question_answer 1) A ladder 2.5 m long and 150 N weight has Us centre of gravity l m from the bottom. A weight 40 N is attached to the Top end. The work required to raise the ladder from the horizontal position to the vertical position is:
A)
190 J
done
clear
B)
250 J
done
clear
C)
285 J
done
clear
D)
475 J
done
clear
View Answer play_arrow
question_answer 2) Two bodies having the same mass 2 kg each, have different surface areas \[50{{m}^{2}}\]and \[100{{m}^{2}}\]in contact with a horizontal plane. If the coefficient of friction is 0.2. the forces of friction that come into play when they are in motion will be in the ratio:
A)
1 : 1
done
clear
B)
1 : 2
done
clear
C)
2 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 3) A stone is tied up to the end of a string and whirled in a vertical circle. The tension of the string is T1 when the stone is at the highest point and T2 ,when it is at the lowest point, then:
A)
\[{{T}_{1}}<2{{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
D)
\[{{T}_{1}}<{{T}_{2}}\]
done
clear
View Answer play_arrow
question_answer 4) A van is moving with a speed of 72 km/h on a level road. Where the coefficient of friction between its tyres and road is 0.5. The minimum radius of curvature, that the road must have, for safe driving of van is\[(g=10m{{s}^{-2}})\]:
A)
80 m
done
clear
B)
40 m
done
clear
C)
20 m
done
clear
D)
4 m
done
clear
View Answer play_arrow
question_answer 5) A thin uniform rod AB of mass m and length l is hinged at one end A to the level floor. Initially if stands vertically and is allowed to fall freely to the floor in the vertical plane. The angular velocity of the rod, when its end B strikes the floor is {g is acceleration due to gravity):
A)
\[\left( \frac{mg}{l} \right)\]
done
clear
B)
\[{{\left( \frac{mg}{3l} \right)}^{1/2}}\]
done
clear
C)
\[\left( \frac{g}{l} \right)\]
done
clear
D)
\[{{\left( \frac{3g}{l} \right)}^{1/2}}\]
done
clear
View Answer play_arrow
question_answer 6) Two satellites of mass 50 kg and 100 kg revolve around the earth in circular orbits of radius 9 R and 16 R respectively, where R is the radius of earth. The speeds of the two satellites will be in the ratio:
A)
3/4
done
clear
B)
4/3
done
clear
C)
9/16
done
clear
D)
16/9
done
clear
View Answer play_arrow
question_answer 7) The escape velocities on two planets of masses m1 and m2 having same radius, are v1 and v2 respectively, Then:
A)
\[\frac{{{v}_{1}}}{{{v}_{2}}}=\frac{{{m}_{1}}}{{{m}_{2}}}\]
done
clear
B)
\[\frac{{{v}_{2}}}{{{v}_{1}}}=\frac{{{m}_{1}}}{{{m}_{2}}}\]
done
clear
C)
\[\frac{{{v}_{1}}}{{{v}_{2}}}={{\left( \frac{{{m}_{1}}}{{{m}_{2}}} \right)}^{2}}\]
done
clear
D)
\[\frac{{{v}_{1}}}{{{v}_{2}}}=\sqrt{\frac{{{m}_{1}}}{{{m}_{2}}}}\]
done
clear
View Answer play_arrow
question_answer 8) he displacement of a particle executing simple harmonic motion, from its mean position is given by \[x=0.5\,\sin \,(10\pi l+1.5x)\cos (10\pi l+1.5\pi ).\] The ratio of the maximum velocity to the maximum acceleration of the body is given by:
A)
\[20\pi \]
done
clear
B)
\[\frac{1}{20\,\pi }\]
done
clear
C)
\[\frac{1}{10\,\pi }\]
done
clear
D)
\[10\,\pi \]
done
clear
View Answer play_arrow
question_answer 9) The percentage change in the time period of a seconds pendulum when its amplitude is reduced by 50% is:
A)
75%
done
clear
B)
25%
done
clear
C)
0%
done
clear
D)
50%
done
clear
View Answer play_arrow
question_answer 10) Y, K and \[\eta \] represent respectively the Youngs modulus, bulk modulus and rigidity modulus of a body. If rigidity modulus is twice the bulk modulus, then:
A)
\[Y=\frac{5}{18}K\]
done
clear
B)
\[Y=\frac{5}{8}\eta \]
done
clear
C)
\[Y=\frac{9}{5}K\]
done
clear
D)
\[Y=\frac{18}{5}K\]
done
clear
View Answer play_arrow
question_answer 11) If m is mass, Q is charge and B is magnetic induction, then \[\frac{m}{BQ}\]has the same dimensions as:
A)
frequency
done
clear
B)
\[\frac{1}{\text{frequency}}\]
done
clear
C)
velocity
done
clear
D)
acceleration
done
clear
View Answer play_arrow
question_answer 12) Then the angle between \[\overrightarrow{A}\] and \[\overrightarrow{B}\] is: If \[\overrightarrow{A}+\overrightarrow{B}=\overrightarrow{C}\] and \[\left| \overrightarrow{A} \right.\left| = \right.\left| \overrightarrow{B} \right.=\left| \overrightarrow{C} \right.\left| {} \right.\]
A)
\[45{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[120{}^\circ \]
done
clear
View Answer play_arrow
question_answer 13) The distances travelled by a body starting from rest and travelling with uniform acceleration, in successive intervals of time of equal duration will be in the ratio:
A)
1 : 2 : 3
done
clear
B)
1 : 2 : 4
done
clear
C)
1 : 3 : 5
done
clear
D)
1 : 5 : 9
done
clear
View Answer play_arrow
question_answer 14) A stone is dropped freely, while another is thrown vertically downward with an initial velocity at 2 m/s from the same point, simultaneously. The time required by them to have a distance of separation 22 m between them is:
A)
11 s
done
clear
B)
5.5 s
done
clear
C)
44 s
done
clear
D)
22 s
done
clear
View Answer play_arrow
question_answer 15) The angle of projection of a projectile, for which the horizontal range and the maximum height are equal, is:
A)
\[{{\tan }^{-1}}\left( \sqrt{(3)} \right)\]
done
clear
B)
\[{{\tan }^{-1}}\left( 4 \right)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \sqrt{2} \right)\]
done
clear
D)
\[{{\tan }^{-1}}\left( \frac{1}{\sqrt{3}} \right)\]
done
clear
View Answer play_arrow
question_answer 16) A person of 70 kg mass is lifted by a helicopter with the help of rope, hanging from it. If the helicopter is rising up with an acceleration \[4\text{ }m{{s}^{-2}},\] the tension in the rope is (taking\[g=10m{{s}^{-2}}\]):
A)
250 N
done
clear
B)
490 N
done
clear
C)
980 N
done
clear
D)
1960 N
done
clear
View Answer play_arrow
question_answer 17) A force of 100 N acts on a body of mass 2 kg for 10 s. The change in momentum of the body is:
A)
100 Ns
done
clear
B)
250 Ns
done
clear
C)
500 Ns
done
clear
D)
1000 Ns
done
clear
View Answer play_arrow
question_answer 18) In an elastic collision between two bodies. the physical quantity that is conserved is:
A)
kinetic energy
done
clear
B)
momentum
done
clear
C)
potential energy
done
clear
D)
kinetic energy and momentum
done
clear
View Answer play_arrow
question_answer 19) The kinetic energy of a body is four limes its momentum. Its velocity is:
A)
\[2\text{ }m{{s}^{-1}}\]
done
clear
B)
\[4m{{s}^{-1}}\]
done
clear
C)
\[8\text{ }m{{s}^{-1}}\]
done
clear
D)
\[16m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 20) When a capillary tube is lowered into water, the mass of water, raised in the tube, above the outside water level is 5 g. If the radius of the tube is doubled, the mass of water that rises in the capillary tube, above the outside water level is:
A)
1.25 g
done
clear
B)
5 g
done
clear
C)
10 g
done
clear
D)
20 g
done
clear
View Answer play_arrow
question_answer 21) Two copper rods of same length, but of different diameters of cross-section are given same amount of heat. Assuming no heat losses:
A)
thick rod expands more
done
clear
B)
thin rod expands more
done
clear
C)
both rods expand equally
done
clear
D)
thick rods expand more tor large quantity of heat and thin rods expand more for small quantity of heal
done
clear
View Answer play_arrow
question_answer 22) When water is heated from \[0{}^\circ C\] to \[10{}^\circ C\], its volume:
A)
decreases continuously
done
clear
B)
first decreases and then increases
done
clear
C)
first increases and then decreases
done
clear
D)
increases continuously
done
clear
View Answer play_arrow
question_answer 23) An air bubble doubles its radius on raising from the bottom of water reservoir to the surface of water in it. if the atmospheric pressure is equal to 10 m of water, the height of water in the reservoir is:
A)
10 m
done
clear
B)
20 m
done
clear
C)
70 m
done
clear
D)
80 m
done
clear
View Answer play_arrow
question_answer 24) For an adiabatic change in a gas. if P, V, T denote pressure, volume and absolute temperature of gas at any time and 7 is ratio of specific heats of gas, which of the following equations is true?
