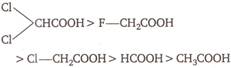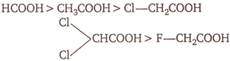question_answer 1)
The value of current \[I\] in the circuit will be
A)
1.7 A
done
clear
B)
2.1 A
done
clear
C)
3 A
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 2) Two plane mirrors are inclined to each other at angle \[60{}^\circ \]. Ray incident on first mirror parallel to the second mirror becomes parallel to first mirror after reflection. Angle of deviation is
A)
\[30{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[120{}^\circ \]
done
clear
View Answer play_arrow
question_answer 3)
The equivalent capacitance of the circuit will be
A)
\[1.\mu F\]
done
clear
B)
\[3\mu F\]
done
clear
C)
\[9\mu F\]
done
clear
D)
\[4\mu F\]
done
clear
View Answer play_arrow
question_answer 4)
If the equivalent capacitance between A and B is \[1\mu F,\] then the value of C will be
A)
\[2\mu F\]
done
clear
B)
\[4\mu F\]
done
clear
C)
\[3\mu F\]
done
clear
D)
\[6\mu F\]
done
clear
View Answer play_arrow
question_answer 5)
If the input is given between A and C, then the output at the ends of R will be
A)
fully rectified
done
clear
B)
half rectified
done
clear
C)
AC
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 6)
If the acceleration-displacement graph of simple harmonic motion of a particle is given, then the time period of the particle will be
A)
2n
done
clear
B)
3n
done
clear
C)
An
done
clear
D)
5n
done
clear
View Answer play_arrow
question_answer 7)
Figure shows four, p-V diagram for the given sample of gas. In which case no exchange of eat occurs with the sample?
A)
P
done
clear
B)
Q
done
clear
C)
R
done
clear
D)
S
done
clear
View Answer play_arrow
question_answer 8) The charge on a particle Y is double the charge on particle X. These two particles X and Y after being accelerated through the same potential difference enter a region of uniform magnetic field and describe circular paths of radii J? and \[{{R}_{2}}\] respectively. The ratio of the mass of X to that of V is
A)
\[{{\text{(}{{\text{R}}_{\text{1}}}\text{/}{{\text{R}}_{\text{2}}}\text{)}}^{\text{2}}}\]
done
clear
B)
\[{{\text{(}{{\text{R}}_{\text{1}}}\text{/2}{{\text{R}}_{\text{2}}}\text{)}}^{\text{2}}}\]
done
clear
C)
\[\text{R}\,_{1}^{2}\text{/2R}_{2}^{2}\]
done
clear
D)
\[\text{2}{{\text{R}}_{\text{1}}}\text{/}{{\text{R}}_{\text{2}}}\]
done
clear
View Answer play_arrow
question_answer 9) In a step up transformer the turn ratio is 1:10. A resistance of 200\[\Omega \] connected across the secondary is drawing a current of 0.5 A. What is the primary voltage and current?
A)
50 V, 1 A
done
clear
B)
10 V, 5 A
done
clear
C)
25V, 4 A
done
clear
D)
20V, 2 A
done
clear
View Answer play_arrow
question_answer 10) Dimensions of \[{{\varepsilon }_{0}}\] are
A)
\[\text{ }\!\![\!\!\text{ M}{{\text{L}}^{\text{-1}}}{{\text{T}}^{\text{2}}}\text{A }\!\!]\!\!\text{ }\]
done
clear
B)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{2}}{{\text{L}}^{\text{-3}}}{{\text{T}}^{\text{2}}}\text{A }\!\!]\!\!\text{ }\]
done
clear
C)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{-2}}{{\text{L}}^{\text{-3}}}{{\text{T}}^{4}}{{\text{A}}^{2}}\text{ }\!\!]\!\!\text{ }\]
done
clear
D)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{2}}{{\text{L}}^{\text{3}}}{{\text{T}}^{\text{-2}}}\text{A }\!\!]\!\!\text{ }\]
done
clear
View Answer play_arrow
question_answer 11) Dimensions of \[\frac{R}{L}\] are
A)
\[\text{ }\!\![\!\!\text{ }{{\text{T}}^{\text{2}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
B)
\[\text{ }\!\![\!\!\text{ T }\!\!]\!\!\text{ }\]
done
clear
C)
\[\text{ }\!\![\!\!\text{ }{{\text{T}}^{-2}}\text{ }\!\!]\!\!\text{ }\]
done
clear
D)
\[\text{ }\!\![\!\!\text{ }{{\text{T}}^{-2}}\text{ }\!\!]\!\!\text{ }\]
done
clear
View Answer play_arrow
question_answer 12) The unit of magnetic moment is
A)
A-m2
done
clear
B)
A-m
done
clear
C)
A-m3
done
clear
D)
kg-m3
done
clear
View Answer play_arrow
question_answer 13) In \[\text{SI}\] system the unit of dipole moment is
A)
C-m
done
clear
B)
C-m2
done
clear
C)
C/m2
done
clear
D)
C/m
done
clear
View Answer play_arrow
question_answer 14) Electron volt is a unit of
A)
potential
done
clear
B)
charge
done
clear
C)
power
done
clear
D)
energy
done
clear
View Answer play_arrow
question_answer 15) Which of the following can be written as \[\text{kg}\,\text{-}\,{{\text{m}}^{\text{2}}}{{\text{A}}^{\text{-2}}}{{\text{T}}^{\text{-2}}}\,\text{?}\]
A)
Inductance
done
clear
B)
Reactance
done
clear
C)
Capacitance
done
clear
D)
Resistance
done
clear
View Answer play_arrow
question_answer 16) If the frequency of \[{{K}_{a,}}\] .X-ray of the element of atomic number 31 is/, then the frequency of\[{{K}_{a,}}\] X-ray for atomic number 51 is
A)
\[\text{25/9}f\]
done
clear
B)
\[\text{16/25}f\]
done
clear
C)
\[\text{9/25}f\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 17) A bird weighing 1 kg sitting on the base of a wire mesh cage weighing 1.5 kg. The bird starts flying in the cage. The weight of the bird cage assembly will be
A)
1250 g
done
clear
B)
1500 g
done
clear
C)
1750g
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 18) If the escape velocity for a monkey from the earth surface is 11.2 km/s, then the escape velocity for the elephant is
A)
less than 11.2
done
clear
B)
more than 11.2
done
clear
C)
11.2 km/s
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 19) If the sphere of iron is heated, then its
A)
density decreases
done
clear
B)
volume increases
done
clear
C)
radius decreases
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 20) If a bar magnet is droping through the copper ring, then its velocity
A)
decreases
done
clear
B)
increases
done
clear
C)
remain unaffected
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 21) The power factor of a good choke coil is
A)
nearly zero
done
clear
B)
exactly zero
done
clear
C)
nearly one
done
clear
D)
exactly one
done
clear
View Answer play_arrow
question_answer 22) If two spheres of same masses and radius are brought in contact, then the force of attraction between them will be proportional to
A)
r2
done
clear
B)
r3
done
clear
C)
r6
done
clear
D)
r4
done
clear
View Answer play_arrow
question_answer 23) Capacitors are used for
A)
AC
done
clear
B)
DC
done
clear
C)
Both AC and DC
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 24) The ratio of current flowing through primary and secondary of a transformers depends on
A)
number of turns
done
clear
B)
resistance
done
clear
C)
capacitance
done
clear
D)
inductance
done
clear
View Answer play_arrow
question_answer 25) A proton, a deuteron and an a-particle having the same momentum, enters a region of uniform electric field between the parallel plates of a capacitor. If the electric field is perpendicular to the initial direction of the particles. Then, the ratio of deviations of these particles
A)
1:2:8
done
clear
B)
1:2:4
done
clear
C)
1:2:2
done
clear
D)
1:1:2
done
clear
View Answer play_arrow
question_answer 26) If a radioactive substance decays \[\frac{1}{16}\]th of its original amount in 2 h, then the half-life of that substance is
A)
15 min
done
clear
B)
30 min
done
clear
C)
45 min
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 27) If on applying the potential of 20 V on a conductor its conductance becomes 8 \[{{(\Omega )}^{-1}},\] then the current flowing through it will be
A)
120 A
done
clear
B)
160 A
done
clear
C)
90 A
done
clear
D)
80 A
done
clear
View Answer play_arrow
question_answer 28)
Equivalent resistance of the given circuit is
A)
R
done
clear
B)
R/2
done
clear
C)
R/4
done
clear
D)
R/6
done
clear
View Answer play_arrow
question_answer 29) In a biprism experiment by using light of wavelength \[5000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\], 5 mm wide fringes are obtained on a screen 1.0 m away from the coherent sources. The separation between the two coherent sources is
A)
1.0 mm
done
clear
B)
0.1 mm
done
clear
C)
0.05 mm
done
clear
D)
0.01 mm
done
clear
View Answer play_arrow
question_answer 30) The power of lens used by a short-sighted person is -2 D. Find the maximum distance of an object, which he can see without spectacles
A)
25 cm
done
clear
B)
50 cm
done
clear
C)
100cm
done
clear
D)
10cm
done
clear
View Answer play_arrow
question_answer 31) Kirchhoffs first law\[(\Sigma i=0)\] expresses the conservation of
A)
energy
done
clear
B)
charge
done
clear
C)
momentum
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 32) In p-type semiconductors majority charge carriers are
A)
electrons
done
clear
B)
holes
done
clear
C)
neutrons
done
clear
D)
protons
done
clear
View Answer play_arrow
question_answer 33) A 1 µF capacitance of TV is subjected to 4000 V potential difference. The energy stored in the capacitor is
A)
8 J
done
clear
B)
16 J
done
clear
C)
\[4\times {{10}^{-3}}\text{J}\]
done
clear
D)
\[2\times {{10}^{-3}}\text{J}\]
done
clear
View Answer play_arrow
question_answer 34) A body moves a distance of 10 m along a straight line under the action of a force of 5 N. If the work done is 25 J the angle which the force makes with the direction of motion of the body is
A)
\[0{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[90{}^\circ \]
done
clear
View Answer play_arrow
question_answer 35) A particle of mass m is projected with a velocity making an angle of \[45{}^\circ \] with the horizontal. The magnitude of angular momentum of the projectile about the axis of projection when the particle is at maximum height
A)
zero
done
clear
B)
\[\frac{m{{v}^{3}}}{4\sqrt{2g}}\]
done
clear
C)
\[\frac{m{{v}^{3}}}{\sqrt{2}g}\]
done
clear
D)
\[\frac{m{{v}^{3}}}{2g}\]
done
clear
View Answer play_arrow
question_answer 36) If the moment of inertia of a disc about an axis tangential and parallel to its surface be \[I,\] then what will be the moment of inertia about the axis tangential but perpendicular to the surface?
A)
\[\frac{6}{5}I\]
done
clear
B)
\[\frac{3}{4}I\]
done
clear
C)
\[\frac{3}{2}I\]
done
clear
D)
\[\frac{5}{4}I\]
done
clear
View Answer play_arrow
question_answer 37) The magnetic needle of a tangent galvanometer is deflected by an angle \[30{}^\circ \] due to a magnet. The horizontal component of earth's magnetic field is \[0.34\times {{10}^{-4}}T\] along the plane of the coil. The intensity of magnetic field of magnet is
A)
\[1.96\times {{10}^{-4}}T\]
done
clear
B)
\[1.96\times {{10}^{-5}}T\]
done
clear
C)
\[1.96\times {{10}^{-4}}T\]
done
clear
D)
\[1.96\times {{10}^{-5}}T\]
done
clear
View Answer play_arrow
question_answer 38) A coil of resistance R and inductance L is connected to a battery of emf E volt. The final current in the coil is
A)
E/R
done
clear
B)
E/L
done
clear
C)
\[\sqrt{\text{E/}({{R}^{2}}+{{L}^{2}})}\]
done
clear
D)
\[\sqrt{\frac{EL}{({{R}^{2}}+{{L}^{2}})}}\]
done
clear
View Answer play_arrow
question_answer 39) The refractive indices of violet and red light are 1.54 and 1.52 respectively. If the angle of the prism is \[10{}^\circ \], the angular dispersion is
A)
\[0.02{}^\circ \]
done
clear
B)
\[0.2{}^\circ \]
done
clear
C)
\[3.06{}^\circ \]
done
clear
D)
\[30.6{}^\circ \]
done
clear
View Answer play_arrow
question_answer 40) The focal lengths of the objective and eye-piece of a telescope are respectively 100 cm and 2cm. The moon substends an angle of \[0.5{}^\circ \] at the eye. If it is looked through the telescope, the angle subtended by the moon's image will be
A)
\[100{}^\circ \]
done
clear
B)
\[50{}^\circ \]
done
clear
C)
\[25{}^\circ \]
done
clear
D)
\[10{}^\circ \]
done
clear
View Answer play_arrow
question_answer 41) The temperature of a body on Kelvin scale is found to be x K. When it is measured by Fahrenheit thermometer, it is found to be \[x{}^\circ \] F, then the value of x is
A)
30
done
clear
B)
313
done
clear
C)
574.25
done
clear
D)
301.25
done
clear
View Answer play_arrow
question_answer 42) Two containers of equal volume contain the same gas at pressure \[{{P}_{1}}\] and \[{{P}_{2}}\] and absolute temperature \[{{T}_{1}}\] and \[{{T}_{2}}\] respectively. On joining the vessels the gas reaches a common pressure and common temperature T. The ratio p/T is equal to
A)
\[\frac{{{P}_{1}}}{{{T}_{1}}}+\frac{{{P}_{2}}}{{{T}_{2}}}\]
done
clear
B)
\[\frac{{{P}_{1}}{{T}_{1}}+{{P}_{2}}{{T}_{2}}}{{{({{T}_{1}}+{{T}_{2}})}^{2}}}\]
done
clear
C)
\[\frac{{{P}_{1}}{{T}_{2}}+{{P}_{2}}{{T}_{1}}}{{{({{T}_{1}}+{{T}_{2}})}^{2}}}\]
done
clear
D)
\[\frac{{{P}_{1}}}{2{{T}_{1}}}+\frac{{{P}_{2}}}{2{{T}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 43) \[1c{{m}^{3}}\] of water at its boiling point absorbs 540 cal of heat to become steam with a volume of \[1671\text{ }c{{m}^{3}}.\] If the atmospheric pressure \[=1.013\times {{10}^{5}}N/{{m}^{2}}\] and the mechanical equivalent of heat = 4.19 J/cal, the energy spent in this process in overcoming intermolecular forces is
A)
540 cal
done
clear
B)
40cal
done
clear
C)
500 cal
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 44) N atoms of a radioactive element emit n-alpha particles per second. The half-life of the element is
A)
\[\frac{n}{N}s\]
done
clear
B)
\[\frac{N}{n}s\]
done
clear
C)
\[\frac{0.693N}{n}s\]
done
clear
D)
\[\frac{0.693n}{N}s\]
done
clear
View Answer play_arrow
question_answer 45) Two long parallel wires P and Q are both perpendicular to the plane of the paper with distance 5 m between them. If P and Q carry current of 2.5 A and 5 A respectively in the same direction, then the magnetic field at a point half way between the wires is
A)
\[\frac{\sqrt{3}{{\mu }_{0}}}{2\pi }\]
done
clear
B)
\[\frac{{{\mu }_{0}}}{\pi }\]
done
clear
C)
\[\frac{3{{\mu }_{0}}}{2\pi }\]
done
clear
D)
\[\frac{{{\mu }_{0}}}{2\pi }\]
done
clear
View Answer play_arrow
question_answer 46) An alternating potential of frequency \[f\] is applied on a circuit containing a resistance R and a choke L in series. The impedance of this current is
A)
\[R+2\pi fL\]
done
clear
B)
\[\sqrt{{{R}^{2}}+4{{\pi }^{2}}{{f}^{2}}{{L}^{2}}}\]
done
clear
C)
\[\sqrt{{{R}^{2}}+{{L}^{2}}}\]
done
clear
D)
\[\sqrt{{{R}^{2}}+2\pi fL}\]
done
clear
View Answer play_arrow
question_answer 47) There is a voltmeter in a circuit. In order to triple its range, the resistance of how much value should be used?
