}^{1/2002}},\] then \[{{D}_{f}}\] is
A)
\[R-[-1,\,1]\]
done
clear
B)
\[\{-\infty ,1\}\]
done
clear
C)
\[\{-\infty ,-1\}\cup (0,1)\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 158) If \[\vec{a},\vec{b},\vec{c}\] are the unit vectors such that \[\vec{a}\] is perpendicular to the plane \[\vec{b},\vec{c}\] and the angle between \[\vec{b},\vec{c}\] is \[\frac{\pi }{3},\] then \[|\vec{a}+\vec{b}+\vec{c}|\]is equal to
A)
\[0\]
done
clear
B)
\[\pm 1\]
done
clear
C)
\[\pm \,2\]
done
clear
D)
\[\pm \,3\]
done
clear
View Answer play_arrow
question_answer 159) \[\int_{\alpha }^{\beta }{\sqrt{\frac{x-\alpha }{\beta -x}}}\,dx\] is equal to
A)
\[\frac{\pi }{2}(\alpha -\beta )\]
done
clear
B)
\[\frac{\pi }{2}(\beta -\alpha )\]
done
clear
C)
\[\pi (\alpha -\beta )\]
done
clear
D)
\[\pi (\beta -\alpha )\]
done
clear
View Answer play_arrow
question_answer 160) If \[{{a}_{1}},{{a}_{2}}{{a}_{3}},{{a}_{4}},{{a}_{5}},{{a}_{6}}\] are in, AP with common difference \[d\ne 0,\] .then the system of equations \[{{a}_{1}}x+{{a}_{2}}y={{a}_{3}},{{a}_{4}}x+{{a}_{5}}y={{a}_{6}}\]has
A)
infinite number of solutions
done
clear
B)
unique solution
done
clear
C)
no solution
done
clear
D)
cannot say anything
done
clear
View Answer play_arrow
question_answer 161) If the value of \[{{\int_{0}^{\pi }{\left( \frac{x}{1+\sin \,x} \right)}}^{2}}dx=1,\] then the value of the integral \[\int_{0}^{\pi }{\left[ \frac{2{{x}^{2}}\,{{\cos }^{2}}\,x/2}{{{(1+\sin \,x)}^{2}}} \right]}dx\] is equal to
A)
\[1+\pi (2-\pi )\]
done
clear
B)
\[1-\pi (\pi -2)\]
done
clear
C)
\[1+\pi (2+\pi )\]
done
clear
D)
\[1-\pi (2+\pi )\]
done
clear
View Answer play_arrow
question_answer 162) \[\int_{-1}^{1}{(x-[x])\,dx}\] is equal to
A)
\[0\]
done
clear
B)
\[1\]
done
clear
C)
\[2\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 163) If the normal at ?t? on the parabola \[{{y}^{2}}=4ax\] meets the parabola again at t', then
A)
\[t'=-t-\frac{2}{t}\]
done
clear
B)
\[t'=-t+\frac{2}{t}\]
done
clear
C)
\[t'=-t'-\frac{2}{t'}\]
done
clear
D)
\[t'+t'+2tt'\]
done
clear
View Answer play_arrow
question_answer 164) The complex numbers having positive argument and satisfying \[|2-3i|\,\,<\] is
A)
\[\frac{12}{5}+\frac{16}{5}\,i\]
done
clear
B)
\[\frac{4}{5}+\frac{6}{5}\,i\]
done
clear
C)
\[\frac{6}{5}-\frac{5}{2}\,i\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 165) The value of \[\int_{0}^{\pi /2}{\log \,\,\tan \,x\,\,dx}\] is
A)
\[\frac{\pi }{8}\]
done
clear
B)
\[\infty \]
done
clear
C)
\[1\]
done
clear
D)
\[0\]
done
clear
View Answer play_arrow
question_answer 166) Given that. \[\tan \,\,A\] and \[\tan \,\,B\] are the roots of \[{{x}^{2}}-px+q=0,\] then the value of \[{{\sin }^{2}}(A+B)\]is
A)
\[\frac{{{p}^{2}}}{\{{{p}^{2}}+{{(1-q)}^{2}}\}}\]
done
clear
B)
\[\frac{{{q}^{2}}}{({{p}^{2}}+{{q}^{2}})}\]
done