A)
\[{{T}^{\gamma }}{{P}^{1-\gamma }}\]= constant
done
clear
B)
\[{{T}^{\gamma -1}}{{P}^{\gamma }}\]= constant
done
clear
C)
\[{{T}^{\gamma -1}}{{V}^{\gamma }}\]= constant
done
clear
D)
\[{{T}^{\gamma }}{{V}^{\gamma }}\]= constant
done
clear
View Answer play_arrow
question_answer 25) Boiling water at \[100{}^\circ C\] and cold water at \[t{}^\circ C\] are mixed in the ratio 1:3 and the resultant maximum temperature was \[37{}^\circ C\]. Assuming no heat losses, the value of t is:
A)
\[4{}^\circ C\]
done
clear
B)
\[9{}^\circ C\]
done
clear
C)
\[12{}^\circ C\]
done
clear
D)
\[16{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 26) 20 g of ice and 20 g of hoi water are mixed. When the ice is melted the temperature of the mixture was found to be \[0{}^\circ C\]. The temperature of hot water taken should (L = 80 cal/g):
A)
\[40{}^\circ C\]
done
clear
B)
\[72{}^\circ C\]
done
clear
C)
\[80{}^\circ C\]
done
clear
D)
\[96{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 27) A wall has two layers A and B, made of two different materials. The thermal conductivity of material A is twice that of B. If two layers have the same thickness and under thermal equilibrium, the temperature difference across the wall is \[48{}^\circ C\], the temperature difference across layer B is:
A)
\[40{}^\circ C\]
done
clear
B)
\[32{}^\circ C\]
done
clear
C)
\[16{}^\circ C\]
done
clear
D)
\[24{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 28) The frequency of transverse vibration in a stretched string is 200 Hz. If the tension is increased four times and the length is reduced to one-fourth, the original values, the frequency of vibrations will be:
A)
25 Hz
done
clear
B)
200 Hz
done
clear
C)
400 Hz
done
clear
D)
1600 Hz
done
clear
View Answer play_arrow
question_answer 29) Equation of a stationary wave is given by:
A)
\[y=A\,\sin \,k(x-vt)\]
done
clear
B)
\[y=2A\,\sin \,kx\cos \omega t\]
done
clear
C)
\[y=A\,\cos \,2\pi \left( \frac{kx}{\lambda }-\frac{t}{T} \right)\]
done
clear
D)
\[y=A\,\cos \frac{2\pi vt}{\lambda }\]
done
clear
View Answer play_arrow
question_answer 30) The ratio of densities of nitrogen and oxygen is 14:16. The temperature at which the speed of sound in nitrogen will be same as that of oxygen at \[55{}^\circ C\] is
A)
\[35{}^\circ C\]
done
clear
B)
\[48{}^\circ C\]
done
clear
C)
\[65{}^\circ C\]
done
clear
D)
\[14{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 31) Two tuning forks A and B produce 6 beats per second. If the frequency of A is 512 Hz. The fork B is loaded with wax and they produce 4 beats per second- The frequency of B is:
A)
506 Hz
done
clear
B)
516 Hz
done
clear
C)
508 Hz
done
clear
D)
518 Hz
done
clear
View Answer play_arrow
question_answer 32) In a compound microscope the image produced by the objective is:
A)
real, enlarged and erect
done
clear
B)
real, enlarged and inverted
done
clear
C)
virtual, enlarged and erect
done
clear
D)
virtual, enlarged and inverted
done
clear
View Answer play_arrow
question_answer 33) An astronomical telescope has a length of 1.1 m and a magnifying power 10. The focal lengths of objective and eye lenses are:
A)
0. 1 m and 1.0 m
done
clear
B)
1.0 m and 0.1 m
done
clear
C)
0.6 m and 0.5 m
done
clear
D)
0.5 m and 0.6 m
done
clear
View Answer play_arrow
question_answer 34) A ray of light is incident on one of the refracting faces of an equilateral prism at an angle of incidence \[48{}^\circ C\] in the minimum deviation position. The angle of deviation produced by the prism is:
A)
\[48{}^\circ C\]
done
clear
B)
\[36{}^\circ C\]
done
clear
C)
\[24{}^\circ C\]
done
clear
D)
\[18{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 35) Two sources of light are said to be coherent, if light waves produced by them have:
A)
the same wavelength
done
clear
B)
the same wavelength and constant phase difference
done
clear
C)
the same amplitude
done
clear
D)
the same wavelength and same amplitude
done
clear
View Answer play_arrow
question_answer 36) The null points are on the axial line of a bar magnet, when it is placed such that its south pole points:
A)
south
done
clear
B)
east
done
clear
C)
north
done
clear
D)
west
done
clear
View Answer play_arrow
question_answer 37) If two bar magnets of different lengths have equal moments, then the pole strength is:
A)
equal for both the magnets
done
clear
B)
lesser for shorter magnet
done
clear
C)
more for longer magnet
done
clear
D)
more for shorter magnet
done
clear
View Answer play_arrow
question_answer 38) If a diamagnetic material is placed in a magnetic field, the flux density inside material compared to that outside will be:
A)
slightly less
done
clear
B)
slightly more
done
clear
C)
very much more
done
clear
D)
same
done
clear
View Answer play_arrow
question_answer 39) Two equal unlike charges, placed 3 cm. Apart in air. attract each other with a force of 40 N. The magnitude of each charge in micro coulombs is, take \[\frac{1}{4\pi {{\varepsilon }_{0}}}=9\times {{10}^{9}}\,SI\]units:
A)
0.2
done
clear
B)
2
done
clear
C)
20
done
clear
D)
200
done
clear
View Answer play_arrow
question_answer 40) At any point on the right bisector of line joining two equal and opposite charges:
A)
the electric field is zero
done
clear
B)
the electric potential is zero
done
clear
C)
the electric potential decreases with increasing distance from centre
done
clear
D)