A)
2R
done
clear
B)
R/2
done
clear
C)
3R
done
clear
D)
4R
done
clear
View Answer play_arrow
question_answer 48)
A wire is bent in the form of a triangle now the equivalent resistance R between its one end and (he midpoint of the side is
A)
\[\frac{5R}{12}\]
done
clear
B)
\[\frac{7R}{12}\]
done
clear
C)
\[\frac{3R}{12}\]
done
clear
D)
\[\frac{R}{12}\]
done
clear
View Answer play_arrow
question_answer 49) Which of the following is incorrect?
A)
\[{{C}_{V}}=\frac{R}{\gamma -1}\]
done
clear
B)
\[{{C}_{P}}=\frac{\gamma R}{\gamma -1}\]
done
clear
C)
\[\frac{{{C}_{P}}}{{{C}_{v}}}=\gamma \]
done
clear
D)
\[{{C}_{v}}-{{C}_{P}}=2R\]
done
clear
View Answer play_arrow
question_answer 50) 50. A mass of 20 kg moving with a speed of 10 m/ s collides with another stationary mass of 5 kg. As a result of the collision both masses s; together. The kinetic energy of the compos. mass will be
A)
600 J
done
clear
B)
800 J
done
clear
C)
1000J
done
clear
D)
1200J
done
clear
View Answer play_arrow
question_answer 51) The electric field between two parallel plates of a capacitor is \[2.1\times {{10}^{-5}}.\] If a medium is inserted between the plates than the electric field becomes \[1.0\times {{10}^{-5}}.\] Now, the value of dielectric will be
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 52) A ray of light propagates from glass (refracts index = 3/2) to water (refractive index = 4/3). The value of the critical angle is
A)
\[{{\sin }^{1}}\left( \frac{1}{2} \right)\]
done
clear
B)
\[{{\sin }^{-1}}\left( \sqrt{\frac{8}{9}} \right)\]
done
clear
C)
\[{{\sin }^{-1}}\left( \frac{8}{9} \right)\]
done
clear
D)
\[{{\sin }^{-1}}\left( \frac{5}{7} \right)\]
done
clear
View Answer play_arrow
question_answer 53) An electric heater of resistance \[6\,\Omega \] is run are 10 min on a 120 V line. The energy liberated in this period of time is
A)
\[7.2\times {{10}^{3}}J\]
done
clear
B)
\[14.4\times {{10}^{5}}J\]
done
clear
C)
\[43.2\times {{10}^{4}}J\]
done
clear
D)
\[28.8\times {{10}^{4}}J\]
done
clear
View Answer play_arrow
question_answer 54) Lenz's law gives
A)
the magnitude of the induced emf
done
clear
B)
the direction of the induced current
done
clear
C)
both the magnitude and direction of the induced current
done
clear
D)
the magnitude of the induced current
done
clear
View Answer play_arrow
question_answer 55) When two bodies collide elastically, then
A)
kinetic energy of the system alone is conserved
done
clear
B)
only momentum is conserved
done
clear
C)
both energy and momentum are conserved
done
clear
D)
neither energy nor momentum is conserved
done
clear
View Answer play_arrow
question_answer 56) A satellite moves around the earth in a circular orbit of radius r with speed v. If the mass of the satellite is M, its total energy is
A)
\[-1/2M{{v}^{2}}\]
done
clear
B)
\[1/2M{{v}^{2}}\]
done
clear
C)
\[3/2M{{v}^{2}}\]
done
clear
D)
\[M{{v}^{2}}\]
done
clear
View Answer play_arrow
question_answer 57) A telescope, consisting of an objective of focal length 60 cm and a single eye lens of focal length 5 cm is focussed on a distant object is such a way that parallel rays comes out from the eye lens. If the object subtends an angle \[2{}^\circ \] at the objective the angle subtended by the image at the eye is
A)
\[10{}^\circ \]
done
clear
B)
\[24{}^\circ \]
done
clear
C)
\[50{}^\circ \]
done
clear
D)
\[1/6{}^\circ \]
done
clear
View Answer play_arrow
question_answer 58) If the steel bob of a simple pendulum is replaced by a wooden bob, then its time period will
A)
increase
done
clear
B)
decrease
done
clear
C)
remains the same
done
clear
D)
first increases and then decreases
done
clear
View Answer play_arrow
question_answer 59) An electric dipole is placed along the x-axis at the origin 0. A point P is at a distance of 20 cm from this origin such that OP makes an angle \[\text{ }\!\!\pi\!\!\text{ /3}\]with the x-axis. If the electric field at P makes an angle \[\text{ }\!\!\theta\!\!\text{ }\] with the x-axis, the value of \[\text{ }\!\!\theta\!\!\text{ }\] would be
A)
\[\frac{\pi }{3}\]
done
clear
B)
\[\frac{\pi }{3}+{{\tan }^{-1}}\left( \frac{\sqrt{3}}{2} \right)\]
done
clear
C)
\[\frac{2\pi }{3}\]
done
clear
D)
\[{{\tan }^{-1}}\left( \frac{\sqrt{3}}{2} \right)\]
done
clear
View Answer play_arrow
question_answer 60) The wavelength of light in two liquids x and y are \[3500\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\] and \[7000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\], then the critical angle of x relative toy will be
A)
\[60{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[30{}^\circ \]
done
clear
D)
\[15{}^\circ \]
done
clear
View Answer play_arrow
question_answer 61) An ideal transformer has 500 turns in the primary and 2500 in the secondary. The meters of the secondary are indicating 200 V, 8 A, under these conditions. What would the meters of the primary read?
A)
100 V, 16 A
done
clear
B)
40 V, 40 A
done
clear
C)
160 V, 10 A
done
clear
D)
80 V, 20 A
done
clear
View Answer play_arrow
question_answer 62) Sensitivity of potentiometer can be increased by
A)
increasing the emf of the cell
done
clear
B)
increasing the length of the potentiometer wire
done
clear
C)
decreasing the length of the potentiometer wire
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 63) The following four wires are made of same material and same tension is applied on them. Which one will have maximum increase in length?
A)
Length =100 cm. Diameter = 1 mm
done
clear
B)
Length = 50 cm, Diameter = 0.5 mm
done
clear
C)
Length = 200 cm, Diameter = 2 mm
done
clear
D)
Length = 300 cm. Diameter = 3 mm
done
clear
View Answer play_arrow
question_answer 64) Radius of a capillary is \[2\times {{10}^{-3}}m.\text{ }A\] liquid of weight \[6.28\times {{10}^{-4}}N\] may remain in the capillary, then the surface tension of liquid will be
A)
\[5\times {{10}^{-3}}N/m\]
done
clear
B)
\[5\times {{10}^{-2}}N/m\]
done
clear
C)
5 N/m
done
clear
D)
50 N/m
done
clear
View Answer play_arrow
question_answer 65) If two tuning forks A and B are sounded together, they produce 4 beats/s. A is then slightly loaded with wax, they produce 2 beats/s when sounded again. The frequency of A is 256 Hz. The frequency of B will be
A)
250 Hz
done
clear
B)
252 Hz
done
clear
C)
260 Hz
done
clear
D)
262 Hz
done
clear
View Answer play_arrow
question_answer 66) Two waves are given by \[{{y}_{1}}=a\,\sin \,\]\[(\omega t-kx)\]and \[{{y}_{2}}=a\,\cos \]\[(\omega t-kx).\]The phase difference between the two waves is
A)
\[\text{ }\!\!\pi\!\!\text{ /4}\]
done
clear
B)
\[\text{ }\!\!\pi\!\!\text{ }\]
done
clear
C)
\[\text{ }\!\!\pi\!\!\text{ /8}\]
done
clear
D)
\[\text{ }\!\!\pi\!\!\text{ /2}\]
done
clear
View Answer play_arrow
question_answer 67) Period of oscillation of mass attached to a spring and performing SHM is T. The spring is now cut into four equal pieces and the same mass attached to one piece. Now, the period of its simple harmonic oscillations is
A)
2T
done
clear
B)
T/4
done
clear
C)
T/2
done
clear
D)
T
done
clear
View Answer play_arrow
question_answer 68) An electron makes a transition from orbit n = 4 to the orbit n = 2 of a hydrogen atom. The wave number of the emitted radiation (R = Rydberg's constant) will be
A)
16/3R
done
clear
B)
2R/16
done
clear
C)
3R/16
done
clear
D)
4R/16
done
clear
View Answer play_arrow
question_answer 69) A uniform rope of length \[I\] lies on a table. If the coefficient of friction is \[\mu \] then the maximum length \[{{l}_{1}}\] of the part of this rope which can overhang from the edge of the table without sliding down is
A)
\[\frac{1}{\mu }\]
done
clear
B)
\[\frac{1}{\mu +1}\]
done
clear
C)
\[\frac{\mu 1}{1+\mu }\]
done
clear
D)
\[\frac{\mu l}{\mu -1}\]
done
clear
View Answer play_arrow
question_answer 70) The magnetic flux linked with a coil at any instant t is given by \[\phi =5{{t}^{3}}-100t+300,\] the emf induced in the coil after t = 2 s is
A)
-40V
done
clear
B)
40V
done
clear
C)
140V
done
clear
D)
300V
done
clear
View Answer play_arrow
question_answer 71) An ideal heat engine working between temperature \[{{T}_{1}}\] and \[{{T}_{2}}\] has an efficiency \[\eta ,\] the new efficiency if temperature of both the source and sink are doubled, will be
A)
\[\eta /2\]
done
clear
B)
\[\eta \]
done
clear
C)
\[2\eta \]
done
clear
D)
\[3\eta \]
done
clear
View Answer play_arrow
question_answer 72) A 50 g bullet moving with velocity 10 m/s strikes a block of mass 950 g at rest and gets embedded into it. The loss in kinetic energy will be
A)
100%
done
clear
B)
95%
done
clear
C)
5%
done
clear
D)
50%
done
clear
View Answer play_arrow
question_answer 73) The sun radiates energy in all directions. The average radiation received on the earth surface from the sun per second is \[1.4\text{ }kW/{{m}^{3}}.\] The average distance between sun and earth is \[1.5\times {{10}^{11}}m.\] The mass lost by the sun per day
A)
\[4.4\times {{10}^{10}}kg\]
done
clear
B)
\[7.6\times {{10}^{14}}kg\]
done
clear
C)
\[3.8\times {{10}^{11}}kg\]
done
clear
D)
\[3.8\times {{10}^{14}}kg\]
done
clear
View Answer play_arrow
question_answer 74) Every gas behaves as an ideal gas
A)
at high temperature and low pressure
done
clear
B)
at low temperature and high pressure
done
clear
C)
at normal temperature and pressure
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 75) In an AC circuit, the current is given by \[I=5\,\sin (100t-\text{ }\!\!\pi\!\!\text{ /2)}\] and the alternating potential is V = 200 sin (100)C volt. The power consumed in the circuit is
A)
20 W
done
clear
B)
40 W
done
clear
C)
1000 W
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 76) The maximum wavelength of radiation that can produce photoelectric effect in a certain metal is 200 nm. The maximum kinetic energy acquired by electron due to radiation of wavelength 100 nm will be
A)
12.4 eV
done
clear
B)
6.2 eV
done
clear
C)
100 eV
done
clear
D)
200 eV
done
clear
View Answer play_arrow
question_answer 77) The material of wire of potentiometer is
A)
copper
done
clear
B)
steel
done
clear
C)
manganin
done
clear
D)
aluminium
done
clear
View Answer play_arrow
question_answer 78) A liquid cools down from \[70{}^\circ C\] to \[60{}^\circ C\] in 5 min. The time taken to cool it from \[60{}^\circ C\] to \[50{}^\circ C\] will be
A)
5 min
done
clear
B)
lesser than 5 min
done
clear
C)
greater than 5 min
done
clear
D)
lesser or greater than 5 min depending upon the density of the liquid
done
clear
View Answer play_arrow
question_answer 79) There are 50 turns of a wire in every cm length of a long solenoid. If 4 A current is flowing in the solenoid, the approximate value of magnetic field along its axis at an internal point and at one end will be respectively
A)
\[\begin{align} & 12.6\times {{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ & 6.3\times {{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ \end{align}\]
done
clear
B)
\[\begin{align} & 12.6\times {{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ & 25.1\times {{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ \end{align}\]
done
clear
C)
\[\begin{align} & 25.1\times {{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ & 12.6\times {{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ \end{align}\]
done
clear
D)
\[\begin{align} & 25.1\times {{10}^{-5}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ & 12.6\times {{10}^{-5}}\text{Wb/}{{\text{m}}^{\text{2}}} \\ \end{align}\]
done
clear
View Answer play_arrow
question_answer 80) Monochromatic light of frequency \[5\times {{10}^{14}}Hz\] travelling in vacuum enters medium of refractive index 1.5. Its wavelength in the medium is