clear
C)
\[\frac{{{q}^{2}}}{\{{{p}^{2}}-(1-{{q}^{2}})\}}\]
done
clear
D)
\[\frac{{{p}^{2}}}{({{p}^{2}}+{{q}^{2}})}\]
done
clear
View Answer play_arrow
question_answer 167) The least positive non-integral solution of \[\sin \,\,\pi ({{x}^{2}}+x)-\sin \,\pi {{x}^{2}}=0\]is
A)
rational
done
clear
B)
irrational of the from \[\sqrt{P}\]
done
clear
C)
irrational of the from \[\frac{\sqrt{P}-1}{4}\]when p is an odd integer
done
clear
D)
irrational of the from \[\frac{\sqrt{P}+1}{4}\] where p is an even integer
done
clear
View Answer play_arrow
question_answer 168) The general solution of \[\frac{dy}{dx}=\frac{2x-y}{x+2y}\] is
A)
\[{{x}^{2}}-xy+{{y}^{2}}=c\]
done
clear
B)
\[{{x}^{2}}-xy-{{y}^{2}}=c\]
done
clear
C)
\[{{x}^{2}}+xy-{{y}^{2}}=c\]
done
clear
D)
\[{{x}^{2}}+x{{y}^{2}}=c\]
done
clear
View Answer play_arrow
question_answer 169) A function \[y=f(x)\] has second order derivative \[f''(x)=6(x-1)\]. If its graph passes through the point \[(2,1)\] and at that point the tangent to the graph is \[y=3x-5,\] then the function is
A)
\[{{(x-1)}^{2}}\]
done
clear
B)
\[{{(x-1)}^{3}}\]
done
clear
C)
\[{{(x+1)}^{3}}\]
done
clear
D)
\[{{(x+1)}^{2}}\]
done
clear
View Answer play_arrow
question_answer 170) If \[f(x)\] is a function satisfying \[f\left( \frac{1}{x} \right)+{{x}^{2}}f(x)=0\]for all non-zero x, then \[\int_{\sin \,\theta }^{\text{cosec }\theta }{f(x)\,\,dx}\]is equal to
A)
\[0\]
done
clear
B)
\[1\]
done
clear
C)
\[2\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 171) The area bounded by the curves \[{{x}^{2}}+{{y}^{2}}=25,\] \[4y=|4-{{x}^{2}}|\] and \[x=0\] above the x- axis is
A)
\[24\,\,{{\sin }^{-1}}\left( \frac{4}{5} \right)\]
done
clear
B)
\[25\,\,{{\sin }^{-1}}\left( \frac{4}{5} \right)\]
done
clear
C)
\[2+\frac{25}{2}{{\sin }^{-1}}\left( \frac{4}{5} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 172) The curve in which the sub tangent is always bisected at the origin is
A)
A parabola
done
clear
B)
A circle
done
clear
C)
A hyperbola
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 173) The value of \[\int_{0}^{\pi /2}{{{\sin }^{8}}\,x\,\,dx}\] is
A)
\[\frac{105\,\pi }{32(4!)}\]
done
clear
B)
\[\frac{105\,\pi }{14\,(4!)}\]
done
clear
C)
\[\frac{105\,}{16\pi \,(4!)}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 174) A particle is moving in a straight line such that the distance described ?s' and the time taken ?t? are given by \[t=a{{s}^{2}}+bs+c,\,\,\,a>0.\]If v is the velocity of the particle at any time t, then acceleration is
A)
\[-2av\]
done
clear
B)
\[-2av\]
done
clear
C)
\[-2av\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 175) If the vectors \[\vec{a}=(c\,\,{{\log }_{2}}\,x)\,\hat{i}-6\hat{j}+3\hat{k}\] and \[\vec{b}=({{\log }_{2}}x)\hat{i}+2\hat{j}+(2c\,{{\log }_{2}}\,x)\hat{k}\] make an obtuse angle for any \[x\,\in \,(0,\infty ),\] then the interval of which 'c? belongs
