question_answer 1) A force \[\overset{\to }{\mathop{\text{F}}}\,=\left( 5\hat{i}+3\hat{j} \right)\,\,\text{N}\] is applied over a particle which displaces it from its original to the point\[\overset{\to }{\mathop{s}}\,=\left( 5\overset{\to }{\mathop{\text{i}}}\,+3\overset{\wedge }{\mathop{\text{j}}}\, \right)\,\,m\]. The work done on the particle is:
A)
\[+11\,\,j\]
done
clear
B)
\[+7\,\,j\]
done
clear
C)
\[+13\,\,j\]
done
clear
D)
\[-7\,\,j\]
done
clear
View Answer play_arrow
question_answer 2) The dimensional formula for Planck's constant and angular momentum is:
A)
\[\left[ M{{L}^{2}}{{T}^{-2}} \right]\,and\,\left[ ML{{T}^{-1}} \right]\]
done
clear
B)
\[\left[ M{{L}^{2}}{{T}^{-1}} \right]\,and\,\left[ M{{L}^{2}}{{T}^{-1}} \right]\]
done
clear
C)
\[\left[ M{{L}^{2}}{{T}^{1}} \right]\,and\,\left[ M{{L}^{2}}{{T}^{-2}} \right]\]
done
clear
D)
\[\left[ ML{{T}^{-1}} \right]\,and\,\left[ ML{{T}^{-2}} \right]\]
done
clear
View Answer play_arrow
question_answer 3) A elevator car whose floor to ceiling distance is 2.7 m starts ascending with a constant acceleration of \[1.2\,\,m/{{s}^{2}}\], 2 s after the start a bolt is begin to fall from the ceiling of the car. The free fall time of the bolt is \[\left( g=9.8\,\,m/{{s}^{2}} \right):\]
A)
\[\sqrt{\frac{2.7}{9.8}s}\]
done
clear
B)
\[\sqrt{\frac{5.4}{9.8}s}\]
done
clear
C)
\[\sqrt{\frac{5.4}{8.6}s}\]
done
clear
D)
\[\sqrt{\frac{5.4}{11}s}\]
done
clear
View Answer play_arrow
question_answer 4) Two items, each of 50 m length are moving parallel on parallel lines towards each other with speeds of\[10\,\,m/s\,\,and\,\,15\,\,m/{{s}^{2}}\]. The pass each other in.
A)
\[6\,\,s\]
done
clear
B)
\[2\,\,s\]
done
clear
C)
\[4\,\,s\]
done
clear
D)
\[5\sqrt{\frac{2}{3}}\,\,s\]
done
clear
View Answer play_arrow
question_answer 5) A body of mass m moving with velocity u collides with a stationary body of mass 2m. The speed of the system after collision, is:
A)
\[2u\]
done
clear
B)
\[\frac{u}{3}\]
done
clear
C)
\[2u\]
done
clear
D)
\[\frac{u}{4}\]
done
clear
View Answer play_arrow
question_answer 6) A wet open umbrella is held vertical and it whirled about the handle at a uniform rate of 21 revolutions in 44 s. If the rim of the umbrella is a circle of 1 m in diameter and the height of the rim above the flour is 4.9 m, the locus of the drop is a circle of radius:
A)
\[\sqrt{2.5}\,m\]
done
clear
B)
\[1\,m\]
done
clear
C)
\[3\,\,m\]
done
clear
D)
\[1.5\,\,m\]
done
clear
View Answer play_arrow
question_answer 7) The moment of inertia of a body about a given axis is\[1.2\,\,kg\,-{{m}^{2}}\]. To produce a rotational kinetic energy of \[1500\,\,J\] an angular acceleration of \[25\,\,rad/{{s}^{2}}\] must be applied for:
A)
\[8.5\,\,s\]
done
clear
B)
\[5\,\,s\]
done
clear
C)
\[2\,\,s\]
done
clear
D)
\[1\,\,s\]
done
clear
View Answer play_arrow
question_answer 8) A running man has half the kinetic energy of that of a boy of half of his mass. The man speeds up by\[1\,\,m/s\], so as the have same kinetic energy as that of the boy. The original speed of the man is:
A)
\[\left( \sqrt{2}-1 \right)m/s\]
done
clear
B)
\[\sqrt{2}\,m/s\]
done
clear
C)
\[\frac{1}{\sqrt{2}-1}\,m/s\]
done
clear
D)
\[\frac{1}{\sqrt{2}}\,m/s\]
done
clear
View Answer play_arrow
question_answer 9) An earth satellites \[S\] has an orbit radius which is 4 times that of communication satellite C. The period of revolution of \[S\]will be:
A)
\[32\,\,day\]
done
clear
B)
\[18\,\,day\]
done
clear
C)
\[8\,\,day\]
done
clear
D)
\[9\,\,day\]
done
clear
View Answer play_arrow
question_answer 10) \[n\] small balls, each of mass \[m\]impinge elastically each second on a surface with a velocity \[u\], then the force experienced by the surface in one second, will be:
A)
\[4\,\,mnu\]
done
clear
B)
\[2\,\,mnu\]
done
clear
C)
\[1.5\,\,mnu\]
done
clear
D)
\[0.8\,\,mnu\]
done
clear
View Answer play_arrow
question_answer 11) If the potential energy of a body on a planet is numerically \[U\] and the escape velocity for the same body be \[{{v}_{e}}\] for the same planet then \[U/{{v}_{e}}\] will be:
A)
\[\frac{U}{{{v}_{e}}}=m\sqrt{\frac{GM}{2R}}\]
done
clear
B)
\[\frac{U}{{{v}_{e}}}=m\sqrt{\frac{GM}{2R}}\]
done
clear
C)
\[\frac{U}{{{v}_{e}}}=m\sqrt{\frac{2GM}{R}}\]
done
clear
D)
\[\frac{U}{{{v}_{e}}}=m\frac{GM}{R}\]
done
clear
View Answer play_arrow
question_answer 12) A car of mass \[m\] moves in a horizontal circular path of radius \[r\] meter. At an instant its speed os \[v\,\,m/s\] and is increasing at a rate a \[m/{{s}^{2}}\], then the acceleration of the car is:
A)
\[\sqrt{a\left( \frac{{{v}^{2}}}{r} \right)}\]
done
clear
B)
\[\sqrt{{{a}^{2}}+{{\left( \frac{{{v}^{2}}}{r} \right)}^{2}}}\]
done
clear
C)
\[\frac{{{v}^{2}}}{r}\]
done
clear
D)
\[a\]
done
clear
View Answer play_arrow
question_answer 13) An object of mass 40 kg and having a velocity 4 m/s collides with another object \[\left( m=60\,kg \right)\] having velocity 2 m/s. The collision is perfectly inelastic. The loss in energy is:
A)
\[110\,\,J\]
done
clear
B)
\[48\,\,J\]
done
clear
C)
\[392\,\,J\]
done
clear
D)
\[440\,\,J\]
done
clear
View Answer play_arrow
question_answer 14) Dimensions of surface tension are:
A)
\[\left[ {{M}^{2}}{{L}^{2}}{{T}^{-2}} \right]\]
done
clear
B)
\[\left[ {{M}^{2}}L{{T}^{-2}} \right]\]
done
clear
C)
\[\left[ M{{T}^{-2}} \right]\]
done
clear
D)
\[\left[ ML{{T}^{-2}} \right]\]
done
clear
View Answer play_arrow
question_answer 15) An iron rod of length 2 m and area of cross-section \[50\,\,m{{m}^{2}}\] stretches by\[0.5\,\,mm\], when a mass of 250 kg is hung from its lower end. The Young?s of iron rod is:
A)
\[19.6\times {{10}^{20}}\,\,N/{{m}^{2}}\]
done
clear
B)
\[19.6\times {{10}^{18}}\,\,N/{{m}^{2}}\]
done
clear
C)
\[19.6\times {{10}^{15}}\,\,N/{{m}^{2}}\]
done
clear
D)
\[19.6\times {{10}^{10}}\,\,N/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 16) A car of mass m is moving on a rough road with momentum p. If the coefficient of friction between the tyres and road is u, then the stopping distance is :
A)
\[\frac{{{p}^{2}}}{2\mu {{m}^{2}}g}\]
done
clear
B)
\[\frac{p}{2\mu {{m}^{2}}g}\]
done
clear
C)
\[\frac{{{p}^{2}}}{2\mu mg}\]
done
clear
D)
\[\frac{p}{2\mu mg}\]
done
clear
View Answer play_arrow
question_answer 17) The time period of revolution of planet X around the sun is 8 times that of Y. The distance of X from the sun is how many times greater than that of the sun?