A)
\[4000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[5000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[6000\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[5500\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 81) The vector form of Biot-Savart's law for a current carrying element is
A)
\[d\,\mathbf{\vec{B}}=\frac{{{\mu }_{0}}}{4\pi }\frac{Id\mathbf{\vec{l}}\,\sin \,\phi }{{{r}^{2}}}\]
done
clear
B)
\[d\,\mathbf{\vec{B}}=\frac{{{\mu }_{0}}}{4\pi }\frac{Idl\times \,\mathbf{\hat{r}}}{{{r}^{2}}}\]
done
clear
C)
\[d\,\mathbf{\vec{B}}=\frac{{{\mu }_{0}}}{4\pi }\frac{Id\mathbf{\vec{l}}\times \,\mathbf{\hat{r}}}{{{r}^{3}}}\]
done
clear
D)
\[d\,\mathbf{\vec{B}}=\frac{{{\mu }_{0}}}{4\pi }\frac{Id\mathbf{\vec{l}}\times \,\mathbf{\hat{r}}}{{{r}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 82) The potential energy of a body is given by \[U=A-B{{x}^{2}}\] (where x is the displacement). The magnitude of force acting on the particle is
A)
constant
done
clear
B)
proportional to
done
clear
C)
proportional to x2
done
clear
D)
proportional to 1/x
done
clear
View Answer play_arrow
question_answer 83) Spherical ball of radius R is falling in a viscous fluid of viscosity \[\eta \] with a velocity v. The retarding viscous force acting on the spherical ball is
A)
directly proportional to radius JR but inversely proportional to velocity v
done
clear
B)
directly proportional to both radius R and to velocity v
done
clear
C)
inversely proportional to both radius R and velocity v
done
clear
D)
inversely proportional to radius R but directly proportional to velocity v
done
clear
View Answer play_arrow
question_answer 84) Even Carnot engine cannot give 100% efficiency because we cannot
A)
prevent radiation
done
clear
B)
find ideal sources
done
clear
C)
reach absolute zero temperature
done
clear
D)
eliminate friction
done
clear
View Answer play_arrow
question_answer 85) The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photoelectron emission from this substance is approximately
A)
540 nm
done
clear
B)
400 nm
done
clear
C)
310 nm
done
clear
D)
220 nm
done
clear
View Answer play_arrow
question_answer 86) A particle of charge \[-16\times {{10}^{-18}}C\]moving with velocity \[10\text{ }m{{s}^{-1}}\] along the x-axis enters a region where a magnetic field of induction B is along the y-axis and an electric field of magnitude \[{{10}^{4}}/m\]is along the negative z-axis. If the charged particle continues moving along the x-axis, the magnitude of B is
A)
\[{{10}^{16}}\text{Wb/}{{\text{m}}^{\text{2}}}\]
done
clear
B)
\[{{10}^{5}}\text{Wb/}{{\text{m}}^{\text{2}}}\]
done
clear
C)
\[{{10}^{3}}\text{Wb/}{{\text{m}}^{\text{2}}}\]
done
clear
D)
\[{{10}^{-3}}\text{Wb/}{{\text{m}}^{\text{2}}}\]
done
clear
View Answer play_arrow
question_answer 87) If No is the original mass of the substance of half-life period \[{{T}_{1/2}}=5\] yr, then the amount of substance left after 15 yr is
A)
\[{{N}_{0}}\text{/8}\]
done
clear
B)
\[{{N}_{0}}\text{/16}\]
done
clear
C)
\[{{N}_{0}}\text{/2}\]
done
clear
D)
\[{{N}_{0}}\text{/4}\]
done
clear
View Answer play_arrow
question_answer 88)
A charged ball B hangs from a silk thread S, which makes an angle \[\text{ }\!\!\theta\!\!\text{ }\] with a large charged conducting sheet P, as shown in the figure. The surface charge density \[\sigma \] of the sheet is proportional to
A)
\[\cot \,\text{ }\!\!\theta\!\!\text{ }\]
done
clear
B)
\[\cot \,\text{ }\!\!\theta\!\!\text{ }\]
done
clear
C)
\[\tan \,\text{ }\!\!\theta\!\!\text{ }\]
done
clear
D)
\[\sin \,\text{ }\!\!\theta\!\!\text{ }\]
done
clear
View Answer play_arrow
question_answer 89) If \[\,{{\text{ }\!\!\theta\!\!\text{ }}_{i}}\] is the inversion temperature, \[\,{{\text{ }\!\!\theta\!\!\text{ }}_{n}}\] is the neutral temperature, \[\,{{\text{ }\!\!\theta\!\!\text{ }}_{c}}\] is the temperature of the cold junction for thermocouple, then
A)
\[\,{{\text{ }\!\!\theta\!\!\text{ }}_{i}}+{{\text{ }\!\!\theta\!\!\text{ }}_{c}}={{\text{ }\!\!\theta\!\!\text{ }}_{n}}\]
done
clear
B)
\[\,{{\text{ }\!\!\theta\!\!\text{ }}_{i}}\text{-}{{\text{ }\!\!\theta\!\!\text{ }}_{c}}=2{{\text{ }\!\!\theta\!\!\text{ }}_{n}}\]
done
clear
C)
\[\,\frac{{{\text{ }\!\!\theta\!\!\text{ }}_{i}}\text{+}\,{{\text{ }\!\!\theta\!\!\text{ }}_{c}}}{2}={{\text{ }\!\!\theta\!\!\text{ }}_{n}}\]
done
clear
D)
\[\,{{\text{ }\!\!\theta\!\!\text{ }}_{c}}\text{-}{{\text{ }\!\!\theta\!\!\text{ }}_{i}}=2{{\text{ }\!\!\theta\!\!\text{ }}_{n}}\]
done
clear
View Answer play_arrow
question_answer 90) Speeds of two identical cars are u are 4u at a specific instant. The ratio of the respective distances in which the two cars are stopped in the same time
A)
1:1
done
clear
B)
1: 4
done
clear
C)
1:8
done
clear
D)
1:16
done
clear
View Answer play_arrow
question_answer 91) Before using the tangent galvanometer, its coil is set up in
A)
magnetic meridian
done
clear
B)
perpendicular to magnetic meridian
done
clear
C)
at angle of \[45{}^\circ \] to magnetic meridian
done
clear
D)
it does not require any setting
done
clear
View Answer play_arrow
question_answer 92)
Which of the following plots may represent reactance of a series LC combination?
A)
a
done
clear
B)
b
done
clear
C)
c
done
clear
D)
d
done
clear
View Answer play_arrow
question_answer 93) Three identical capacitors are connected together differently. For the same voltage to every combination the one that stores maximum energy is
A)
the three in series
done
clear
B)
the three in parallel
done
clear
C)
two in series and the third in parallel with it
done
clear
D)
two in parallel and the third in series with it
done
clear
View Answer play_arrow
question_answer 94)
Each resistance shown in the circuit is of \[10\,\Omega .\] The equivalent resistance between the points X and V is
A)
\[10\,\Omega \]
done
clear
B)
\[20\,\Omega \]
done
clear
C)
\[25\,\Omega \]
done
clear
D)
\[30\,\Omega \]
done
clear
View Answer play_arrow
question_answer 95) A L-R circuit consists of an inductance of 8 mH and a resistance of \[4\,\Omega .\] The time constant of the circuit is
A)
2ms
done
clear
B)
12ms
done
clear
C)
32ms
done
clear
D)
500s
done
clear
View Answer play_arrow
question_answer 96)
Three identical resistances A, B and C are connected as shown in the figure. The heat produced will be maximum in
A)
B
done
clear
B)
same for A, B and C
done
clear
C)
A
done
clear
D)
B and C
done
clear
View Answer play_arrow
question_answer 97) A kilowatt hour is equal to
A)
\[3.6\times {{10}^{6}}\text{J}\]
done
clear
B)
\[3.6\times {{10}^{4}}\text{J}\]
done
clear
C)
\[3.6\times {{10}^{3}}\text{J}\]
done
clear
D)
\[6\times {{10}^{-4}}\text{J}\]
done
clear
View Answer play_arrow
question_answer 98) Wire P and Q have the same resistance at ordinary (room) temperature. When heated, resistance of? increases and that of Q decreases. We conclude that
A)
P and Q are conductors of different materials.
done
clear
B)
P is n-type semiconductor and Q is p-type semiconductor
done
clear
C)
P is semiconductor and Q is conductor
done
clear
D)
P is conductor and Q is semiconductor
done
clear
View Answer play_arrow
question_answer 99) In the disintegration series \[_{92}^{238}U\xrightarrow[{}]{\alpha }X\xrightarrow[{}]{\text{ }\!\!\beta\!\!\text{ -}}_{\text{Z}}^{\text{A}}\text{Y}\] the values of Z and A respectively will be
A)
92,236
done
clear
B)
88,230
done
clear
C)
90, 234
done
clear
D)
91, 234
done
clear
View Answer play_arrow
question_answer 100) The plane faces of two identical plano-convex lenses, each having focal length of 40 cm, are placed against each other to form a common convex lens. The distance from this lens at which an object must be placed to obtain a real inverted image with magnification equal to unity is
A)
80cm
done
clear
B)
40cm
done
clear
C)
20cm
done
clear
D)
160cm
done
clear
View Answer play_arrow
question_answer 101) If an organic compound has \[C=40%,\] \[H=13.3%,\]\[N=46.67%\text{ },\] then the empirical formula for this compound is
A)
\[C{{H}_{4}}N\]
done
clear
B)
\[{{C}_{2}}{{H}_{8}}{{N}_{2}}\]
done
clear
C)
\[C{{H}_{3}}N\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 102) TEL is
A)
petrol fuel
done
clear
B)
anti knocking compound
done
clear
C)
gasoline
done
clear
D)
insecticide
done
clear
View Answer play_arrow
question_answer 103) DDT is related to
A)
insecticide
done
clear
B)
fungicide
done
clear
C)
chlorination
done
clear
D)
florination
done
clear
View Answer play_arrow
question_answer 104) Coordination number \[8:8\] is of
A)
\[CsCl\]
done
clear
B)
\[KCl\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[ZnS\]
done
clear
View Answer play_arrow
question_answer 105) Which of the following inert gas is soluble in water?
A)
\[He\]
done
clear
B)
\[Ne\]
done
clear
C)
\[Ar\]
done
clear
D)
\[Xe\]
done
clear
View Answer play_arrow
question_answer 106) Number of peptide bonds in dipeptide is
A)
\[2\]
done
clear
B)
\[1\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 107) Cupellation method is used for the extraction of
A)
\[Cu\]
done
clear
B)
\[Ag\]
done
clear
C)
\[Na\]
done
clear
D)
\[Al\]
done
clear
View Answer play_arrow
question_answer 108) Blood sugar is
A)
fructose
done
clear
B)
maltose
done
clear
C)
haemoglobin
done
clear
D)
glucose
done
clear
View Answer play_arrow
question_answer 109) Lactose sugar is formed from
A)
d-galactose
done
clear
B)
d-glucose
done
clear
C)
d-galactose and d-glucose
done
clear
D)
d-glucose and d-fructose
done
clear
View Answer play_arrow
question_answer 110) The values for all the quantum numbers for 15th electron of chlorine are
A)
\[n=3,l=1,m=0,s=-1/2\]
done
clear
B)
\[n=4,l=2,m=0,s=+1/2\]
done
clear
C)
\[n=3,l=1,m=+1,s=+1/2\]
done
clear
D)
\[n=2,l=0,m=0,s=+1/2\]
done
clear
View Answer play_arrow
question_answer 111) The IUPAC name of the compound is\[\text{C}{{\text{H}}_{\text{3}}}-[C{{H}_{2}}]s-CH-\underset{\begin{smallmatrix} | \\ \underset{\underset{\underset{C{{H}_{3}}}{\overset{C{{H}_{2}}}{\mathop{|}}}\,}{\overset{\beta CHC{{H}_{3}}}{\mathop{|}}}\,}{\overset{C{{H}_{2}}}{\mathop{|}}}\, \end{smallmatrix}}{\mathop{\text{CH}}}\,-\text{C}{{\text{H}}_{\text{2}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \underset{\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}} \end{smallmatrix}}{\mathop{\text{C}{{\text{H}}_{\text{2}}}}}\,}{\overset{\text{ }\!\!\alpha\!\!\text{ CHC}{{\text{H}}_{\text{3}}}}{\mathop{\text{ }\!\!|\!\!\text{ }}}}\, \end{smallmatrix}}{\mathop{\text{CH}}}\,-{{\text{ }\!\![\!\!\text{ C}{{\text{H}}_{\text{2}}}\text{ }\!\!]\!\!\text{ }}_{\text{3}}}-\text{C}{{\text{H}}_{\text{3}}}\]
A)
\[7\text{ }\!\![\!\!\text{ }\beta \text{ }methyl\text{ }butyl],\text{ }9\text{ }butyl\text{ }tridecane\]
done
clear
B)
\[3[\beta \text{ }ethyl\text{ }butyl],9\text{ }ethyl\text{ }tridecane\]
done
clear
C)
\[2[\beta \text{ }ethyl\text{ }ethenyl],\text{ }8\text{ }propyl\text{ }decane\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
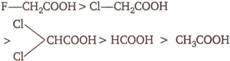
A)
\[(III)>(II)>(I)>(IV)\]
done
clear
B)
\[(II)>(III)>(I)>(IV)\]
done
clear
C)
\[(I)>(II)>(III)>(IV)\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 113) Outermost configuration for \[Z=25,\] is
A)
\[4{{s}^{2}}3{{d}^{5}}\]
done
clear
B)
\[5{{s}^{2}}4{{d}^{5}}\]
done
clear
C)
\[4{{s}^{2}}3{{d}^{3}}\]
done
clear
D)
\[4{{s}^{2}}3{{d}^{1}}\]
done
clear
View Answer play_arrow
question_answer 114) The colour of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]in acidic medium is
A)
red
done
clear
B)
green
done
clear
C)
yellow
done
clear
D)
pink
done
clear
View Answer play_arrow
question_answer 115) Baeyer's reagent decolourises which of the following?