A)
\[\left( \frac{4}{3},0 \right)\]
done
clear
B)
\[\left( -\frac{4}{3},0 \right)\]
done
clear
C)
\[\left( \frac{3}{4},0 \right)\]
done
clear
D)
\[\left( -\frac{3}{4},0 \right)\]
done
clear
View Answer play_arrow
question_answer 176) Two forces of magnitude 5 and 3 units acting in the directions \[6\hat{i}+2\hat{j}+3\hat{k}\]and \[3\hat{i}-2\hat{j}+6\hat{k}\] respectively act on a particle which is displaced from the point \[(2,2,\,-1)\] to \[(4,3,1)\]. The work done by the forces is
A)
\[\frac{149}{7}\]
done
clear
B)
\[\frac{148}{6}\]
done
clear
C)
\[\frac{148}{7}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 177) The image of the point \[P(1,3,4)\] in the plane \[2x-y+z+3=0\] is
A)
\[(3,\,5,\,-2)\]
done
clear
B)
\[(-3,\,5,2)\]
done
clear
C)
\[(3,-\,5,2)\]
done
clear
D)
\[(3,\,5,2)\]
done
clear
View Answer play_arrow
question_answer 178) If E and F are two events with \[P(E)\le P(F)>0,\]then
A)
occurrence of \[E\Rightarrow \]occurrence of F
done
clear
B)
Occurrence of \[F\Rightarrow \]occurrence of£
done
clear
C)
Non-occurrence of \[E\Rightarrow \]non-occurrence of F
done
clear
D)
None of the above implications hold
done
clear
View Answer play_arrow
question_answer 179) If the normal to the parabola \[{{x}^{2}}=-4ay\] at the points \[({{x}_{1}},\,{{y}_{1}}),\,({{x}_{2}},{{y}_{2}})\] and \[({{x}_{3}},{{y}_{3}})\] are concurrent, then
A)
\[{{y}_{1}}+{{y}_{2}}+{{y}_{3}}=0\]
done
clear
B)
\[{{x}_{1}}{{x}_{2}}+{{x}_{2}}{{x}_{2}}+{{x}_{3}}{{x}_{1}}=0\]
done
clear
C)
\[{{x}_{1}}+{{x}_{2}}+{{x}_{3}}=0\]
done
clear
D)
\[{{x}_{1}}{{y}_{1}}+{{x}_{2}}{{y}_{2}}+{{x}_{3}}{{y}_{3}}=0\]
done
clear
View Answer play_arrow
question_answer 180) If the sum of n terms of the series \[1+\frac{4}{5}+\frac{7}{{{5}^{2}}}+\frac{10}{{{5}^{3}}}+.....\] is \[l+\frac{15}{16}\left( 1-\frac{1}{{{5}^{n-1}}} \right)-\frac{(3n-2)}{4({{5}^{n-1}})},\] then is
A)
\[\frac{4}{5}\]
done
clear
B)
\[\frac{5}{4}\]
done
clear
C)
\[\frac{6}{5}\]
done
clear
D)
\[\frac{5}{6}\]
done
clear
View Answer play_arrow
question_answer 181) It the sides of a triangle are in GP and the largest angle is twice the smallest angle, then the common ratio which is greater than one lies in the interval
A)
\[(1,\,\sqrt{3})\]
done
clear
B)
\[(1,\,\sqrt{2})\]
done
clear
C)
\[\left( 1,\frac{\sqrt{5}+1}{2} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 182) If \[f(x)\] is a differentiable function, then \[\underset{x\to a}{\mathop{\lim }}\,\frac{af\,(x)-x\,f(a)}{x-a}\] is equal to
A)
\[af'\,(a)-f(a)\]
done
clear
B)
\[af\,\,(a)+f'(a)\]
done
clear
C)
\[af'\,(a)+f(a)\]
done
clear
D)
\[af\,(a)-f'(a)\]
done
clear
View Answer play_arrow
question_answer 183) The locus represented by the equation \[|z-1|=|z-i|\] is
A)
a circle of radius 1
