question_answer 1) Two bodies A and B having temperatures \[0.31\] and \[0.81\] are radiating heat to the surrounding. The surrounding temperature is \[0.42\]. The ratio of rate of heat radiation of A to that of B is
A)
\[{{r}_{A}}\]
done
clear
B)
\[{{r}_{B}}\]
done
clear
C)
\[{{v}_{A}}\]
done
clear
D)
\[{{v}_{B}}\]
done
clear
View Answer play_arrow
question_answer 2) Two bulbs 40 W and 60 W and rated voltage 240 V are connected in series across a potential difference of 420 V. Which bulb will work at above its rated voltage?
A)
40 W bulb
done
clear
B)
60 W bulb
done
clear
C)
Both will work
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 3) Two cars A and B move along a concentric circular path of radius \[\frac{{{v}_{A}}}{{{v}_{B}}}\] and \[\frac{{{r}_{B}}}{{{r}_{A}}}\] with velocities \[\frac{{{r}_{A}}}{{{r}_{B}}}\] and \[\frac{r_{A}^{2}}{R_{B}^{2}}\] maintaining constant distance, then \[\frac{R_{B}^{2}}{R_{A}^{2}}\] is equal to
A)
\[3m/{{s}^{2}}\]
done
clear
B)
\[-3m/{{s}^{2}}\]
done
clear
C)
\[0.3m/{{s}^{2}}\]
done
clear
D)
\[-0.3m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 4) A body of mass 10 kg is moving with a constant velocity of 10 m/s. When a constant force acts for 4 s on it, it moves with a velocity 2 m/s in the opposite directions. The acceleration produced in it is
A)
\[4:1\]
done
clear
B)
\[1:3\]
done
clear
C)
\[3:1\]
done
clear
D)
\[1:9\]
done
clear
View Answer play_arrow
question_answer 5) The ratio of maximum and minimum intensities of two sources is \[1:16\]. The ratio of their amplitudes is
A)
\[6m/s\]
done
clear
B)
\[4m/s\]
done
clear
C)
\[2m/s\]
done
clear
D)
\[1m/s\]
done
clear
View Answer play_arrow
question_answer 6) A weight w is suspended from the mid-point of a rope, whose ends are at the same level. In order to make the rope perfectly horizontal, the force applied to each of its ends must be
A)
less than w
done
clear
B)
equal to w
done
clear
C)
equal to 2w
done
clear
D)
infinitely large
done
clear
View Answer play_arrow
question_answer 7) The velocity of a particle at an instant is 10 m/s. After 3 s its velocity will become 16 m/s. The velocity at 2 s, before the given instant will be
A)
\[10m/s\]
done
clear
B)
\[7m/s\]
done
clear
C)
\[12m/s\]
done
clear
D)
\[\frac{2v(n-1)}{n}\]
done
clear
View Answer play_arrow
question_answer 8) A heavy stone hanging from a massless string of length 15 m is projected horizontally with speed 147 m/s. The speed of the particle at the point where the tension in the string equals the weight of the particle is?
A)
\[\frac{v(n-1)}{n}\]
done
clear
B)
\[\frac{v(n+1)}{n}\]
done
clear
C)
\[\frac{v(n+1)}{n}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 9) When a body moves with constant speed in a circular path, then
A)
work done will be zero
done
clear
B)
acceleration will be zero
done
clear
C)
no force acts on the body
done
clear
D)
its velocity remains constant
done
clear
View Answer play_arrow
question_answer 10) A body initially at rest is moving with uniform acceleration a. Its velocity after n second is v. The displacement of the body in
A)
\[{{327}^{o}}C\]
done
clear
B)
\[{{427}^{o}}C\]
done
clear
C)
\[{{27}^{o}}C\]
done
clear
D)
\[0.52\]
done
clear
View Answer play_arrow
question_answer 11) The magnetic force on a point charge is \[0.31\] Here, q = electric charge v = velocity of point charge B = magnetic field The dimensions of B is
A)
\[0.81\]
done
clear
B)
\[0.42\]
done
clear
C)
\[{{r}_{A}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 12) The first diffraction minimum due to single slit diffraction is \[\theta \], for a light of wavelength \[{{r}_{B}}\]. If the width of the slit is \[{{v}_{A}}\], then the value of \[\theta \] is
A)
\[{{v}_{B}}\]
done
clear
B)
\[\frac{{{v}_{A}}}{{{v}_{B}}}\]
done
clear
C)
\[\frac{{{r}_{B}}}{{{r}_{A}}}\]
done
clear
D)
\[\frac{{{r}_{A}}}{{{r}_{B}}}\]
done
clear
View Answer play_arrow
question_answer 13) A stone of mass m tied to a string of length l is rotated in a circle with the other end of the string as the centre. The speed of the stone is v. If the string breaks, the stone will
A)
move towards the centre
done
clear
B)
move away from the centre
done
clear
C)
move along tangent
done
clear
D)
stop
done
clear
View Answer play_arrow
question_answer 14) A body of mass 2 kg is placed on rough horizontal plane. The coefficient of friction between body and plane is 0.2. Then,
A)
body will move in forward direction if F= 5N
done
clear
B)
body will move in backward direction with acceleration \[\frac{r_{A}^{2}}{R_{B}^{2}}\], if force F = 3N
done
clear
C)
If F = 3 N, then body will be in rest condition
done
clear
D)
Both (a) and (b) are correct
done
clear
View Answer play_arrow
question_answer 15) A particle moves with a velocity \[\frac{R_{B}^{2}}{R_{A}^{2}}\] under the influence of a constant force \[3m/{{s}^{2}}\]. The instantaneous power applied to the particle is
A)
\[-3m/{{s}^{2}}\]
done
clear
B)
\[0.3m/{{s}^{2}}\]
done
clear
C)
\[-0.3m/{{s}^{2}}\]
done
clear
D)
\[4:1\]
done
clear
View Answer play_arrow
question_answer 16) The potential energy of a particle of mass 5 kg moving in the xy-plane is given by \[1:3\], x and y being in metre. If the particle starts from rest from origin, then speed of particle at t= 2 s is
A)
\[3:1\]
done
clear
B)
\[1:9\]
done
clear
C)
\[1:16\]
done
clear
D)
\[6m/s\]
done
clear
View Answer play_arrow