A)
Alkane
done
clear
B)
Alkene
done
clear
C)
Alkene and alkyne both
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 116) When \[Mn{{O}_{2}}\] is fused with \[KOH,\]a coloured compound is formed. The product and its colouris
A)
\[{{K}_{2}}Mn{{O}_{4}},\] purple green
done
clear
B)
\[KMn{{O}_{4}},\] purple
done
clear
C)
\[M{{n}_{2}}{{O}_{3}},\] brown
done
clear
D)
\[M{{n}_{2}}{{O}_{4}},\] black
done
clear
View Answer play_arrow
question_answer 117) The type of hybridization present in each carbon of benzene is
A)
\[s{{p}^{3}}\]
done
clear
B)
\[s{{p}^{2}}\]
done
clear
C)
\[sp\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 118) Total number of isomers of \[{{C}_{4}}{{H}_{10}}O\] is
A)
\[7\]
done
clear
B)
\[4\]
done
clear
C)
\[3\]
done
clear
D)
\[8\]
done
clear
View Answer play_arrow
question_answer 119) Vitamin \[{{B}_{12}}\] contains
A)
\[Co\]
done
clear
B)
\[Fe\]
done
clear
C)
\[Zn\]
done
clear
D)
\[Ca\]
done
clear
View Answer play_arrow
question_answer 120) Night blindness is caused due to the deficiency of
A)
\[{{B}_{12}}\]
done
clear
B)
\[A\]
done
clear
C)
\[C\]
done
clear
D)
\[D\]
done
clear
View Answer play_arrow
question_answer 121) RNA does not contain
A)
Thymine
done
clear
B)
Adenine
done
clear
C)
Guanine
done
clear
D)
Uracil
done
clear
View Answer play_arrow
question_answer 122) Reaction of dry \[HCl\]with acetone gives
A)
aldol
done
clear
B)
chloritone
done
clear
C)
isopropyi alcohol
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 123) Which of the following is incorrect for radial distribution curve? n I Node
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 124) If \[{{H}^{+}}\] concentration of fruit juice is \[3.3\times {{10}^{-2}},\]then its pH will be
A)
\[4.8\text{ }basic\]
done
clear
B)
\[4.8\text{ }neutral\]
done
clear
C)
\[4.8\text{ }acidic\]
done
clear
D)
\[1.6\text{ }acidic\]
done
clear
View Answer play_arrow
question_answer 125) \[{{C}_{2}}{{H}_{5}}OH\xrightarrow[NaOH]{{{I}_{2}}}A\xrightarrow[KOH]{Aqueous}B\]In equation given above A and B are respectively
A)
\[CH{{I}_{3}}\]and \[HCOOK\]
done
clear
B)
\[CH{{I}_{3}}\] and \[C{{H}_{3}}COOK\]
done
clear
C)
\[C{{H}_{4}}\]and \[HCOOK\]
done
clear
D)
\[CH{{I}_{3}}\] and \[{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 126) Formula of washing soda is
A)
\[N{{a}_{2}}C{{O}_{3}}.10{{H}_{2}}O\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}.10{{H}_{2}}O\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 127) \[AgN{{O}_{3}}\] gives red colour with
A)
\[NaCl\]
done
clear
B)
\[NaBr\]
done
clear
C)
\[NaI\]
done
clear
D)
\[{{K}_{2}}Cr{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 128) If the value of bond order is zero, then
A)
Molecule will be stable
done
clear
B)
Molecule will be unstable
done
clear
C)
Molecule will be in ionic state
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 129) Which is responsible for the diagonal relation of lithium with magnesium?
A)
Less ionic radii
done
clear
B)
High polarizing power
done
clear
C)
Approximately equal electronegativity and affinity
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 130)
Trioxane has the formula
A)
formaldehyde
done
clear
B)
methanol
done
clear
C)
dichloromethane
done
clear
D)
vinyl alcohol
done
clear
View Answer play_arrow
question_answer 131) Natural rubber is
A)
butadiene
done
clear
B)
isoprene
done
clear
C)
neoprene
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 132) Monomer of polythene is
A)
ethylene
done
clear
B)
propylene
done
clear
C)
vinyl chloride
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 133) Bond order of \[{{N}_{2}}\] molecule is
A)
3
done
clear
B)
2
done
clear
C)
1
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 134) Which of the following is a proper match? \[\frac{{{r}^{+}}}{{{r}^{-}}}\] Shape \[0.115-0.225\] (i) Triangular \[0.225-0.414\] (ii) Tetrahedral \[0.414-0.732\] (iii) Cubic \[0.732-1\] (iv) Octahedral
A)
\[A-i,B-ii,C-iv,D-iii\]
done
clear
B)
\[A-iii,B-ii,C-iv,D-i\]
done
clear
C)
\[A-i,B-iii,C-i,D-iv\]
done
clear
D)
\[A-ii,B-iv,C-i,D-iii\]
done
clear
View Answer play_arrow
question_answer 135)
The correct order of acidity of following acids is \[C{{H}_{3}}COOH,HCOOH,Cl-C{{H}_{2}}COOH\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 136) \[C{{O}_{2}}\] gas obtained by the combustion of \[12\text{ }mL\]butane gas is
A)
\[\text{3 }mL\]
done
clear
B)
\[12\text{ }mL\]
done
clear
C)
\[\text{24 }mL\]
done
clear
D)
\[\text{48 }mL\]
done
clear
View Answer play_arrow
question_answer 137) Calomel is
A)
\[H{{g}_{2}}C{{l}_{2}}\]
done
clear
B)
\[Hg+H{{g}_{2}}C{{l}_{2}}\]
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 138) In an adiabatic process which of the following is true?
A)
\[q=+W\]
done
clear
B)
\[q=0\]
done
clear
C)
\[\Delta E=q\]
done
clear
D)
\[p\Delta V=0\]
done
clear
View Answer play_arrow
question_answer 139) The number of paired electrons in \[Xe\] atom of \[Xe{{F}_{2}}\] is
A)
\[3\]
done
clear
B)
\[5\]
done
clear
C)
\[4\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 140) Hydrolysis of \[Xe{{F}_{6}}\] gives
A)
\[Xe{{O}_{3}}\]
done
clear
B)
\[Xe{{O}_{6}}\]
done
clear
C)
\[Xe{{O}_{2}}\]
done
clear
D)
\[Xe{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 141) Zwitter ion is
A)
Dipolar ion
done
clear
B)
Ion formed from amino acid
done
clear
C)
Internal salt
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 142) IUPAC name of \[[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]is
A)
Diamine dichloro platinum (II)
done
clear
B)
Amine, chloro platinum (III)
done
clear
C)
Chloro diamine platinum (II)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 143) Nylon obtained by the condensation of adipic acid with hexamethylene diamine
A)
Nylon \[6,6\]
done
clear
B)
Nylon \[7,6\]
done
clear
C)
Nylon \[6,5\]
done
clear
D)
Nylon \[9,7\]
done
clear
View Answer play_arrow
question_answer 144) Equation log \[\frac{{{K}_{2}}}{{{K}_{1}}}=\frac{\Delta H}{2.303R}\left[ \frac{{{T}_{2}}-{{T}_{1}}}{{{T}_{1}}.{{T}_{2}}} \right],\] n is
A)
van der Waals' equation
done
clear
B)
Kirchoff's equation
done
clear
C)
Gas equation
done
clear
D)
Van?t Hoff equation
done
clear
View Answer play_arrow
question_answer 145) Which of the following is a colligative property?
A)
Surface tension
done
clear
B)
Viscosity
done
clear
C)
Osmotic pressure
done
clear
D)
Vapour pressure
done
clear
View Answer play_arrow
question_answer 146) Number of a and p emitted in the reaction \[_{92}{{U}^{238}}{{\xrightarrow{{}}}_{82}}P{{b}^{206}}\]are
A)
\[6\alpha \]and \[8\beta \]
done
clear
B)
\[8\alpha \] and \[6\beta \]
done
clear
C)
\[6\alpha \] and \[4\beta \]
done
clear
D)
\[4\beta \] and \[6\beta \]
done
clear
View Answer play_arrow
question_answer 147) A chemical reaction is catalysed by 'X' therefore, 'X'
A)
Increases the activation energy
done
clear
B)
Does not affect the equilibrium constant of the reaction
done
clear
C)
Decreases the enthalpy of the reaction
done
clear
D)
Decreases the velocity constant of the reaction
done
clear
View Answer play_arrow
question_answer 148) The half-life of a radioactive isotope is\[3\text{ }h\]. If the initial mass of the isotope were \[256\text{ }g,\] the mass of it remaining undecayed after \[18\text{ }h\]would be
A)
\[8.0\text{ }g\]
done
clear
B)
\[4.0\text{ }g\]
done
clear
C)
\[12.0\text{ }g\]
done
clear
D)
\[16.0\text{ }g\]
done
clear
View Answer play_arrow
question_answer 149) According to MOT which of the following is correct for potassium hexa cyano ferrate (III)?
A)
It is a octahedral complex
done
clear
B)
\[{{t}_{2g}}\] Orbital contains the \[{{e}^{-}}\] of metal
done
clear
C)
For iron, overlapping between empty orbitals and ligand orbitals takes place
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 150) Radioactive hydrogen is
A)
Tritium
done
clear
B)
Deuterium
done
clear
C)
Ortho hydrogen
done
clear
D)
Para hydrogen
done
clear
View Answer play_arrow
question_answer 151) Percentage of oxygen in phenol is
A)
\[17.02\]
done
clear
B)
\[15.2\]
done
clear
C)
\[16\]
done
clear
D)
\[15\]
done
clear
View Answer play_arrow
question_answer 152) Which of the following differs in BAN?
A)
\[[Co{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]
done
clear
B)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
C)
\[{{K}_{2}}[ZnC{{l}_{4}}]\]
done
clear
D)
\[{{K}_{2}}[Hg{{I}_{4}}]\]
done
clear
View Answer play_arrow
question_answer 153) Which of the following complex neutralises three molecules of\[AgN{{O}_{3}}\]?
A)
\[[Co{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{5}}Cl]C{{l}_{2}}\]
done
clear
C)
\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl\]
done
clear
D)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}]\]
done
clear
View Answer play_arrow
question_answer 154) \[Zn\underset{C=1}{\mathop{|Z{{n}^{2+}}|}}\,\underset{C=1}{\mathop{|C{{u}^{2+}}|}}\,Cu\] If the standard reduction potential of zinc electrode and copper half-cell is \[-0.76\text{ }V\]and \[0.34\text{ }V\]respectively then the emf will be
A)
\[1.1V\]
done
clear
B)
\[1.4V\]
done
clear
C)
\[1.34V\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 155) Which of the following does not show optical isomerisation?
A)
\[[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]
done
clear
B)
\[{{[Co{{(oX)}_{3}}]}^{3-}}\]
done
clear
C)
\[{{[Co{{(en)}_{3}}]}^{3+}}\]
done
clear
D)
\[{{[Cr{{(diph)}_{3}}]}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 156) Solubility product of pure \[PbC{{l}_{2}}\] will be
A)
\[{{K}_{sp}}={{S}^{2}}\]
done
clear
B)
\[{{K}_{sp}}=4{{S}^{3}}\]
done
clear
C)
\[{{K}_{sp}}=108{{S}^{5}}\]
done
clear
D)
\[{{K}_{sp}}=S\]
done
clear
View Answer play_arrow
question_answer 157) The expression for velocity constant for the second order reaction is
A)
\[k=\frac{2.303}{t}{{\log }_{10}}\frac{a}{a-x}\]
done
clear
B)
\[k=\frac{1}{t}\frac{x}{a[a-x]}\]
done
clear
C)
\[k=\frac{1}{t}\frac{x}{{{a}^{2}}{{[a-x]}^{2}}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 158) The unit of rate constant for second order reaction is
A)
\[{{s}^{-1}}\]
done
clear
B)
\[mol/{{L}^{-1}}\]
done
clear
C)
\[L\,\,mo{{l}^{-1}}{{s}^{-1}}\]
done
clear
D)
\[{{L}^{2}}mo{{l}^{-2}}s\]
done
clear
View Answer play_arrow
question_answer 159) Which of the following, on heating with ammonia gives urotropin?
A)
Formaldehyde
done
clear
B)
Acetaldehyde
done
clear
C)
Acetone
done
clear
D)
Benzaldehyde
done
clear
View Answer play_arrow
question_answer 160) Which of the following statement is correct?
A)
Basic nature increases on increasing pH
done
clear
B)
Basic nature decreases on increasing pH
done
clear
C)
Acidic nature increases on increasing pH
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 161) For the redox reaction \[Zn(s)+C{{u}^{2+}}(0.1M)\xrightarrow{{}}Z{{n}^{2+}}(1M)+Cu(s)\] taking place in a cell, \[E_{cell}^{o}\] is \[1.10V.\] \[{{E}_{cell}}\] for the cell will be \[\left( 2.303\frac{RT}{F}=0.0591 \right)\]
A)
\[2.14V\]
done
clear
B)
\[1.80V\]
done
clear
C)
\[0.82V\]
done
clear
D)
\[1.07V\]
done
clear
View Answer play_arrow
question_answer 162) \[{{C}_{6}}{{H}_{6}}+C{{l}_{2}}\xrightarrow{Sunlight}\] product
A)
BHC
done
clear
B)
Chloro benzene
done
clear
C)
Dichloro benzene
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 163) The required condition for an ideal solution is
A)
\[\Delta {{H}_{mix}}=0\]
done
clear
B)
\[\Delta {{V}_{mix}}\ne 0\]
done
clear
C)
\[{{p}_{A}}\ne p_{A}^{o}{{X}_{A}}\]
done
clear
D)
\[\Delta {{H}_{mix}}>1\]
done
clear
View Answer play_arrow
question_answer 164) Interionic distance for \[CsCl\]crystal is
A)
\[a\]
done
clear
B)
\[a/2\]
done
clear
C)
\[\frac{\sqrt{3}}{4}a\]
done
clear
D)
\[\frac{2a}{\sqrt{3}}\]
done
clear
View Answer play_arrow
question_answer 165) How many space lattices are obtained from the different crystal systems?
A)
\[7\]
done
clear
B)
\[14\]
done
clear
C)
\[32\]
done
clear
D)
\[230\]
done
clear
View Answer play_arrow
question_answer 166) Which of the following occurs at anode?
A)
Reduction
done
clear
B)
Oxidation
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 167) Parke's process is used for the extraction of
A)
Iron
done
clear
B)
sodium
done
clear
C)
Silver
done
clear
D)
zinc
done
clear
View Answer play_arrow
question_answer 168) The common method of extraction of metals from oxide ores is
A)
Reduction with carbon
done
clear
B)
Reduction with hydrogen
done
clear
C)
Reduction with aluminium
done
clear
D)
Electrolytic method
done
clear
View Answer play_arrow
question_answer 169) The transition metals mostly are
A)
Diamagnetic
done
clear
B)
Paramagnetic
done
clear
C)
Neither diamagnetic nor paramagnetic
done
clear
D)
Both diamagnetic and paramagnetic
done
clear
View Answer play_arrow
question_answer 170) Which of the following is an electron deficient compound?