done
clear
B)
an ellipse with foci at 1 and \[-i\]
done
clear
C)
a line through the origin
done
clear
D)
a circle on the line joining 1 and \[-i\] as diameter
done
clear
View Answer play_arrow
question_answer 184) The rank of the matrix \[\left| \begin{matrix} 4 & 2 & (1-x) \\ 5 & k & 1 \\ 6 & 3 & (1+x) \\ \end{matrix} \right|\] is 2, then
A)
\[k=\frac{5}{2},\,x=\frac{1}{5}\]
done
clear
B)
\[k=\frac{5}{2},\,x\ne \frac{1}{5}\]
done
clear
C)
\[k=\frac{1}{5},\,x=\frac{5}{2}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 185) The largest interval lying in \[\left( -\frac{\pi }{2},\frac{\pi }{2} \right)\] n in which the function \[f(x)={{3}^{-{{x}^{2}}}}+{{\cos }^{-1}}\left( \frac{x}{2}-1 \right)+\log \,\,\cos \,x\]is defined as
A)
\[[0,\pi ]\]
done
clear
B)
\[\left[ -\frac{\pi }{2},\frac{\pi }{2} \right]\]
done
clear
C)
\[\left[ 0,\frac{\pi }{2} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 186) If a and B are two fixed points, then the locus of a point which moves in such a way that the angle APB is a right angle is
A)
a circle
done
clear
B)
an ellipse
done
clear
C)
a parabola
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 187) Equation of the circle which of the mirror image of the circle \[{{x}^{2}}+{{y}^{2}}-2x=0\]in the line \[x+y=2\]is
A)
\[{{x}^{2}}+{{y}^{2}}-2x+4y+3=0\]
done
clear
B)
\[2({{x}^{2}}+{{y}^{2}})+x+y+1=0\]
done
clear
C)
\[{{x}^{2}}+{{y}^{2}}-4x-2y+4=0\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 188) Let \[y=p+q\]where p varies directly as x and q varies inversely as\[{{x}^{2}}\]. If \[y=19\]when \[x=2\] or 3, then y in terms of x is
A)
\[36x+\frac{5}{{{x}^{2}}}\]
done
clear
B)
\[\frac{5}{x}\,36{{x}^{2}}\]
done
clear
C)
\[5x+\frac{36}{{{x}^{2}}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 189) Seven digits from the digits 1, 2, 3, 4, 5, 6, 7, 8, 9 are written in a random order. The probability that these seven digit number is divisible by 9, is
A)
\[\frac{2}{9}\]
done
clear
B)
\[\frac{1}{5}\]
done
clear
C)
\[\frac{1}{3}\]
done
clear
D)
\[\frac{1}{9}\]
done
clear
View Answer play_arrow
question_answer 190) The orthocenter of a triangle formed by the lines \[x+y=1,\,\,2x+3y=6\]and \[4x-y+4=0\] lies in the
A)
Ist quadrant
done
clear
B)
IInd quadrant
done
clear
C)
IIIrd quadrant
done
clear
D)
IVth quadrant
done
clear
View Answer play_arrow
question_answer 191) If a circle and a parabola intersect at four points, then the y am of the ordinates is equal to
A)
AM of radius is of the circle and latusrectum of the par abola
done
clear
B)
GM of the radius and latusrectum
done
clear
C)
HM of the radius and latusrectum
done
clear
D)
Zero
done
clear
View Answer play_arrow
question_answer 192) In \[\Delta \,ABC\] if \[a=3,\,\,b=4,\,c=5,\] then the value of sin 2B s
A)
\[\frac{4}{5}\]
done
clear
B)