A)
\[5\frac{1}{2}times\]
done
clear
B)
\[4\,\,times\]
done
clear
C)
\[3\,\,times\]
done
clear
D)
\[None\,\,of\,\,these\]
done
clear
View Answer play_arrow
question_answer 18) \[16\,\,c{{m}^{3}}\]Water flows per second through a capillary tube of radius a cm and of length \[l\]and when connected a pressure head of \[H\]cm of water. If a tube of same length and radius \[a/2\] cm is connected to the same pressure head the quantity of water flowing through the tube per second is:
A)
\[18\,\,c{{m}^{3}}\]
done
clear
B)
\[1\,\,c{{m}^{3}}\]
done
clear
C)
\[16\,\,c{{m}^{3}}\]
done
clear
D)
\[4\,\,c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 19) Pressure inside two soap bubbles are 1.01 and 1.03 atm. Ratio between their volumes is :
A)
\[27:1\]
done
clear
B)
\[3:1\]
done
clear
C)
\[127:101\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 20) A man measures time period of a pendulum (T) in stationary lift. If the lift moves upward with p acceleration \[\frac{g}{4}\], then new time period will be :
A)
\[\sqrt{\frac{2}{5}T}\]
done
clear
B)
\[\sqrt{\frac{5}{2}T}\]
done
clear
C)
\[\frac{\sqrt{5}\,T}{2}\]
done
clear
D)
\[\frac{2T}{\sqrt{5}}\]
done
clear
View Answer play_arrow
question_answer 21) A stationary object at \[4{}^\circ C\]and weighing 3.5 kg falls from a height of 2000 m on a snow mountain at\[0{}^\circ \,C\]. If the temperature of the object just before hitting the snow is \[0{}^\circ \,C\] and the object comes to rest immediately \[\left( g=10\,\,m/{{s}^{2}}\,\,and\,\,latent\,\,heat\,\,of\,\,ice=3.5\times {{10}^{5}}\,J/s \right),\] then the object will melt:
A)
\[2\,\,kg\,\,of\,ice\]
done
clear
B)
\[200\,\,g\,\,of\,ice\]
done
clear
C)
\[20\,\,g\,\,\,ice\]
done
clear
D)
\[2\,\,g\,\,\,ice\]
done
clear
View Answer play_arrow
question_answer 22) A Carnot enging works between temperatures \[727{}^\circ C\] and \[27{}^\circ C\]. The efficiency of the engine is:
A)
\[70%\]
done
clear
B)
\[30\,%\]
done
clear
C)
\[0\,%\]
done
clear
D)
\[10\,%\]
done
clear
View Answer play_arrow
question_answer 23) A sphere at temperature 600 K is placed in Environment of temperature 200 K. Its cooling Rate is\[H\]. If the temperature is reduced to 400 K, then the cooling in same environment will be:
A)
\[\frac{H}{16}\]
done
clear
B)
\[\left( \frac{9}{27} \right)H\]
done
clear
C)
\[\left( \frac{16}{3} \right)H\]
done
clear
D)
\[\left( \frac{3}{16} \right)H\]
done
clear
View Answer play_arrow
question_answer 24) An ideal gas at \[27{}^\circ C\] is compressed Adiabatically to \[\frac{8}{27}\] of its original volume if \[\gamma =\frac{5}{3}\], Then rise in temperature is:
A)
\[405\,\,K\]
done
clear
B)
\[225\,\,K\]
done
clear
C)
\[375\,\,K\]
done
clear
D)
\[450\,\,K\]
done
clear
View Answer play_arrow
question_answer 25) The root mean square velocity of the molecules in a sample of helium\[\frac{5}{7}\] th that of the molecules in a Sample of hydrogen at\[{{0}^{\circ }}C\]. Then, the Temperature of helium sample is about:
A)
\[{{100}^{\circ }}C\]
done
clear
B)
\[{{273}^{\circ }}C\]
done
clear
C)
\[0\,K\]
done
clear
D)
\[{{0}^{\circ }}C\]
done
clear
View Answer play_arrow
question_answer 26) The displacement of the particle from its mean Position (in meter) varies with time (in second) According to the equation: \[y=0.2\,\,\sin \,\,\left( 10\pi t+1.5\pi \right)\cos \left( 10\pi t+1.5\pi \right)\]. The motion of the particle is:
A)
Along a circular path
done
clear
B)
Simple harmonic motion with time period 0.1 s
done
clear
C)
No simple harmonic motion
done
clear
D)
Simple harmonic motion with time period 0.2 s
done
clear
View Answer play_arrow
question_answer 27) Light of frequency \[8\times {{10}^{15}}\,\,Hz\] is incident on a substance of photoelectric work function\[6.125\,eV\]. The maximum kinetic energy of the emitted photoelectrons will be:
A)
\[39\,\,eV\]
done
clear
B)
\[27\,\,eV\]
done
clear
C)
\[54\,\,eV\]
done
clear
D)
\[13.5\,\,eV\]
done
clear
View Answer play_arrow
question_answer 28) A cylindrical tube, open at both ends has a fundamental frequency \[f\] in air. The tube dipped vertically in water, so that half of it is in water, the fundamental frequency of the air colon is now:
A)
\[2\,\,f\]
done
clear
B)
\[f\]
done
clear
C)
\[\frac{3f}{4}\]
done
clear
D)
\[\frac{f}{2}\]
done
clear
View Answer play_arrow
question_answer 29) The equation of plane wave is given by as: \[y=2\,\sin \,\pi \left( 200\,t\frac{x}{150} \right)\] When displacement \[y\] is given in cms and time \[t\] in second, then the velocity of the wave is:
A)
\[30000\,\,cm/s\]
done
clear
B)
\[200\,\,cm/s\]
done
clear
C)
\[150\,\,cm/s\]
done
clear
D)
\[2\,\,cm/s\]
done
clear
View Answer play_arrow
question_answer 30) The distance between two point?s difference in phase by \[{{60}^{\circ }}\] on a wave having a wave velocity \[360\,\,m\text{/}s\] and frequency \[500\,\,Hz\] is:
A)
0.36 m
done
clear
B)
0.12 m
done
clear
C)
0.18 m
done
clear
D)
0.72 m
done
clear
View Answer play_arrow
question_answer 31) The heat generated in a circuit is dependent upon the resistance, current and time for which the current is flown. If the error in measuring the above are\[1%,\,2%\] and\[1%\]respectively. The maximum error in measuring the heat is:
A)
\[8\,%\]
done
clear
B)
\[6\,%\]
done
clear
C)
\[18\,%\]
done
clear
D)
\[12\,%\]
done
clear
View Answer play_arrow
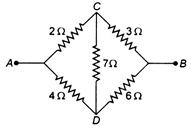
question_answer 32)
Five resistors are connected as shown in the given figure. The effective resistance between \[A\] and \[B\]is:
A)
\[7\,\Omega \]
done
clear
B)
\[\frac{54}{15}\Omega \]
done
clear
C)
\[\frac{10}{3}\Omega \]
done
clear
D)
\[17\,\Omega \]
done
clear
View Answer play_arrow
question_answer 33) A particle having charge 100 times that of an electron is revolving in a circular path of radius 0.8 m with one rotation per second. The magnetic field produced at the center will be:
A)
\[{{10}^{-17}}\,\,{{\mu }_{0}}\]
done
clear
B)
\[{{10}^{-11}}\,\,{{\mu }_{0}}\]
done
clear
C)
\[{{10}^{-7}}\,\,{{\mu }_{0}}\]
done
clear
D)
\[{{10}^{-3}}\,\,{{\mu }_{0}}\]
done
clear
