question_answer 1) The position of the particle moving along y-axis is given as \[\text{y}=\text{A}{{\text{t}}^{\text{2}}}-\text{B}{{\text{t}}^{\text{3}}},\]where y is measured in metre and t in second. Then, the dimensions of B are
A)
\[\left[ \text{L}{{\text{T}}^{-\text{2}}} \right]\]
done
clear
B)
\[\left[ \text{L}{{\text{T}}^{-\text{1}}} \right]\]
done
clear
C)
\[\left[ \text{L}{{\text{T}}^{-\text{3}}} \right]\]
done
clear
D)
\[\left[ \text{ML}{{\text{T}}^{-\text{2}}} \right]\]
done
clear
View Answer play_arrow
question_answer 2) A conveyor belt is moving horizontally at a speed of 4 m/s. A box of mass 20 kg is gently laid on it. It takes O.ls for the box to come to rest. If the belt continues to move uniformly, then the distance moved by the box on the conveyor belt is
A)
zero
done
clear
B)
0.2 m
done
clear
C)
0.4 m
done
clear
D)
0.8 m
done
clear
View Answer play_arrow
question_answer 3) If retardation produced by air resistance to the particle is one-tenth of acceleration due to gravity, the time to return from the maximum height
A)
increases by 9%
done
clear
B)
decreases by 9 %
done
clear
C)
increases by 11%
done
clear
D)
decreases by 11%
done
clear
View Answer play_arrow
question_answer 4) An open knife edge of mass m is dropped from a height h on a wooden floor. If the blade penetrates distance s into the wood, the average resistance offered by the wood to the blade is
A)
\[mg\]
done
clear
B)
\[mg\frac{h}{s}\]
done
clear
C)
\[mg\left( 1-\frac{h}{s} \right)\]
done
clear
D)
\[mg{{\left( 1+\frac{h}{s} \right)}^{2}}\]
done
clear
View Answer play_arrow
question_answer 5) Energy required to accelerate a car from \[\text{1}0\text{m}/\text{s to 2}0\text{ m}/\text{s}\]compared with that required to accelerate it from 0 to \[\text{1}0\text{ m}/\text{s}\]is
A)
twice
done
clear
B)
thrice
done
clear
C)
four times
done
clear
D)
same
done
clear
View Answer play_arrow
question_answer 6) A solid sphere of mass 2 kg rolls up a \[30{}^\circ \] incline with an initial speed of 10 m/s. The maximum height reached by the sphere is\[\left( \text{g}=\text{1}0\text{m}/{{\text{s}}^{\text{2}}} \right)\]
A)
3.5 m
done
clear
B)
7.0 m
done
clear
C)
10.5 m
done
clear
D)
14.0 m
done
clear
View Answer play_arrow
question_answer 7) Two satellites are moving in the same circular orbit around the earth. They must have the same
A)
mass
done
clear
B)
angular momentum
done
clear
C)
kinetic energy
done
clear
D)
speed
done
clear
View Answer play_arrow
question_answer 8) A planet having average surface temperature \[{{T}_{0}}\]at an average distance d from the sun. Assuming that the planet receives radiant energy from the sun only and it loses radiant energy only from the surface and neglecting all other atmospheric effects we conclude
A)
\[{{T}_{0}}\propto {{d}^{2}}\]
done
clear
B)
\[{{T}_{0}}\propto {{d}^{-2}}\]
done
clear
C)
\[{{T}_{0}}\propto {{d}^{1/2}}\]
done
clear
D)
\[{{T}_{0}}\propto {{d}^{-1/2}}\]
done
clear
View Answer play_arrow
question_answer 9) Time period of a simple pendulum is T. If its length increases by 2%, the new time period becomes
A)
0.98 T
done
clear
B)
1.02 T
done
clear
C)
0.99 T
done
clear
D)
1.01 T
done
clear
View Answer play_arrow
question_answer 10) A boat is rocked by waves of velocity 25 m/s having crests 100 m apart. They reach the boat once every
A)
4.0 s
done
clear
B)
8.0 s
done
clear
C)
2.0 s
done
clear
D)
0.25 s
done
clear
View Answer play_arrow
question_answer 11) Two point charges +2C and + 6C repel each other with a force of 12 N. If a charge of- 2C is given to each of these charges the force will now be
A)
zero
done
clear
B)
8 N (attractive)
done
clear
C)
8 N (repulsive)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
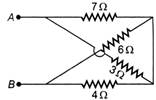
question_answer 12)
The equivalent resistance between A and B (of the circuit shown) is
A)
4.5\[\Omega \]
done
clear
B)
12\[\Omega \]
done
clear
C)
5.4\[\Omega \]
done
clear
D)
20\[\Omega \]
done
clear
View Answer play_arrow
question_answer 13) A 2 kW boiler used for 1 h/day consumes the following electrical energy in thirty days
A)
60 unit
done
clear
B)
120 unit
done
clear
C)
15 unit
done
clear
D)
\[6\times {{10}^{4}}\]unit
done
clear
View Answer play_arrow
question_answer 14) Two long straight wires are set parallel to each other at separation r and each carries a current \[I\] in the same direction. The strength of the magnetic field at any point midway between the two wires is
A)
\[\frac{{{\mu }_{o}}I}{\pi r}\]
done
clear
B)
\[\frac{2{{\mu }_{o}}I}{\pi r}\]
done
clear
C)
\[\frac{{{\mu }_{o}}I}{2\pi r}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 15) An inductive coil has a resistance of 100\[\Omega \]. When an AC signal of frequency 1000 Hz is applied to the coil, the voltage leads the current by 45°. The inductance of the coil is
A)
\[\frac{1}{10\pi }\]
done
clear
B)
\[\frac{1}{20\pi }\]
done
clear
C)
\[\frac{1}{40\pi }\]
done
clear
D)
\[\frac{1}{60\pi }\]
done
clear
View Answer play_arrow
question_answer 16) The intensity of a plane electromagnetic wave is \[\text{5W}/{{\text{m}}^{\text{2}}}.\] It is incident on a perfectly reflecting surface. Find the radiation pressure.