\[\frac{3}{20}\]
done
clear
C)
\[\frac{24}{25}\]
done
clear
D)
\[\frac{1}{50}\]
done
clear
View Answer play_arrow
question_answer 193) The equation of the circle of radius 5 in the first quadrant which touches x-axis and the line \[4y=3x\]is
A)
\[{{x}^{2}}+{{y}^{2}}-24x-y-25=0\]
done
clear
B)
\[{{x}^{2}}+{{y}^{2}}-30x-10y+225=0\]
done
clear
C)
\[{{x}^{2}}+{{y}^{2}}-16x-18y+64=0\]
done
clear
D)
\[{{x}^{2}}+{{y}^{2}}-20x-12y+144=0\]
done
clear
View Answer play_arrow
question_answer 194) The line \[lx+my+n=0\] is a normal to the ellipse \[\frac{{{x}^{2}}}{{{a}^{2}}}+\frac{{{y}^{2}}}{{{b}^{2}}}=1\]
A)
\[\frac{{{a}^{2}}}{{{l}^{2}}}+\frac{{{b}^{2}}}{{{m}^{2}}}=\frac{{{({{a}^{2}}-{{b}^{2}})}^{2}}}{{{n}^{2}}}\]
done
clear
B)
\[\frac{{{a}^{2}}}{{{m}^{2}}}+\frac{{{b}^{2}}}{{{l}^{2}}}=\frac{{{({{a}^{2}}-{{b}^{2}})}^{2}}}{{{n}^{2}}}\]
done
clear
C)
\[{{a}^{2}}{{l}^{2}}+{{b}^{2}}{{m}^{2}}={{({{a}^{2}}-{{b}^{2}})}^{2}}{{n}^{2}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 195) The equation \[\left| \sqrt{{{x}^{2}}+{{(y-1)}^{2}}}-\sqrt{{{x}^{2}}+{{(y+1)}^{2}}} \right|=k\]will represent a hyperbola for
A)
\[k\in (0,2)\]
done
clear
B)
\[k\in (0,1)\]
done
clear
C)
\[k\in (1,\infty )\]
done
clear
D)
\[k\in R'\]
done
clear
View Answer play_arrow
question_answer 196) Two forces \[{{\vec{F}}_{1}}=3\hat{i}-2\hat{j}+\hat{k}\] and \[{{\vec{F}}_{2}}=\hat{i}-3\hat{j}+5\hat{k}\] acting on a particle at A, move to .6. The work done if the position vector of \[\vec{A}\] and \[\vec{B}\]are \[-2\hat{i}+5\hat{k}\]and \[-3\hat{i}-7\hat{j}+2\hat{k}\]is
A)
\[25\]
done
clear
B)
\[13\]
done
clear
C)
\[26\]
done
clear
D)
\[28\]
done
clear
View Answer play_arrow
question_answer 197) If the three angles' of a quadrilateral are \[{{60}^{o}},\] \[{{80}^{o}}\] and \[\frac{5\pi }{6},\]then the fourth angle is
A)
\[{{70}^{o}}\]
done
clear
B)
\[{{79}^{o}}\]
done
clear
C)
\[{{80}^{o}}\]
done
clear
D)
\[{{81}^{o}}\]
done
clear
View Answer play_arrow
question_answer 198) Which one is not periodic
A)
\[|\sin \,3x|+{{\sin }^{2}}\,x\]
done
clear
B)
\[\cos \,\,\sqrt{x}\,+{{\cos }^{2}}\,x\]
done
clear
C)
\[\cos \,4x+{{\tan }^{2}}x\]
done
clear
D)
\[{{\cos }^{2}}x+\sin \,x\]
done
clear
View Answer play_arrow
question_answer 199) Let R be the relation on the set R of all real numbers defined by aRb if \[|a-b|\le 1,\]then R is
A)
Reflexive and symmetric
done
clear
B)
Symmetric only
done
clear
C)
Transitive only
done
clear
D)
Anti symmetric only
done
clear
View Answer play_arrow
question_answer 200) The expression \[(1+\tan \,x+{{\tan }^{2}}x)\,\,(1-\cot \,x+{{\cot }^{2}}x)\] has the positive values for x, given by
A)
\[0\le x\le \frac{\pi }{2}\]
done
clear
B)
\[0\le x\le \pi \]
done
clear
C)
For all \[x\in R\]
done
clear
D)
\[x\ge 0\]
done
clear
View Answer play_arrow