the electric field is perpendicular to the line joining the charges
done
clear
View Answer play_arrow
question_answer 41) A double ionized He+2 atom travels at right angles lo a magnetic field of induction 0.4 T at a velocity of 10 m/s describing a circle of radius r. A proton travelling with the same speed in the same direction in the same field will describe a circle radius:
A)
\[\frac{r}{4}\]
done
clear
B)
\[\frac{r}{2}\]
done
clear
C)
\[r\]
done
clear
D)
\[2\,r\]
done
clear
View Answer play_arrow
question_answer 42) For a thermo couple if the cold junction is maintained at \[0{}^\circ C\] the inversion temperature is \[680{}^\circ C\]. its neutral temperature is:
A)
\[1360{}^\circ C\]
done
clear
B)
\[680{}^\circ C\]
done
clear
C)
\[340{}^\circ C\]
done
clear
D)
\[170{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 43) A dielectric of thickness 5 cm and dielectric constant 10, is introduced in between the plates of a parallel plate capacitor having plate area 500 cm2 and separation between plates 10 cm. The capacitance of the, capacitor is \[(If\,{{\varepsilon }_{0}}=8.8\times {{10}^{-12}}\,SI\,units):\]
A)
8 pF
done
clear
B)
6 pF
done
clear
C)
4 pF
done
clear
D)
20 pF
done
clear
View Answer play_arrow
question_answer 44) The total number of spectral lines expected to be emitted by atomic hydrozen excited to the 5th energy level:
A)
4
done
clear
B)
5
done
clear
C)
10
done
clear
D)
20
done
clear
View Answer play_arrow
question_answer 45) The threshold frequency for photo electric effect for a metal surface is found to be \[4.8\times {{10}^{16}}Hz,\] the stopping potential required when the metal is irradiated by radiation of frequency \[5.6\times {{10}^{16}}Hz\]is: (taking \[h=6.6\times {{10}^{-34}}J-s\]and\[e=6.6\times {{10}^{-19}}C\])
A)
22.4 V
done
clear
B)
33 V
done
clear
C)
66 V
done
clear
D)
198 V
done
clear
View Answer play_arrow
question_answer 46) If the operating voltage of an X-ray tube is increased:
A)
X-rays intensity increases
done
clear
B)
X-rays wavelength limit on the maximum side increases
done
clear
C)
X-rays wavelength limit on minimum side decreases
done
clear
D)
X-rays intensity decreases
done
clear
View Answer play_arrow
question_answer 47) In each fission of \[{{U}^{235}},200\]MeV of energy is released. If a reactor produces 100 MW power the rate of fission in ii will be:
A)
\[3.125\times {{10}^{18}}\]per min
done
clear
B)
\[3.125\times {{10}^{17}}\]per sec
done
clear
C)
\[3.125\times {{10}^{17}}\]per min
done
clear
D)
\[3.125\times {{10}^{18}}\]per sec
done
clear
View Answer play_arrow
question_answer 48) The nuclear force between two protons is \[{{F}_{pp}}\]between two neutrons is \[{{F}_{nn}}\]and between a proton and a neutron is F , then:
A)
\[{{F}_{pp}}>{{F}_{pn}}>{{F}_{nn}}\]
done
clear
B)
\[{{F}_{pp}}>{{F}_{nn}}>{{F}_{pn}}\]
done
clear
C)
\[{{F}_{pp}}={{F}_{pn}}={{F}_{mn}}\]
done
clear
D)
\[{{F}_{nn}}>\text{ }{{F}_{pp}}>~{{F}_{pn}}\]
done
clear
View Answer play_arrow
question_answer 49) The width of forbidden gap in silicon crystal is 1.1 eV. When the crystal is converted into n- type semiconductor the distance of Fermi level from conduction band is:
A)
greater than 0.55 eV
done
clear
B)
equal to 0.55 eV
done
clear
C)
lesser than 0.55 eV
done
clear
D)
equal 1.1 eV
done
clear
View Answer play_arrow
question_answer 50) The donor impurity to be added for doping germanium crystal will be of valency:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 51) In the nuclear reaction \[_{13}A{{l}^{27}}(d,\alpha )x,X\]is:
A)
\[{{\,}_{6}}{{C}^{12}}\]
done
clear
B)
\[{{\,}_{14}}S{{i}^{30}}\]
done
clear
C)
\[{{\,}_{12}}M{{g}^{25}}\]
done
clear
D)
\[{{\,}_{11}}N{{a}^{24}}\]
done
clear
View Answer play_arrow
question_answer 52) The conjugate acid of \[\text{N}_{3}^{-}\] (Azide ion) is :
A)
\[N{{H}_{3}}\]
done
clear
B)
\[H{{N}_{3}}\]
done
clear
C)
\[NH_{2}^{-}\]
done
clear
D)
\[N_{2}^{-}\]
done
clear
View Answer play_arrow
question_answer 53) Which of the following form of sulphur can be prepared by pouring liquid sulphur into cold water?
A)
Monoclinic
done
clear
B)
Rhombic
done
clear
C)
Plastic
done
clear
D)
Colloidal
done
clear
View Answer play_arrow
question_answer 54) Stainless steel vessels are made from the steel containing:
A)
Ni
done
clear
B)
Mn
done
clear
C)
W
done
clear
D)
Cr
done
clear
View Answer play_arrow
question_answer 55) The dielectric constants of \[{{\text{H}}_{\text{2}}}\text{O}\]and \[{{\text{D}}_{\text{2}}}\text{O}\]are 82 and 80.5 respectively. Therefore, the solubility of NaCI in \[{{\text{D}}_{\text{2}}}\text{O}\]is ...... than in water:
A)
equal to
done
clear
B)
slightly less
done
clear
C)
slightly higher
done
clear
D)
very high
done
clear
View Answer play_arrow
question_answer 56) Metals having \[\text{n}{{\text{s}}^{\text{1}}}\]the valence electronic configuration:
A)
are strong oxidising agents
done
clear
B)
are highly electronegative
done
clear
C)
are highly electropositive
done
clear
D)
have a first ionisation potential of more than 10 eV/atom
done
clear
View Answer play_arrow
question_answer 57) A vessel contains equal volumes of \[\text{S}{{\text{O}}_{\text{2}}}\]and \[\text{C}{{\text{H}}_{4}}.\]Through a small hole the gases effused into vacuum. After 200 seconds the total volume is reduced to half. What is the ratio of \[\text{S}{{\text{O}}_{\text{2}}}\]and \[\text{C}{{\text{H}}_{\text{4}}}\]remaining in the vessel?