question_answer 17) The ratio of radii of gyration of a circular disc and a circular ring of the same radii and same mass about a tangential axis in the plane is
A)
\[4m/s\]
done
clear
B)
\[2m/s\]
done
clear
C)
\[1m/s\]
done
clear
D)
\[10m/s\]
done
clear
View Answer play_arrow
question_answer 18) A particle performs uniform circular motion with an angular momentum L. If the frequency of particle motion is doubled and its KE is halved, the angular momentum becomes
A)
\[7m/s\]
done
clear
B)
\[12m/s\]
done
clear
C)
\[\frac{2v(n-1)}{n}\]
done
clear
D)
\[\frac{v(n-1)}{n}\]
done
clear
View Answer play_arrow
question_answer 19) The minimum energy required to launch a m kg satellite from earths surface in a circular orbit at an altitude of 2R, R is the radius of earth, will be
A)
\[\frac{v(n+1)}{n}\]
done
clear
B)
\[\frac{2v(n+1)}{n}\]
done
clear
C)
\[\frac{2v(n+1)}{n}\]
done
clear
D)
\[{{327}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 20) A satellite is moving on a circular path of radius r around the earth has a time period T. If its radius slightly increases by \[{{427}^{o}}C\], the change in its time period is
A)
\[{{27}^{o}}C\]
done
clear
B)
\[0.52\]
done
clear
C)
\[0.31\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 21) A particle executes SHM, its time period is 16 s. If its passes through the centre of oscillation then its velocity is 2 m/s at time 2 s. The amplitude will be
A)
\[0.81\]
done
clear
B)
\[0.42\]
done
clear
C)
\[{{r}_{A}}\]
done
clear
D)
\[{{r}_{B}}\]
done
clear
View Answer play_arrow
question_answer 22) A simple harmonic oscillator has amplitude A, angular velocity co, and mass m. Then, average energy in one time period will be
A)
\[{{v}_{A}}\]
done
clear
B)
\[{{v}_{B}}\]
done
clear
C)
\[\frac{{{v}_{A}}}{{{v}_{B}}}\]
done
clear
D)
\[\frac{{{r}_{B}}}{{{r}_{A}}}\]
done
clear
View Answer play_arrow
question_answer 23) The wave front due to a source situated at infinity is
A)
spherical
done
clear
B)
cylindrical
done
clear
C)
planar
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 24) An astronaut is approaching the moon. He sends out a radio signal of frequency 5000 MHz and the frequency of echo is different from that of the original frequency by 100 kHz. His velocity of approach with respect to the moon is
A)
\[\frac{{{r}_{A}}}{{{r}_{B}}}\]
done
clear
B)
\[\frac{r_{A}^{2}}{R_{B}^{2}}\]
done
clear
C)
\[\frac{R_{B}^{2}}{R_{A}^{2}}\]
done
clear
D)
\[3m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 25) When a sphere is taken to bottom of sea 1 km deep, it contracts by 0.01%. The bulk modulus of elasticity of the material of sphere is (Given : Density of water\[-3m/{{s}^{2}}\])
A)
\[0.3m/{{s}^{2}}\]
done
clear
B)
\[-0.3m/{{s}^{2}}\]
done
clear
C)
\[4:1\]
done
clear
D)
\[1:3\]
done
clear
View Answer play_arrow
question_answer 26) The temperature of \[3:1\] at which the rms velocity of its molecules is seven times the rms velocity of the molecules of nitrogen at 300 K is
A)
\[1:9\]
done
clear
B)
\[1:16\]
done
clear
C)
\[6m/s\]
done
clear
D)
\[4m/s\]
done
clear
View Answer play_arrow
question_answer 27) Gas exerts pressure on the walls of the container because
A)
gas has weight
done
clear
B)
gas molecules have momentum
done
clear
C)
gas molecules collide with each other
done
clear
D)
gas molecules collide with the walls of the container
done
clear
View Answer play_arrow
question_answer 28) The inside and outside temperatures of a refrigerator are 273 K and 303 K respectively. Assuming that refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be
A)
\[2m/s\]
done
clear
B)
\[1m/s\]
done
clear
C)
\[10m/s\]
done
clear
D)
\[7m/s\]
done
clear
View Answer play_arrow
question_answer 29) The wave number of stokes and anti-stokes lines in Raman spectrum are \[12m/s\] and \[\frac{2v(n-1)}{n}\]respectively. The wave number of exciting line is
A)
\[\frac{v(n-1)}{n}\]
done
clear
B)
\[\frac{v(n+1)}{n}\]
done
clear
C)
\[\frac{2v(n+1)}{n}\]
done
clear
D)
\[{{327}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 30) The surface temperature of the sun is T K and the solar constant for a plate is S. The sun subtends an angle 9 at the planet. Then,
A)
\[{{427}^{o}}C\]
done
clear
B)
\[{{27}^{o}}C\]
done
clear
C)
\[0.52\]
done
clear
D)
\[0.31\]
done
clear
View Answer play_arrow
question_answer 31) A body at a temperature of \[0.81\] and has surface area\[0.42\], radiates 300 J of energy each minute. The emissivity is (Given : Boltzman constant \[{{r}_{A}}\])
A)
e = 0.18
done
clear
B)
e = 0.02
done
clear
C)
e = 0.2
done
clear
D)
e = 0.15
done
clear
View Answer play_arrow
question_answer 32) If at NTP velocity of sound in a gas is 1150 m/s, then the rms velocity of gas molecules at NTP is (Given \[{{r}_{B}}\])
A)
\[{{v}_{A}}\]
done
clear
B)
\[{{v}_{B}}\]
done
clear
C)
\[\frac{{{v}_{A}}}{{{v}_{B}}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 33) If \[\frac{{{r}_{B}}}{{{r}_{A}}}\] = surface charge density, E = electric permittivity, the dimensions of \[\frac{{{r}_{A}}}{{{r}_{B}}}\] are same as
A)
electric force
done
clear
B)
electric field intensity
done
clear
C)
pressure
done
clear
D)
electric charge
done
clear
View Answer play_arrow
question_answer 34)
For circuit the equivalent capacitance between P and Q is
A)
\[\frac{r_{A}^{2}}{R_{B}^{2}}\]
done
clear
B)
\[\frac{R_{B}^{2}}{R_{A}^{2}}\]
done
clear
C)
\[3m/{{s}^{2}}\]
done
clear
D)
\[-3m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 35) Masses m and M on pulley move 0.6 m in 4 s. What is ratio of \[0.3m/{{s}^{2}}\]?