View Answer play_arrow
question_answer 34) The energy of an X-ray photon is 2 keV, then its Frequency will be:
A)
\[3.2\times {{10}^{-6}}\] Per sec
done
clear
B)
\[5\times {{10}^{17}}\,Per\,\sec \]
done
clear
C)
\[2\times {{10}^{17}}\,Per\,\sec \]
done
clear
D)
\[2\times {{10}^{18}}\,Per\,\sec \]
done
clear
View Answer play_arrow
question_answer 35) A refracting angle of a prism is A and the \[A\] Refractive index of the prism is cot\[\left( \frac{A}{2} \right)\]. Then, Angle of minimum deviation is:
A)
\[{{180}^{\circ }}-2A\]
done
clear
B)
\[{{90}^{\circ }}-A\]
done
clear
C)
\[{{180}^{\circ }}+2A\]
done
clear
D)
\[{{180}^{\circ }}-3A\]
done
clear
View Answer play_arrow
question_answer 36) If the critical angle for total internal reflection from medium to vacuum is 30°. The velocity in the medium will be:
A)
\[\sqrt{3}\times {{10}^{8}}\,\,m/s\]
done
clear
B)
\[6\times {{10}^{8}}\,\,m/s\]
done
clear
C)
\[1.5\times {{10}^{8}}\,\,m/s\]
done
clear
D)
\[3\times {{10}^{8}}\,\,m/s\]
done
clear
View Answer play_arrow
question_answer 37) Given, \[_{a}{{\mu }_{8}}=\frac{3}{2},{{\,}_{a}}{{\mu }_{w}}=\frac{4}{3}\], if a convex lens of Focal length 10 cm is placed in water, then it's Focal length in water is:
A)
Equal to 40 cm
done
clear
B)
Equal to 20 cm
done
clear
C)
Equal to 10 cm
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 38) The length of an astronomical telescope adjusted for parallel light is 100 cm. If the magnification of the telescope is 19. The focal length of two lenses, i.e., objective and eye piece are:
A)
76 cm and 4 cm
done
clear
B)
95 cm and 5 cm
done
clear
C)
85 cm and 15 cm
done
clear
D)
82 cm and 18 cm
done
clear
View Answer play_arrow
question_answer 39) Doubly ionised helium atoms and hydrogen ions are accelerated from rest through the same potential drop. The ratio of the final velocities of helium and the hydrogen ions are:
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{1}{\sqrt{2}}\]
done
clear
C)
\[\sqrt{2}\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 40) In a transformer, the number of turns of primary coil and secondary coil are 5 and 4 respectively. If 220 V is applied on the primary coil, then the ratio of primary current to the secondary current will be:
A)
\[9:5\]
done
clear
B)
\[5:9\]
done
clear
C)
\[5:4\]
done
clear
D)
\[4:5\]
done
clear
View Answer play_arrow
question_answer 41) If the distance between parallel plates of a Capacitor is halved and dielectric constant is Doubled then the capacitance will:
A)
remain the same
done
clear
B)
increase 4 times
done
clear
C)
increase 2 times
done
clear
D)
decrease 2 times
done
clear
View Answer play_arrow
question_answer 42) In a nuclear fission. 0-1% mass is converted into energy. The energy released by the fission of 1 kg mass will be:
A)
\[9\times {{10}^{19}}\,\,J\]
done
clear
B)
\[9\times {{10}^{17}}\,\,J\]
done
clear
C)
\[9\times {{10}^{16}}\,\,J\]
done
clear
D)
\[9\times {{10}^{13}}\,\,J\]
done
clear
View Answer play_arrow
question_answer 43) The energy of hydrogen atom in the nth orbit is\[{{E}_{n}},\]Then the energy in the nth orbit of single Ionized helium atom is:
A)
\[\frac{{{E}_{n}}}{2}\]
done
clear
B)
\[2{{E}_{n}}\]
done
clear
C)
\[4{{E}_{n}}\]
done
clear
D)
\[\frac{{{E}_{n}}}{4}\]
done
clear
View Answer play_arrow
question_answer 44) Suppose. 1 mg of radioactive substance is taken Initially. After 2 h, it is found that 0.25 mg of the Substance is left behind. The mean life of the Substance is:
A)
\[\frac{1}{0.693}h\]
done
clear
B)
\[0.693\times 2h\]
done
clear
C)
\[0.693\times \frac{1}{4}h\]
done
clear
D)
\[0.693\times 8h\]
done
clear
View Answer play_arrow
question_answer 45) Force between two identical bar magnets whose Centres \[r\]meter apart is 4.8 N when their axis are in the same line. If the separation is increased to 2r meter, the force between them is reduced to:
A)
0.3 N
done
clear
B)
0.6 N
done
clear
C)
2.4 N
done
clear
D)
1.2 N
done
clear
View Answer play_arrow
question_answer 46) An ideal transformer has a primary power input of 10 kW. The secondary current when the Transformer is on load is 25 A. If the primary Secondary turns ratio is 8 : 1, then the potential Difference applied in the primary coil will be:
A)
\[\frac{{{\left( 10 \right)}^{4}}}{25{{\left( 8 \right)}^{2}}}V\]
done
clear
B)
\[\frac{{{10}^{4}}}{\left( 25 \right)\left( 8 \right)}V\]
done
clear
C)
\[\frac{{{\left( 10 \right)}^{4}}\,8}{25}V\]
done
clear
D)
\[\frac{{{\left( 10 \right)}^{4}}\,{{\left( 8 \right)}^{2}}}{25}V\]
done
clear
View Answer play_arrow
question_answer 47) An air capacitor of capacity \[C=10\mu F\]is connected to a constant voltage battery of 12V. Now, the space between the plates is filled with a Liquid of dielectric constant 5. The additional Charge that flows now from battery to the Capacitor is:
A)
\[24\mu C\]
done
clear
B)
\[480\,\mu C\]
done
clear
C)
\[600\,\mu C\]
done
clear
D)
\[120\,\mu C\]
done
clear
View Answer play_arrow
question_answer 48) Pure silicon at 300 K has equal electrons\[{{n}_{e}}\], and Holes \[{{n}_{h}}\]concentration of\[1.5\times {{10}^{16}}/{{m}^{3}}\].Doping by Indium increases number of holes\[{{n}_{h}}\] to\[4.5\times {{10}^{22}}/{{m}^{3}}\,\,then\,\,{{n}_{h}}\]?doped in silicon will be:
A)
\[3.0\times {{10}^{-19}}/{{m}^{3}}\]
done
clear
B)
\[5\times {{10}^{9}}/{{m}^{3}}\]
done
clear
C)
\[4.5\times {{10}^{22}}/{{m}^{3}}\]
done
clear
D)
\[1.5\times {{10}^{16}}/{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 49) In an atom bomb the reaction which occurs is:
A)
Thermo nuclear
done
clear
B)
Uncontrolled fission
done
clear
C)
Controlled fission
done
clear
D)
Fusion
done
clear
View Answer play_arrow
question_answer 50) If 1 g of hydrogen is converted into 0.993 g of Helium, in the thermo nuclear reaction, the Energy released in the reaction is:
A)
\[63\times {{10}^{20}}\,J\]
done
clear
B)
\[63\times {{10}^{14}}\,J\]
done
clear
C)
\[63\times {{10}^{10}}\,J\]
done
clear
D)
\[63\times {{10}^{7}}\,J\]
done
clear
View Answer play_arrow