A)
\[3.33\times {{10}^{-9}}N/{{m}^{2}}\]
done
clear
B)
\[3.33\times {{10}^{-8}}N/{{m}^{2}}\]
done
clear
C)
\[8.0\times {{10}^{-8}}N/{{m}^{2}}\]
done
clear
D)
\[8.0\times {{10}^{-9}}N/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 17) A boy stands straight in front of a mirror at a distance of 30 cm from it. He sees his erect image whose height is - of his real height. The mirror he is using, is
A)
plane
done
clear
B)
convex
done
clear
C)
concave
done
clear
D)
plano-concave
done
clear
View Answer play_arrow
question_answer 18) If the wavelength of incident radiation in a photoelectric experiment is decreased, then the
A)
photoelectric current will decrease
done
clear
B)
photoelectric current will increase
done
clear
C)
stopping potential will decrease
done
clear
D)
stopping potential will increase
done
clear
View Answer play_arrow
question_answer 19) The energy associated with a thermal neutron is of the order of
A)
10 MeV
done
clear
B)
1 keV
done
clear
C)
0.1 MeV
done
clear
D)
0.01 eV
done
clear
View Answer play_arrow
question_answer 20) In the depletion region of an unbiased p-n junction diode, there are
A)
only electrons
done
clear
B)
only holes
done
clear
C)
both electrons and holes
done
clear
D)
only fixed ions
done
clear
View Answer play_arrow
question_answer 21) The condition under which vectors \[(\overset{\to }{\mathop{\text{a}}}\,+\overset{\to }{\mathop{\text{b}}}\,)\] and \[(\overset{\to }{\mathop{\text{a}}}\,-\overset{\to }{\mathop{\text{b}}}\,)\] should be at right angles to each other is
A)
\[\overset{\to }{\mathop{\text{a}}}\,\ne \overset{\to }{\mathop{\text{b}}}\,\]
done
clear
B)
\[\overset{\to }{\mathop{\text{a}}}\,.\overset{\to }{\mathop{\text{b}}}\,=0\]
done
clear
C)
\[|\overset{\to }{\mathop{\text{a}}}\,|=|\overset{\to }{\mathop{\text{b}}}\,|\]
done
clear
D)
\[\overset{\to }{\mathop{\text{a}}}\,.\overset{\to }{\mathop{\text{b}}}\,=1\]
done
clear
View Answer play_arrow
question_answer 22) The error in the measurement of radius of the sphere is 0.3%. What is the permissible error in its surface area?
A)
0.6%
done
clear
B)
1.2%
done
clear
C)
1.8%
done
clear
D)
2.4%
done
clear
View Answer play_arrow
question_answer 23) A fielder in a cricket match throws ball from the boundary line to the wicket keeper. The ball describes a parabolic path. Which of the following quantities remain constant during the motion in air? (Neglecting air resistance)
A)
Kinetic energy
done
clear
B)
Vertical component of velocity
done
clear
C)
Horizontal component of velocity
done
clear
D)
Speed
done
clear
View Answer play_arrow
question_answer 24) A thin rod of mass m and length 21 is made to rotate about an axis passing through its centre and perpendicular to it. If its angular velocity changes from 0 to \[\omega \]time t, the torque acting on it is
A)
\[\frac{m{{l}^{2}}\omega }{12t}\]
done
clear
B)
\[\frac{m{{l}^{2}}\omega }{3t}\]
done
clear
C)
\[\frac{m{{l}^{2}}\omega }{t}\]
done
clear
D)
\[\frac{4m{{l}^{2}}\omega }{3t}\]
done
clear
View Answer play_arrow
question_answer 25) A gas is heated at constant pressure. The fraction of heat supplied used for external work is
A)
\[\frac{1}{y}\]
done
clear
B)
\[\left( 1-\frac{1}{y} \right)\]
done
clear
C)
\[y-1\]
done
clear
D)
\[\left( 1-\frac{1}{{{y}^{2}}} \right)\]
done
clear
View Answer play_arrow
question_answer 26) The maximum displacement of the particle executing SHM is 1 cm and the maximum acceleration is \[{{\left( \text{1}.\text{57 } \right)}^{\text{2}}}\text{ cm}/{{\text{s}}^{\text{2}}}.\] Its time period is
A)
0.25 s
done
clear
B)
4.0 s
done
clear
C)
1.57 s
done
clear
D)
3.14 s
done
clear
View Answer play_arrow
question_answer 27) The equation of a wave travelling on a string is \[y=4\sin \left[ \frac{\pi }{2}\left( 8t-\frac{x}{8} \right) \right],\]where x, y are in cm and t in second. The velocity of the wave is
A)
64 cm /s, in - x direction
done
clear
B)
32 cm /s, in - x direction
done
clear
C)
32 cm /s, in + x direction
done
clear
D)
64 cm /s, in + x direction
done
clear
View Answer play_arrow
question_answer 28)
In the figure, the equivalent capacitance between A and B is
A)
\[\text{3}.\text{75 }\mu \text{F}\]
done
clear
B)
\[\text{5}.\text{25 }\mu \text{F}\]
done
clear
C)
\[\text{6}.\text{5 }\mu \text{F}\]
done
clear
D)
\[\text{1}0.\text{5 }\mu \text{F}\]
done
clear
View Answer play_arrow
question_answer 29) Two bulbs 100 W, 250 V and 200 W, 250 V are connected in parallel across a 500 V line. Then,
A)
100 W bulb will be fused
done
clear
B)
200 W bulb will be fused
done
clear
C)
both bulbs will be fused
done
clear
D)
no bulb will be fused
done
clear
View Answer play_arrow
question_answer 30) A long solenoid has 20 turns /cm. The current necessary to produce a magnetic field of 20 mT inside the solenoid is approximately