A)
1:2
done
clear
B)
2:1
done
clear
C)
1 :1
done
clear
D)
1:4
done
clear
View Answer play_arrow
question_answer 58) The Lassaignes solution of an organic compound with nitrogen; sulphur and chlorine contains:
A)
\[NaCN,N{{a}_{2}}S,NaCl\]
done
clear
B)
\[KCN,{{K}_{2}}S,KCl\]
done
clear
C)
\[AgCl,NaCN,{{K}_{2}}S\]
done
clear
D)
\[NaNC,FeS{{O}_{4}},KCl\]
done
clear
View Answer play_arrow
question_answer 59) A mixture of carbon monoxide, hydrogen and hydrocarbon is known as:
A)
water gas
done
clear
B)
carburetted water gas
done
clear
C)
semi water gas
done
clear
D)
producer gas
done
clear
View Answer play_arrow
question_answer 60) Hydrolysis of \[\text{SC}{{\text{l}}_{\text{4}}}\] forms :
A)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[{{H}_{2}}S{{O}_{3}}\]
done
clear
C)
\[S{{O}_{3}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 61) The r. m. s velocity of a gas at a particular temperature is found to be 12240 cm \[{{\sec }^{-1}}.\]What is its most probable velocity (in cm \[{{\sec }^{-1}}.\]) at the same temperature?
A)
\[{{10}^{5}}\]
done
clear
B)
\[{{10}^{4}}\]
done
clear
C)
11280
done
clear
D)
1128
done
clear
View Answer play_arrow
question_answer 62) Which of the following is used to reduce pure haematite to iron?
A)
\[{{H}_{2}}\]
done
clear
B)
CO
done
clear
C)
\[LiAl{{H}_{4}}\]
done
clear
D)
\[{{H}_{2}}/Pd\]
done
clear
View Answer play_arrow
question_answer 63) Which of the following is used in the preparation of ammonal?
A)
\[F{{e}_{2}}O3\]
done
clear
B)
Al powder
done
clear
C)
\[CaC{{N}_{2}}+C\] (graphite)
done
clear
D)
\[NaOH+CaO\]
done
clear
View Answer play_arrow
question_answer 64) What is formed when 3 parts of HCl is mixed with one part of\[\text{HN}{{\text{O}}_{\text{3}}}\]?
A)
\[NO\]
done
clear
B)
\[NC{{l}_{3}}\]
done
clear
C)
\[NOCl\]
done
clear
D)
\[N{{H}_{4}}Cl\]
done
clear
View Answer play_arrow
question_answer 65) Which one of the following ions has the same number of unpaired electrons as that of \[{{\text{V}}^{\text{3+}}}\] ion?
A)
\[C{{r}^{3+}}\]
done
clear
B)
\[M{{n}^{2+}}\]
done
clear
C)
\[N{{i}^{2+}}\]
done
clear
D)
\[F{{e}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 66) 18 C-H and 7 C-C sigma bonds are present in:
A)
cyclo hexane
done
clear
B)
3, 3-dimethyl pentane
done
clear
C)
2, 2, 3-trimethyl pentane
done
clear
D)
\[n-\] heptane
done
clear
View Answer play_arrow
question_answer 67) How many electrons are present in the M shell of the atom of an element with atomic number 24?
A)
5
done
clear
B)
6
done
clear
C)
12
done
clear
D)
13
done
clear
View Answer play_arrow
question_answer 68) The pH of 1 litre \[\text{ }\!\!~\!\!\text{ NaOH}\] solution is 12. What is the amount in grams of \[\text{ }\!\!~\!\!\text{ NaOH}\] present in this solution?
A)
40
done
clear
B)
4
done
clear
C)
0.4
done
clear
D)
20
done
clear
View Answer play_arrow
question_answer 69) \[\text{Si}{{\text{O}}_{2}}\] is soluble in:
A)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[HN{{O}_{3}}\]
done
clear
C)
\[HF\]
done
clear
D)
\[HCl\]
done
clear
View Answer play_arrow
question_answer 70) Which of the following does not exhibit allotropy?
A)
P
done
clear
B)
Bi
done
clear
C)
Sb
done
clear
D)
As
done
clear
View Answer play_arrow
question_answer 71) The pH of an aqueous solution of \[{{H}_{2}}{{O}_{2}}\] is \[6.0\text{ }.\]Some chlorine is bubbled through this solution. The pH of the resultant solution will be:
A)
6
done
clear
B)
7
done
clear
C)
less than 6
done
clear
D)
more than 7
done
clear
View Answer play_arrow
question_answer 72) Electron is an alloy of:
A)
\[Mg+Ni\]
done
clear
B)
\[Ni+Al\]
done
clear
C)
\[Mg+Al\]
done
clear
D)
\[Mg+Zn\]
done
clear
View Answer play_arrow
question_answer 73) Which of the following can form compounds with xenon?
A)
\[{{I}_{2}}\]
done
clear
B)
\[{{F}_{2}}\]
done
clear
C)
\[B{{r}_{2}}\]
done
clear
D)
Al
done
clear
View Answer play_arrow
question_answer 74) The test used for carbon-carbon double bond in an alkene is :
A)
Baeyers
done
clear
B)
carbyl amine
done
clear
C)
Lassaignes
done
clear
D)
silver mirror
done
clear
View Answer play_arrow
question_answer 75) Which of the following atomic orbitals is not directed along the axis?
A)
\[{{p}_{x}}\]
done
clear
B)
\[{{d}_{{{x}^{2}}-{{y}^{2}}}}\]
done
clear
C)
\[{{d}_{xy}}\]
done
clear
D)
\[{{d}_{{{z}^{2}}}}\]
done
clear
View Answer play_arrow
question_answer 76) In which of the following compounds the angle between two covalent bonds is highest?
A)
\[C{{H}_{4}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 77) How many grams of phosphoric acid is required to completely neutralise 120g of sodium hydroxide?
A)
49
done
clear
B)
98
done
clear
C)
196
done
clear
D)
9.8
done
clear
View Answer play_arrow
question_answer 78) During the extraction of zinc, ZnO is obtained from zinc blende by :
A)
roasting
done
clear
B)
cupellation
done
clear
C)
poling
done
clear
D)
electrolytic reduction
done
clear
View Answer play_arrow
question_answer 79) Which of the following is more stable?
A)
HF
done
clear
B)
HI
done
clear
C)
HCl
done
clear
D)
HBr
done
clear
View Answer play_arrow
question_answer 80) The IUPAC name of \[{{\text{(}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}\text{)}}_{\text{4}}}\text{C}\]is:
A)
tetraethyl methane
done
clear
B)
2, 2-dimethyl pentane
done
clear
C)
3, 3-diethyl pentane
done
clear
D)
neopentane
done
clear
View Answer play_arrow
question_answer 81) The number of periods present in the long form of the periodic table i :
A)
6
done
clear
B)
7
done
clear
C)
8
done
clear
D)
18
done
clear
View Answer play_arrow
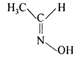
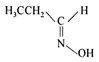
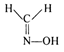
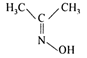
question_answer 82) Which of the following is acetone oxime?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 83) 6 g of urea is dissolved in 16.2 g of water at \[\text{25}{{\,}^{\text{o}}}\text{C}\text{.}\] What is the vapour pressure (in mm) of resulting solution? The vapour pressure of water at \[\text{25}{{\,}^{\text{o}}}\text{C}\text{.}\]is 24 mm:
A)
24
done
clear
B)
21.3
done
clear
C)
23.6
done
clear
D)
18
done
clear
View Answer play_arrow
question_answer 84) Which of the following compounds liberates hydrogen gas on reacting with sodium metal?
A)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 85) Standard reduction potentials of \[S{{n}^{2+}}/Sn\]and \[Z{{n}^{2+}}/Zn\]electrodes are 0.14 V and \[-\text{ }0.74\text{ V}\] respectively. What is the \[{{\text{E}}^{\text{o}}}\] of the following cell in volts \[\text{Zn/Z}{{\text{n}}^{2+}}||S{{n}^{2+}}/Sn\]?