question_answer 51) Nitration of benzene, produce nitrobenzene. During nitration the role of cone.\[HN{{O}_{3}}\]in nitrating mixture is as a:
A)
catalyst
done
clear
B)
reducing agent
done
clear
C)
acid
done
clear
D)
base
done
clear
View Answer play_arrow
question_answer 52) Steam distillation is used for the purification of:
A)
covalent compounds
done
clear
B)
electrovalent compounds
done
clear
C)
co-ordinate compounds
done
clear
D)
volatile compounds
done
clear
View Answer play_arrow
question_answer 53) The number of isomers shown by the compound with molecular formula\[{{C}_{7}}{{H}_{8}}O\]is:
A)
2
done
clear
B)
3
done
clear
C)
5
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 54) Chromium is represented by the electronic configuration:
A)
\[[Ne]3{{s}^{2}}\text{ }3{{p}^{6}}\text{ }3{{d}^{1}}\text{ }4{{s}^{2}}\]
done
clear
B)
\[[Ne]3{{s}^{2}}\text{ }3{{p}^{6}}\text{ }3{{d}^{2}}\text{ }4{{s}^{1}}\]
done
clear
C)
\[[Ne]3{{s}^{2}}\text{ }3{{p}^{6}}\text{ }3{{d}^{5}}\,4{{s}^{1}}\]
done
clear
D)
\[[Ne]3{{s}^{2}}\text{ }3{{p}^{6}}\text{ }4{{s}^{2}}\text{ }3{{d}^{4}}\]
done
clear
View Answer play_arrow
question_answer 55) \[2HCl(g){{H}_{2}}(g)+C{{l}_{2}}(g)\] For the above reaction, the equilibrium constant is\[1.2\times {{10}^{-4}}M,\]. If the equilibrium concentration of\[{{H}_{2}}\]and\[C{{l}_{2}}\]is\[1.2\times {{10}^{-3}}M\]and \[1.2\times {{10}^{-4}}M\] respectively, then the concentration of\[HCl\]is:
A)
\[12\times {{10}^{-4}}M\]
done
clear
B)
\[12\times {{10}^{-3}}M\]
done
clear
C)
\[12\times {{10}^{-2}}M\]
done
clear
D)
\[12\times {{10}^{-1}}M\]
done
clear
View Answer play_arrow
question_answer 56) An asymmetric carbon atom is that in which :
A)
three similar and one dissimilar groups are attached
done
clear
B)
two similar and two dissimilar groups are attached
done
clear
C)
all the different groups are attached
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 57) The optical inactivity due to internal compensation can be exhibited by:
A)
dextro-tartaric acid
done
clear
B)
laevo-tartaric acid
done
clear
C)
racemic-tartaric acid
done
clear
D)
meso-tartaric acid
done
clear
View Answer play_arrow
question_answer 58) The common donor atom in ligands:
A)
nitrogen
done
clear
B)
oxygen
done
clear
C)
arsenic
done
clear
D)
both and
done
clear
View Answer play_arrow
question_answer 59) Isotopes are the elements having same:
A)
mass number
done
clear
B)
atomic number
done
clear
C)
number of atoms
done
clear
D)
number of neutrons
done
clear
View Answer play_arrow
question_answer 60) By passing 0.50 A current to an aqueous solution half gram of an element is liberated. The time of passing the current in seconds is: (eq. wt. = 96.5)
A)
100s
done
clear
B)
500s
done
clear
C)
1000s
done
clear
D)
2000s
done
clear
View Answer play_arrow
question_answer 61) The values of\[\Delta H\]for the combustion of ethane and acetylene are\[-341.1\]and\[-310.0\] kcal, respectively. The better fuel is:
A)
ethane
done
clear
B)
acetylene
done
clear
C)
both
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 62) Bauxite is an oxide mineral of:
A)
Al
done
clear
B)
B
done
clear
C)
Ba
done
clear
D)
Bi
done
clear
View Answer play_arrow
question_answer 63) Marsh gas is:
A)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
B)
\[{{H}_{2}}S\]
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 64) Red lead is:
A)
\[PbO\]
done
clear
B)
\[Pb{{O}_{2}}\]
done
clear
C)
\[P{{b}_{3}}{{O}_{4}}\]
done
clear
D)
\[P{{b}_{4}}{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 65) Dipole moment is shown by:
A)
2-methyl propane
done
clear
B)
trans-butene-2
done
clear
C)
cis-butene-2
done
clear
D)
carbon tetrachloride
done
clear
View Answer play_arrow
question_answer 66) Concentrated hydrochloric acid contains 37% \[HCl\](by mass). The density of this solution is 1.18 g/mL. The molarity of the solution is:
A)
10 M
done
clear
B)
12 M
done
clear
C)
13 M
done
clear
D)
14 M
done
clear
View Answer play_arrow
question_answer 67) The osmotic pressure at\[{{17}^{o}}C\]of an aqueous solution containing 1.75g of sucrose per 150 mL solution is:
A)
0.08 atm
done
clear
B)
8.1 atm
done
clear
C)
0.8 atm
done
clear
D)
9.1 aim
done
clear
View Answer play_arrow
question_answer 68) Phenol reacts with excess of bromine water to produce:
A)
o-bromophenol
done
clear
B)
p-bromophenol
done
clear
C)
2, 4, 6-tribromophenol
done
clear
D)
mixture of o-and p-bromophenol
done
clear
View Answer play_arrow
question_answer 69) The IUPAC name of \[C{{H}_{3}}\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,==C{{H}_{2}}C{{H}_{2}}OH\]is:
A)
2-methyl-3-butenol
done
clear
B)
3-methyl-2-butenol
done
clear
C)
3-methyl-3-butenol
done
clear
D)
2-methyl-2-butenol
done
clear
View Answer play_arrow
question_answer 70) Natural rubber is a polymer of:
A)
styrene
done
clear
B)
isoprene
done
clear
C)
butadiene
done
clear
D)
ethylene
done
clear
View Answer play_arrow
question_answer 71) In Friedel-Craft's reaction, anhydride. \[AlC{{l}_{3}}\] acts to:
A)
produce electrophile
done
clear
B)
produce nucleophile
done
clear
C)
absorb water
done
clear
D)
absorb\[HCl\]
done
clear
View Answer play_arrow
question_answer 72) Mullikan's oil drop method is used to determine:
A)
velocity of electron
done
clear
B)
mass of electron
done
clear
C)
charge of electron
done
clear
D)
e/m ratio of electron
done
clear
View Answer play_arrow
question_answer 73) Wavelength of spectral line emitted is inversely proportional to:
A)
energy
done
clear
B)
velocity
done
clear
C)
radius
done
clear
D)
quantum number
done
clear
View Answer play_arrow
question_answer 74) To explain the structure of atom, the first use of quantum theory was made by:
A)
Heisenberg
done
clear
B)
Bohr
done
clear
C)
Planck
done
clear
D)
Einstein
done
clear
View Answer play_arrow
question_answer 75) If 0.44 g of substance dissolved in 22.2 g of benzene lowers the freezing point of benzene by\[0.567{}^\circ C,\]then the molecular mass of substance is : (the molal depression constant\[={{5.12}^{o}}C\,mo{{l}^{-1}}\])
A)
128.4
done
clear
B)
156.6
done
clear
C)
178.9
done
clear
D)
232.4
done