A)
1A
done
clear
B)
2 A
done
clear
C)
4 A
done
clear
D)
8 A
done
clear
View Answer play_arrow
question_answer 31) The inductance of a coil is proportional to
A)
its length
done
clear
B)
the number of turns
done
clear
C)
the resistance of the coil
done
clear
D)
the square of the number of turns
done
clear
View Answer play_arrow
question_answer 32) A photosensitive surface is receiving light of wavelength \[5000\overset{\text{o}}{\mathop{\text{A}}}\,\] at the rate of \[\text{1}{{0}^{-7}}\text{ J}/\text{s}.\] The number of photons ejected per second is
A)
\[\text{2}.\text{5}\times \text{1}{{0}^{12}}\]
done
clear
B)
\[\text{2}.\text{5}\times \text{1}{{0}^{11}}\]
done
clear
C)
\[\text{2}.\text{5}\times \text{1}{{0}^{10}}\]
done
clear
D)
\[\text{2}.\text{5}\times \text{1}{{0}^{9}}\]
done
clear
View Answer play_arrow
question_answer 33) A ball is projected horizontally with a velocity of 4 m/s. The velocity of the ball after 0.7 s is \[\left( \text{g}=\text{1}0\text{m}/{{\text{s}}^{\text{2}}} \right)\]
A)
11 m/s
done
clear
B)
10 m/s
done
clear
C)
8 m/s
done
clear
D)
3 m/s
done
clear
View Answer play_arrow
question_answer 34) Unification of weak and electromagnetic interaction was done by
A)
Einstein
done
clear
B)
Raman
done
clear
C)
Salam
done
clear
D)
Hubble
done
clear
View Answer play_arrow
question_answer 35) Two discs have same mass and same thickness. Their materials are of densities \[{{\rho }_{1}} and {{\rho }_{2}}\] . The ratio of their moments of inertia about central axis will be
A)
\[{{\rho }_{1}}{{\rho }_{2}}:1\]
done
clear
B)
\[1:{{\rho }_{1}}{{\rho }_{2}}\]
done
clear
C)
\[{{\rho }_{1}}:{{\rho }_{2}}\]
done
clear
D)
\[{{\rho }_{2}}:{{\rho }_{1}}\]
done
clear
View Answer play_arrow
question_answer 36) The Youngs modulus of the material of the wire of length L and radius r is \[\text{Y N}/{{\text{m}}^{\text{2}}}.\]If the length is reduced to L/2 and radius r/2, the Youngs modulus will be
A)
\[\text{Y}/\text{2}\]
done
clear
B)
\[\text{Y}\]
done
clear
C)
2\[\text{Y}\]
done
clear
D)
4\[\text{Y}\]
done
clear
View Answer play_arrow
question_answer 37) A body floats with one-third of its volume outside water and 3/4 of its volume outside another liquid. The density of the other liquid is
A)
\[\frac{9}{4}g/cc\]
done
clear
B)
\[\frac{4}{3}g/cc\]
done
clear
C)
\[\frac{8}{3}g/cc\]
done
clear
D)
\[\frac{3}{8}g/cc\]
done
clear
View Answer play_arrow
question_answer 38) Two soap bubbles have radii in the ratio of 2 : 1. What is the ratio of excess pressures inside them?
A)
1 : 2
done
clear
B)
1 : 4
done
clear
C)
2 : 1
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
question_answer 39) The terminal velocity of small-sized spherical body of radius r falling vertically in a viscous liquid is given by the following proportionality
A)
\[1{{r}^{2}}\]
done
clear
B)
\[1\,r\]
done
clear
C)
\[\,r\]
done
clear
D)
\[{{r}^{2}}\]
done
clear
View Answer play_arrow
question_answer 40) The reading of a manometer fitted to a closed tap is \[\text{3}.\text{5}\times \text{l}{{0}^{\text{5}}}\text{ N}/{{\text{m}}^{\text{2}}}.\] If the valve is opened, the reading of the manometer falls to \[\text{3}\times \text{1}{{0}^{\text{5}}}\text{ N}/{{\text{m}}^{\text{2}}}.\]The velocity of water is
A)
1m/s
done
clear
B)
10 m/s
done
clear
C)
100 m/s
done
clear
D)
0.1 m/s
done
clear
View Answer play_arrow
question_answer 41) 1 L of an ideal gas at \[27{}^\circ C\] is heated at a constant pressure to \[297{}^\circ C\]. Then, the final volume is approximately
A)
1.2 L
done
clear
B)
1.9 L
done
clear
C)
19 L
done
clear
D)
2.4 L
done
clear
View Answer play_arrow
question_answer 42) Coefficient of cubical expansion of water is zero at
A)
\[0{}^\circ C\]
done
clear
B)
\[4{}^\circ C\]
done
clear
C)
\[15.5{}^\circ C\]
done
clear
D)
\[100{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 43) A cylindrical conductor is placed near another positively charged conductor. The net charge acquired by the cylindrical conductor will be
A)
positive only
done
clear
B)
negative only
done
clear
C)
zero
done
clear
D)
either positive or negative
done
clear
View Answer play_arrow
question_answer 44) The ratio of areas between the electron orbits for the first excited state to the ground state for the hydrogen atom is
A)
2 : 1
done
clear
B)
4 : 1
done
clear
C)
8 : 1
done
clear
D)
16 : 1
done
clear
View Answer play_arrow
question_answer 45) An electron, accelerated by a potential difference V, has de-Broglie wavelength\[\lambda \]. If the electron is accelerated by a potential difference 4V, its de-Broglie wavelength will be
A)
\[2\lambda \]
done
clear
B)
\[4\lambda \]
done
clear
C)
\[\frac{\lambda }{2}\]
done
clear
D)
\[\frac{\lambda }{4}\]
done
clear
View Answer play_arrow