A)
\[-0.6\]
done
clear
B)
\[-0.88\]
done
clear
C)
0.88
done
clear
D)
0.6
done
clear
View Answer play_arrow
question_answer 86) Benzene is a monocyclic compound. It is aromatic because it contains :
A)
6 carbons and 6 hydrogens
done
clear
B)
3\[\pi -\]bonds
done
clear
C)
\[6p-\] orbitals
done
clear
D)
\[(4n+2)\pi \]electrons
done
clear
View Answer play_arrow
question_answer 87) What is the volume in litres of\[\text{0}\text{.1}\,\text{M}\,{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\] required to completely neutralise 1 L of 0.5 \[\text{M}\,\text{NaOH}\]?
A)
5
done
clear
B)
2.5
done
clear
C)
0.5
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 88) Which of the following compounds exhibit functional isomerism?
A)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{3}}{{H}_{6}}O\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}Cl\]
done
clear
D)
\[{{C}_{5}}{{H}_{12}}\]
done
clear
View Answer play_arrow
question_answer 89) Radioactivity of an isotope falls to 1/8 of its original value in 48 days. Its half life period is (in days):
A)
48
done
clear
B)
16
done
clear
C)
12
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 90) The number of sigma bonds in ethane formed by the overlap of \[s{{p}^{3}}\] and s orbitals is
A)
5
done
clear
B)
6
done
clear
C)
7
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 91) From the following data for the decomposition of \[{{\text{N}}_{\text{2}}}{{\text{O}}_{\text{4}}}\] at \[\text{30}{{\,}^{\text{o}}}\text{C}\] find out the rate constant (in \[\text{mi}{{\text{n}}^{-1}}\]) Volume of \[{{\text{O}}_{\text{2}}}\] after 10 minutes of the reaction = 90 mL Volume of \[{{\text{O}}_{\text{2}}}\] after completion of the reaction =100 mL:
A)
2.303
done
clear
B)
0.2303
done
clear
C)
0.02303
done
clear
D)
23.03
done
clear
View Answer play_arrow
question_answer 92) The two enzymes present in yeast that an responsible for the formation of ethyl alcohol from molasses in the fermentatiol process are:
A)
invertase, diastase
done
clear
B)
invertase, zymase
done
clear
C)
zymase, diastase
done
clear
D)
invertase, maltase
done
clear
View Answer play_arrow
question_answer 93) What is the amount of heat liberated (in kcal when one mole of aqueous NaOH reacted with one mole of aqueous HCl solution?
A)
22.4
done
clear
B)
13.7
done
clear
C)
2.303
done
clear
D)
0.693
done
clear
View Answer play_arrow
question_answer 94) Which one of the following reaction conditions are used for preparing diethyl ether from ethyl alcohol?
A)
\[HI/200{{\,}^{o}}C\]
done
clear
B)
Conc. \[{{H}_{2}}S{{O}_{4}}/140{{\,}^{o}}C\]
done
clear
C)
\[NaOH/70{{\,}^{o}}C\]
done
clear
D)
\[NaO{{C}_{2}}{{H}_{5}}/27{{\,}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 95) The equilibrium constant for the reaction \[{{H}_{2}}O(g)+CO(g)\rightleftharpoons {{H}_{2}}(g)+C{{O}_{2}}(g)\] 64. The rate constant for the forward reaction is 160, the rate constant for the backward reaction is:
A)
0.4
done
clear
B)
2.5
done
clear
C)
6.2
done
clear
D)
\[10.24\times {{10}^{3}}\]
done
clear
View Answer play_arrow
question_answer 96) Aniline reacts with chloroform in the presence of alkali to give a product with very bad smell. The test is called:
A)
Isocyanide test
done
clear
B)
Silver mirror test
done
clear
C)
Baeyers test
done
clear
D)
Lucas test
done
clear
View Answer play_arrow
question_answer 97) An organic compound reacts (i) with metallic sodium to liberate hydrogen and(ii) with \[\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\]solution to liberate \[\text{C}{{\text{O}}_{2}}.\] compound is:
A)
alcohol
done
clear
B)
carboxylic acid
done
clear
C)
ether
done
clear
D)
an ester
done
clear
View Answer play_arrow
question_answer 98) The compound formed when nitrobenzene is reduced with \[\text{LiAl}{{\text{H}}_{\text{4}}}\] is:
A)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}-NHOH\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}N=N{{C}_{6}}{{H}_{5}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}NHNH{{C}_{6}}{{H}_{5}}\]
done
clear
View Answer play_arrow
question_answer 99) At \[\text{27}{{\,}^{\text{o}}}\text{C}\] one mole of an ideal gas exerted a pressure of 0.821 atmosphere. What is its volume in liters (\[\text{R = 0}\text{.0821}\,\,\text{L}\]atm \[\text{mo}{{\text{l}}^{-1}}{{K}^{-1}}\]?
A)
300
done
clear
B)
30
done
clear
C)
0.3
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 100) What is the oxidation number of chromium in potassium dichromate?
A)
+7
done
clear
B)
+5
done
clear
C)
+6
done
clear
D)
+4
done
clear
View Answer play_arrow
question_answer 101) An example of terrestrial protozoan is:
A)
Vorliceiia
done
clear
B)
Giardia
done
clear
C)
Didymium
done
clear
D)
Plasmodium
done
clear
View Answer play_arrow
question_answer 102) The infective form of malarial parasite is:
A)
multinucleate sporozoite
done
clear
B)
cryptomerozoite
done
clear
C)
cryptozoite
done
clear
D)
uninucieate sporozoite
done
clear
View Answer play_arrow
question_answer 103) The band of cilia present on the dorsal wall of vestibule of Paramecium is celled:
A)
penniculus
done
clear
B)
ambulacrum
done
clear
C)
kinety
done
clear
D)
quadrulus
done
clear
View Answer play_arrow
question_answer 104) The duration of cycle of golgi in Plasmodium malariae:
A)
24 hours
done
clear
B)
48 hours
done
clear
C)
36 to 48 hours
done
clear
D)
72 hours
done
clear
View Answer play_arrow
question_answer 105) In Euglena, the proteinaceous substance that surrounds the slender part of chromatophore:
A)
pyrenophore
done
clear
B)
pyrenoid
done
clear
C)
paramylem
done
clear
D)
pyrethroid
done
clear
View Answer play_arrow
question_answer 106) Phlebotomus argenlipus aids in the spread of:
A)
kala- azar
done
clear
B)
malignent tertian fever
done
clear
C)
gamlia fever
done
clear
D)
plague
done
clear
View Answer play_arrow
question_answer 107) In sponges, circulation of waler in flagellated chamber is facilitated by:
A)
amoebocytes
done
clear
B)
pinacocytes
done
clear
C)
clioanocytes
done
clear
D)
miocyles
done
clear
View Answer play_arrow
question_answer 108) An animal, which is diploblastic and radially symmertrical is:
A)
Meanderma
done
clear
B)
Pherelima
done
clear
C)
Ascaris
done
clear
D)
Spongilla
done
clear
View Answer play_arrow
question_answer 109) In coeienterates, the mouth in medusa is located on:
A)
hypostome
done
clear
B)
manubrium
done
clear
C)
velum
done
clear
D)
basal disc
done
clear
View Answer play_arrow
question_answer 110) The number of myonemes in muscle tails of epitheliomuscular cells:
A)
2
done
clear
B)
4
done
clear
C)
8
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 111) The disease caused by a helminth parasite of man that has a gastropoda member as an intermediate host:
A)
Ascariasis
done
clear
B)
Biiharzsasis
done