A)
\[{{B}_{2}}{{H}_{6}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
question_answer 171) Aniline reacts with chloroform in the presence of \[KOH\] to give
A)
Phenol
done
clear
B)
Chlorobenzene
done
clear
C)
Phenyl cyanide
done
clear
D)
Phenyl isocyanide
done
clear
View Answer play_arrow
question_answer 172) Which one of the following is conjugate acid of water in the reaction? \[{{H}_{2}}S{{O}_{4}}+{{H}_{2}}O\rightleftharpoons {{H}_{3}}{{O}^{+}}+HSO_{4}^{-}\]
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{3}}{{O}^{+}}\]
done
clear
C)
\[SO_{4}^{2-}\]
done
clear
D)
\[HSO_{4}^{-}\]
done
clear
View Answer play_arrow
question_answer 173) The product of \[{{H}^{+}}\] and \[O{{H}^{-}}\] of water will be
A)
\[{{K}_{w}}={{10}^{-12}}\]
done
clear
B)
\[{{K}_{w}}={{10}^{-14}}\]
done
clear
C)
\[{{K}_{w}}={{10}^{-11}}\]
done
clear
D)
\[{{K}_{w}}={{10}^{-10}}\]
done
clear
View Answer play_arrow
question_answer 174) When methyl cyanide is hydrolysed in presence of alkali, the product is
A)
Acetamide
done
clear
B)
methane
done
clear
C)
\[C{{O}_{2}}+{{H}_{2}}O\]
done
clear
D)
acetic acid
done
clear
View Answer play_arrow
question_answer 175) For \[NaCl,\]the \[{{K}_{sp}}=36\,\,mo{{l}^{2}}{{L}^{-2}},\] the molar concentration of it will be
A)
\[\frac{1}{36}M\]
done
clear
B)
\[\frac{1}{16}M\]
done
clear
C)
\[36\text{ }M\]
done
clear
D)
\[6M\]
done
clear
View Answer play_arrow
question_answer 176) Pour solutions A,B,C,D has glucose \[0.5\text{ }M,\] \[NaCl\text{ }0.1\text{ }M,\]\[BaC{{l}_{2}}\text{ }0.5\text{ }M\] and \[MgC{{l}_{2}}\text{ }0.1\text{ }M,\]then which of the following will have highest osmotic pressure?
A)
Glucose
done
clear
B)
\[BaC{{l}_{2}}\]
done
clear
C)
\[MgC{{l}_{2}}\]
done
clear
D)
\[NaCl\]
done
clear
View Answer play_arrow
question_answer 177) The highest boiling point is of
A)
\[FeC{{l}_{2}}\]
done
clear
B)
\[FeC{{l}_{3}}\]
done
clear
C)
\[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
D)
\[AlC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 178) For an ionic crystal of the general formula AX and coordination number is 6, the value of radius ratio will be
A)
greater than \[0.73\]
done
clear
B)
in between \[0.73\] and \[0.41\]
done
clear
C)
in between \[0.41\] and \[0.22\]
done
clear
D)
less than \[0.22\]
done
clear
View Answer play_arrow
question_answer 179) Which of the following can be used to convert \[_{7}^{14}N\]into \[_{8}^{17}O\]?
A)
Deutron
done
clear
B)
Proton
done
clear
C)
\[\alpha \]-particle
done
clear
D)
Neutron
done
clear
View Answer play_arrow
question_answer 180) \[{{N}_{2}}{{O}_{4}}\rightleftharpoons 2N{{O}_{2}}-Q\] The unit of \[{{K}_{p}}\] for the given reaction is
A)
atmosphere
done
clear
B)
\[atmospher{{e}^{2}}\]
done
clear
C)
\[atmospher{{e}^{-1}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 181) Number of ions for the complex \[[Co{{(N{{H}_{3}})}_{4}}Cl]C{{l}_{2}}\]shown by the conductivity measurement, is
A)
\[2\]
done
clear
B)
\[4\]
done
clear
C)
\[3\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
question_answer 182) Rusting on iron needs
A)
dry air
done
clear
B)
air and water
done
clear
C)
distilled water and carbon dioxide
done
clear
D)
oxygen and carbon dioxide
done
clear
View Answer play_arrow
question_answer 183) First ionization potential is highest for
A)
\[Na\]
done
clear
B)
\[Mg\]
done
clear
C)
\[Al\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 184) Which compound is not a lipid?
A)
Lecithin
done
clear
B)
Lysine
done
clear
C)
Cerebroside
done
clear
D)
Cephalin
done
clear
View Answer play_arrow
question_answer 185) Which one of the following notations shows product incorrectly?
A)
\[_{96}^{242}Cm(\alpha ,2n)_{97}^{243}Bk\]
done
clear
B)
\[_{5}^{10}B(\alpha ,n)_{7}^{13}N\]
done
clear
C)
\[_{7}^{14}N(n,p)_{6}^{14}C\]
done
clear
D)
\[_{14}^{28}Si(d,n)_{15}^{29}P\]
done
clear
View Answer play_arrow
question_answer 186) In the equilibrium reaction \[2HI(g)\rightleftharpoons {{H}_{2}}+{{I}_{2}}\]which of the following expressions is true?
A)
\[{{K}_{p}}={{K}_{c}}\]
done
clear
B)
\[{{K}_{c}}=2{{K}_{p}}\]
done
clear
C)
\[{{K}_{p}}>{{K}_{c}}\]
done
clear
D)
\[{{K}_{c}}={{K}_{p}}{{(RT)}^{2}}\]
done
clear
View Answer play_arrow
question_answer 187) For a reaction of the type \[A+B\to \] products, it is observed that doubling the concentration of A causes the reaction rate to be four times as great, but doubling the amount of B does not affect the rate. The rate equation is
A)
\[Rate=k[A][B]\]
done
clear
B)
\[Rate=k{{[A]}^{2}}\]
done
clear
C)
\[Rate=k{{[A]}^{2}}[B]\]
done
clear
D)
\[Rate=k{{[A]}^{2}}{{[B]}^{2}}\]
done
clear
View Answer play_arrow
question_answer 188) In the following, the element with the highest electro positivity is
A)
copper
done
clear
B)
cesium
done
clear
C)
barium
done
clear
D)
chromium
done
clear
View Answer play_arrow
question_answer 189) The reaction \[C{{H}_{2}}=C{{H}_{2}}+{{H}_{2}}\xrightarrow[{{250}^{o}}-{{300}^{o}}C]{Ni}C{{H}_{3}}-C{{H}_{3}}\] is called
A)
Wurtz reaction
done
clear
B)
Kolbe's reaction
done
clear
C)
Sabatier-Senderens reaction
done
clear
D)
Carbylamine reaction
done
clear
View Answer play_arrow
question_answer 190) Unit of first order rate constant is
A)
\[mol\,\,L\,s\]
done
clear
B)
\[mo{{l}^{-1}}\,\,{{L}^{-1}}\,{{s}^{-1}}\]
done
clear
C)
\[mol\,\,{{L}^{-1}}\,{{s}^{-1}}\]
done
clear
D)
\[{{s}^{-1}},{{\min }^{-1}}etc.\]
done
clear
View Answer play_arrow
question_answer 191) Which of the following describes the shape of orbital?
A)
Principal quantum number
done
clear
B)
Azimuthal quantum number
done
clear
C)
Magnetic quantum number
done
clear
D)
Spin quantum number
done
clear
View Answer play_arrow
question_answer 192) The formula of a phosphate of a metal M, is \[MP{{O}_{4}}\]. The formula of its nitrate will be
A)
\[MN{{O}_{3}}\]
done
clear
B)
\[M{{(N{{O}_{3}})}_{2}}\]
done
clear
C)
\[M{{(N{{O}_{3}})}_{3}}\]
done
clear
D)
\[{{M}_{2}}{{(N{{O}_{3}})}_{3}}\]
done
clear
View Answer play_arrow
question_answer 193) The d-block element consists mostly of
A)
monovalent metals
done
clear
B)
all non-metals
done
clear
C)
elements which generally form stoichiometric metal oxide
done
clear
D)
many metals with catalytic properties
done
clear
View Answer play_arrow
question_answer 194) A mixture containing \[C{{u}^{2+}}\] and \[N{{i}^{2+}}\] can be separated for identification by
A)
passing \[{{H}_{2}}S\] in acid medium
done
clear
B)
passing \[{{H}_{2}}S\] in alkaline medium
done
clear
C)
passing \[{{H}_{2}}S\] in neutral medium
done
clear
D)
passing \[{{H}_{2}}S\] in dry mixture
done
clear
View Answer play_arrow
question_answer 195) Strongest Breasted base among the following anions is
A)
\[Cl{{O}^{-}}\]
done
clear
B)
\[ClO_{2}^{-}\]
done
clear
C)
\[ClO_{3}^{-}\]
done
clear
D)
\[ClO_{4}^{-}\]
done
clear
View Answer play_arrow
question_answer 196) Which one of the following is a good conductor of electricity?
A)
Diamond
done
clear
B)
Graphite
done
clear
C)
Silicon
done
clear
D)
Amorphous carbon
done
clear
View Answer play_arrow
question_answer 197) Which one of the following is an example of non-typical transition elements?
A)
\[Li,K,Na\]
done
clear
B)
\[Be,Al,Pb\]
done
clear
C)
\[Zn,Cd,Hg\]
done
clear
D)
\[Ba,Ca,Sr\]
done
clear
View Answer play_arrow
question_answer 198) Which of the following has zero dipole moment?
A)
\[ClF\]
done
clear
B)
\[PC{{l}_{3}}\]
done
clear
C)
\[Si{{F}_{4}}\]
done
clear
D)
\[CFC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 199) In galvanic cell, the salt bridge is used to
A)
complete the circuit
done
clear
B)
reduce the electric resistance in Ae cell
done
clear
C)
separate cathode from anode
done
clear
D)
carry salts for the chemical reaction
done
clear
View Answer play_arrow
question_answer 200) Reduction of methyl isocyanide gives
A)
ethylamine
done
clear
B)
methylamine
done
clear
C)
dimethylamine
done
clear
D)
trimethylamine
done
clear
View Answer play_arrow
question_answer 201) Polar bodies are formed during
A)
oviposition
done
clear
B)
oogenesis
done
clear
C)
fertilization
done
clear
D)
sperm to genesis
done
clear
View Answer play_arrow
question_answer 202) Node of Ranvier can be seen in
A)
right atrium
done
clear
B)
dendrites
done
clear
C)
cell body
done
clear
D)
axon
done
clear
View Answer play_arrow
question_answer 203) Mutation is
A)
sudden change in morphology
done
clear
B)
change in character
done
clear
C)
change in heritable characters
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 204) Amniocentesis is a technique used for
A)
estimating amino acid content in the amino
done
clear
B)
measuring size of the amino
done
clear
C)
determining sex of the foetus
done
clear
D)
determining position of the foetus
done
clear
View Answer play_arrow
question_answer 205) The science of ageing is called
A)
Chronology
done
clear
B)
Odontology
done
clear
C)
Gynaecology
done
clear
D)
Gerontology
done
clear
View Answer play_arrow
question_answer 206) Which is correct about human brain?
A)
It is covered by two membranes
done
clear
B)
There is no blood-brain barrier
done
clear
C)
Largest number of cranial nerves originate form cerebral hemisphere
done
clear
D)
Cerebral cortex is highly developed
done
clear
View Answer play_arrow
question_answer 207) The placenta of human beings belong to the category
A)
epitheliochorial
done
clear
B)
syndesmochorial
done
clear
C)
endotheliochorial
done
clear
D)
haemochorial
done
clear
View Answer play_arrow
question_answer 208) Which of the blood group does not contain any antigen ?
A)
Blood group O
done
clear
B)
Blood group A
done
clear
C)
Blood group B
done
clear
D)
Blood group AB
done
clear
View Answer play_arrow
question_answer 209) AIDS related complex (ARC) is a disease, which leads to fever, swollen lymph nodes, night sweats, loss in weight etc present
A)
sever form of AIDS
done
clear
B)
initial form of AIDS
done
clear
C)
single stranded DNA
done
clear
D)
double stranded DNA
done
clear
View Answer play_arrow
question_answer 210) External ears are characteristics of
A)
mammals
done
clear
B)
reptiles
done
clear
C)
amphibians
done
clear
D)
pisces
done
clear
View Answer play_arrow
question_answer 211) Pace-maker of the heart is situated
A)
in wall of right atrium
done
clear
B)
on the inter auricular septum
done
clear
C)
on inter ventricular septum
done
clear
D)
in wall of left atrium
done
clear
View Answer play_arrow
question_answer 212) Electric organs are found in
A)
sharks
done
clear
B)
porpoises
done
clear
C)
goldfish
done
clear
D)
rays
done
clear
View Answer play_arrow
question_answer 213) Acromion process is found in rabbit in
A)
pectoral girdle
done
clear
B)
femur
done
clear
C)
vertebral column
done
clear
D)
pelvic girdle
done
clear
View Answer play_arrow
question_answer 214) Mammalian lungs contain a large number of minute alveoli (air sacs). This is to allow
A)
more space for increasing the total volume of inspired air
done
clear
B)
more surface area for diffusion of gases
done
clear
C)
more spongy texture for keeping lungs in proper shape
done
clear
D)
more nerve supply to keep the organs working more efficiently
done
clear
View Answer play_arrow
question_answer 215) Name of nervous band connecting cerebral hemispheres in rabbit is
A)
corpus callosum
done
clear
B)
corpus albicans
done
clear
C)
corpus striatum
done
clear
D)
corpus spongiosum
done
clear
View Answer play_arrow
question_answer 216) Lateral ventricles are found in
A)
brain
done
clear
B)
heart
done
clear
C)
thyroid
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
question_answer 217) Biological name of insect (vector) carrying the plague
A)
Xenopey cheopis
done
clear
B)
Anopheles mosquito
done
clear
C)
Bacillus pestis
done
clear
D)
Pediculus humanus
done
clear
View Answer play_arrow
question_answer 218) Which cell types secretes \[\text{HCl}\] in stomach?
A)
Chief cells
done
clear
B)
Oxyntic cells
done
clear
C)
Ppneth cells
done
clear
D)
Goblet cells
done
clear
View Answer play_arrow
question_answer 219) Nissl's granules are found in
A)
mast cells
done
clear
B)
osteoblasts
done
clear
C)
chondro blasts
done
clear
D)
neurons
done
clear
View Answer play_arrow
question_answer 220) What for the ascending limb of loop of Henie is permeable?