A)
\[-0.3m/{{s}^{2}}\]
done
clear
B)
\[4:1\]
done
clear
C)
\[1:3\]
done
clear
D)
\[3:1\]
done
clear
View Answer play_arrow
question_answer 36) n-alpha particles per second are emitted from N atoms of a radioactive element. The half-life of radioactive element is
A)
\[1:9\]
done
clear
B)
\[1:16\]
done
clear
C)
\[6m/s\]
done
clear
D)
\[4m/s\]
done
clear
View Answer play_arrow
question_answer 37) In nuclear reaction, \[2m/s\] where, M denotes
A)
electron
done
clear
B)
positron
done
clear
C)
proton
done
clear
D)
neutron
done
clear
View Answer play_arrow
question_answer 38) In terms of Rydberg constant R, the wave number of the first Banner line is
A)
R
done
clear
B)
\[1m/s\]
done
clear
C)
\[10m/s\]
done
clear
D)
\[7m/s\]
done
clear
View Answer play_arrow
question_answer 39) Three particles having charges in the ratio of 2:3:5, produce the same point on the photographic film in Thomson experiment. Their masses are in the ratio of
A)
\[12m/s\]
done
clear
B)
\[\frac{2v(n-1)}{n}\]
done
clear
C)
\[\frac{v(n-1)}{n}\]
done
clear
D)
\[\frac{v(n+1)}{n}\]
done
clear
View Answer play_arrow
question_answer 40) Polarisation of light proves
A)
corpuscular nature of light
done
clear
B)
quantum nature of light
done
clear
C)
transverse wave nature of light
done
clear
D)
longitudinal wave nature of light
done
clear
View Answer play_arrow
question_answer 41) In Youngs double slit experiment, the spacing between the slits is d and wavelength of light used is \[\frac{2v(n+1)}{n}\]. If the angular width of a fringe formed on a distant screen is 1°, then value of d is
A)
\[{{327}^{o}}C\]
done
clear
B)
\[{{427}^{o}}C\]
done
clear
C)
\[{{27}^{o}}C\]
done
clear
D)
\[0.52\]
done
clear
View Answer play_arrow
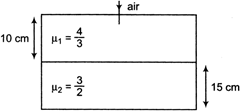
question_answer 42)
Considering normal incidence of ray, the equivalent refractive index of combination of two slabs shown in figure is
A)
\[0.31\]
done
clear
B)
\[0.81\]
done
clear
C)
\[0.42\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 43) The electric field E and magnetic field B in electromagnetic waves are
A)
parallel to each other
done
clear
B)
inclined at an angle of \[{{r}_{A}}\]
done
clear
C)
perpendicular to each other
done
clear
D)
opposite to each other
done
clear
View Answer play_arrow
question_answer 44) The correct option, if speed of gamma rays, X-rays and micro waves are \[{{r}_{B}}\] and \[{{v}_{A}}\] respectively will be
A)
\[{{v}_{B}}\]
done
clear
B)
\[\frac{{{v}_{A}}}{{{v}_{B}}}\]
done
clear
C)
\[\frac{{{r}_{B}}}{{{r}_{A}}}\]
done
clear
D)
\[\frac{{{r}_{A}}}{{{r}_{B}}}\]
done
clear
View Answer play_arrow
question_answer 45) The wavelength of a radio wave of frequency of 1 MHz is
A)
\[\frac{r_{A}^{2}}{R_{B}^{2}}\]
done
clear
B)
\[\frac{R_{B}^{2}}{R_{A}^{2}}\]
done
clear
C)
\[3m/{{s}^{2}}\]
done
clear
D)
\[-3m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 46) An AC is represented by \[0.3m/{{s}^{2}}\] V and is applied over a resistance of \[-0.3m/{{s}^{2}}\]. The heat produced in 7 min is
A)
\[4:1\]
done
clear
B)
\[1:3\]
done
clear
C)
\[3:1\]
done
clear
D)
\[1:9\]
done
clear
View Answer play_arrow
question_answer 47) The couple acting on a magnet of length 10 cm and pole strength 15 Am, kept in a field of \[1:16\] at an angle of \[6m/s\], is
A)
\[4m/s\]
done
clear
B)
\[2m/s\]
done
clear
C)
\[1m/s\]
done
clear
D)
\[10m/s\]
done
clear
View Answer play_arrow
question_answer 48) At a point on the right bisector of a magnetic dipole, the magnetic
A)
potential varies as \[7m/s\]
done
clear
B)
potential is zero at all points on the right bisector
done
clear
C)
field varies as \[12m/s\]
done
clear
D)
field is perpendicular to the axis of dipole
done
clear
View Answer play_arrow
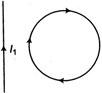
question_answer 49)
In the given figure, the loop is fixed but straight wire can move. The straight wire will
A)
remain stationary
done
clear
B)
move towards the loop
done
clear
C)
move away from the loop
done
clear
D)
rotates about the axis
done
clear
View Answer play_arrow
question_answer 50) If two identical heaters each rated as (1000 W, 220 V) are connected in parallel to 220 V, then the total power consumed is
A)
200 W
done
clear
B)
2500 W
done
clear
C)
250 W
done
clear
D)
2000 W
done
clear
View Answer play_arrow
question_answer 51) Formula of gypsum is
A)
\[{{(CaS{{O}_{4}})}_{2}}.{{H}_{2}}O\]
done
clear
B)
\[CaS{{O}_{4}}.\frac{1}{2}{{H}_{2}}O\]
done
clear
C)
\[CaS{{O}_{4}}.\,\,2{{H}_{2}}O\]
done
clear
D)
\[CaS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 52) \[\text{ }\!\!\alpha\!\!\text{ -}\]sulphur and \[\text{ }\!\!\beta\!\!\text{ -}\]sulphur are in a state of equilibrium at
A)
critical temperature
done
clear
B)
Boyles temperature
done
clear
C)
any temperature
done
clear
D)
transition temperature
done
clear
View Answer play_arrow
question_answer 53) The purification method that is represented by \[Ti+2{{I}_{2}}\xrightarrow{{}}Ti{{I}_{4}}\xrightarrow{\Delta }Ti+2{{I}_{2}}\] is called
A)
Monds process
done
clear
B)
Zone-refining
done
clear
C)
liquation
done
clear
D)
van-Arkel process
done
clear
View Answer play_arrow
question_answer 54) Which of the following substances consists of only one element?
A)
Marble
done
clear
B)
Sand
done
clear
C)
Diamond
done
clear
D)
Glass
done
clear
View Answer play_arrow
question_answer 55) Which one of the following cations gives a brick red flame by flame test?
A)
\[B{{a}^{2+}}\]
done
clear
B)
\[S{{r}^{2+}}\]
done
clear
C)
\[C{{a}^{2+}}\]
done
clear
D)
\[Z{{n}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 56) For which element of first transition series, the oxidation potential value \[(M\to {{M}^{2+}}+2{{e}^{-}})\] is lowest?
A)
\[Mn\]
done
clear
B)
\[Fe\]
done
clear
C)
\[Ni\]
done
clear
D)
\[Cu\]
done
clear
View Answer play_arrow
question_answer 57) A compound of zinc which is white in cold state and yellow in hot state, is
A)
\[ZnS\]
done
clear
B)
\[ZnO\]
done
clear
C)
\[ZnC{{l}_{2}}\]
done
clear
D)
\[ZnS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 58) Which one of the following is wrongly matched?