clear
C)
Taeniasis
done
clear
D)
Enterobiasis
done
clear
View Answer play_arrow
question_answer 112) The number of tastes in each mature proglottid of Taenia is:
A)
one pair
done
clear
B)
two pairs
done
clear
C)
three pairs
done
clear
D)
many pairs
done
clear
View Answer play_arrow
question_answer 113) The function of Mehalis glands in Taeniais:
A)
production of vitalline cells
done
clear
B)
storage of sperm
done
clear
C)
formation ofembryophore
done
clear
D)
lubrication of uterus
done
clear
View Answer play_arrow
question_answer 114) An example of a free living platy helminth with a ciliated body:
A)
Fasciola
done
clear
B)
Schistosoma
done
clear
C)
Ditgesia
done
clear
D)
Enlerobmm
done
clear
View Answer play_arrow
question_answer 115) The host animals for the members of Myzostomida belong to the class:
A)
Holothuroidea
done
clear
B)
Echinoidea
done
clear
C)
Crinoidea
done
clear
D)
Stella roidea
done
clear
View Answer play_arrow
question_answer 116) Leech with setae is:
A)
Hirudinaia
done
clear
B)
Hirude
done
clear
C)
Acanthobdella
done
clear
D)
Pantobdela
done
clear
View Answer play_arrow
question_answer 117) The structural adaptation found in the alimentary canal of cockroach for the conservation of water:
A)
presence of cuticle in the gigsand
done
clear
B)
peritrophic membrane in masenthon
done
clear
C)
stomodael wall
done
clear
D)
rectal papiilae
done
clear
View Answer play_arrow
question_answer 118) In one of the below given the first pair of legs modified into poisonous claws:
A)
Limulus
done
clear
B)
Spiroin- eplus
done
clear
C)
Scolopendra
done
clear
D)
Peripolus
done
clear
View Answer play_arrow
question_answer 119) The pincer like structures present on the stipes of maxillaof cockroach are:
A)
galea
done
clear
B)
cardo
done
clear
C)
lacinia
done
clear
D)
para glossa
done
clear
View Answer play_arrow
question_answer 120) In cockroach, the gland that helps in the formation of oolheca is:
A)
shell gland
done
clear
B)
mushroom gland
done
clear
C)
conglobate gland
done
clear
D)
collaterial glands
done
clear
View Answer play_arrow
question_answer 121) Spiroslreptus belongs to the class:
A)
Chiiopoda
done
clear
B)
Crustacea
done
clear
C)
Insecia
done
clear
D)
Diplopoda
done
clear
View Answer play_arrow
question_answer 122) In cockroach, the female genital pore is located at:
A)
between 9 - 10 terga
done
clear
B)
in the centre of 8th sternum
done
clear
C)
between 9 - 10 sternum
done
clear
D)
below 10th sternum as between - paraprocts
done
clear
View Answer play_arrow
question_answer 123) A cephalopod without a shell:
A)
Sepia
done
clear
B)
Loligo
done
clear
C)
Octopus
done
clear
D)
Nautilus
done
clear
View Answer play_arrow
question_answer 124) Which of the following is a sea urchin:
A)
Emerobhim
done
clear
B)
Echis
done
clear
C)
Eclmms
done
clear
D)
Echneis
done
clear
View Answer play_arrow
question_answer 125) Development of blastopore into anus is seen in:
A)
Hydra
done
clear
B)
Octopus
done
clear
C)
Nereis
done
clear
D)
Ophiolhrix
done
clear
View Answer play_arrow
question_answer 126) Presence of claspers is an important character in:
A)
Sphyrna
done
clear
B)
Hippocampus
done
clear
C)
Cyanoglossus
done
clear
D)
Echenis
done
clear
View Answer play_arrow
question_answer 127) In one of the following fishes, dorsal fin is modified into a sucker:
A)
Neoceratodus
done
clear
B)
Hippocampus
done
clear
C)
Torpedo
done
clear
D)
Echeneis
done
clear
View Answer play_arrow
question_answer 128) In frog coracoid fenestra is associated with:
A)
pelvic girdle
done
clear
B)
pectoral girdle
done
clear
C)
skull
done
clear
D)
vertebral column
done
clear
View Answer play_arrow
question_answer 129) In frog the innominate vein is formed by the union of:
A)
lingual vein and mandibular vein
done
clear
B)
sub scapular and internal jugular vein
done
clear
C)
brachial vein and musenio cuianeous vein
done
clear
D)
external jugular and sub clavian vein
done
clear
View Answer play_arrow
question_answer 130) In frog astragalus is a part of:
A)
wrist
done
clear
B)
pectoral girdle
done
clear
C)
ankle
done
clear
D)
pelvic girdle
done
clear
View Answer play_arrow
question_answer 131) In frog skull, the bone that connects sphenethmoid with maxilla:
A)
pterygoid
done
clear
B)
squamosal
done
clear
C)
palatine
done
clear
D)
pre maxilla
done
clear
View Answer play_arrow
question_answer 132) In frog, the nerve that innervates the retractor bulli muscles of the eye:
A)
optic nerves
done
clear
B)
oculomotor
done
clear
C)
pathetic nerve
done
clear
D)
abducens nerve
done
clear
View Answer play_arrow
question_answer 133) In frog, stratum spongiosum is made up of:
A)
dense connective tissue
done
clear
B)
loose fibrous connective tissue
done
clear
C)
fasienti
done
clear
D)
stratified epithelium
done
clear
View Answer play_arrow
question_answer 134) The blood supply to the eyes of frog is through:
A)
lingual artery
done
clear
B)
internal carotid artery
done
clear
C)
palatine
done
clear
D)
occipito ventral artery
done
clear
View Answer play_arrow
question_answer 135) In frog the hypoglossal nerves are:
A)
1stpair of spinal nerves
done
clear
B)
12thpair of cranial nerves
done
clear
C)
9th pair of spinal nerves
done
clear
D)
7th pair of cranial nerves
done
clear
View Answer play_arrow
question_answer 136) The animal unique to the country where Apteryx is the national bird:
A)
Hatteriu
done
clear
B)
Lepidosiren
done
clear
C)
Latimeria
done
clear
D)
Tylolriton
done
clear
View Answer play_arrow
question_answer 137) One of following pairs of animals is found in country where Neoceralodus is found:
A)
Myrmecobius and Emu
done
clear
B)
Rhea and Didelphis
done
clear
C)
Tinarnus and Lepidosiren
done
clear
D)
Naja and Bungarus
done
clear
View Answer play_arrow
question_answer 138) The animal in which mammary glands does not have teats:
A)
Tachyglossus
done
clear
B)
Thylacinus
done
clear
C)
Noioiyclus
done
clear
D)
Armadillo
done
clear
View Answer play_arrow
question_answer 139) Ratchet movement is associated with:
A)
muscle contraction
done
clear
B)
ciliary movement
done
clear
C)
protoplasmic movement
done
clear
D)
flagellar movement
done
clear
View Answer play_arrow
question_answer 140) Tigroid granules are present in:
A)
muscle cell
done
clear
B)
nerve cell
done
clear
C)
gland cell
done
clear
D)
sperm cell
done
clear
View Answer play_arrow
question_answer 141) The variation in the quantity of cream and fat in the milk produced by the off- springs of two oxen. born of common parents is:
A)
independent assortment of genes
done
clear
B)
mutation
done
clear
C)
environmental effect
done
clear
D)
food habit
done
clear
View Answer play_arrow
question_answer 142) In andalusian fowls, if a cross is made between black fowl and splashed white, what will be the phenolypic ratio in F2:
A)
1: 1: 2
done
clear
B)
2: 1: 1
done
clear
C)
3: 1
done
clear
D)
1: 2: 1
done
clear
View Answer play_arrow
question_answer 143) Coenozoic era refers to the age of:
A)
fishes and reptiles
done
clear
B)
amphibians and reptriles
done
clear
C)
birds and mammals
done
clear
D)
amphibians and fishes
done
clear
View Answer play_arrow
question_answer 144) Eusthernopteron connects:
A)
fishes and amphibians
done
clear
B)
amphibians and reptiles
done
clear
C)
reptiles and birds
done
clear
D)
birds and mammals
done
clear
View Answer play_arrow
question_answer 145) One of the following is known as "oil sardine":
A)
Rastrelliger kanegunta
done
clear
B)
Atroplus surtensis
done
clear
C)
Harpodon rechenus
done
clear
D)
Surdmella longiceps
done
clear
View Answer play_arrow
question_answer 146) Identify the cat fish from the below given fishes:
A)
Calla catla
done
clear
B)
Cyprinus carpeo
done
clear
C)
Cirrhina nirigala
done
clear
D)
Wallago attu
done
clear
View Answer play_arrow
question_answer 147) Lymphoid leucosis in poultry is a:
A)
viral disease
done
clear
B)
bacterial disease
done
clear
C)
fungal disease
done
clear
D)
helminth disease
done
clear
View Answer play_arrow
question_answer 148) Chicks of the first 2 weeks in the brooder hoven are usually susceptible to one of the following:
A)
Mareks disease
done
clear
B)
fowl pox
done
clear
C)
ranikhet disease
done
clear
D)
gomboora disease
done
clear
View Answer play_arrow
question_answer 149) The metal pollutant held responsible for minimata disease is:
A)
mercury
done
clear
B)
cadmium
done
clear
C)
arsenic
done
clear
D)
zinc
done
clear
View Answer play_arrow
question_answer 150) Sterility is caused by this pollutant in water:
A)
cadmium
done
clear
B)
mercury
done
clear
C)
copper
done
clear
D)
manganese
done
clear
View Answer play_arrow
question_answer 151) The earlier institute established at Pusa is now called:
A)
BIS
done
clear
B)
lCAR
done
clear
C)
lARl
done
clear
D)
CSIR
done
clear
View Answer play_arrow
question_answer 152) Balancing roots areseen in :
A)
Dolichos
done
clear
B)
Pislia
done
clear
C)
Piper
done
clear
D)
Vanda
done
clear
View Answer play_arrow
question_answer 153) Root, stem and leaf are modified in:
A)
Dracaena
done
clear
B)
Aloe
done
clear
C)
Asparagus
done
clear
D)
Opuntia
done
clear
View Answer play_arrow
question_answer 154) The following feature, is common in both Balanophora and Casuarina:
A)
parasitism
done
clear
B)
tree habit
done
clear
C)
seale leaves
done
clear
D)
phyllode
done
clear
View Answer play_arrow
question_answer 155) The following plant produces both offsets and bulbils:
A)
Chrysanthemum
done
clear
B)
Oxalis
done
clear
C)
Agave
done
clear
D)
Bryophyllum
done
clear
View Answer play_arrow
question_answer 156) Inflorescence in Jatropha is:
A)
monochasialscorpioid cyme
done
clear
B)
polychasial cyme
done
clear
C)
dischasial cyme
done
clear
D)
solitary flower
done
clear
View Answer play_arrow
question_answer 157) In Fabaceae the floral structures which encloses the essential organs are:
A)
posterior petals
done
clear
B)
lateral petals
done
clear
C)
sepals
done
clear
D)
anterior petals
done
clear
View Answer play_arrow
question_answer 158) Geitonogamyis found in:
A)
Sorghum
done
clear
B)
Erythrina
done
clear
C)
Hydrilla
done
clear
D)
Maize
done
clear
View Answer play_arrow
question_answer 159) Modification of bract in Typhonium is:
A)
seale
done
clear
B)
spa the
done
clear
C)
involucre
done
clear
D)
glume
done
clear
View Answer play_arrow
question_answer 160) Free central placentation is seen in:
A)
Raphanus
done
clear
B)
Dianthus
done
clear
C)
Dolichos
done
clear
D)
Helianthus
done
clear
View Answer play_arrow
question_answer 161) The position of microphyle found in an orthotropous ovule are turned down about160° with reference to funicle. In addition, the ovule and the embryosac are curved. Such a type of ovule is:
A)
anatropous
done
clear
B)
campylotropous
done
clear
C)
hemitropous
done
clear
D)
amphitropous
done
clear
View Answer play_arrow
question_answer 162) Scale bark is seen in:
A)
Terminalia
done
clear
B)
Mangifera
done
clear
C)
Psidium
done
clear
D)
neem
done
clear
View Answer play_arrow
question_answer 163) The scientific term which is not related to the primary constriction of a chromosome is:
A)
centromere
done
clear
B)
kinomere
done
clear
C)
kinetochore
done
clear
D)
chromo mere
done
clear
View Answer play_arrow
question_answer 164) Asafoetidaisa:
A)
tannin
done
clear
B)
oleoresin
done
clear
C)
latex
done
clear
D)
gum resin
done
clear
View Answer play_arrow
question_answer 165) The haploid chromosome number in Succharum is as many times greater in the following as that of Zea mays:
A)
4 fold
done
clear
B)
2 fold
done
clear
C)
6 fold
done
clear
D)
3 fold
done
clear
View Answer play_arrow
question_answer 166) Tyloses are observed in the lumen of:
A)
tracheids
done
clear
B)
sieve tubes
done
clear
C)
fibretracheids
done
clear
D)
xylem vessels
done
clear
View Answer play_arrow
question_answer 167) The genetical importance of raisinghaploids in higher plants through tissue liestheir use in the production of:
A)
homozygous diploid
done
clear
B)
heterozygous diploids
done
clear
C)
plants that do not produce flowers
done
clear
D)
hybrids
done
clear
View Answer play_arrow
question_answer 168) As compared to sclernchyma, parenchymadoes not function in :
A)
gas exchange
done
clear
B)
nutrient transport
done
clear
C)
mechanical support
done
clear
D)
support and gas exchange
done
clear
View Answer play_arrow
question_answer 169) The stele, in the root of the following is tetrarch :
A)
Nicotiana
done
clear
B)
Helianthus
done
clear
C)
Gossypium
done
clear
D)
Trapa
done
clear
View Answer play_arrow
question_answer 170) Trigonella belongs to the family:
A)
Brassicaceae
done
clear
B)
Caesalpinaceae
done
clear
C)
Fabaceae
done
clear
D)
Asteraceae
done
clear
View Answer play_arrow
question_answer 171) The inflorescence in case of Yucca belonging to Liliaceous is:
A)
umbel
done
clear
B)
calkin
done
clear
C)
panicle
done
clear
D)
compound umbel
done
clear
View Answer play_arrow
question_answer 172) Virus cause swollen shoot disease in:
A)
tomato
done
clear
B)
brinjal
done
clear
C)
potato
done
clear
D)
cocoa
done
clear
View Answer play_arrow
question_answer 173) The plant without tuberous roots:
A)
Raphanussativus
done
clear
B)
Asparagus racemoisus
done
clear
C)
Solanumtuberosum
done
clear
D)
Ipomoea butatas
done
clear
View Answer play_arrow
question_answer 174) The general method of propagation for cultivated varieties of Musa is Through :
A)
sucker
done
clear
B)
stem cuttings
done
clear
C)
corus
done
clear
D)
seeds
done
clear
View Answer play_arrow
question_answer 175) LRG 30 is an important variety of the following crop plant grown in A.P.