A)
Glucose
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
Na
done
clear
D)
Water
done
clear
View Answer play_arrow
question_answer 221) Parathormone deficiency in man causes
A)
hypercalcemia
done
clear
B)
hypocalcemia
done
clear
C)
goiter
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 222) The eggs of placental mammals are described as
A)
microlecithal
done
clear
B)
telolecithal
done
clear
C)
centrolecithal
done
clear
D)
alecithal
done
clear
View Answer play_arrow
question_answer 223) If a haemophilic man marries a normal lady, the of fsprings will be
A)
all haemophilic
done
clear
B)
all normal
done
clear
C)
haemophilic daughters
done
clear
D)
haemophilic sons
done
clear
View Answer play_arrow
question_answer 224) "Origin of life' was written by
A)
\[\text{S}\,\,\text{I}\] Miller
done
clear
B)
\[\text{A}\,\,\text{I}\] Oparin
done
clear
C)
de Vries
done
clear
D)
Charles Darwin
done
clear
View Answer play_arrow
question_answer 225) Improvement of human race by improving the environment is called
A)
Euphenics
done
clear
B)
Eugenics
done
clear
C)
Euthenics
done
clear
D)
Ecology
done
clear
View Answer play_arrow
question_answer 226) The carrier of virus causing human yellow fever is
A)
mosquito
done
clear
B)
bug
done
clear
C)
louse
done
clear
D)
beetle
done
clear
View Answer play_arrow
question_answer 227) Heart-beat originates from
A)
pace-maker
done
clear
B)
cardiac muscles
done
clear
C)
left atrium
done
clear
D)
right ventricle
done
clear
View Answer play_arrow
question_answer 228) Smoking addiction is harmful because it produces polycyclic aromatic hydrocarbons which cause
A)
reduction in oxygen transport
done
clear
B)
increase in blood pressure
done
clear
C)
cancer
done
clear
D)
retardation of growth of foetus
done
clear
View Answer play_arrow
question_answer 229) Main function of cerebellum is
A)
balancing
done
clear
B)
to see
done
clear
C)
to hear
done
clear
D)
remembering
done
clear
View Answer play_arrow
question_answer 230) The centre for sense of smell in brain is
A)
cerebellum
done
clear
B)
cerebrum
done
clear
C)
olfactory lobes
done
clear
D)
midbrain
done
clear
View Answer play_arrow
question_answer 231) The nerve related with diaphragm is
A)
vagus
done
clear
B)
phrenic
done
clear
C)
trigeminal
done
clear
D)
glossopharyngeal
done
clear
View Answer play_arrow
question_answer 232) The disease caused by deficiency of parathormone is
A)
cretinism
done
clear
B)
tetany
done
clear
C)
hypercalcemia
done
clear
D)
myxedema
done
clear
View Answer play_arrow
question_answer 233) In human females at the time of birth there are two million ova; how many of them normally reach maturity in the course of normal reproductive life?
A)
500
done
clear
B)
1,000
done
clear
C)
2,000
done
clear
D)
5,000
done
clear
View Answer play_arrow
question_answer 234) Which of the following -belongs to phylum Arthropoda ?
A)
Starfish
done
clear
B)
Goldfish
done
clear
C)
Silver fish
done
clear
D)
Cuttlefish
done
clear
View Answer play_arrow
question_answer 235) During replication of a bacterial chromosome DNA synthesis starts from a replication origin site and
A)
RNA primers are involved
done
clear
B)
is facilitated by telomerase
done
clear
C)
moves in one direction of the site
done
clear
D)
moves in bi-directional way
done
clear
View Answer play_arrow
question_answer 236) Ear drum is known as
A)
tympanic membrane
done
clear
B)
tensor tympani
done
clear
C)
scala tympani
done
clear
D)
scala vestibule
done
clear
View Answer play_arrow
question_answer 237) Silkworm is a
A)
fly
done
clear
B)
worm
done
clear
C)
moth
done
clear
D)
beetle
done
clear
View Answer play_arrow
question_answer 238) A normal woman, whose father was colourblind is married to a normal man. The sons would be
A)
75% colourblind
done
clear
B)
50% colourblind
done
clear
C)
all normal
done
clear
D)
All colourblind
done
clear
View Answer play_arrow
question_answer 239) In contrast to annelids the platyhelminths show
A)
radial symmetry
done
clear
B)
presence of pseudocoel
done
clear
C)
bilateral symmetry
done
clear
D)
absence of body cavity
done
clear
View Answer play_arrow
question_answer 240) Which of the following unicellular organism has a macronucleus for trophic function and one or more micronuclei for reproduction?
A)
Euglena
done
clear
B)
Amoeba
done
clear
C)
Paramecium
done
clear
D)
Trypanosoma
done
clear
View Answer play_arrow
question_answer 241) In human, muscular coordination is controlled by
A)
bone marrow
done
clear
B)
medula oblongata
done
clear
C)
cerebellum
done
clear
D)
cerebrum
done
clear
View Answer play_arrow
question_answer 242) Increase in human race by using rules of Genetics is known as
A)
Eugenics
done
clear
B)
Euphenics
done
clear
C)
Euthenics
done
clear
D)
Dysgenics
done
clear
View Answer play_arrow
question_answer 243) What kind of evidence suggested that man is more closely related with chimpanzee than with other Hominoid apes?
A)
Evidence from DNA from sex chromosomes only
done
clear
B)
Comparison of chromosomes morphologically only
done
clear
C)
Evidence from fossil remains and the fossil mitochondrial DNA alone
done
clear
D)
Evidence from DNA extracted from sex chromosomes, autosomes and mitochondria
done
clear
View Answer play_arrow
question_answer 244) Presence of gills in the tadpole of frog indicates that
A)
fishes were amphibious in the past
done
clear
B)
fishes evolved from frog-like ancestors
done
clear
C)
frogs will have gills in future
done
clear
D)
frogs evolved from gilled ancestors
done
clear
View Answer play_arrow
question_answer 245) Which one of the following phenomena supports Darwin's concept of natural selection in organic evolution?
A)
Development of transgenic animals
done
clear
B)
Production of 'Dolly* the sheep by cloning
done
clear
C)
Prevalence of pesticide resistant insects
done
clear
D)
Development of organs from stem cells for organ transplantation
done
clear
View Answer play_arrow
question_answer 246) de Vries gave his mutation theory on organic evolution, while working on
A)
Althea rosea
done
clear
B)
Drosophila melanogaster
done
clear
C)
Oenothera lamarckiana
done
clear
D)
Pisum sativum
done
clear
View Answer play_arrow
question_answer 247) There are two opposing views about origin of modem man. According to one view. Homo erectus in Asia were the ancestors of Modern man. A study of variations of DNA however suggested African origin of Modern man. What kind of observation on DNA variation could suggest this?
A)
Greater variation in Africa than in Asia
done
clear
B)
Variation only in Asia and no variation in Africa
done
clear
C)
Greater variation in Asia than in Africa
done
clear
D)
Similar variation in Africa and Asia
done
clear
View Answer play_arrow
question_answer 248) Mast cells of connective tissue contain
A)
vasopressin and relaxin
done
clear
B)
heparin and histamine
done
clear
C)
heparin and calcitonin
done
clear
D)
serotonin and melanin
done
clear
View Answer play_arrow
question_answer 249) In the resting state of the neural membrane, diffusion due to concentration gradients, if allowed would drive
A)
into the cell
done
clear
B)
and
out of the cell
done
clear
C)
into the cell
done
clear
D)
out of the cell
done
clear
View Answer play_arrow
question_answer 250) Which of the following substances, if introduce the blood stream, would cause coagulation at the site of its introduction?
A)
Fibrinogen
done
clear
B)
Prothrombin
done
clear
C)
Heparin
done
clear
D)
Thromboplastin
done
clear
View Answer play_arrow
question_answer 251) Duodenum has characteristic Brunner's glands, which secrete two hormones called
A)
kinase, estrogen
done
clear
B)
secretin, cholecystokinin
done
clear
C)
prolactin, parathormone
done
clear
D)
estradiol, progesterone
done
clear
View Answer play_arrow
question_answer 252) Which one of the following is the correct matching of a vitamin, its nature and ist deficiency disease?
A)
Vitamin-A-Fat soluble-Nightblindness
done
clear
B)
Vitamin-K-Fat-soluble-Beri-beri
done
clear
C)
Vitamin-A-Fat-soluble-Beri-beri
done
clear
D)
Vitamin-K-Water-soluble-pellagra
done
clear
View Answer play_arrow
question_answer 253)
The richest sources of vitamin
A)
goat's liver and Spirulina
done
clear
B)
chocolate and green gram
done
clear
C)
rice and hen's egg
done
clear
D)
carrot and chicken's breast
done
clear
View Answer play_arrow
question_answer 254) Epithelial cells of the intestine involved in food absorption have on their surface
A)
pinocytic vesicles
done
clear
B)
phagocytic vesicles
done
clear
C)
zymogen granules
done
clear
D)
microvilli
done
clear
View Answer play_arrow
question_answer 255) A patient is generally advised to specially. consume more meat, lentils, milk and eggs in diet only when he suffers from
A)
kwashiorkor
done
clear
B)
rickets
done
clear
C)
anaemia
done
clear
D)
scurvy
done
clear
View Answer play_arrow
question_answer 256) A person is undergoing prolonged fasting. His urine will be found to contain abnormal quantities of
A)
fats
done
clear
B)
ketones
done
clear
C)
amino acids
done
clear
D)
glucose
done
clear
View Answer play_arrow
question_answer 257) Which group of three of the following five statements (1-5) contains all three correct statements regarding beri-beri? 1. A crippling disease prevalent among the native population of sub-Sahara Africa. 2. A deficiency disease caused by lack of thiamine (vitamin-Bi). 3. A nutritional disorder in infants and young children when the diet is persistently deficient in essential protein. 4. Occurs in those countries where the staple diet is polished rice. 5. The symptoms are pain from neuritis, paralysis, muscle wasting, progressive oedema, mental deterioration and finally heart failure.
A)
1,2 and 4
done
clear
B)
2, 3 and 5
done
clear
C)
1,3 and 5
done
clear
D)
2, 4 and S
done
clear
View Answer play_arrow
question_answer 258)
When
A)
shallower and slow
done
clear
B)
there is no effect on breathing
done
clear
C)
slow and deep
done
clear
D)
faster and deeper
done
clear
View Answer play_arrow
question_answer 259) Blood analysis of a patient reveals an unusually high quantity of car boxy hemoglobin content. Which of the following conclusions is most likely to be correct? The patient has been inhaling polluted air containing unusually high content of
A)
carbon disulphide
done
clear
B)
chloroform
done
clear
C)
carbon dioxide
done
clear
D)
carbon monoxide
done
clear
View Answer play_arrow
question_answer 260) During which stage in the complete oxidation of glucose are the greatest number of ATP molecules formed?
A)
Conversion of pyruvic add to acetyl Co-A
done
clear
B)
Electron transport chain
done
clear
C)
Glycolysis
done
clear
D)
Krebs' cycle
done
clear
View Answer play_arrow
question_answer 261) to the ABO system of blood groups, if both antigens are present but no antibody, the blood group of the individual would be
A)
B
done
clear
B)
0
done
clear
C)
AB
done
clear
D)
A
done
clear
View Answer play_arrow
question_answer 262) You are required to draw blood from a patient and to keep it in a test tube for analysis of blood corpuscles and plasma. You are also provided with the following four types of test tubes. Which of them will you not use for the purpose?
A)
Test tube containing calcium bicarbonate
done
clear
B)
Chilled test tube
done
clear
C)
Test tube containing heparin
done
clear
D)
Test tube containing sodium oxalate
done
clear
View Answer play_arrow
question_answer 263) The cardiac pace-maker in a patient fails to function normally. The doctors find that an artificial pace-maker is to be grafted in him. It is likely that it will be grafted at the site of
A)
atrioventricular bundle
done
clear
B)
Purkinje system
done
clear
C)
sinuatrial node
done
clear
D)
atrioventricular node
done
clear
View Answer play_arrow
question_answer 264) Uricotelism is found in
A)
mammals and birds
done
clear
B)
fishes and fresh water protozoans
done
clear
C)
birds, reptiles and insects
done
clear
D)
frogs and toads
done
clear
View Answer play_arrow
question_answer 265) A terrestrial animal must be able to
A)
excrete large amount of water in urine
done
clear
B)
conserve water
done
clear
C)
actively pump salts out through the skin
done
clear
D)
excrete large amount of salts in urine
done
clear
View Answer play_arrow
question_answer 266) In ornithine cycle, which of the following wastes are removed from the blood?
A)
Urea and urine
done
clear
B)
Ammonia and urea
done
clear
C)
and ammonia
done
clear
D)
and urea
done
clear
View Answer play_arrow
question_answer 267) The net pressure gradient that causes the fluid to filter out of the glomeruli into the capsule is
A)
20mmHg
done
clear
B)
75 mm Hg
done
clear
C)
30mmHg
done
clear
D)
50 mm Hg
done
clear
View Answer play_arrow
question_answer 268) ATP ase enzyme needed for muscle contraction is located in
A)
acrinin
done
clear
B)
troponin
done
clear
C)
myosin
done
clear
D)
actin
done
clear
View Answer play_arrow
question_answer 269) An acromian process is characteristically found in the
A)
pelvic girdle of mammals
done
clear
B)
skull of frog
done
clear
C)
pectoral girdle of mammals
done
clear
D)
sperm of mammals
done
clear
View Answer play_arrow
question_answer 270) Which of the following pairs is correctly matched?
A)
Cartilaginous - Skull bones joint
done
clear
B)
Hinge joint - Between vertebrae
done
clear
C)
Fibrous joint - Between phalanges
done
clear
D)
Gliding joint - Between zygapophyses of the successive vertebrae
done
clear
View Answer play_arrow
question_answer 271) Injury to vagus nerve in humans is not likely to affect
A)
tongue movements
done
clear
B)
gastrointestinal movements
done
clear
C)
pancreatic secretion
done
clear
D)
cardiac movements
done
clear
View Answer play_arrow
question_answer 272) In a man, abducens nerve is injured. Which one of the following functions will be affected?
A)
Movement of the eye ball
done
clear
B)
Swallowing
done
clear
C)
Movement of the tongue
done
clear
D)
Movement of the neck
done
clear
View Answer play_arrow
question_answer 273) One of the examples of the action of the autonomous nervous system is
A)
knee-jerk response
done
clear
B)
pupillary reflex
done
clear
C)
swallowing of food
done
clear
D)
peristalsis of the intestine
done
clear
View Answer play_arrow
question_answer 274) Parkinson's disease (characterized by tremors and progressive rigidity of limbs) is caused by degeneration of brain neurons that are involved in movement, control and make use of neurotransmitter
A)
acetylcholine
done
clear
B)
nor-epinephrine
done
clear
C)
dopamine
done
clear
D)
GABA
done
clear
View Answer play_arrow
question_answer 275) Chemically hormones are
A)
biogenic amines only
done
clear
B)
proteins, steroids and biogenic amines
done
clear
C)
proteins only
done
clear
D)
steroids only
done
clear
View Answer play_arrow
question_answer 276) Which one of the following pairs correctly matches a hormone with a diseases resulting from its deficiency?
A)
Luteinizing - Failure of ovulation hormone
done
clear
B)
Insulin - Diabetes insipidus
done
clear
C)
Thyroxine - Tetany
done
clear
D)
Parathyroid - Diabetes mellitus hormone
done
clear
View Answer play_arrow
question_answer 277) Which of the following hormones is not a secretion product of human placenta?