question_answer 46) A convex lens of focal length 40 cm is in contact with a concave lens of focal length 25 cm. The power of the combination in dioptre is
A)
-1.5
done
clear
B)
-6.5
done
clear
C)
+ 6.5
done
clear
D)
+ 6.67
done
clear
View Answer play_arrow
question_answer 47) A material particle with a rest mass mo is moving with speed of light c. The de-Broglie wavelength associated is given by
A)
\[\frac{h}{{{m}_{0}}c}\]
done
clear
B)
\[\frac{{{m}_{0}}c}{h}\]
done
clear
C)
Zero
done
clear
D)
infinity
done
clear
View Answer play_arrow
question_answer 48) The impedance of a circuit consists of 3\[\Omega \] resistance and 4\[\Omega \] reactance. The power factor of the circuit is
A)
0.4
done
clear
B)
0.6
done
clear
C)
0.8
done
clear
D)
1.0
done
clear
View Answer play_arrow
question_answer 49) A glass has refractive index 3/2 and water has refractive index 4/3. If the speed of light in glass is \[\text{2}\times \text{1}{{0}^{\text{8}}}\text{ m}/\text{s},\] the speed of light in water in m/s is
A)
\[\text{1}.\text{5}\times \text{l}{{0}^{\text{6}}}\]
done
clear
B)
\[\text{1}.\text{78}\times \text{l}{{0}^{\text{8}}}\]
done
clear
C)
\[\text{2}.\text{25}\times \text{l}{{0}^{\text{8}}}\]
done
clear
D)
\[\text{2}.\text{67}\times \text{l}{{0}^{\text{8}}}\]
done
clear
View Answer play_arrow
question_answer 50) A bulb is placed between two plane mirrors inclined at an angle of 60°.The number of images formed is
A)
5
done
clear
B)
6
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 51) The mass of a mole of proton and electron is
A)
\[6.023\times {{10}^{23}}g\]
done
clear
B)
1.008 g and 0.55 mg
done
clear
C)
\[9.1\times {{10}^{-28}}kg\]
done
clear
D)
2 g
done
clear
View Answer play_arrow
question_answer 52) Correct set of four quantum numbers for valence electron of rubidium (Z = 37) is
A)
5, 0, 0, \[+\frac{1}{2}\]
done
clear
B)
5,1, 0, \[+\frac{1}{2}\]
done
clear
C)
5, 1, 1, \[+\frac{1}{2}\]
done
clear
D)
6, 0, 0, \[+\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 53) The reaction \[2NO(g)+{{O}_{2}}(g)2N{{O}_{2}}(g)\] is of first order. If volume of reaction vessel is reduced to the rate of reaction would be
A)
\[\frac{1}{3}\] times
done
clear
B)
\[\frac{2}{3}\] times
done
clear
C)
3 times
done
clear
D)
6 times
done
clear
View Answer play_arrow
question_answer 54) Which of the following species has the highest electron affinity?
A)
F
done
clear
B)
O
done
clear
C)
\[{{O}^{-}}\]
done
clear
D)
\[N{{a}^{+}}\]
done
clear
View Answer play_arrow
question_answer 55) The largest number of molecules is in
A)
34 g of water
done
clear
B)
28 g of \[C{{O}_{2}}\]
done
clear
C)
46 g of \[C{{H}_{3}}OH\]
done
clear
D)
54 g of \[{{N}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 56) Gold is extracted by hydrometallurgical process based on its property
A)
of being electropositive
done
clear
B)
of being less reactive
done
clear
C)
to form complexes which are water soluble
done
clear
D)
to form salts which are water soluble
done
clear
View Answer play_arrow
question_answer 57) Argentite is a mineral of
A)
copper
done
clear
B)
silver
done
clear
C)
platinum
done
clear
D)
aluminium
done
clear
View Answer play_arrow
question_answer 58) As the s-character of hybridised orbital increases, the bond angle
A)
increases
done
clear
B)
decrease
done
clear
C)
becomes zero
done
clear
D)
does not change
done
clear
View Answer play_arrow
question_answer 59) In the calcium fluoride structure the coordination number of the cation and anions are respectively
A)
6, 6
done
clear
B)
8, 4
done
clear
C)
4,4
done
clear
D)
4,8
done
clear
View Answer play_arrow
question_answer 60) Which of the following liquid pairs shows a positive deviation from Raoults law?
A)
Water-nitric acid
done
clear
B)
Benzene-methanol
done
clear
C)
Water-hydrochloric acid
done
clear
D)
Acetone-chloroform
done
clear
View Answer play_arrow
question_answer 61) What should be the freezing point of aqueous solution containing 17 g of \[{{C}_{2}}{{H}_{5}}OH\] in 1000 g of water (water \[{{k}_{f}}={{1.86}^{o}}kg\,mo{{l}^{-1}}\]) ?
A)
\[-{{0.69}^{o}}C\]
done
clear
B)
\[{{0.34}^{o}}C\]
done
clear
C)
\[{{0.0}^{o}}C\]
done
clear
D)
\[{{0.24}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 62) A radioisotope will not emit
A)
gamma and alpha rays simultaneously
done
clear
B)
gamma rays only
done
clear
C)
alpha and beta rays simultaneously
done
clear
D)
beta and gamma rays simultaneously
done
clear
View Answer play_arrow
question_answer 63) Hardness of water is due to presence of salts of
A)
\[N{{a}^{+}}\] and \[{{K}^{+}}\]
done
clear
B)
\[C{{a}^{2+}}\] and \[M{{g}^{2+}}\]
done
clear
C)
\[C{{a}^{2+}}\] and \[{{K}^{+}}\]
done
clear
D)
\[C{{a}^{2+}}\] and \[N{{a}^{+}}\]
done
clear
View Answer play_arrow
question_answer 64) What is the role of a catalyst in a reaction?