question_answer 201) The equation of a circle with origin as a centre and passing through an equilateral triangle whose median is of length 3a, is
A)
\[{{x}^{2}}+{{y}^{2}}=9{{a}^{2}}\]
done
clear
B)
\[{{x}^{2}}+{{y}^{2}}=16{{a}^{2}}\]
done
clear
C)
\[{{x}^{2}}+{{y}^{2}}=4{{a}^{2}}\]
done
clear
D)
\[{{x}^{2}}+{{y}^{2}}={{d}^{2}}\]
done
clear
View Answer play_arrow
question_answer 202) The point on the curve \[3{{x}^{2}}-4{{y}^{2}}=72\]which is nearest to the line \[3x+2y-1=0,\] is
A)
\[(6,3)\]
done
clear
B)
\[(6,-3)\]
done
clear
C)
\[(6,-6)\]
done
clear
D)
\[(6,5)\]
done
clear
View Answer play_arrow
question_answer 203) A circle is inscribed in a equilateral triangle of side a. The area of the circle is
A)
\[3\pi {{a}^{2}}\] sq unit
done
clear
B)
\[2{{a}^{2}}\] sq unit
done
clear
C)
\[{{a}^{2}}\]sq unit
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 204) If a, b, c are distinct positive real numbers and \[{{a}^{2}}+{{b}^{2}}+{{c}^{2}}=1,\]then \[3{{({{a}^{2}}{{b}^{2}}{{c}^{2}})}^{1/3}}\] is
A)
Less than 1
done
clear
B)
Equal to 1
done
clear
C)
Greater than 1
done
clear
D)
any real number
done
clear
View Answer play_arrow
question_answer 205) If \[\alpha ,\beta ,\gamma ,\delta \] are the smallest positive angles in ascending order of magnitude which have their sines equal to the positive quantity k, then the value of \[4\,\sin \frac{\alpha }{2}+3\sin \frac{\beta }{2}+2\sin \frac{\gamma }{2}+\sin \frac{\delta }{2}\]is equal to
A)
\[2\sqrt{1-k}\]
done
clear
B)
\[2\sqrt{1-k}\]
done
clear
C)
\[\frac{\sqrt{1+k}}{2}\]
done
clear
D)
\[\sqrt{1+k}\]
done
clear
View Answer play_arrow
question_answer 206) If \[x+9\] is a root of \[\left| \begin{matrix} x & 3 & 7 \\ 2 & x & 2 \\ 7 & 6 & x \\ \end{matrix} \right|=0,\]then other roots are
A)
\[-9,\,2,\,7\]
done
clear
B)
\[9,\,2,\,7\]
done
clear
C)
\[9,\,2,-\,7\]
done
clear
D)
\[9,-\,2,\,7\]
done
clear
View Answer play_arrow
question_answer 207) The value of \[\underset{x\to \pi /2}{\mathop{\lim }}\,\frac{\cot \,x-\cos x}{{{(\pi -2x)}^{3}}}\]
A)
\[1\]
done
clear
B)
\[\frac{1}{16}\]
done
clear
C)
\[16\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 208) If the tangent \[P(1,1)\] on \[{{y}^{2}}=x{{(2-x)}^{2}}\] meets the curve again at Q, then Q is
A)
\[(2,2)\]
done
clear
B)
\[(-1,-2)\]
done
clear
C)
\[\left( \frac{9}{4},\frac{3}{8} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 209) For the equation \[\frac{1}{x+a}-\frac{1}{x+b}=\frac{1}{x+c},\]if the product of the ratio is zero, then the sum of (he roots is
A)
\[0\]
done
clear
B)
\[\frac{2ab}{b+c}\]
done
clear
C)
\[\frac{2ac}{b+c}\]
done
clear
D)
\[-\frac{2ac}{b+c}\]
done
clear
View Answer play_arrow
question_answer 210) If \[n=2002,\] evaluate \[\frac{1}{{{\log }_{2}}n!}+\frac{1}{{{\log }_{3}}n!}+\frac{1}{{{\log }_{4}}n!}+.....+\frac{1}{{{\log }_{2002}}n!}\]