clear
View Answer play_arrow
question_answer 76) In 1,2-butadiene, all the carbon are hybridized as:
A)
\[sp\]
done
clear
B)
\[s{{p}^{2}}\]
done
clear
C)
\[s{{p}^{3}}\]
done
clear
D)
\[s{{p}^{2}}\]and\[s{{p}^{3}}\]
done
clear
View Answer play_arrow
question_answer 77) Distribution law cannot be applicable for a system in which\[{{I}_{2}}\]is distributed in between:
A)
water and alcohol
done
clear
B)
water and benzene
done
clear
C)
water and chloroform
done
clear
D)
water and acetone
done
clear
View Answer play_arrow
question_answer 78) \[N{{H}_{3}}\]can be dried by:
A)
cone.\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
anhyd.\[CaC{{l}_{2}}\]
done
clear
C)
\[{{P}_{2}}{{O}_{5}}\]
done
clear
D)
\[CaO\]
done
clear
View Answer play_arrow
question_answer 79) \[AlC{{l}_{3}}\]acts as a strong Lewis acid because \[AlC{{l}_{3}}\]is:
A)
easily hydrolysed
done
clear
B)
a covalent compound
done
clear
C)
an ionic compound
done
clear
D)
an electron deficient compound
done
clear
View Answer play_arrow
question_answer 80) \[O_{2}^{2+}\]has a bond order of:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 81) The main structure feature of proteins is:
A)
glycoside linkage
done
clear
B)
ester linkage
done
clear
C)
ether linkage
done
clear
D)
peptide linkage
done
clear
View Answer play_arrow
question_answer 82) According to Raoult's law, relative lowering of vapour pressure for a solution is equal to:
A)
mole fraction of a solute
done
clear
B)
mole fraction of a solvent
done
clear
C)
moles of a solute
done
clear
D)
moles of a solvent
done
clear
View Answer play_arrow
question_answer 83) Which of the following react with\[KMn{{O}_{4}}\]to produce pyruvic acid?
A)
Butyne
done
clear
B)
Propyne
done
clear
C)
2-pentyne
done
clear
D)
Ethyne
done
clear
View Answer play_arrow
question_answer 84) 1,3-butadiene and ethylene reacts to produce:
A)
cyclohexane
done
clear
B)
cyclohexmonoene
done
clear
C)
cyclohexdiene
done
clear
D)
cyclohextriene
done
clear
View Answer play_arrow
question_answer 85) Lithopone is a mixture of:
A)
\[ZnC{{O}_{3}},BaC{{O}_{3}}\]
done
clear
B)
\[ZnS,N{{a}_{2}}S{{O}_{4}}\]
done
clear
C)
\[ZnS{{O}_{4}},BaS{{O}_{4}}\]
done
clear
D)
\[ZnS,BaS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 86) Glucose comes under the class:
A)
ketohexose
done
clear
B)
aldohexose
done
clear
C)
disaccharide
done
clear
D)
polysaccharide
done
clear
View Answer play_arrow
question_answer 87) Penicillin is an:
A)
antibiotic
done
clear
B)
anesthetic
done
clear
C)
antiseptic
done
clear
D)
antipyretic
done
clear
View Answer play_arrow
question_answer 88) The compound used as antiknock in petrol fuel is:
A)
basic lead sulphate
done
clear
B)
tetraethyl lead
done
clear
C)
lead tetraacetate
done
clear
D)
lead carbonate
done
clear
View Answer play_arrow
question_answer 89) The test used for the distinction of\[HCOOH\]and\[C{{H}_{3}}COOH\]is treatment with:
A)
sodium
done
clear
B)
sodium hydroxide
done
clear
C)
Tollen's reagent
done
clear
D)
alcohol
done
clear
View Answer play_arrow
question_answer 90) Which one of the following is a metalloid?
A)
Phosphorus
done
clear
B)
Antimony
done
clear
C)
Nitrogen
done
clear
D)
Bismuth
done
clear
View Answer play_arrow
question_answer 91) Chloroform, on exposing to air and sunlight produce:
A)
chloropicrin
done
clear
B)
phosgene
done
clear
C)
carbon tetrachloride
done
clear
D)
benzene
done
clear
View Answer play_arrow
question_answer 92) The vapour density of a gas A is four times than B. If molecular mass of B is 1, then molecular mass of A is:
A)
1
done
clear
B)
2
done
clear
C)
1/4
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 93) When a gas is allowed to expand from a region of high pressure to a region of low pressure, there occurs a change in temperature. This phenomenon is:
A)
Joule-Thomson effect
done
clear
B)
First law of thermodynamics
done
clear
C)
Gibbs-Helmholtz equation
done
clear
D)
Second law of thermodynamics
done
clear
View Answer play_arrow
question_answer 94) The amphoteric oxide is:
A)
\[T{{i}_{2}}{{O}_{2}}\]
done
clear
B)
\[MgO\]
done
clear
C)
\[C{{l}_{2}}{{O}_{2}}\]
done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 95) Which one of the following is independent of temperature?
A)
Molarity
done
clear
B)
Molality
done
clear
C)
Normality
done
clear
D)
Formality
done
clear
View Answer play_arrow
question_answer 96) Grignard reagent reacts with formaldehyde to produce:
A)
an acid
done
clear
B)
primary alcohol
done
clear
C)
an anhydride
done
clear
D)
secondary alcohol
done
clear
View Answer play_arrow
question_answer 97) The percentage of carbon atom in acetic acid is:
A)
15%
done
clear
B)
20%
done
clear
C)
40%
done
clear
D)
45%
done
clear
View Answer play_arrow
question_answer 98) The acid which cannot be kept in glass bottles is:
A)
\[HCl\]
done
clear
B)
\[HBr\]
done
clear
C)
\[HF\]
done
clear
D)
\[HI\]
done
clear
View Answer play_arrow
question_answer 99) \[C{{H}_{3}}COOH\xrightarrow[Heat]{N{{H}_{3}}}A\xrightarrow[(-{{H}_{2}}O)]{{{P}_{2}}{{O}_{5}}}B\xrightarrow[(-{{H}_{2}}O)]{{{P}_{2}}{{O}_{5}}}C\] The compound 'C? is:
A)
acetamide
done
clear
B)
ammonium acetate
done
clear
C)
methyl cyanide
done
clear
D)
aniline
done
clear
View Answer play_arrow
question_answer 100) Thermite is a mixture of iron oxide and:
A)
Al-powder
done
clear
B)
Zn-powder
done
clear
C)
Mg-powder
done
clear
D)
Cu-powder
done
clear
View Answer play_arrow
question_answer 101) Maltose, lactose and sucrose are:
A)
disaccharides
done
clear
B)
trisaccharides
done
clear
C)
polysaccharides
done
clear
D)
monosaccharides
done
clear
View Answer play_arrow
question_answer 102) Select the specific pairs of DNA:
A)
adenine and cytosine
done
clear
B)
adenine and thymine
done
clear
C)
guanine and adenine
done
clear
D)
guanine and uracil
done
clear
View Answer play_arrow
question_answer 103) Ribosomes were discovered by :
A)
Palade
done
clear
B)
Porter
done
clear
C)
Brown
done
clear
D)
Watson
done
clear
View Answer play_arrow
question_answer 104) Which of the following does not possess contractile vacuole ?