A)
Human chorionic gonadotropin
done
clear
B)
Prolactin
done
clear
C)
Estrogen
done
clear
D)
Progesterone
done
clear
View Answer play_arrow
question_answer 278) Which one of the following hormones is a modified amino acid?
A)
Epinephrine
done
clear
B)
Progesterone
done
clear
C)
Prostaglandin
done
clear
D)
Estrogen
done
clear
View Answer play_arrow
question_answer 279) Ovulation in the human female normally takes place during the menstrual cycle
A)
at the mid secretory phase
done
clear
B)
just before the end of the secretory phase
done
clear
C)
at the beginning of the proliferative phase
done
clear
D)
at the end of the proliferative phase
done
clear
View Answer play_arrow
question_answer 280) If mammalian ovum fails to get fertilized, which one of the following is unlikely?
A)
Corpus luteum will disintegrate
done
clear
B)
Estrogen secretion further decreases
done
clear
C)
Primary follicle starts developing
done
clear
D)
Progesterone secretion rapidly declines
done
clear
View Answer play_arrow
question_answer 281) Grey crescent is the area
A)
at the point of entry of sperm into ovum
done
clear
B)
just opposite to the site of entry of sperm into ovum
done
clear
C)
at the animal pole
done
clear
D)
at the vegetal pole
done
clear
View Answer play_arrow
question_answer 282) The maximum growth rate occurs in
A)
stationary phase
done
clear
B)
senescent phase
done
clear
C)
lag phase
done
clear
D)
exponential phase
done
clear
View Answer play_arrow
question_answer 283) In a mutational event, when adenine is replaced by guanine, it is a case of
A)
frameshift mutation
done
clear
B)
transcription
done
clear
C)
transition
done
clear
D)
transversion
done
clear
View Answer play_arrow
question_answer 284) DNA fingerprinting refers to
A)
molecular analysis of profiles of DNA samples
done
clear
B)
analysis of DNA samples using imprinting devices
done
clear
C)
techniques used for molecular analysis of different specimens of DNA
done
clear
D)
techniques used for identification of fingerprints of individuals
done
clear
View Answer play_arrow
question_answer 285) Lack of independent assortment of two genes A and B in fruit fly Drosophila is due to
A)
repulsion
done
clear
B)
recombination
done
clear
C)
linkage
done
clear
D)
crossing over
done
clear
View Answer play_arrow
question_answer 286) During transcription, if the nucleotide sequence of the DNA strand that is being coded as ATACG. Then the nucleotide sequence in the mRNA would be
A)
TATGC
done
clear
B)
TCTGG
done
clear
C)
UAUGC
done
clear
D)
UATGC
done
clear
View Answer play_arrow
question_answer 287) Extra nuclear inheritance is a consequence of presence of genes in
A)
mitochondria and chloroplasts
done
clear
B)
endoplasmic reticulum and mitochondria
done
clear
C)
ribosomes and chloroplast
done
clear
D)
lysosomes and ribosomes
done
clear
View Answer play_arrow
question_answer 288) A man and a woman, who do not show any apparent signs of a certain inherited disease have seven children (2 daughters and 5 sons). Three of the sons suffer from the given disease but none of the daughters are affected. Which of the following mode of inheritance do you suggest for this disease?
A)
Autosomal dominant
done
clear
B)
Sex-linked dominant
done
clear
C)
Sex-limited recessive
done
clear
D)
Sex-linked recessive
done
clear
View Answer play_arrow
question_answer 289) Telomerase is an enzyme, which is a
A)
repetitive DNA
done
clear
B)
RNA
done
clear
C)
simple protein
done
clear
D)
rib nucleoprotein
done
clear
View Answer play_arrow
question_answer 290) During transcription holoenzyme RNA polymerase binds to a DNA sequence and the DNA assumes a saddle like structure at that point. What is that sequence called?
A)
CAAT box
done
clear
B)
GGTT box
done
clear
C)
AAAT box
done
clear
D)
TATA box
done
clear
View Answer play_arrow
question_answer 291) Minerals deposition in bones and teeth is due to vitamin
A)
D
done
clear
B)
A
done
clear
C)
C
done
clear
D)
E
done
clear
View Answer play_arrow
question_answer 292) BHC and DDT belong to which class of pesticides?
A)
Organophosphates
done
clear
B)
Organochlorines
done
clear
C)
Carbonates
done
clear
D)
Triazines
done
clear
View Answer play_arrow
question_answer 293) Production of a human protein in bacteria by genetic engineering is possible because
A)
bacterial cell can carry out the RNA splicing reactions
done
clear
B)
the human chromosome can replicate in bacterial cell
done
clear
C)
the mechanism of gene regulation is identicp.1 in humans and bacteria
done
clear
D)
the genetic code is universal
done
clear
View Answer play_arrow
question_answer 294) From the following statements select the wrong one
A)
millepedes have two pairs of appendages in each segment of the body
done
clear
B)
prawn has two pairs of antennae
done
clear
C)
animals belonging to phylum-Porifera are exclusively marine
done
clear
D)
nematocysts are characteristic of the phylum-Cnidaria
done
clear
View Answer play_arrow
question_answer 295) Barophilic prokaryotes
A)
grow slowly in highly alkaline frozen lakes at high altitudes
done
clear
B)
occur in water containing high concentrations of barium hydroxide
done
clear
C)
grow and multiply in very deep marine sediments
done
clear
D)
readily grown and divides in sea water enriched in any soluble salt of barium
done
clear
View Answer play_arrow
question_answer 296) Cancer cells are more easily damaged by radiation than normal cells because they are
A)
starved of mutation
done
clear
B)
undergoing rapid division
done
clear
C)
different in structure
done
clear
D)
non-dividing
done
clear
View Answer play_arrow
question_answer 297) Which of the following in not true for a species?
A)
Members of a species can interbreed
done
clear
B)
Variations occur among members of a species
done
clear
C)
Each species is reproductively isolated from every other species
done
clear
D)
Gene flow does not occur between the populations of a species
done
clear
View Answer play_arrow
question_answer 298) Biodiversity act of India was passed by the Parliament in the year
A)
1996
done
clear
B)
1992
done
clear
C)
2002
done
clear
D)
2000
done
clear
View Answer play_arrow
question_answer 299) Which one of the following characters is not typical of the class-Mammalia?
A)
Seven cervical vertebrae
done
clear
B)
The codont dentition
done
clear
C)
Ten pairs of cranial nerves
done
clear
D)
Alveolar lungs
done
clear
View Answer play_arrow
question_answer 300) Cutaneous glands are
A)
gastric glands
done
clear
B)
sebaceous glands
done
clear
C)
thyroid glands
done
clear
D)
thymus glands
done
clear
View Answer play_arrow
question_answer 301) Fruit of coconut is
A)
drupe
done
clear
B)
berry
done
clear
C)
nut
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 302) Edible part of mango is
A)
epicarp
done
clear
B)
endocarp
done
clear
C)
mesocarp
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 303) System of classification proposed by Linnaeus
A)
botanical system of classification
done
clear
B)
sexual system of classification
done
clear
C)
natural system of classification
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 304) Highest unit of classification
A)
phylum
done
clear
B)
kingdom
done
clear
C)
class
done
clear
D)
series
done
clear
View Answer play_arrow
question_answer 305) Angiosperms differs from gymnosperms because
A)
embryo is present in it
done
clear
B)
tissue is present in it
done
clear
C)
vessel is present in its xylem
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 306) Common bread mould is
A)
yeast
done
clear
B)
Mucor
done
clear
C)
bacteria
done
clear
D)
virus
done
clear
View Answer play_arrow
question_answer 307) Which one of the following organisms may respire in the absence of oxygen?
A)
Azotobacter
done
clear
B)
Clostridium
done
clear
C)
Rhizobium
done
clear
D)
Lactobadllus
done
clear
View Answer play_arrow
question_answer 308) Micorrhiza is a
A)
parasitic relation between fungus and algae
done
clear
B)
symbiotic relation between fungus and algae
done
clear
C)
symbiotic relation between fungus and roots
done
clear
D)
parasitic relation between fungus and roots
done
clear
View Answer play_arrow
question_answer 309) Earliest settlers on barren land and rocks are
A)
mosses
done
clear
B)
lichens
done
clear
C)
ferns
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 310) Number of neck canal cells is archegonium of Funaria
A)
1
done
clear
B)
3
done
clear
C)
5
done
clear
D)
6-10
done
clear
View Answer play_arrow
question_answer 311) Nostoc is known to perform
A)
only photosynthesis
done
clear
B)
photosynthesis and nitrogen fix am simultaneously
done
clear
C)
only nitrogen fixation
done
clear
D)
either photosynthesis or nitrogen fixatior a time
done
clear
View Answer play_arrow
question_answer 312) In Funaria, haploid structure is
A)
capsule
done
clear
B)
protonema
done
clear
C)
collumella
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 313) Terpentine is obtained from
A)
pteridophyte
done
clear
B)
gymnosperm wood
done
clear
C)
bacteria
done
clear
D)
angiosperm flower
done
clear
View Answer play_arrow
question_answer 314) Tunica corpus theory was proposed by
A)
Schmidt
done
clear
B)
Hanstein
done
clear
C)
Nageli
done
clear
D)
Hoffmeister
done
clear
View Answer play_arrow
question_answer 315) Companion cells are associated with
A)
sieve tubes
done
clear
B)
xylem
done
clear
C)
collenchyma
done
clear
D)
cambium
done
clear
View Answer play_arrow
question_answer 316) Wood is the common name of
A)
phloem
done
clear
B)
vascular bundles
done
clear
C)
cambium
done
clear
D)
secondary xylem
done
clear
View Answer play_arrow
question_answer 317) Embryo is found in
A)
Ulothrix
done
clear
B)
Spirogyra
done
clear
C)
Funaria
done
clear
D)
Chlorella
done
clear
View Answer play_arrow
question_answer 318) Hydathode are found in
A)
leaf
done
clear
B)
stem
done
clear
C)
flower
done
clear
D)
root
done
clear
View Answer play_arrow
question_answer 319) Enzymes are basically
A)
vitamins
done
clear
B)
fat
done
clear
C)
protein
done
clear
D)
carbon
done
clear
View Answer play_arrow
question_answer 320) Which of the following is a C4-plant
A)
sugarcane
done
clear
B)
mango
done
clear
C)
apple
done
clear
D)
tomato
done
clear
View Answer play_arrow
question_answer 321) The first compound formed in photosynthesis of C3 plants is
A)
phosphoglyceric acid
done
clear
B)
starch
done
clear
C)
pyruvic acid
done
clear
D)
ribulose diphosphate
done
clear
View Answer play_arrow
question_answer 322) Ferredoxin is found in
A)
\[\text{PS-I}\]
done
clear
B)
\[\text{PS-II}\]
done
clear
C)
Hill reaction
done
clear
D)
\[{{\text{P}}_{680}}\]
done
clear
View Answer play_arrow
question_answer 323) Correct percentage of \[C{{O}_{2}}\] in atmosphere is
A)
0.03%
done
clear
B)
0.3%
done
clear
C)
1%
done
clear
D)
1.1%
done
clear
View Answer play_arrow
question_answer 324) The cotton thread fibres are
A)
fibres taken out from stem
done
clear
B)
epidermal hair of seed
done
clear
C)
epidermal hair of fruits
done
clear
D)
fibres taken out from roots
done
clear
View Answer play_arrow
question_answer 325) Which of the following is a test cross?
A)
\[Tt\times Tt\]
done
clear
B)
\[TT\times Tt\]
done
clear
C)
\[TT\times tt\]
done
clear
D)
\[tt\times Tt\]
done
clear
View Answer play_arrow
question_answer 326) Gibberellins were first discovered in fungal genus
A)
Mucor
done
clear
B)
Khizopus
done
clear
C)
Agaricus
done
clear
D)
Fusarium
done
clear
View Answer play_arrow
question_answer 327) Stem of Opuntia is
A)
phyllode
done
clear
B)
phylloclade
done
clear
C)
cladode
done
clear
D)
bulbils
done
clear
View Answer play_arrow
question_answer 328) Which of the following is a biofertilizer?
A)
Funaria
done
clear
B)
Fern
done
clear
C)
Anabaena
done
clear
D)
Fungus
done
clear
View Answer play_arrow
question_answer 329) RQ of succulents is
A)
1
done
clear
B)
4
done
clear
C)
0
done
clear
D)
\[\infty \]
done
clear
View Answer play_arrow
question_answer 330) Chemical added fro preservation of fruit juice is
A)
potassium meta-bisulphite
done
clear
B)
potassium permanganate
done
clear
C)
potassium chloride
done
clear
D)
potassium chlorate
done
clear
View Answer play_arrow
question_answer 331) The drug to control high blood pressure is obtained from
A)
Rauwolfia serpentine
done
clear
B)
Atropa belladonna
done
clear
C)
Colchicum autumnale
done
clear
D)
Digitalis purpurea
done
clear
View Answer play_arrow
question_answer 332) The botanical name of 'jute' is
A)
Linum eusitaissimum
done
clear
B)
Corchorus capsularis
done
clear
C)
Hibiscus sabdariffa
done
clear
D)
Crotalaria juncea
done
clear
View Answer play_arrow
question_answer 333) The genetic material in viruses are
A)
in some viruses both DNA and RNA
done
clear
B)
only RNA
done
clear
C)
only DNA
done
clear
D)
Both [b] and [c]
done
clear
View Answer play_arrow
question_answer 334) Food poisoning is caused by the infection of
A)
Bacillus megatherium
done
clear
B)
Salmonella typhi
done
clear
C)
E. coli
done
clear
D)
Clostridium botulinum
done
clear
View Answer play_arrow
question_answer 335) Cyanobacteria are included in
A)
Monera
done
clear
B)
Protista
done
clear
C)
Migae
done
clear
D)
Bryophyta
done
clear
View Answer play_arrow
question_answer 336) Isotopes used in photosynthesis are
A)
\[{{\text{C}}_{\text{11}}}\text{and }{{\text{P}}^{\text{32}}}\]
done
clear
B)
\[{{\text{C}}^{\text{15}}}\text{and }{{\text{P}}^{\text{32}}}\]
done
clear
C)
\[{{\text{C}}^{\text{16}}}\text{and }{{\text{N}}^{\text{15}}}\]
done
clear
D)
\[{{\text{C}}^{\text{14}}}\text{and}\,{{\text{O}}^{\text{18}}}\]
done
clear
View Answer play_arrow
question_answer 337) Mendel observed that certain characters did assort independently. Later researchers found it to be due to
A)
amitosis
done
clear
B)
linkage of characters
done
clear
C)
dominance of one trait over the other
done
clear
D)
crossing-over
done
clear
View Answer play_arrow
question_answer 338) Fertility of soil reduces from
A)
crop rotation
done
clear
B)
intensive cultivation
done
clear
C)
mixed cropping
done
clear
D)
nitrogen fixing bacteria
done
clear
View Answer play_arrow
question_answer 339) Stomata can open at night also in
A)
xerophyte
done
clear
B)
gametophyte
done
clear
C)
hydrophyte
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 340) Stomata open during day because the guard cells have
A)
outer walls thin
done
clear
B)
kidney shape
done
clear
C)
chlorophyll
done
clear
D)
large nuclei
done
clear
View Answer play_arrow
question_answer 341) \[{{\text{P}}_{\text{680}}}\] is related with
A)
\[\text{PS-I}\]
done
clear
B)
\[\text{PS-II}\]
done
clear
C)
Hill reaction
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 342) Organisms without any specific shape are
A)
mycoplasma
done
clear
B)
bacteria
done
clear
C)
viruses
done
clear
D)
cyanobacteria
done
clear
View Answer play_arrow
question_answer 343) In an aquatic environment, microscopic animals and plants are collectively known as
A)
commensals
done
clear
B)
herbivores
done
clear
C)
fauna and flora
done
clear
D)
planktons
done
clear
View Answer play_arrow
question_answer 344) Cell theory was proposed by
A)
Schleiden and Schwann
done
clear
B)
Watson and Crick
done
clear
C)
Darwin and Wallace
done
clear
D)
Mendel and Morgan
done
clear
View Answer play_arrow
question_answer 345) The tobacco mosaic virus was crystallized for the first time by
A)
W M Stanley
done
clear
B)
Louis Pasteur
done
clear
C)
Edward Jenner
done
clear
D)
Andre Lwoff
done
clear
View Answer play_arrow
question_answer 346) The main function of Golgi complex is
A)
translocation
done
clear
B)
phosphorylation
done
clear
C)
glyco-oxidation
done
clear
D)
fermentation
done
clear
View Answer play_arrow
question_answer 347) 5th June is
A)
World Environment Day
done
clear
B)
World AIDS Day
done
clear
C)
World Women Day
done
clear
D)
World Polio Day
done
clear
View Answer play_arrow
question_answer 348) Four daugher cells formed after meiosis are
A)
genetically similar
done
clear
B)
genetically different
done
clear
C)
anucleate
done
clear
D)
multinucleate
done
clear
View Answer play_arrow
question_answer 349) Which one of the following nutrients serve as microelements for plant growth?