A)
\[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] - Square planar
done
clear
B)
\[[Ni{{(CO)}_{4}}]\] - Neutral ligand
done
clear
C)
\[{{[Fe{{(C{{N}_{6}})}_{4}}]}^{3-}}\] \[-s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[{{[Co{{(en)}_{3}}]}^{3+}}\] - Follows EAN rule
done
clear
View Answer play_arrow
question_answer 59) The compound which does not show par magnetism, is
A)
\[[Cu{{(N{{H}_{3}})}_{4}}]C{{l}_{2}}\]
done
clear
B)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
C)
\[NO\]
done
clear
D)
\[N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 60) The activation energy for a reaction at the temperature T K was found to be 2.303 RT J \[mo{{l}^{-1}}\]. The ratio of the rate constant to Arrhenius factor is
A)
\[{{10}^{-1}}\]
done
clear
B)
\[{{10}^{-2}}\]
done
clear
C)
\[2\times {{10}^{-3}}\]
done
clear
D)
\[2\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 61) An element forms a solid oxide which when dissolved in water, forms an acidic solution. The element is
A)
neon
done
clear
B)
sodium
done
clear
C)
phosphorus
done
clear
D)
sulphur
done
clear
View Answer play_arrow
question_answer 62) What is the product obtained when \[MnS{{O}_{4}}\] in solution is boiled with \[Pb{{O}_{2}}\]and concentrated \[HN{{O}_{3}}\]?
A)
\[Mn{{O}_{2}}\]
done
clear
B)
\[HMn{{O}_{4}}\]
done
clear
C)
\[M{{n}_{3}}{{O}_{4}}\]
done
clear
D)
\[PbMn{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 63) Lead nitrate on heating gives lead oxide, nitrogen dioxide and oxygen. This reaction is known as
A)
combustion
done
clear
B)
combination
done
clear
C)
displacement
done
clear
D)
decomposition
done
clear
View Answer play_arrow
question_answer 64) The number of d-electrons in \[[Cr{{({{H}_{2}}O)}_{4}}]\] is
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 65) Coordination number for copper \[(Cu)\] is
A)
1
done
clear
B)
6
done
clear
C)
8
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 66) Silver nitrate on heating gives
A)
\[AgO\] and \[N{{O}_{2}}\]
done
clear
B)
\[AgO\] , \[NO\] and \[{{O}_{2}}\]
done
clear
C)
\[Ag\] and \[N{{O}_{2}}\]
done
clear
D)
\[Ag\], \[N{{O}_{2}}\] and \[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 67) Which one of the following is an example of a true peroxide?
A)
\[N{{O}_{2}}\]
done
clear
B)
\[Mn{{O}_{2}}\]
done
clear
C)
\[Ba{{O}_{2}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 68) The molarity of 98% \[{{H}_{2}}S{{O}_{4}}\] (d= 1.8 g/mL) by weight, is
A)
\[6M\]
done
clear
B)
\[18M\]
done
clear
C)
\[10M\]
done
clear
D)
\[4M\]
done
clear
View Answer play_arrow
question_answer 69) 20 mL of 10 N \[HCl\] are mixed with 10 mL of 36 N \[{{H}_{2}}S{{O}_{4}}\] and the mixture is made 1 L. Normality of the mixture will be
A)
\[0.56N\]
done
clear
B)
\[0.50N\]
done
clear
C)
\[0.40N\]
done
clear
D)
\[0.35N\]
done
clear
View Answer play_arrow
question_answer 70) The energy of an electron in the 3rd orbit of an atom is \[-E\]. The energy of an electron in the first orbit will be
A)
\[-3E\]
done
clear
B)
\[-\frac{E}{3}\]
done
clear
C)
\[-\frac{E}{9}\]
done
clear
D)
\[-9E\]
done
clear
View Answer play_arrow
question_answer 71) Which emits P-particles?
A)
\[_{1}^{3}H\]
done
clear
B)
\[_{6}^{14}C\]
done
clear
C)
\[_{19}^{40}K\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 72) What is X in the nuclear reaction\[_{7}^{14}N+_{1}^{1}H\xrightarrow{{}}_{8}^{15}O+X\]?
A)
\[_{1}^{1}H\]
done
clear
B)
\[_{0}^{1}n\]
done
clear
C)
\[\gamma \]
done
clear
D)
\[_{-1}^{0}e\]
done
clear
View Answer play_arrow
question_answer 73) Which one of the following does not involve coagulation?
A)
Formation of delta regions
done
clear
B)
Peptization
done
clear
C)
Treatment of drinking water by potash alum
done
clear
D)
Clotting of blood by the use of ferric chloride
done
clear
View Answer play_arrow
question_answer 74) Benzaldehyde and acetone can be best distinguished using
A)
Fehlings solution
done
clear
B)
Sodium hydroxide solution
done
clear
C)
2, 4-DNP
done
clear
D)
Tollens reagent
done
clear
View Answer play_arrow
question_answer 75) For the chemical reaction \[A\xrightarrow{{}}E\], it is found that the rate of the reaction doubles when the concentration of A is increased four times. The order in terms of A for this reaction is
A)
two
done
clear
B)
one
done
clear
C)
zero
done
clear
D)
half
done
clear
View Answer play_arrow
question_answer 76) Consider the following gaseous equilibria with equilibrium constants \[{{K}_{1}}\] and \[{{K}_{2}}\] respectively \[S{{O}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)S{{O}_{3}}(g)\]\[2S{{O}_{3}}(g)2S{{O}_{2}}(g)+{{O}_{2}}(g)\] The equilibrium constants are related as
A)
\[K_{1}^{2}=\frac{1}{{{K}_{2}}}\]
done
clear
B)
\[2{{K}_{1}}=K_{2}^{2}\]
done
clear
C)
\[{{K}_{2}}=\frac{2}{K_{1}^{2}}\]
done
clear
D)
\[K_{2}^{2}=\frac{1}{{{K}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 77) Which one of the following is paramagnetic?
A)
\[{{O}_{2}}\]
done
clear
B)
\[{{N}_{2}}\]
done
clear
C)
\[He\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 78) The compound which is non-linear is
A)
\[C{{O}_{2}}\]
done
clear
B)
\[C{{S}_{2}}\]
done
clear
C)
\[HgC{{l}_{2}}\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 79) The valency of the element having atomic number 9 is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 80) From the knowledge of the position of radium in the Periodic Table, which of the following statements would you expect to be false?
A)
\[RaS{{O}_{4}}\] is insoluble in water
done
clear
B)
\[RaS{{O}_{4}}\] is insoluble in \[HN{{O}_{3}}\]
done
clear
C)
\[RaS{{O}_{4}}\] is a white solid
done
clear
D)
\[RaS{{O}_{4}}\] is a colourless liquid
done
clear
View Answer play_arrow
question_answer 81) Hexa-2-ene-4-yne is
A)
\[C{{H}_{3}}-C{{H}_{2}}-C\equiv C-CH\equiv C{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}-C\equiv C-CH=CH-C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}-CH=CH-C\equiv CH\]
done
clear
D)
\[C{{H}_{3}}-C\equiv C-C{{H}_{2}}-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 82) The number of unpaired electrons in carbon atom is
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
question_answer 83) The end product of 4n series is
A)
\[_{82}^{208}Pb\]
done
clear
B)
\[_{82}^{207}Pb\]
done
clear
C)
\[_{82}^{209}Pb\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 84) Which statement is correct?
A)
Ethanol is more acidic than phenol
done
clear
B)
Phenol is more acidic than ethanol
done
clear
C)
Phenol is more acidic than benzoic acid
done
clear
D)
Acidity of phenol and benzoic acid is about equal
done
clear
View Answer play_arrow
question_answer 85) Which chloride is least reactive with the hydrolysis point of view?