A)
It lowers the activation energy
done
clear
B)
It increases the activation energy
done
clear
C)
It affects the free energy change
done
clear
D)
It affects the enthalpy change
done
clear
View Answer play_arrow
question_answer 65) Noble gas are adsorbed by
A)
anhydrous calcium chloride
done
clear
B)
ferric hydroxide
done
clear
C)
cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
activated coconut charcoal
done
clear
View Answer play_arrow
question_answer 66) A cylinder of 5 L capacity, filled with air at NTP is connected with another evacuated cylinder of 30 L of capacity. The resultant air pressure in both the cylinders will
A)
10.8 cm of Hg
done
clear
B)
14.9 cm of Hg
done
clear
C)
21.8 cm of Hg
done
clear
D)
38.8 cm of Hg
done
clear
View Answer play_arrow
question_answer 67) The right order of the solubility of sulphates of alkaline earth metals in water, is
A)
Be > Ca > Mg > Ba > Sr
done
clear
B)
Mg > Be > Ba > Ca > Sr
done
clear
C)
Be > Mg > Ca > Sr > Ba
done
clear
D)
Mg > Ca > Ba > Be > Sr
done
clear
View Answer play_arrow
question_answer 68) In Halls process, the main reagent is mixed with
A)
NaF
done
clear
B)
\[N{{a}_{3}}Al{{F}_{6}}\]
done
clear
C)
\[Al{{F}_{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 69) Inorganic graphite is
A)
\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\]
done
clear
B)
\[{{B}_{3}}{{N}_{3}}\]
done
clear
C)
SiC
done
clear
D)
\[Fe{{(CO)}_{5}}\]
done
clear
View Answer play_arrow
question_answer 70) Which of the following halides is least stable and has doubtful existence?
A)
\[C{{I}_{4}}\]
done
clear
B)
\[Ge{{I}_{4}}\]
done
clear
C)
\[Sn{{I}_{4}}\]
done
clear
D)
\[Pb{{I}_{4}}\]
done
clear
View Answer play_arrow
question_answer 71) Which of the following ions has the least magnetic moment?
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[N{{i}^{2+}}\]
done
clear
C)
\[C{{o}^{2+}}\]
done
clear
D)
\[F{{e}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 72) In solid \[CuS{{O}_{4}}\,.\,\,5{{H}_{2}}O\], copper is coordinated to
A)
five water molecules
done
clear
B)
four water molecules
done
clear
C)
one sulphate anion
done
clear
D)
one water molecule
done
clear
View Answer play_arrow
question_answer 73) Which of the following is a local anesthetic?
A)
Diazepam
done
clear
B)
Procaine
done
clear
C)
Chlorophenicol
done
clear
D)
Penicillin-G
done
clear
View Answer play_arrow
question_answer 74) Which of the following is not an actinide?
A)
Curium
done
clear
B)
Californium
done
clear
C)
Uranium
done
clear
D)
Terbium
done
clear
View Answer play_arrow
question_answer 75) The oxidation state of chlorine in \[KCl{{O}_{4}}\] is
A)
-1
done
clear
B)
+1
done
clear
C)
+7
done
clear
D)
-7
done
clear
View Answer play_arrow
question_answer 76) Glucose and mannose are
A)
epimers
done
clear
B)
anomers
done
clear
C)
ketohexoses
done
clear
D)
disaccharide
done
clear
View Answer play_arrow
question_answer 77) Proteins when heated with cone. \[HN{{O}_{3}}\], give a yellow colour. This is
A)
oxidising test
done
clear
B)
xanthoprotic test
done
clear
C)
Hoppes test
done
clear
D)
acid-base test
done
clear
View Answer play_arrow
question_answer 78) Which reagent can be used to identify nickel ion?
A)
Resorcinol
done
clear
B)
Dimethyl glyoxime [DMG]
done
clear
C)
Diphenyl benzidine
done
clear
D)
Potassium ferrocyanide
done
clear
View Answer play_arrow
question_answer 79) \[Mn{{O}_{2}}\] and \[{{H}_{2}}S{{O}_{4}}\] are added to NaCl. The greenish yellow gas liberated is
A)
\[C{{l}_{2}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[{{N}_{3}}\]
done
clear
D)
\[{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 80) Lassaignes test for the detection of nitrogen fails in
A)
\[N{{H}_{2}}CONHN{{H}_{2}}.\,HCl\]
done
clear
B)
\[N{{H}_{2}}.\,\,N{{H}_{2}}.\,\,HCl\]
done
clear
C)
\[N{{H}_{2}}CON{{H}_{2}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}NHN{{H}_{2}}.\,\,HCl\]
done
clear
View Answer play_arrow
question_answer 81) The IUPAC name of the compound \[C{{H}_{3}}-CH({{C}_{2}}{{H}_{5}})-CH=CH-C{{H}_{3}}\] is
A)
4-ethyl-2-pentene
done
clear
B)
4-methyl-2-hexene
done
clear
C)
3-ethyl-2-pentene
done
clear
D)
2-ethyl-3-pentene
done
clear
View Answer play_arrow
question_answer 82) Which is the most stable carbocation?
A)
iso-propyl
done
clear
B)
triphenyl methyl cation
done
clear
C)
Ethyl cation
done
clear
D)
\[\pi \] propyl cation
done
clear
View Answer play_arrow
question_answer 83) Which one of the following is least reactive in a nucleophilic substitution reaction?