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 211) A and B are the independent events. The probability that both occur simultaneously is \[\frac{1}{6}\]and the probability that neither occur is \[\frac{1}{3}\]The probability of occurrence of the events A and B
A)
\[\frac{1}{2},\frac{3}{2}\]
done
clear
B)
\[\frac{1}{2},\frac{1}{3}\]
done
clear
C)
not possible
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 212) A card from a pack of 52 cards is lost. From the remaining cards of the pack, two cards are drawn and are found to be hearts. The probability of the missing card to be a heart is
A)
\[\frac{50}{11}\]
done
clear
B)
\[\frac{51}{11}\]
done
clear
C)
\[\frac{11}{50}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 213) \[\int_{0}^{\pi /2}{\frac{{{\sin }^{2}}x}{{{\sin }^{2}}x+{{\cos }^{2}}x}}\] is equal to
A)
\[\frac{\pi }{4}\]
done
clear
B)
\[2\pi \]
done
clear
C)
\[{{\pi }^{2}}\]
done
clear
D)
\[\frac{1}{2}{{\pi }^{2}}\]
done
clear
View Answer play_arrow
question_answer 214) The centre and the radius of the circle \[z\bar{z}+(2+3i)\,\,\bar{z}-2(2-3i)z+12=0\] is
A)
\[-(2+3i),\,(1)\]
done
clear
B)
\[(3+2i),(1)\]
done
clear
C)
\[(3+6i),(3)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 215) The weight (in Kilogram) of 15 students are as follows 31, 35, 27, 29, 32, 43, 37, 41, 34, 28, 36,44, 45, 42, 30. If the weight 44 kg is replaced by 46 kg and \[27\text{ }kg\]is by \[25\text{ }kg,\] then new median is
A)
\[32\]
done
clear
B)
\[33\]
done
clear
C)
\[34\]
done
clear
D)
\[35\]
done
clear
View Answer play_arrow
question_answer 216) If \[1,\,{{\log }_{9}}({{3}^{1-x}}+2),{{\log }_{3}}({{4.3}^{x}}-1)\] are in AP, then x equals
A)
\[{{\log }_{3}}4\]
done
clear
B)
\[1-{{\log }_{3}}4\]
done
clear
C)
\[1-{{\log }_{4}}3\]
done
clear
D)
\[{{\log }_{4}}3\]
done
clear
View Answer play_arrow
question_answer 217) If the roots of the equation \[3{{x}^{2}}-6x+5=0\] are \[\alpha \] and \[\beta ,\] then the equation whose roots are \[\alpha +\beta \]and\[\frac{2}{\alpha +\beta }\]will be
A)
\[{{x}^{2}}+3x-1=0\]
done
clear
B)
\[{{x}^{2}}+3x-2=0\]
done
clear
C)
\[{{x}^{2}}+3x+2=0\]
done
clear
D)
\[{{x}^{2}}-3x+2=0\]
done
clear
View Answer play_arrow
question_answer 218) The equation of the locus of a point equidistant from the points \[({{a}_{1}},\,{{b}_{1}})\] and \[({{a}_{2}},\,{{b}_{2}})\] is \[({{a}_{1}}-{{a}_{2}})x+({{b}_{1}}-{{b}_{2}})y+c=0,\] then the value of c is
A)
\[\sqrt{(a_{1}^{2}+b_{1}^{2}+c_{1}^{2})}\]
done
clear
B)
\[a_{1}^{2}-b_{1}^{2}-c_{1}^{2}\]
done
clear
C)
\[\frac{1}{2}(a_{2}^{2}+b_{2}^{2}-a_{1}^{2}-b_{1}^{2})\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 219) The general solution of the equation \[{{2}^{\cos \,2x}}+1={{3.2}^{-\sin \,x}}\]is
A)
\[n\,\pi \]
done
clear
B)
\[n\,\pi -\pi \]
done
clear
C)