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}CCl\]
done
clear
D)
\[C{{H}_{2}}=CH-Cl\]
done
clear
View Answer play_arrow
question_answer 86) The reaction of acetone with \[PC{{l}_{5}}\] gives
A)
\[C{{H}_{3}}COC{{H}_{2}}Cl\]
done
clear
B)
\[C{{H}_{3}}COCHC{{l}_{2}}\]
done
clear
C)
done
clear
D)
\[ClC{{H}_{2}}COC{{H}_{2}}Cl\]
done
clear
View Answer play_arrow
question_answer 87) Towards electrophonic reagents
A)
ethene is more reactive than ethyne
done
clear
B)
ethene is less reactive than ethyne
done
clear
C)
both have equal reactivity
done
clear
D)
the reactivity of both cannot be predicted
done
clear
View Answer play_arrow
question_answer 88) On heating a mixture of sodium benzoate and soda lime, ...... is formed.
A)
toluene
done
clear
B)
phenol
done
clear
C)
benzene
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 89) In the following reaction,\[2C{{H}_{2}}=C{{H}_{2}}+{{S}_{2}}C{{l}_{2}}\to \Pr oduct\]The product is
A)
mustard gas
done
clear
B)
lewisite
done
clear
C)
polythene
done
clear
D)
Teflon
done
clear
View Answer play_arrow
question_answer 90) What is the initial product of the acidic hydrolysis of a cyanide?
A)
A primary amide
done
clear
B)
An isocyanide
done
clear
C)
An isocyanate
done
clear
D)
A nitrile
done
clear
View Answer play_arrow
question_answer 91) In the preparation of nylon-6 from cyclohexanone oxime, use is made of a rearrangement reaction. This rearrangement reaction is called
A)
Wolff rearrangement
done
clear
B)
Amadori rearrangement
done
clear
C)
Curtius rearrangement
done
clear
D)
Beckmann rearrangement
done
clear
View Answer play_arrow
question_answer 92) In the reaction,\[C{{l}_{2}}+C{{H}_{4}}\xrightarrow{hv}C{{H}_{3}}Cl+HCl\]presence of a small amount of oxygen
A)
increases the rate of reaction for a brief period of time
done
clear
B)
decreases the rate of reaction for a brief period of time
done
clear
C)
does not affect the rate of reaction
done
clear
D)
completely stops the reaction
done
clear
View Answer play_arrow
question_answer 93) An example of a lipid is
A)
lard
done
clear
B)
keratin
done
clear
C)
glutathione
done
clear
D)
oxytocin
done
clear
View Answer play_arrow
question_answer 94) Which of the following reagents can be used for distinguishing the three classes of alcohols?
A)
Fentons reagent
done
clear
B)
Lucas reagent
done
clear
C)
Schiffs reagent
done
clear
D)
Tollens reagent
done
clear
View Answer play_arrow
question_answer 95) Natural rubber is
A)
a polymer of 1, 3-butadiene
done
clear
B)
a polymer of ethylene
done
clear
C)
a polymer of 2-methyl-l, 3-butadiene
done
clear
D)
a polymer of styrene
done
clear
View Answer play_arrow
question_answer 96) In alkaline medium, alanine exists predominantly as
A)
anion
done
clear
B)
Zwitter ion
done
clear
C)
cation
done
clear
D)
covalent form
done
clear
View Answer play_arrow
question_answer 97) Number of resonating structures for Dewars benzene will be
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
question_answer 98) Which of the following reactions can be used for the synthesis of an alkene?
A)
Chugaev reaction
done
clear
B)
Dakin reaction
done
clear
C)
Reimer-Tiemann reaction
done
clear
D)
Wurtz-Fittig reaction
done
clear
View Answer play_arrow
question_answer 99) The reaction,\[{{R}_{2}}C=NN{{H}_{2}}\xrightarrow{{{C}_{2}}{{H}_{5}}ONa}{{R}_{2}}C{{H}_{2}}+{{N}_{2}}\] is called
A)
Clemmensen reduction
done
clear
B)
Hunsdiecker reaction
done
clear
C)
Tischenko reaction
done
clear
D)
Wolff-Kishner reduction
done
clear
View Answer play_arrow
question_answer 100) Molecules of a noble gas do not possess vibrational energy because a noble gas
A)
is monoatomic
done
clear
B)
is chemically inert
done
clear
C)
has completely filled shells
done
clear
D)
is diamagnetic
done
clear
View Answer play_arrow
question_answer 101) The number of hydrogen bonds between adenine and thymine in a DNA molecule is
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
eight
done
clear
View Answer play_arrow
question_answer 102) The enzyme, which combines with non-protein part to form a functional enzyme known as
A)
co-enzyme
done
clear
B)
holoenzyme
done
clear
C)
apoenzyme
done
clear
D)
prosthetic group
done
clear
View Answer play_arrow
question_answer 103) Which of the following enzyme digest protein in stomach?
A)
Trypsin
done
clear
B)
Pepsin
done
clear
C)
Crepsin
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 104) Passive food ingestion in Amoeba is known as
A)
import
done
clear
B)
invagination
done
clear
C)
circumfluence
done
clear
D)
circumvallation
done
clear
View Answer play_arrow
question_answer 105) The slime moulds are characterized by the presence of
A)
elaters
done
clear
B)
pseudoelaters
done
clear
C)
capillitium
done
clear
D)
capitulum
done
clear
View Answer play_arrow
question_answer 106) Ecdysone is secreted from
A)
insecta
done
clear
B)
trematoda
done
clear
C)
nematoda
done
clear
D)
polycheta
done
clear
View Answer play_arrow
question_answer 107) In the life cycle of mosquito, common-shaped stage is
A)
larval stage
done
clear
B)
pupal stage
done
clear
C)
imago stage
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 108) Haemocoel is found in
A)
Hydra and Aurelia
done
clear
B)
Taenia and Ascaris
done
clear
C)
Cockroach and Pila
done
clear
D)
Balanoglossus and Herdmania
done
clear
View Answer play_arrow
question_answer 109) The group of anamniota includes
A)
reptiles and birds
done
clear
B)
birds and mammals
done
clear
C)
fishes and amphibians
done
clear
D)
reptiles and mammals
done
clear
View Answer play_arrow
question_answer 110) The excretory material of bony fish is
A)
urea
done
clear
B)
protein
done
clear
C)
ammonia
done
clear
D)
amino acid
done
clear
View Answer play_arrow
question_answer 111) Different colours of frog skin are controlled by
A)
hormones
done
clear
B)
melanocytes
done
clear
C)
nervous system
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 112) Blastula of frog has
A)
blastopore
done
clear
B)
blastocoel
done
clear
C)
archenteron
done
clear
D)
gastropore
done
clear
View Answer play_arrow
question_answer 113) Carotene pigment is found in the cells of
A)
dermis
done
clear
B)
epidermis
done
clear
C)
adipose cell
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 114) Deboves membrane is a layer of
A)
muscular tissue
done
clear
B)
epithelial tissue
done
clear
C)
connective tissue
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 115) Achilles tendon is associated with
A)
gluteus muscle
done
clear
B)
hamstring muscle
done
clear
C)
quadriceps muscle
done
clear
D)
gastrocnemius muscle
done
clear
View Answer play_arrow
question_answer 116) The leucocytes contain, which of the following in large quantity?