A)
Euglena
done
clear
B)
Paramecium
done
clear
C)
Amoeba
done
clear
D)
Plasmodium
done
clear
View Answer play_arrow
question_answer 105) ?Venus flower basket? is the name of dried skeleton of:
A)
Euspongia
done
clear
B)
Spongilla
done
clear
C)
Euplectella
done
clear
D)
Leucosolema
done
clear
View Answer play_arrow
question_answer 106) AIDS was first noticed in :
A)
Russia
done
clear
B)
USA
done
clear
C)
France
done
clear
D)
Germany
done
clear
View Answer play_arrow
question_answer 107) Polio is caused by :
A)
bacteriophage
done
clear
B)
bacteria
done
clear
C)
virus with single stranded DNA
done
clear
D)
virus with single stranded RNA
done
clear
View Answer play_arrow
question_answer 108) Transduction in bacteria is mediated by
A)
plasmid
done
clear
B)
phage vector
done
clear
C)
F-factor
done
clear
D)
pili
done
clear
View Answer play_arrow
question_answer 109) Parthenogenesis is common in :
A)
house fly
done
clear
B)
honey bee
done
clear
C)
Drosophila
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 110) The common method of DNA replication is :
A)
disruptive
done
clear
B)
semiconservative
done
clear
C)
conservative
done
clear
D)
random
done
clear
View Answer play_arrow
question_answer 111) Lower jaw of mammal is made up of:
A)
maxilla
done
clear
B)
dentary
done
clear
C)
squamosal
done
clear
D)
collumella
done
clear
View Answer play_arrow
question_answer 112) Which of the following organ remain unchanged during metamorphosis of frog?s tadpole ?
A)
Heart
done
clear
B)
Lung
done
clear
C)
Nervous system
done
clear
D)
Intestine
done
clear
View Answer play_arrow
question_answer 113) Which of the following hormone is related with iodine ?
A)
Oxytocin
done
clear
B)
Thyroxin
done
clear
C)
Pituterin
done
clear
D)
Secretin
done
clear
View Answer play_arrow
question_answer 114) Germplasm theory was proposed by :
A)
Darwin
done
clear
B)
Lamarck
done
clear
C)
Weismann
done
clear
D)
de Vries
done
clear
View Answer play_arrow
question_answer 115) The Ornithine cycle removes two waste products from blood in liver. These are :
A)
\[C{{O}_{2}}\]and urea
done
clear
B)
\[C{{O}_{2}}\]and ammonia
done
clear
C)
ammonia and urea
done
clear
D)
ammonia and uric acid
done
clear
View Answer play_arrow
question_answer 116) The main function of centromere is :
A)
formation of RNA
done
clear
B)
movement of chromosomes
done
clear
C)
formation of DNA
done
clear
D)
duplication of DNA
done
clear
View Answer play_arrow
question_answer 117) Anabolism is :
A)
exergonic process
done
clear
B)
destructive process
done
clear
C)
endergonic process
done
clear
D)
bidirectional process
done
clear
View Answer play_arrow
question_answer 118) Abiogenesis means :
A)
spontaneous generation
done
clear
B)
origin of life from living organisms
done
clear
C)
origin of life from living organisms
done
clear
D)
origin of life from microbes
done
clear
View Answer play_arrow
question_answer 119) Bone marrow is absent in bones of :
A)
fish
done
clear
B)
birds
done
clear
C)
reptiles
done
clear
D)
frog
done
clear
View Answer play_arrow
question_answer 120) Secretions of ductless glands is called :
A)
pheromone
done
clear
B)
enzymes
done
clear
C)
mucous
done
clear
D)
hormone
done
clear
View Answer play_arrow
question_answer 121) In earthworm arrangement of vessels is :
A)
different in last 15 segments
done
clear
B)
different in 1st 13 segments
done
clear
C)
same throughout body
done
clear
D)
different in 1st 10 and last 10 segments
done
clear
View Answer play_arrow
question_answer 122) Which of the following phylum belongs to Deuterostomia ?
A)
Annelida
done
clear
B)
Parazoa
done
clear
C)
Mollusca
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 123) Cytochromes are present in :
A)
outer membrane of mitochondria
done
clear
B)
matrix of mitochondria
done
clear
C)
matrix of mitochondria
done
clear
D)
cytoplasm of cell
done
clear
View Answer play_arrow
question_answer 124) A coral island with a central shallow lake is called as :
A)
atoll
done
clear
B)
corallite
done
clear
C)
diatom land
done
clear
D)
coral feed
done
clear
View Answer play_arrow
question_answer 125) Protein synthesis takes place in :
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
cytoplasm
done
clear
D)
ribosomes
done
clear
View Answer play_arrow
question_answer 126) Which of the following nerve of human being is both sensory and motor?
A)
Optic
done
clear
B)
Abducens
done
clear
C)
Trigeminal
done
clear
D)
Olfactory
done
clear
View Answer play_arrow
question_answer 127) Lymph vessles are united to form :
A)
lymph heart
done
clear
B)
lymph junctive
done
clear
C)
cisterna chyle
done
clear
D)
lymph jugular veins
done
clear
View Answer play_arrow
question_answer 128) Ascorbic acid is also called as :
A)
thiamine
done
clear
B)
vitamin-C
done
clear
C)
pyridoxine
done
clear
D)
vitamin-\[{{\text{B}}_{\text{12}}}\]
done
clear
View Answer play_arrow
question_answer 129) Hamburger's phenomenon is also called as :
A)
sodium shift
done
clear
B)
hydrogen shift
done
clear
C)
chloride shift
done
clear
D)
bicarbonate shift
done
clear
View Answer play_arrow
question_answer 130) In mammals digestion of starch starts from :
A)
mouth
done
clear
B)
stomach
done
clear
C)
oesophagus
done
clear
D)
duodenum
done
clear
View Answer play_arrow
question_answer 131) Which of the following carries blood rich in food materials from intestine to liver ?
A)
Dorsal aorta
done
clear
B)
Renal portal vein
done
clear
C)
Hepatic portal vein
done
clear
D)
Thoracic artery
done
clear
View Answer play_arrow
question_answer 132) What is common among amylase, renin and trypsin ?