A)
Manganese, copper, calcium
done
clear
B)
Sodium, potassium, boron
done
clear
C)
Sodium, nickel, chlorine
done
clear
D)
Copper, molybdenum, zinc
done
clear
View Answer play_arrow
question_answer 350) Mo is related with
A)
\[{{N}_{3}}\] fixation
done
clear
B)
flower induction
done
clear
C)
chromosome contraction
done
clear
D)
carbon collection
done
clear
View Answer play_arrow
question_answer 351) Which one of the following elements is present in chlorophylls?
A)
Manganese
done
clear
B)
Magnesium
done
clear
C)
Copper
done
clear
D)
Iron
done
clear
View Answer play_arrow
question_answer 352) Cellulose is
A)
monosaccharide
done
clear
B)
disaccharide
done
clear
C)
polysaccharide
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 353) Pulses yielding main family of plants
A)
Poaceae (Gramineae)
done
clear
B)
Cucurbitaceae
done
clear
C)
Liliaceae
done
clear
D)
Papilionaceae
done
clear
View Answer play_arrow
question_answer 354) Constituents of plasma membrane are
A)
Protein-lipid-protein
done
clear
B)
Lipid-protein-lipid
done
clear
C)
Protein-phosphorus
done
clear
D)
Lipid-sugar-protein
done
clear
View Answer play_arrow
question_answer 355) Lichens are
A)
parasitic relation between fungus and algae
done
clear
B)
symbiotic relation between fungus and algae
done
clear
C)
parasitic relation between fungus and root
done
clear
D)
symbiotic relation between fungus and roots
done
clear
View Answer play_arrow
question_answer 356) NADP is reduced into \[NADP{{H}_{2}}\]in
A)
\[\text{PS-I}\]
done
clear
B)
\[\text{PS-II}\]
done
clear
C)
Calvin cycle
done
clear
D)
Non-cyclic phosphorylation
done
clear
View Answer play_arrow
question_answer 357) Hill's reaction is completed in
A)
light
done
clear
B)
dark
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 358) Carbon cycle includes
A)
producer, consumer, decomposer
done
clear
B)
decomposer, consumer, producer
done
clear
C)
producer, decomposer, consumer
done
clear
D)
consumer, producer, decomposer
done
clear
View Answer play_arrow
question_answer 359) The cycling of elements in an ecosystem is called
A)
chemical cycle
done
clear
B)
biogeochemical cycle
done
clear
C)
geological cycle
done
clear
D)
geochemical cycle
done
clear
View Answer play_arrow
question_answer 360) Transcription of DNA is aided by
A)
RNA polymerase
done
clear
B)
DNA polymerase
done
clear
C)
exonuclease
done
clear
D)
recombinase
done
clear
View Answer play_arrow
question_answer 361) In which of the following protonema stage is found ?
A)
Riccia
done
clear
B)
Funaria
done
clear
C)
Selaginella
done
clear
D)
Pinus
done
clear
View Answer play_arrow
question_answer 362) When a seed germinates, while fruit is still attached to plant
A)
vivipary
done
clear
B)
scarification
done
clear
C)
germination
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 363) Emasculation is only achieved by
A)
removal of stigma
done
clear
B)
removal of sepals and petals
done
clear
C)
removal of anthers
done
clear
D)
removal of gynoecium
done
clear
View Answer play_arrow
question_answer 364) If the base sequence in DNA is 5' AAAA 3' thee the base sequence in mRNA will be
A)
5'UUUU3'
done
clear
B)
3'UUUU 5'
done
clear
C)
5'AAAA 3'
done
clear
D)
3TTTT 5'
done
clear
View Answer play_arrow
question_answer 365) Which of the following is a stop codon
A)
AUG. GUG, UUU
done
clear
B)
UGA, UAG, UAA
done
clear
C)
UUU, UAC, CUC
done
clear
D)
CUC, UAC, UAA
done
clear
View Answer play_arrow
question_answer 366) Which of the following is a viral STD disease ?
A)
AIDS
done
clear
B)
Cholera
done
clear
C)
Syphilis
done
clear
D)
Gonnorhoea
done
clear
View Answer play_arrow
question_answer 367) Correct order of molecular weight is
A)
DNA < rRNA < tRNA
done
clear
B)
DNA < mRNA < rRNA
done
clear
C)
rRNA < mRNA < DNA
done
clear
D)
tRNA < DNA < mRNA
done
clear
View Answer play_arrow
question_answer 368) Which of the following is found in fem?
A)
Eustele
done
clear
B)
Atactostele
done
clear
C)
Dictyostele
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 369) Characteristics of Cyanophyieae is
A)
phycocyanin
done
clear
B)
chlorophyll-\[b\]
done
clear
C)
phycoerythrin
done
clear
D)
chlorophyll-\[a\]
done
clear
View Answer play_arrow
question_answer 370) Which is closet to fungi in Protista?
A)
Hypophyte
done
clear
B)
True slime would
done
clear
C)
Actinomycetes
done
clear
D)
Ascomycetes
done
clear
View Answer play_arrow
question_answer 371) Pith and cortex belong to
A)
epidermis
done
clear
B)
ground tissue
done
clear
C)
vascular tissue
done
clear
D)
bundle sheath
done
clear
View Answer play_arrow
question_answer 372) Velamen of orchids are formed from
A)
phelloderm
done
clear
B)
phellogen
done
clear
C)
dermatogen
done
clear
D)
cortex
done
clear
View Answer play_arrow
question_answer 373) Total yield in one Krebs' cycle
A)
\[3\text{ }FAD{{H}_{2,}}~2\text{ }NAD{{H}_{2,}}1\text{ }ATP\]
done
clear
B)
\[2\text{ }FAD{{H}_{2,}}~2\text{ }NA{{D}_{2,}}~~~2\text{ }ATP\]
done
clear
C)
\[2\text{ }NAD{{H}_{2,}}~1\text{ }FAD{{H}_{2,}}2\text{ }ATP\]
done
clear
D)
\[3\text{ }NAD{{H}_{2,}}~1\text{ }FAD{{H}_{2,}}1\text{ }ATP\]
done
clear
View Answer play_arrow
question_answer 374) Edaphic factor is related with
A)
soil
done
clear
B)
water
done
clear
C)
air
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 375) The type of inflorescence in Tulsi
A)
cyathium
done
clear
B)
hypanthodium
done
clear
C)
verticellaster
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 376) The pyramid of energy in a forest ecosystem is
A)
always upright
done
clear
B)
always inverted
done
clear
C)
Both [a] and [b]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 377) The sites for protein synthesis in plants are
A)
chloroplasts
done
clear
B)
ribosomes
done
clear
C)
pyrenoids
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 378) Castor oil is yielded from which of the following?
A)
Brassica campestris
done
clear
B)
Sesamum indicum
done
clear
C)
Ricinus communis
done
clear
D)
Cocos nucifera
done
clear
View Answer play_arrow
question_answer 379) The process involved in the RNA formation on the DNA template is
A)
translation
done
clear
B)
transduction
done
clear
C)
transcription
done
clear
D)
transformation
done
clear
View Answer play_arrow
question_answer 380) The genes are responsible for growth and differentiation in an organism through regulation of
A)
translocation
done
clear
B)
transformation
done
clear
C)
transduction and translation
done
clear
D)
translation and transcription
done
clear
View Answer play_arrow
question_answer 381) DNA is present in
A)
nucleus only
done
clear
B)
mitochondrion only
done
clear
C)
chloroplast only
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 382) Nitrifying bacteria are those which can convert
A)
atmospheric nitrogen into ammonia
done
clear
B)
ammonia into nitrites
done
clear
C)
nitrites into nitrates
done
clear
D)
nitrates into ammonia
done
clear
View Answer play_arrow
question_answer 383) Which one of the following bacteria has potential for nitrogen fixation?
A)
Nitrosomonas
done
clear
B)
Nitrobacter
done
clear
C)
Nitrosococcus
done
clear
D)
Rhizobium
done
clear
View Answer play_arrow
question_answer 384) The importance of ecosystem lies in
A)
flow of energy
done
clear
B)
cycling of materials
done
clear
C)
Both [a] and [b]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 385) Which one of the following is a renewable source of energy?
A)
Petroleum
done
clear
B)
Coal
done
clear
C)
Nuclear fuel
done
clear
D)
Trees
done
clear
View Answer play_arrow
question_answer 386) The number of primary producers within a specified area would be maximum in
A)
grassland ecosystem
done
clear
B)
forest ecosystem
done
clear
C)
pond ecosystem
done
clear
D)
deserts
done
clear
View Answer play_arrow
question_answer 387) Nepenthes is a
A)
primary producer
done
clear
B)
consumer
done
clear
C)
Both [a] and [b]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 388) The smallest organisms, which cause disease among plants are
A)
viruses
done
clear
B)
fungi
done
clear
C)
mycoplasma
done
clear
D)
bacteria
done
clear
View Answer play_arrow
question_answer 389) How many ATP will be produced during the production of 1 molecule of acetyl Co-A from 1 molecule of pyruvic acid?
A)
3 ATP
done
clear
B)
5 ATP
done
clear
C)
8 ATP
done
clear
D)
38 ATP
done
clear
View Answer play_arrow
question_answer 390) End osulphon is a
A)
herbicide
done
clear
B)
weedicide
done
clear
C)
rodenticide
done
clear
D)
pesticide
done
clear
View Answer play_arrow
question_answer 391) In which kind of study is banding done?
A)
Creation of new species
done
clear
B)
Production of disease resistent variety
done
clear
C)
Mapping of chromosomes
done
clear
D)
Artificial pollination
done
clear
View Answer play_arrow
question_answer 392) Nobel Prize for the concept of jumping genes was awarded to
A)
H G Khorana
done
clear
B)
Barbara McClintock
done
clear
C)
Kornberg
done
clear
D)
Watson
done
clear
View Answer play_arrow
question_answer 393) Phytochrome was discovered by
A)
W Went
done
clear
B)
Gamer and Allard
done
clear
C)
F F Blackman
done
clear
D)
R Hill
done
clear
View Answer play_arrow
question_answer 394) Lateral meristem is responsible for which of the following
A)
Growth in length
done
clear
B)
Growth in thickness
done
clear
C)
Growth in parenchyma
done
clear
D)
Growth in cortex
done
clear
View Answer play_arrow
question_answer 395) The presence of auxins in a solution could be tested by
A)
Avena sativa stem tip test
done
clear
B)
carbon tetrachloride test
done
clear
C)
iodine test
done
clear
D)
defoliation test
done
clear
View Answer play_arrow
question_answer 396) Enzymes are sensitive to
A)
cold
done
clear
B)
cell wall
done
clear
C)
heat
done
clear
D)
pressure
done
clear
View Answer play_arrow
question_answer 397) A normal angiosperm embyo sac at the final stage of development has
A)
4 nuclei
done
clear
B)
8 nuclei
done
clear
C)
3 nuclei
done
clear
D)
7 nuclei
done
clear
View Answer play_arrow
question_answer 398) Coir of commerce is obtained from which part of the coconut?
A)
Epicarp
done
clear
B)
Mesocarp
done
clear
C)
Endocarp
done
clear
D)
Seed coat
done
clear
View Answer play_arrow
question_answer 399) Diphtheria is caused by
A)
viruses
done
clear
B)
eukaryotes
done
clear
C)
mycoplasma
done
clear
D)
bacteria
done
clear
View Answer play_arrow
question_answer 400) Reserpine drug is obtained from
A)
Rauwolfia serpentine
done
clear
B)
Withania somnifera
done
clear
C)
Atropa belladonna
done
clear
D)
Papaver sominiferum
done
clear
View Answer play_arrow

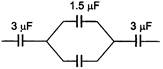
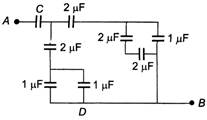
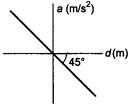
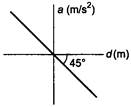
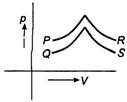
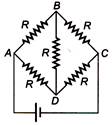
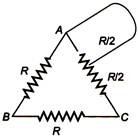
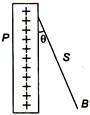
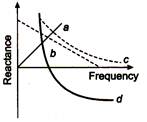
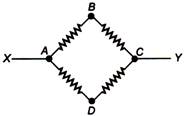

 (II)
(II)