A)
Basophils
done
clear
B)
Neutrophils
done
clear
C)
Eosinophils
done
clear
D)
Monocytes
done
clear
View Answer play_arrow
question_answer 117) Which part of our body secreted the hormone secretin?
A)
Ileum
done
clear
B)
Stomach
done
clear
C)
Duodenum
done
clear
D)
Oesophagus
done
clear
View Answer play_arrow
question_answer 118) During inspiration, the diaphragm
A)
expands
done
clear
B)
shows no change
done
clear
C)
contracts and flattens
done
clear
D)
relaxes to become dome-shaped
done
clear
View Answer play_arrow
question_answer 119) The oxygen toxicity is related with
A)
blood poisoning
done
clear
B)
collapse of alveolar walls
done
clear
C)
failure of ventilation of lungs
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 120) Cardiac output is determined by
A)
heart rate
done
clear
B)
stroke volume
done
clear
C)
blood flow
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 121) The important function of lymph is to
A)
transport oxygen to the brain
done
clear
B)
transport carbon dioxide to the lungs
done
clear
C)
return RBCs to the lymph nodes
done
clear
D)
return interstitial fluid to the blood
done
clear
View Answer play_arrow
question_answer 122) The lining of intestine and kidneys in humans is
A)
keratinized
done
clear
B)
brush border
done
clear
C)
ciliated
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 123) The yellow colour of urine is due to the presence of
A)
urea
done
clear
B)
uric acid
done
clear
C)
urochrome
done
clear
D)
bilirubin
done
clear
View Answer play_arrow
question_answer 124) The Leydig cells secrete
A)
oestrogen
done
clear
B)
testosterone
done
clear
C)
progesterone
done
clear
D)
corticosterone
done
clear
View Answer play_arrow
question_answer 125) The function of pineal body is to
A)
lighten the skin colours
done
clear
B)
control sexual behavior
done
clear
C)
regulates the period of puberty
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 126) Biomagnification refers to
A)
rapid growth due to excessive intake of nutrients
done
clear
B)
increase in population size
done
clear
C)
decrease in population size
done
clear
D)
increase in concentration of non-degradable pollutants as they pass through food chain
done
clear
View Answer play_arrow
question_answer 127) During the conduction of never impulse, the action potential is the result of movement of
A)
\[N{{a}^{+}}\] from intracellular fluid to extracellular fluid
done
clear
B)
\[N{{a}^{+}}\] from extracellular fluid to intracellular fluid
done
clear
C)
\[N{{a}^{+}}\] towards both directions
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 128) The AIDS virus spreads through
A)
killer-T cells
done
clear
B)
helper-T cells
done
clear
C)
suppressor-T cells
done
clear
D)
carrier-T cells
done
clear
View Answer play_arrow
question_answer 129) Apis dorsata refers to
A)
rock bee
done
clear
B)
little bee
done
clear
C)
Indian bee
done
clear
D)
European bee
done
clear
View Answer play_arrow
question_answer 130) Which are the longest cells in the body of man?
A)
Muscle cells of legs
done
clear
B)
Bone cells
done
clear
C)
Nerve cells
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 131) \[CO\] is more toxic than \[C{{O}_{2}}\] because
A)
it affects the nervous system
done
clear
B)
it damages lungs
done
clear
C)
it reduces the oxygen carrying capacity of haemoglobin
done
clear
D)
it forms acid with water
done
clear
View Answer play_arrow
question_answer 132) Leprosy is also called as
A)
Hansens disease
done
clear
B)
sarcoma
done
clear
C)
slim disease
done
clear
D)
leukaemia
done
clear
View Answer play_arrow
question_answer 133) Which of the following is not a character of Chordata?
A)
Dorsal tubular nerve cord
done
clear
B)
Pharyngeal gill slits
done
clear
C)
Presence of notochord
done
clear
D)
Presence of spinal cord
done
clear
View Answer play_arrow
question_answer 134) Vitamin essential for proper functioning of liver and clotting of blood is
A)
vitamin-K
done
clear
B)
vitamin-A
done
clear
C)
vitamin-E
done
clear
D)
vitamin- \[{{B}_{2}}\]
done
clear
View Answer play_arrow
question_answer 135) Cancer causing genes (oncogenes) were discovered by
A)
Watson and Crick
done
clear
B)
Beadle and Tatum
done
clear
C)
Bishop and Varamus
done
clear
D)
Temin and Baltimore
done
clear
View Answer play_arrow
question_answer 136) Beak is toothed in
A)
pelican
done
clear
B)
kiwi
done
clear
C)
ostrich
done
clear
D)
Archaeopteryx
done
clear
View Answer play_arrow
question_answer 137) The sliding filament theory of muscle contraction was proposed by
A)
AF Huxley and AP Pullman
done
clear
B)
AF Huxley and HE Huxley
done
clear
C)
B Pullman and AF Huxley
done
clear
D)
A Pullman and E Pullman
done
clear
View Answer play_arrow
question_answer 138) Which one is uricotelic?
A)
Frog and toads
done
clear
B)
Lizards and birds/cockroach
done
clear
C)
Cattle, monkey and man
done
clear
D)
Molluscs and teleost fishes
done
clear
View Answer play_arrow
question_answer 139) Interacting populations are
A)
symbiotic
done
clear
B)
mutualistic
done
clear
C)
parasitic
done
clear
D)
coevolved
done
clear
View Answer play_arrow
question_answer 140) Salamandra atra is
A)
ovoviviparous
done
clear
B)
oviparous
done
clear
C)
sexually sterile
done
clear
D)
parthenogenetic
done
clear
View Answer play_arrow
question_answer 141) Flying fish is
A)
Torpedo
done
clear
B)
Scoliodon
done
clear
C)
Anguilla
done
clear
D)
Exocoetus
done
clear
View Answer play_arrow
question_answer 142) Tay-Sachs disease is due to
A)
sex-linked recessive gene
done
clear
B)
sex-linked dominant gene
done
clear
C)
autosomal dominant gene
done
clear
D)
autosomal recessive gene
done
clear
View Answer play_arrow
question_answer 143) The earliest fossil form in phylogeny of horse is
A)
Merychippus
done
clear
B)
Mesohippus
done
clear
C)
Eohippus
done
clear
D)
Equus
done
clear
View Answer play_arrow
question_answer 144) What is true about vein?