\[n\,\pi +\pi \]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 220) Let \[0<x<\pi \] and \[y(x)\] be given by \[(1+\sin \,x){{y}^{3}}-(\cos \,x){{y}^{2}}+2(1+\sin \,x)y-2cos\,x=0\] The derivative of y with respect to \[\frac{x}{2}\]at \[x=\frac{\pi }{2}\] is
A)
\[-\frac{1}{2}\]
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[-2\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 221) The value of \[\int_{0}^{\pi }{\left( \sum\limits_{r=0}^{3}{{{a}_{r}}\,\,{{\cos }^{3-r\,}}x\,{{\sin }^{r}}x} \right)}dx\]depends upon
A)
\[{{a}_{1}}\] and \[{{a}_{2}}\]
done
clear
B)
\[{{a}_{0}}\] and \[{{a}_{3}}\]
done
clear
C)
\[{{a}_{2}}\] and \[{{a}_{3}}\]
done
clear
D)
\[{{a}_{1}}\] and \[{{a}_{3}}\]
done
clear
View Answer play_arrow
question_answer 222) If the .function \[f:[1,\,\infty )\to [1,\infty )\] is denned by \[f(x)={{2}^{x(x-1)}},\]then \[{{f}^{-1}}(x)\] is
A)
\[{{\left( \frac{1}{2} \right)}^{x(x-1)}}\]
done
clear
B)
\[\frac{1}{2}(1+\sqrt{1+4{{\log }_{2}}y})\]
done
clear
C)
\[\frac{1}{2}(1-\sqrt{1+4{{\log }_{2}}y})\]
done
clear
D)
\[\infty \]
done
clear
View Answer play_arrow
question_answer 223) If the roots of the given equation \[(\cos \,\,p-1){{x}^{2}}+(\cos \,p)x+\sin \,p=0\] are real, then
A)
\[p\in (-\pi ,0)\]
done
clear
B)
\[p\in \left( -\frac{\pi }{2},\frac{\pi }{2} \right)\]
done
clear
C)
\[p\in (0,\pi )\]
done
clear
D)
\[p\in (0,2\pi )\]
done
clear
View Answer play_arrow
question_answer 224) The angle of depression of a boat m in a river is \[{{30}^{o}}\] from the top of a tower, \[87\text{ }m\]high and the speed of the boat is\[5.8\text{ }km/h\]. The time taken by the boat to reach at the base of the tower is
A)
\[9\,\,\min \]
done
clear
B)
\[\frac{9\sqrt{3}}{10}\,\,\min \]
done
clear
C)
\[25\,\,\min \]
done
clear
D)
\[15\,\,\min \]
done
clear
View Answer play_arrow
question_answer 225) If \[\alpha =\frac{3}{5},\,\,(0<\alpha ,\pi )\]and \[\cos \beta =\frac{5}{13},\] then \[(\alpha -\beta )\] lies in the quadrants
A)
I, II, IV
done
clear
B)
I, III, IV
done
clear
C)
I, II, III
done
clear
D)
I,IV
done
clear
View Answer play_arrow
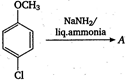 The major product A is
The major product A is
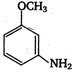
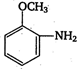
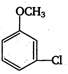
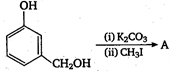
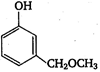
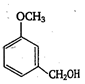
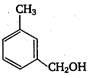
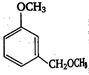
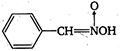
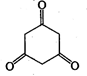
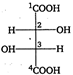 Configuration at \[{{C}_{2}}\]and \[{{C}_{3}}\]atoms are
Configuration at \[{{C}_{2}}\]and \[{{C}_{3}}\]atoms are
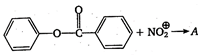 The product A is
The product A is