A)
All are proteins
done
clear
B)
All act at less than 7 pH
done
clear
C)
All are produced in stomach
done
clear
D)
All are hormones
done
clear
View Answer play_arrow
question_answer 133) Typhlop is a :
A)
grass snake
done
clear
B)
sea snake
done
clear
C)
blind snake
done
clear
D)
tree snake
done
clear
View Answer play_arrow
question_answer 134) Limbless amphibians belong to order :
A)
Anura
done
clear
B)
Urodela
done
clear
C)
Gymnophiana
done
clear
D)
Squamata
done
clear
View Answer play_arrow
question_answer 135) Typhlosole in earthworm starts from segment number:
A)
7
done
clear
B)
14
done
clear
C)
47
done
clear
D)
27
done
clear
View Answer play_arrow
question_answer 136) Chemoreceptor in Pila is :
A)
radula
done
clear
B)
osphradium
done
clear
C)
canula
done
clear
D)
spiracle
done
clear
View Answer play_arrow
question_answer 137) Leydig cells secrete :
A)
testosterone
done
clear
B)
oestrogen
done
clear
C)
corticosterone
done
clear
D)
progesterone
done
clear
View Answer play_arrow
question_answer 138) Who coined the term Ecology ?
A)
Darwin
done
clear
B)
Linnaeus
done
clear
C)
Odum
done
clear
D)
Haeckel
done
clear
View Answer play_arrow
question_answer 139) Trophic levels are linked to form :
A)
food web
done
clear
B)
food chain
done
clear
C)
autotrops
done
clear
D)
carnivores
done
clear
View Answer play_arrow
question_answer 140) Adjacent epithelial cells are held together by :
A)
liposomes
done
clear
B)
lysosomes
done
clear
C)
microsome
done
clear
D)
desmosomes
done
clear
View Answer play_arrow
question_answer 141) Heat-islands are produced due to :
A)
air pollution
done
clear
B)
water pollution
done
clear
C)
land pollution
done
clear
D)
noise pollution
done
clear
View Answer play_arrow
question_answer 142) Colchicine is used for :
A)
polyploidy
done
clear
B)
aneuploidy
done
clear
C)
monosomy
done
clear
D)
trisomy
done
clear
View Answer play_arrow
question_answer 143) Sting apparatus of honey bee is a modified form of:
A)
ovipositor
done
clear
B)
sex spine
done
clear
C)
podical gland
done
clear
D)
wax gland
done
clear
View Answer play_arrow
question_answer 144) Blood clotting is prevented by :
A)
vitamin-K
done
clear
B)
calcium ions
done
clear
C)
potassium ions
done
clear
D)
heparin
done
clear
View Answer play_arrow
question_answer 145) Phallic organs in cockroach are related to :
A)
male reproductive system
done
clear
B)
male excretory system
done
clear
C)
male digestive system
done
clear
D)
male circulatory system
done
clear
View Answer play_arrow
question_answer 146) Haemocyanin is present in :
A)
Chordates
done
clear
B)
Annelida
done
clear
C)
Mollusca
done
clear
D)
Porifera
done
clear
View Answer play_arrow
question_answer 147) Who coined the term biocoenosis ?
A)
Karl Mobius
done
clear
B)
Haeckel
done
clear
C)
Ambust
done
clear
D)
Lindmann
done
clear
View Answer play_arrow
question_answer 148) True nucleus is absent in :
A)
man
done
clear
B)
rabbit
done
clear
C)
camel
done
clear
D)
E. coli
done
clear
View Answer play_arrow
question_answer 149) Coelome is found between :
A)
ectoderm and endoderm
done
clear
B)
mesoderm and ectoderm
done
clear
C)
body wall and ectoderm
done
clear
D)
mesoderm and endoderm
done
clear
View Answer play_arrow
question_answer 150) DNA of bacteria is :
A)
with histones
done
clear
B)
without histones
done
clear
C)
with sugar base
done
clear
D)
without sugar base
done
clear
View Answer play_arrow
question_answer 151) Anemophilous flowers have :
A)
scented, large, showy flowers
done
clear
B)
large feathery stigma
done
clear
C)
stickey pollen grains
done
clear
D)
sessile stigma
done
clear
View Answer play_arrow
question_answer 152) A stress hormone is:
A)
IAA
done
clear
B)
NAA
done
clear
C)
ABA
done
clear
D)
IBA
done
clear
View Answer play_arrow
question_answer 153) When water enters in roots due to diffusion, it is termed as :
A)
osmosis
done
clear
B)
passive absorption
done
clear
C)
active absorption
done
clear
D)
imbibition
done
clear
View Answer play_arrow
question_answer 154) Social foresting species with multiple utility is :
A)
Rosa indica
done
clear
B)
Mangiferaindica
done
clear
C)
Acacia nelotica
done
clear
D)
Leucaenaleucocephala
done
clear
View Answer play_arrow
question_answer 155) Vascular cambium of the root is an example of:
A)
primary meristem
done
clear
B)
secondary meristem
done
clear
C)
apical meristem
done
clear
D)
lateral meristem
done
clear
View Answer play_arrow
question_answer 156) Photosystem-II works in :
A)
grana of chloroplast
done
clear
B)
stroma of chloroplast
done
clear
C)
periplastidial space
done
clear
D)
cytoplasm
done
clear
View Answer play_arrow
question_answer 157) Tapetum is :
A)
protective
done
clear
B)
reproductive
done
clear
C)
nutritive
done
clear
D)
respiratory
done
clear
View Answer play_arrow
question_answer 158) Indirect transfer of genetic material from one bacterium to another through a bacteriophage is called :
A)
transformation
done
clear
B)
conjugation
done
clear
C)
translation
done
clear
D)
transduction
done
clear
View Answer play_arrow
question_answer 159) The latest explanation for closure of stomata comes from :
A)
Munch theory
done
clear
B)
starch glucose theory
done
clear
C)
Steward?s theory
done
clear
D)
active K+ transport theory
done
clear
View Answer play_arrow
question_answer 160) Aggregate fruit is found in :
A)
Custard apple
done
clear
B)
Narvelia
done
clear
C)
Rubus
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 161) Sugarcane is propagated vegetatively because :
A)
it does not produce seeds
done
clear
B)
the seeds are not viable
done
clear
C)
it helps in maintaining genetic purity
done
clear
D)
it becomes more juicy
done
clear
View Answer play_arrow
question_answer 162) Agriculture was originated in mesolithic age about:
A)
1000-5000 years ago
done
clear
B)
5000-10000 years ago
done
clear
C)
7000-15000 years ago
done
clear
D)
15000-20000 years ago
done
clear
View Answer play_arrow
question_answer 163) Fertilizers providing N, P, K all essential elements to plants are called :
A)
complete fertilizers
done
clear
B)
incomplete fertilizers
done
clear
C)
manure
done
clear
D)
compost
done
clear
View Answer play_arrow
question_answer 164) Crescograph was invented by :
A)
F.W. Went
done
clear
B)
C.V. Raman
done
clear
C)
J.C. Bose
done
clear
D)
Charles Darwin
done
clear
View Answer play_arrow
question_answer 165) Guttation is direct evidence for the presence of:
A)
transpiration
done
clear
B)
absorption
done
clear
C)
root pressure
done
clear
D)
ascent of sap
done
clear
View Answer play_arrow
question_answer 166) Mycorrhiza is useful association because :
A)
it fixes atmospheric nitrogen
done
clear
B)
it enhances absorption of nutrients from soil
done
clear
C)
it kills pathogens
done
clear
D)
it provides resistance against drought
done
clear
View Answer play_arrow
question_answer 167) Spores can be liberated from sporophyte only after the decay of gametophyte in :
A)
Riccia
done