A)
All veins carry deoxygenated blood
done
clear
B)
All veins carry oxygenated blood
done
clear
C)
They carry blood from organs towards heart
done
clear
D)
They carry blood from heart towards organs
done
clear
View Answer play_arrow
question_answer 145) Autosomes in humans are
A)
11 pairs
done
clear
B)
22 pairs
done
clear
C)
23 pairs
done
clear
D)
43 pairs
done
clear
View Answer play_arrow
question_answer 146) Haeckels biogenetic law or recapitulation theory states that
A)
life history of an animal reflects evolutionary history of the same
done
clear
B)
progeny resembles parents
done
clear
C)
mutations are acquired characters
done
clear
D)
all organisms begin their life from zygote
done
clear
View Answer play_arrow
question_answer 147) A colourblind mother and normal father would have
A)
colour blind sons and normal/carrier daughters
done
clear
B)
colour blind sons and daughters
done
clear
C)
all colour blind
done
clear
D)
all normal
done
clear
View Answer play_arrow
question_answer 148) Cranial capacity of modem man is
A)
450-650 cc
done
clear
B)
660-1000 cc
done
clear
C)
900-1100 cc
done
clear
D)
1200-1600 cc
done
clear
View Answer play_arrow
question_answer 149) Inheritance of ABO blood groups illustrates
A)
polyploidy
done
clear
B)
multiple allelism
done
clear
C)
euploidy
done
clear
D)
dominance
done
clear
View Answer play_arrow
question_answer 150) The most primitive cell like chemical aggregates capable of growth and division were
A)
chemoautotrophs
done
clear
B)
eobionts
done
clear
C)
prokaryotes
done
clear
D)
microspheres
done
clear
View Answer play_arrow
question_answer 151) Plasmid is
A)
small extra chromosomal circular self replicating DNA that can carry genes into host organism
done
clear
B)
bacteriophage
done
clear
C)
DNA found in mitochondria
done
clear
D)
DNA incorporated in bacterial chromosome
done
clear
View Answer play_arrow
question_answer 152) A group of plants or animals with similar traits of any rank is
A)
species
done
clear
B)
genus
done
clear
C)
order
done
clear
D)
taxon
done
clear
View Answer play_arrow
question_answer 153) Fermentation products of yeast are
A)
\[{{H}_{2}}O+C{{O}_{2}}\]
done
clear
B)
Methyl alcohol \[+C{{O}_{2}}\]
done
clear
C)
Methyl alcohol + Water
done
clear
D)
Ethyl alcohol \[+C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 154) Grafting is not possible in monocots because they
A)
lack cambium
done
clear
B)
have scattered vascular bundles
done
clear
C)
have parallel venation
done
clear
D)
are herbaceous
done
clear
View Answer play_arrow
question_answer 155) What is true about a gamospermous apospory?
A)
Formation of embryo from egg of embryo sac proliferated from a nucellar cell
done
clear
B)
Formation of embryo from egg of embryo sac formed directly from megaspore mother cell
done
clear
C)
Formation of embryo directly from nucellus
done
clear
D)
Formation of embryo directly from integument
done
clear
View Answer play_arrow
question_answer 156) RQ for fatty substance/fat is
A)
unit
done
clear
B)
less than one
done
clear
C)
more than one
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 157) Seed which are able to withstand reduction in moisture and temperature are called
A)
dormant seeds
done
clear
B)
recalcitrant seeds
done
clear
C)
orthodox seeds
done
clear
D)
non-viable seeds
done
clear
View Answer play_arrow
question_answer 158) Enzyme required for transcription is
A)
RNA se
done
clear
B)
End nuclease
done
clear
C)
RNA polymerase
done
clear
D)
DNA polymerase
done
clear
View Answer play_arrow
question_answer 159) The synthetic hormone used as a weedicide is
A)
indole-3-acetic acid
done
clear
B)
gibberellic acid
done
clear
C)
2, 4-dichlorophenoxyacetic acid
done
clear
D)
maleic hydrazide
done
clear
View Answer play_arrow
question_answer 160) Which is present in monocot flowers?
A)
Sepals
done
clear
B)
Petals
done
clear
C)
Tepals
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 161) Anthesis means
A)
growth of pollen tube inside the carpel
done
clear
B)
dehiscence of anthers
done
clear
C)
opening of floral bud
done
clear
D)
emergence of anthers
done
clear
View Answer play_arrow
question_answer 162) Which one of the following contain agranal chloroplasts?
A)
\[{{C}_{3}}\]-plants
done
clear
B)
Succulants
done
clear
C)
\[{{C}_{4}}\]-plants
done
clear
D)
Hydrophytes
done
clear
View Answer play_arrow
question_answer 163) The process of multiplication of DNA from DNA is known as
A)
replication
done
clear
B)
transversion
done
clear
C)
transcription
done
clear
D)
translation
done
clear
View Answer play_arrow
question_answer 164) Phycobilins absorb light of wavelength
A)
\[670-700nm\]
done
clear
B)
\[610-650mn\]
done
clear
C)
\[500-650nm\]
done
clear
D)
\[520-520nm\]
done
clear
View Answer play_arrow
question_answer 165) For synthesis of one hexose sugar, how many times Calvin cycle is turned?