A)
sorghum
done
clear
B)
red gram
done
clear
C)
bajra
done
clear
D)
groundnut
done
clear
View Answer play_arrow
question_answer 176) The bacterium Eolelovibriobacterio virus is:
A)
autotroph
done
clear
B)
parasite
done
clear
C)
saprophyte
done
clear
D)
symbiont
done
clear
View Answer play_arrow
question_answer 177) Which of the following pathogen do not cause smut disease:
A)
Ustilago
done
clear
B)
Phyllactmia
done
clear
C)
Sphacelotheca
done
clear
D)
Tiilelia
done
clear
View Answer play_arrow
question_answer 178) Butler extensively studied the following disease:
A)
red rot of sugarcane
done
clear
B)
smut disease of sorghum
done
clear
C)
blast disease of rice
done
clear
D)
tikka disease of ground nut
done
clear
View Answer play_arrow
question_answer 179) In the classification of plants, the presence of the following is a primitive character:
A)
zygomorphic flower
done
clear
B)
epigynous flower
done
clear
C)
unisexual flower
done
clear
D)
bisexual flower
done
clear
View Answer play_arrow
question_answer 180) Protoxylem in Selaginella stem is:
A)
endarch
done
clear
B)
mesarch
done
clear
C)
exarch
done
clear
D)
monarch
done
clear
View Answer play_arrow
question_answer 181) In Agaricus during its developmental stages, the cells of the following contain one nucleus:
A)
secondary mycelium
done
clear
B)
primary mycelium only
done
clear
C)
secondary and tertiary mycelium
done
clear
D)
primary and secondary mycelium
done
clear
View Answer play_arrow
question_answer 182) The Calyptra in Funaria is formed from:
A)
archegonium
done
clear
B)
antheridium
done
clear
C)
capsule
done
clear
D)
zygote
done
clear
View Answer play_arrow
question_answer 183) The following species of Cycas is recorded in the forests of A.P.
A)
Cycasrevoluta
done
clear
B)
Cycaspectinata
done
clear
C)
Cycasbeddomei
done
clear
D)
Cycasthomsoni
done
clear
View Answer play_arrow
question_answer 184) Wheat variety introduced from Australia:
A)
Ridley
done
clear
B)
Sioux
done
clear
C)
Texas
done
clear
D)
Dixie
done
clear
View Answer play_arrow
question_answer 185) The results obtained by mass selection are temporary because:
A)
heterozygosity leads to seggregation of character
done
clear
B)
varieties are weak and less productive
done
clear
C)
multilocation adaptability is poor
done
clear
D)
cross pollination is controlled
done
clear
View Answer play_arrow
question_answer 186) The scientist, who first carried putexperiments on hybrid vigour in maize:
A)
Mendel
done
clear
B)
Shull
done
clear
C)
Johanson
done
clear
D)
Kobrenth
done
clear
View Answer play_arrow
question_answer 187) An example of planktonic flora of pondecosystem:
A)
diatoms
done
clear
B)
Crustations
done
clear
C)
Hydrilla
done
clear
D)
Chara
done
clear
View Answer play_arrow
question_answer 188) Lotic ecosystem refers to:
A)
static water system
done
clear
B)
ecosystem of flowering water
done
clear
C)
ecosystem of estuaries
done
clear
D)
deep marine water systems
done
clear
View Answer play_arrow
question_answer 189) The following is a xerophyte, containing airspaces in the stem and roots:
A)
Eichhornia
done
clear
B)
Opuntia
done
clear
C)
Aloe
done
clear
D)
Rhizophora
done
clear
View Answer play_arrow
question_answer 190) In the following, the primary consumer is :
A)
diatoms
done
clear
B)
Pediastrum
done
clear
C)
carnivores
done
clear
D)
rotifers
done
clear
View Answer play_arrow
question_answer 191) The morphogenetic property of cytokinin was first experimentally proved by:
A)
Hannig
done
clear
B)
Guhaand Maheshwari
done
clear
C)
Skoogand Miller
done
clear
D)
Went
done
clear
View Answer play_arrow
question_answer 192) In a plant physiology experiment, a green plant kepi in light is found releasing more units of 18 03. Which of the following compounds would have been supplied to the plant?
A)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]containing \[{{O}_{18}}\]
done
clear
B)
\[C{{O}_{2}}\]containing\[{{O}_{18}}\]
done
clear
C)
O zone containing \[{{O}_{18}}\]
done
clear
D)
\[{{H}_{2}}O\]containing\[{{O}_{18}}\]
done
clear
View Answer play_arrow
question_answer 193) The number of oxidation involving NADH in krebs cycle is:
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 194) In non-cyclic photophosphorylation, the site of ATP synthesis Taking place between the following electron carriers is:
A)
PS- II and Q
done
clear
B)
Cyt \[{{b}_{6}}\]and f
done
clear
C)
Cyt \[{{b}_{6}}\] and plast ocyanin
done
clear
D)
PS -I and ferredoxin
done
clear
View Answer play_arrow
question_answer 195) Stomatal closure is induced by :
A)
indole acetic acid
done
clear
B)
naphthalene acetic acid
done
clear
C)
indole butyric acid
done
clear
D)
phenyl mercuric acid
done
clear
View Answer play_arrow
question_answer 196) Which of the following statements is notconsideredasacriteria for essentiality of elements?
A)
In its absent plants fairto reproduce
done
clear
B)
It plays a role that is replaceable
done
clear
C)
It is necessary at all stage of development
done
clear
D)
It affects the metabolism directly
done
clear
View Answer play_arrow
question_answer 197) Munch mass flow hypothesis mainly explain the long distance transport of the following:
A)
mineral salts
done
clear
B)
sugars
done
clear
C)
protein
done
clear
D)
starch
done
clear
View Answer play_arrow
question_answer 198) The enzyme code of enzyme 2, 7, 11 refers to the following main group :
A)
ligase
done
clear
B)
hydrolase
done
clear
C)
transferase
done
clear
D)
lyase
done
clear
View Answer play_arrow
question_answer 199) The following hormone induces dormancyin potatoes :
A)
IAA
done
clear
B)
gibberellin
done
clear
C)
abscisicacid
done
clear
D)
zeatin
done
clear
View Answer play_arrow
question_answer 200) The following is the constituent to nitrogenase:
A)
magnesium
done
clear
B)
molybdenum
done
clear
C)
manganese
done
clear
D)
potassium
done
clear
View Answer play_arrow