clear
B)
Funaria
done
clear
C)
Marchantia
done
clear
D)
Anthoceros
done
clear
View Answer play_arrow
question_answer 168) In Cyeas male gametes are produced in the pollen tube by division of :
A)
body cell
done
clear
B)
stalk cell
done
clear
C)
prothallial cell
done
clear
D)
tube cell
done
clear
View Answer play_arrow
question_answer 169) Asteraceae is the new name given to family :
A)
Cruciferae
done
clear
B)
Gramineae
done
clear
C)
Umbelliferae
done
clear
D)
Compositae
done
clear
View Answer play_arrow
question_answer 170) Causal organism of black rust of wheat is :
A)
Phytophthora
done
clear
B)
Ustilago
done
clear
C)
Puccinia
done
clear
D)
Synchytrium
done
clear
View Answer play_arrow
question_answer 171) Decomposers are :
A)
autotroph
done
clear
B)
saprotroph
done
clear
C)
heterotroph
done
clear
D)
autoheterotroph
done
clear
View Answer play_arrow
question_answer 172) In a food chain, lion is a :
A)
producer
done
clear
B)
primary consumer
done
clear
C)
secondary consumer
done
clear
D)
tertiary consumer
done
clear
View Answer play_arrow
question_answer 173) A 13 celled male gametophyte in Selaginella is :
A)
12 cells of antheridium +1 prothallial cell
done
clear
B)
9 cells of antheridium +4 prothallial cell
done
clear
C)
6 cells of antheridium +7 prothallial cell
done
clear
D)
10 cells of antheridium +3 prothallial cell
done
clear
View Answer play_arrow
question_answer 174) Wheat grain is a :
A)
fruit
done
clear
B)
seed
done
clear
C)
inflorescence
done
clear
D)
bulbil
done
clear
View Answer play_arrow
question_answer 175) The carrying capacity of a population is determined by its :
A)
death rate
done
clear
B)
limiting resources
done
clear
C)
reproductive status
done
clear
D)
birth rate
done
clear
View Answer play_arrow
question_answer 176) ?Club fungi? is included in :
A)
Phycomycetes
done
clear
B)
Ascomycetes
done
clear
C)
Basidiomycetes
done
clear
D)
Deuteromycetes
done
clear
View Answer play_arrow
question_answer 177) Biotic factors of a food chain include :
A)
producer
done
clear
B)
consumers
done
clear
C)
decomposers
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 178) Heterothallism is common in :
A)
Mucor
done
clear
B)
Yeast
done
clear
C)
Spirogyra
done
clear
D)
Ulothrix
done
clear
View Answer play_arrow
question_answer 179) Organisms changed earth?s surface from reducing to oxidizing status are :
A)
autotrophs
done
clear
B)
heterotrophs
done
clear
C)
chemotrophs
done
clear
D)
heteroautotrophs
done
clear
View Answer play_arrow
question_answer 180) Nucleoli are rich in :
A)
RNA
done
clear
B)
DNA
done
clear
C)
protein and RNA
done
clear
D)
protein and DNA
done
clear
View Answer play_arrow
question_answer 181) Dictyosomes are also known as :
A)
ribosomes
done
clear
B)
Golgi bodies
done
clear
C)
lysosomes
done
clear
D)
mesosomes
done
clear
View Answer play_arrow
question_answer 182) The basic unit of classification of plants and animals is :
A)
genus
done
clear
B)
species
done
clear
C)
order
done
clear
D)
family
done
clear
View Answer play_arrow
question_answer 183) Diatomaceous earth is formed due to remains of diatoms :
A)
protoplasm
done
clear
B)
cell wall
done
clear
C)
cell membrane
done
clear
D)
skeleton
done
clear
View Answer play_arrow
question_answer 184) The point at which crossing over occurs is called as :
A)
chiasmata
done
clear
B)
centromere
done
clear
C)
chromosome
done
clear
D)
gene loci
done
clear
View Answer play_arrow
question_answer 185) In the process of respiration, the potential energy stored in organic compounds is released in the form of :
A)
kinetic energy
done
clear
B)
chemical energy
done
clear
C)
radiant energy
done
clear
D)
light energy
done
clear
View Answer play_arrow
question_answer 186) Pili in bacteria represent:
A)
flagella
done
clear
B)
cilia
done
clear
C)
extrachromosomal genetic element
done
clear
D)
outgrowth of cell membrane of donor cell
done
clear
View Answer play_arrow
question_answer 187) Parasitic alga is :
A)
Ulothrix
done
clear
B)
Gelidium
done
clear
C)
Oscillatoria
done
clear
D)
Cephaleuros
done
clear
View Answer play_arrow
question_answer 188) The organelle concerned with photorespiration is
A)
ribosome
done
clear
B)
peroxisome
done
clear
C)
glyoxysome
done
clear
D)
lysosome
done
clear
View Answer play_arrow
question_answer 189) Increase in the concentration of pollutants in higher trophic level is called :
A)
biodegradation
done
clear
B)
biomagnification
done
clear
C)
biodiversity
done
clear
D)
biotism
done
clear
View Answer play_arrow
question_answer 190) When a tall and red flowered individual is crossed with a dwarf and white flowered individual, phenotype of the progeny will be :
A)
homozygous tall and red
done
clear
B)
heterozygous tall and red
done
clear
C)
homozygous tall and white
done
clear
D)
homozygous dwarf and white
done
clear
View Answer play_arrow
question_answer 191) If the chromosome number of leaf cell is 42 the chromosome number of root cell, microspore mother cell and embryo respectively will be :
A)
42, 42, 42
done
clear
B)
42, 21, 21
done
clear
C)
21, 21, 21
done
clear
D)
21, 21, 42
done
clear
View Answer play_arrow
question_answer 192) Saffron from Crocus is obtained from its :
A)
flowers
done
clear
B)
style and stigma
done
clear
C)
stamens
done
clear
D)
petals
done
clear
View Answer play_arrow
question_answer 193) Growth of lateral bud is promoted by :
A)
auxin
done
clear
B)
cytokinin
done
clear
C)
gibberellin
done
clear
D)
abscisic acid
done
clear
View Answer play_arrow
question_answer 194) Pigment associated with photomorphogenesisis :
A)
chlorophyll
done
clear
B)
anthocyanin
done
clear
C)
phytochrome
done
clear
D)
carotenoid
done
clear
View Answer play_arrow
question_answer 195)
A)
Malvaceae
done
clear
B)
Brassicaceae
done
clear
C)
Cucurbitaceae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
question_answer 196) Flowering in short day plants is indicated by :
A)
short night
done
clear
B)
short day and uninterrupted long night
done
clear
C)
long days and interrupted night
done
clear
D)
long night
done
clear
View Answer play_arrow
question_answer 197) Edible part of coconut is :
A)
seed
done
clear
B)
endosperm
done
clear
C)
mesocarp
done
clear
D)
whole
done
clear
View Answer play_arrow
question_answer 198) Fibrous root system is better adopted than tap root system for:
A)
transport of organic matter
done
clear
B)
absorption of water and minerals
done
clear
C)
storage of food
done
clear
D)
anchorage of plant to soil
done
clear
View Answer play_arrow
question_answer 199) Uniform erosion caused by running water is called
A)
gully erosion
done
clear
B)
sheet erosion
done
clear
C)
rillerosinon
done
clear
D)
wind erosion
done
clear
View Answer play_arrow
question_answer 200) Which of the following control flowering in long day plants and sex expression in certain species?
A)
Auxin
done
clear
B)
Cytokinin
done
clear
C)
Gibberellin
done
clear
D)
Ethylene
done
clear
View Answer play_arrow