A)
12
done
clear
B)
2
done
clear
C)
6
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 166) The number of chlorophyll molecules in a quanta some is
A)
\[50-100\]
done
clear
B)
\[200-250\]
done
clear
C)
\[300-400\]
done
clear
D)
\[500-600\]
done
clear
View Answer play_arrow
question_answer 167) A plant having seeds but lacking flowers and fruits belong to
A)
pteridophytes
done
clear
B)
mosses
done
clear
C)
ferns
done
clear
D)
gymnosperms
done
clear
View Answer play_arrow
question_answer 168) Transpiration differ from evaporation in
A)
rate of water loss
done
clear
B)
transpiration is a physiological process whereas, evaporation is physical process
done
clear
C)
transpiration is a physical process, while evaporation is physiological process
done
clear
D)
frequency of water loss
done
clear
View Answer play_arrow
question_answer 169) Pigments common to all algae are
A)
chlorophyll-a and phycobilins
done
clear
B)
chlorophyll-a and carotenoids
done
clear
C)
chlorophyll-a and chlorophyll-b
done
clear
D)
chlorophyll-a) and carotenoids
done
clear
View Answer play_arrow
question_answer 170) An antitranspirant is
A)
cobalt chloride
done
clear
B)
mercury
done
clear
C)
potassium
done
clear
D)
phenyl mercuric acetate
done
clear
View Answer play_arrow
question_answer 171) Crossing over in diploid organisms is responsible for
A)
dominance of genes
done
clear
B)
linkage between genes
done
clear
C)
recombination of linked genes
done
clear
D)
segregation of alleles
done
clear
View Answer play_arrow
question_answer 172) Active K+ exchange mechanism for opening and closing of stomata was given by
A)
Darwin
done
clear
B)
Levit
done
clear
C)
Scarth
done
clear
D)
Khorana
done
clear
View Answer play_arrow
question_answer 173) First geneticist, father of genetics was
A)
deVries
done
clear
B)
Mendel
done
clear
C)
Darwin
done
clear
D)
Morgan
done
clear
View Answer play_arrow
question_answer 174) Quiescent centre is found in root apex and acts as
A)
permanent source of active initiates
done
clear
B)
reservoir of resistant cells
done
clear
C)
reservoir of passive cells to form new root apex if root apex is damaged/killed
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 175) A fruit developed from a condensed inflorescence is
A)
simple fruit
done
clear
B)
aggregate fruit
done
clear
C)
composite fruit
done
clear
D)
exterior
done
clear
View Answer play_arrow
question_answer 176) Acid rain are produced by
A)
excess \[N{{O}_{2}}\] and \[S{{O}_{2}}\] from burning fossil fuels
done
clear
B)
excess production of \[N{{H}_{3}}\] by industry and coal gas
done
clear
C)
excess release of carbon monoxide by incomplete combustion
done
clear
D)
excess formation of \[C{{O}_{2}}\]by combustion and animal respiration
done
clear
View Answer play_arrow
question_answer 177) The principal cereal crop of India/Asia is
A)
Sorghum
done
clear
B)
barley
done
clear
C)
wheat
done
clear
D)
rice
done
clear
View Answer play_arrow
question_answer 178) The stage of cell cycle when cell has undergone differentiation is
A)
\[{{G}_{0}}\]
done
clear
B)
\[{{G}_{1}}\]
done
clear
C)
\[{{G}_{3}}\]
done
clear
D)
\[{{G}_{4}}\]
done
clear
View Answer play_arrow
question_answer 179) Principle protein of cilia and flagella is
A)
globulin
done
clear
B)
fibrin
done
clear
C)
flagellin
done
clear
D)
tubulin
done
clear
View Answer play_arrow
question_answer 180) Photosynthetic bacteria have
A)
pigment system I (only one pigment system)
done
clear
B)
pigment system II
done
clear
C)
Both (a) and (b)
done
clear
D)
some other type
done
clear
View Answer play_arrow
question_answer 181) Major coffee producing state of India is
A)
Tamil Nadu
done
clear
B)
Kerala
done
clear
C)
Karnataka
done
clear
D)
Andhra Pradesh
done
clear
View Answer play_arrow
question_answer 182) DNA/chromosome replication takes place during
A)
\[{{G}_{1}}\]-phase
done
clear
B)
\[{{G}_{2}}\]-phase
done
clear
C)
S-phase
done
clear
D)
Prophase
done
clear
View Answer play_arrow
question_answer 183) Active transport occurs
A)
against concentration gradient and requires ATP
done
clear
B)
against concentration gradient and does not require ATP
done
clear
C)
along concentration gradient but require ATP
done
clear
D)
along concentration gradient but does not require ATP
done
clear
View Answer play_arrow
question_answer 184) Two or more species occupying same or ovar lapping areas are
A)
sympatric
done
clear
B)
sibling
done
clear
C)
sub-species
done
clear
D)
allopatric
done
clear
View Answer play_arrow
question_answer 185) Terminal cytochrome of respiratory chain which donates electrons to oxygen is
A)
cytochrome -b
done
clear
B)
cytochrome-c
done
clear
C)
cytochrome - \[{{a}_{1}}\]
done
clear
D)
cytochrome-\[{{a}_{3}}\]
done
clear
View Answer play_arrow
question_answer 186) Dendrochronology is the study of
A)
height of a tree
done
clear
B)
diameter of a tree
done
clear
C)
age of the tree by counting the number of annual rings in the main stem
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 187) A student wants to study metaphasic behaviour of chromosomes in a living cell. The technique most suitable is
A)
phase contrast microscope
done
clear
B)
X-ray microscope
done
clear
C)
cell fractionation
done
clear
D)
scanning electron microscope
done
clear
View Answer play_arrow
question_answer 188) Protonema is found in the life cycle of
A)
Spirogyra
done
clear
B)
Rhizopus
done
clear
C)
Escherichia
done
clear
D)
Funaria
done
clear
View Answer play_arrow
question_answer 189) Energy currency (reservoir) of the cells is
A)
AMP
done
clear
B)
ATP
done
clear
C)
RNA
done
clear
D)
DNA
done
clear
View Answer play_arrow
question_answer 190) An open collateral bundle is one in which
A)
xylem and phloem are separated by cambium
done
clear
B)
xylem and phloem lie side by side
done
clear
C)
cambium occurs on the out side of bundle
done
clear
D)
cambium does not occur in the bundle
done
clear
View Answer play_arrow
question_answer 191) Agar-agar commonly used in bacterial cultures and medication is obtained from
A)
Sargassum
done
clear
B)
Gelidium
done
clear
C)
Ulothrix
done
clear
D)
Ulva
done
clear
View Answer play_arrow
question_answer 192) Dikaryon formation is characteristic of
A)
Ascomycetes and Basidiomycetes
done
clear
B)
Phycomycetes and Basidiomycetes
done
clear
C)
Ascomycetes and Phycomycetes
done
clear
D)
Phycomycetes and Zygomycetes
done
clear
View Answer play_arrow
question_answer 193) Most effective wavelength of light for photosynthesis is
A)
green
done
clear
B)
violet
done
clear
C)
red
done
clear
D)
yellow
done
clear
View Answer play_arrow
question_answer 194) Divisions characteristic of Korper-Kappe theory are
A)
anticlinal
done
clear
B)
T-type
done
clear
C)
periclinal
done
clear
D)
irregular
done
clear
View Answer play_arrow
question_answer 195) Prothallus of fern has
A)
antheridia and archegonia on lower surface
done
clear
B)
antheridia and archegonia on upper surface
done
clear
C)
antheridia on upper surface and archegonia on lower
done
clear
D)
antheridia on lower surface and archegonia on upper surface
done
clear
View Answer play_arrow
question_answer 196) Male cone of Pinus possesses
A)
mega sporophylls
done
clear
B)
microsporophylls
done
clear
C)
anthers
done
clear
D)
ligules
done
clear
View Answer play_arrow
question_answer 197) \[{{C}_{4}}\]-plants, synthesis of sugars/final \[C{{O}_{2}}\] fixation occurs in
A)
palisade cells
done
clear
B)
spongy cells
done
clear
C)
undifferentiated mesophyll cells
done
clear
D)
bundle sheath cells
done
clear
View Answer play_arrow
question_answer 198) Cork cambium and vascular cambium are
A)
parts of secondary xylem and phloem
done
clear
B)
parts of pericycle
done
clear
C)
lateral meristems
done
clear
D)
apical meristems
done
clear
View Answer play_arrow
question_answer 199) First natural cytokinin was discovered by
A)
Skoog and Miller
done
clear
B)
Letham
done
clear
C)
Benson and Calvin
done
clear
D)
Thimann and Went
done
clear
View Answer play_arrow
question_answer 200) Grassland with scattered trees is
A)
savannah
done
clear
B)
deciduous forest
done
clear
C)
evergreen forest
done
clear
D)
tropical rain forest
done
clear
View Answer play_arrow