A)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
B)
\[C{{H}_{2}}=CHC{{H}_{2}}Cl\]
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}C-Cl\]
done
clear
D)
\[C{{H}_{2}}=CHCl\]
done
clear
View Answer play_arrow
question_answer 84) Octane number can be changed by
A)
isomerisation
done
clear
B)
alkylation
done
clear
C)
cyclisation
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 85) Which one of the following compounds does not form an ozonide?
A)
Ethene
done
clear
B)
Propyne
done
clear
C)
Propene
done
clear
D)
Propane
done
clear
View Answer play_arrow
question_answer 86) The synthetic polymer which resembles natural rubber is
A)
neoprene
done
clear
B)
chloroprene
done
clear
C)
glyptal
done
clear
D)
nylon
done
clear
View Answer play_arrow
question_answer 87) If \[{{K}_{c}}\] is the equilibrium constant for the formation of \[N{{H}_{3}}\]. The dissociation constant of ammonia under the same temperature will be
A)
\[{{K}_{c}}\]
done
clear
B)
\[\sqrt{{{K}_{c}}}\]
done
clear
C)
\[K_{c}^{2}\]
done
clear
D)
\[\frac{1}{{{K}_{c}}}\]
done
clear
View Answer play_arrow
question_answer 88) If solubility product for \[HgS{{O}_{4}}\] is \[6.4\times {{10}^{-5}}\], then solubility of the salt is
A)
\[8\times {{10}^{-3}}\]
done
clear
B)
\[8\times {{10}^{-6}}\]
done
clear
C)
\[6.4\times {{10}^{-5}}\]
done
clear
D)
\[6.4\times {{10}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 89) The atomic weight of Al is 27. When a current of 5 F is passed through a solution of \[A{{l}^{3+}}\]ions, the weight of Al deposited is
A)
27 g
done
clear
B)
36 g
done
clear
C)
45 g
done
clear
D)
39 g
done
clear
View Answer play_arrow
question_answer 90) Given, standard electrode potentials of following reactions, \[F{{e}^{2+}}+2{{e}^{-}}\xrightarrow{{}}Fe\,;\,\,{{E}^{o}}=-0.440\,\,V\] \[F{{e}^{3+}}+3{{e}^{-}}\xrightarrow{{}}Fe\,;\,\,{{E}^{o}}=-0.0.36\,\,V\] The standard electrode potential \[({{E}^{o}})\] for \[F{{e}^{3+}}+{{e}^{-}}\xrightarrow{{}}F{{e}^{2+}}\], is
A)
-0.476 V
done
clear
B)
-0.404 V
done
clear
C)
+ 0.404 V
done
clear
D)
+ 0.772 V
done
clear
View Answer play_arrow
question_answer 91) The value of \[\Delta H-\Delta E\] for the following reaction at \[{{27}^{o}}C\] will be\[2N{{H}_{3}}(g)\xrightarrow{{}}{{N}_{2}}(g)=3{{H}_{2}}(g)\]
A)
\[8.314\times 273\times (-2)\]
done
clear
B)
\[8.314\times 300\times (-2)\]
done
clear
C)
\[8.314\times 27\times (-2)\]
done
clear
D)
\[8.314\times 300\times (-2)\]
done
clear
View Answer play_arrow
question_answer 92) Azo dye is prepared by the coupling of phenol and
A)
diazonium chloride
done
clear
B)
o-nitroaniline
done
clear
C)
benzoic acid
done
clear
D)
chlorobenzene
done
clear
View Answer play_arrow
question_answer 93) Fruity smell is given by
A)
esters
done
clear
B)
alcohols
done
clear
C)
chloroform
done
clear
D)
acid anhydrides
done
clear
View Answer play_arrow
question_answer 94) Reimer-Tiemann reaction involves a
A)
carbonium ion intermediate
done
clear
B)
carbene intermediate
done
clear
C)
carbanion intermediate
done
clear
D)
free radical intermediate
done
clear
View Answer play_arrow
question_answer 95) When acetaldehyde is heated with Fehling solution, it gives a red precipitate of
A)
Cu
done
clear
B)
CuO
done
clear
C)
\[C{{u}_{2}}O\]
done
clear
D)
\[Cu{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 96) Which one of the following undergoes reaction with 50% sodium hydroxide solution to give the corresponding alcohol and acid?
A)
Butanal
done
clear
B)
Benzaldehyde
done
clear
C)
Phenol
done
clear
D)
Benzoic acid
done
clear
View Answer play_arrow
question_answer 97) The alcohol which easily reacts with cone. HCl is
A)
\[C{{H}_{3}}-CHOH-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[{{(C{{H}_{3}})}_{3}}-C-OH\]
done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-OH\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}}-CH-C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 98) At low temperature, phenol reacts with \[B{{r}_{2}}\] in \[C{{S}_{2}}\] to form
A)
m-bromophenol
done
clear
B)
o-and p-bromophenol
done
clear
C)
p-bromophenol
done
clear
D)
2, 4, 6-tribromophenol
done
clear
View Answer play_arrow
question_answer 99)
What will be the product in the following reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 100) The principle buffer present in human blood is
A)
\[N{{a}_{2}}C{{O}_{3}}+N{{a}_{2}}HP{{O}_{4}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{4}}+Na{{H}_{2}}P{{O}_{4}}\]
done
clear
C)
\[{{H}_{2}}C{{O}_{3}}+HCO_{3}^{-}\]
done
clear
D)
\[N{{a}_{2}}HP{{O}_{4}}+N{{a}_{3}}P{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 101) Lysosomes are the reservoirs (store house) of
A)
hydrolytic enzymes
done
clear
B)
secretory glycoproteins
done
clear
C)
RNA and protein
done
clear
D)
fats (or sugars or ATP)
done
clear
View Answer play_arrow
question_answer 102) Which one of the following is correctly matched pair of a certain plant family and its one example?
A)
Malvaceae - Cotton
done
clear
B)
Leguminosae - Mango (or sunflower)
done
clear
C)
Cucurbitaceae - Orange
done
clear
D)
Brassicaceae - Wheat
done
clear
View Answer play_arrow
question_answer 103) Melatonin is secreted by
A)
skin
done
clear
B)
thymus
done
clear
C)
pituitary
done
clear
D)
pineal gland
done
clear
View Answer play_arrow
question_answer 104) Which one of the following is a skull bone?
A)
Coracoid
done
clear
B)
Arytaenoid
done
clear
C)
Adas
done
clear
D)
Pterygoid
done
clear
View Answer play_arrow
question_answer 105) Which one of the following amino acids is an essential part of human diet?
A)
Glycine
done
clear
B)
Phenylalanin
done
clear
C)
Serine
done
clear
D)
Aspartic acid
done
clear
View Answer play_arrow
question_answer 106) A water fern which is used as a green manure in rice fields is
A)
Salvinia
done
clear
B)
Mucor
done
clear
C)
Aspergillus
done
clear
D)
Azolla
done
clear
View Answer play_arrow
question_answer 107) In which of the following animal, post anal tail is found?
A)
Earthworm
done
clear
B)
Lower invertebrate
done
clear
C)
Scorpion
done
clear
D)
Snake
done
clear
View Answer play_arrow
question_answer 108) Apogamy is
A)
reproduction of virus
done
clear
B)
failure of fusion of gametes
done
clear
C)
development of bacteria
done
clear
D)
loss of function of reproduction
done
clear
View Answer play_arrow
question_answer 109) Male XX and female XY sometime occur due to
A)
deletion
done
clear
B)
transfer of segments in X and Y chromosomes
done
clear
C)
aneuploidy
done
clear
D)
hormonal imbalance
done
clear
View Answer play_arrow
question_answer 110) During an injury nasal septum gets damaged. For its recovery which cartilage is preferred?
A)
Elastic cartilage
done
clear
B)
Hyaline cartilage
done
clear
C)
Calcified cartilage
done
clear
D)
Fibrous cartilage
done
clear
View Answer play_arrow
question_answer 111) Darwins finches provide an excellent evidence in favour of organic evolution. These are related to which of the following evidences?
A)
Embryology
done
clear
B)
Palaeontology (or fossils)
done
clear
C)
Anatomy
done
clear
D)
Biogeography (or geographic distribution)
done
clear
View Answer play_arrow
question_answer 112) Which is a reducing sugar?
A)
Galactose
done
clear
B)
Gluconic acid
done
clear
C)
p-methyl galactoside
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
question_answer 113) What will happen if ligaments are cut or broken?
A)
Bones will move freely at joints
done
clear
B)
No movement at joint
done
clear
C)
Bone will become unfixed
done
clear
D)
Bone will become fixed
done
clear
View Answer play_arrow
question_answer 114) Reason of fast speciation in present day crop plants is
A)
mutation
done
clear
B)
isolation
done
clear
C)
polyploidy
done
clear
D)
sexual reproduction
done
clear
View Answer play_arrow
question_answer 115) Which bacteria are utilized in gobar gas plant?
A)
Methanogenes
done
clear
B)
Nitrifying bacteria
done
clear
C)
Ammonifying bacteria
done
clear
D)
Denitrifying bacteria
done
clear
View Answer play_arrow
question_answer 116) In photosynthesis energy from light reaction to dark reaction is transferred in the form of
A)
ADP
done
clear
B)
ATP
done
clear
C)
RuBP
done
clear
D)
Chlorophyll
done
clear
View Answer play_arrow
question_answer 117) In a DNA percentage of thymine is 20%. What is the percentage of guanine?
A)
20%
done
clear
B)
40%
done
clear
C)
30%
done
clear
D)
60%
done
clear
View Answer play_arrow
question_answer 118) ELISA (Enzyme Linked Immuno Sorbent Assay) is an import immunological method which is employed to detect and measure antigens or antibodies. Enzyme reacts on substrate to produce a colour in a positive test. ELISA is used to detect viruses where the key reagent is
A)
DNA probe
done
clear
B)
RNAse
done
clear
C)
alkaline phosphatase
done
clear
D)
catalase
done
clear
View Answer play_arrow
question_answer 119) If Henles loop were absent from mammalian nephron which of the following is to be expected?
A)
The urine will be more concentrated
done
clear
B)
The urine will be more dilute
done
clear
C)
There will be no urine formation
done
clear
D)
There will be hardly any change in the quality and quantity of urine formed
done
clear
View Answer play_arrow
question_answer 120) Degeneration of a genetic code is attributed to the
A)
entrire codon
done
clear
B)
third member of a codon
done
clear
C)
first member of a codon
done
clear
D)
second member of a codon
done
clear
View Answer play_arrow
question_answer 121) You are required to draw blood from patient and to keep it in a test tube for analysis of blood corpuscles and plasma. You are also provided with the following four types of test tubes. Which of them will you not use for the purpose?
A)
Test tube containing calcium bicarbonate
done
clear
B)
Chilled test tube
done
clear
C)
Test tube containing heparin
done
clear
D)
Test tube containing sodium oxalate
done
clear
View Answer play_arrow
question_answer 122) In 1984, the Bhopal gas tragedy took place because methyl isocyanate
A)
reacted with DDT
done
clear
B)
reacted with ammonia
done
clear
C)
reacted with \[C{{O}_{2}}\]
done
clear
D)
reacted with water
done
clear
View Answer play_arrow
question_answer 123) Lack of independent assortment of two genes A and B in fruit fly Drosophila is due to
A)
repulsion
done
clear
B)
recombination
done
clear
C)
linkage
done
clear
D)
crossing over
done
clear
View Answer play_arrow
question_answer 124) In your opinion which is the most effective way to conserve the plant diversity of an area?
A)
By tissue culture method
done
clear
B)
By creating biosphere reserve
done
clear
C)
By creating botanical garden
done
clear
D)
By developing seed bank
done
clear
View Answer play_arrow
question_answer 125) "Continuity of germplasm" theory was given by
A)
Hugo de Vries
done
clear
B)
Weismann
done
clear
C)
Darwin
done
clear
D)
Lamarck
done
clear
View Answer play_arrow
question_answer 126) Grey spots of oat are caused by deficiency of
A)
Mn
done
clear
B)
Fe
done
clear
C)
Cu
done
clear
D)
Zn
done
clear
View Answer play_arrow
question_answer 127) Pattern baldness moustaches and beard in human males are examples of
A)
sex differentiating traits
done
clear
B)
sex determining traits
done
clear
C)
sex linked traits
done
clear
D)
sex limited traits
done
clear
View Answer play_arrow
question_answer 128) For retting of jute the fermenting microbe used is
A)
Helicobastor pylori
done
clear
B)
Methophilic bacteria
done
clear
C)
Streptococcus lactin
done
clear
D)
Butyric acid bacteria
done
clear
View Answer play_arrow
question_answer 129) Ectophloic siphonostele is found in
A)
Adiantum and Cucurbitaceae
done
clear
B)
Osmunda and Equisetum
done
clear
C)
Marsilea and Botrychium
done
clear
D)
Dicksonia and Maiden hair fern
done
clear
View Answer play_arrow
question_answer 130) Which one of the following does not act as a neurotrasmitter?
A)
Acetylcholine
done
clear
B)
Epinephrine
done
clear
C)
Norepinephrine
done
clear
D)
Cortisone
done
clear
View Answer play_arrow
question_answer 131) The respiratory quotient (RQ) of a germinating castor seed is
A)
equal to one
done
clear
B)
greater than one
done
clear
C)
less than one
done
clear
D)
equal to zero
done
clear
View Answer play_arrow
question_answer 132) What is left, when bath-sponges dries up?
A)
Spicules
done
clear
B)
Spongin fibres
done
clear
C)
Tentacles
done
clear
D)
Holdfast
done
clear
View Answer play_arrow
question_answer 133) Enterokinase converts
A)
trypsinogen to trypsin
done
clear
B)
pepsinogen to pepsin
done
clear
C)
chymotrypsin to pepsinogen
done
clear
D)
pepsin to chymotrypsin
done
clear
View Answer play_arrow
question_answer 134) In which of the following stage, the chromo- some is single thin and like long thread?
A)
Leptotene
done
clear
B)
Zygotene
done
clear
C)
Pachytene
done
clear
D)
Diakinesis
done
clear
View Answer play_arrow
question_answer 135) \[{{C}_{4}}\]plants differ from \[{{C}_{3}}\]plants in respect to
A)
number of ATP used
done
clear
B)
substrate which accept the \[C{{O}_{3}}\]molecules
done
clear
C)
the final product
done
clear
D)
number of ATP formed
done
clear
View Answer play_arrow
question_answer 136) Resting potential of a nerve is
A)
+70 millivolt
done
clear
B)
+30 millivolt
done
clear
C)
-30 millivolt
done
clear
D)
-70 millivolt
done
clear
View Answer play_arrow
question_answer 137) Gonads of Obelia occur in
A)
on blastocyst
done
clear
B)
in hydrula stage
done
clear
C)
radial canals of medusa
done
clear
D)
bases of tentacles of medusa
done
clear
View Answer play_arrow
question_answer 138) Bombykol or gyplure is a
A)
drug
done
clear
B)
hormone
done
clear
C)
antibiotic
done
clear
D)
pheromone
done
clear
View Answer play_arrow
question_answer 139) The term "niche" of a species refers to
A)
specific and habitual function
done
clear
B)
specific place where an organism lives
done
clear
C)
competitive power of an organism
done
clear
D)
specific function of an organism
done
clear
View Answer play_arrow
question_answer 140) The correct sequence of food chain is
A)
grass\[\to \] wolf\[\to \] deer\[\to \] buffalo
done
clear
B)
bacteria \[\to \]grass \[\to \]rabbit \[\to \]wolf
done
clear
C)
grass \[\to \]insect \[\to \]bird \[\to \]snake
done
clear
D)
grass \[\to \]snake \[\to \]insect \[\to \]deer
done
clear
View Answer play_arrow
question_answer 141) The number of floating ribs in the human body is
A)
6 pairs
done
clear
B)
5 pairs
done
clear
C)
3 pairs
done
clear
D)
2 pairs
done
clear
View Answer play_arrow
question_answer 142) Myoglobin is present in
A)
all muscle fibres
done
clear
B)
white muscle fibres only
done
clear
C)
red muscle fibres only
done
clear
D)
both white and red muscle fibres
done
clear
View Answer play_arrow
question_answer 143) A couple has 6 children 5 are girls and 1 is boy. The percentage of having a girl on next time is
A)
10%
done
clear
B)
20%
done
clear
C)
50%
done
clear
D)
100%
done
clear
View Answer play_arrow
question_answer 144) The functional xylem of dicot tree is
A)
sap wood
done
clear
B)
hard wood
done
clear
C)
heart wood
done
clear
D)
autumn wood
done
clear
View Answer play_arrow
question_answer 145) In which of the following there is no defect in the sex chromosome?
A)
Turners syndrome
done
clear
B)
Downs syndrome
done
clear
C)
Colour blindness
done
clear
D)
Klinefelters syndrome
done
clear
View Answer play_arrow
question_answer 146) Botanical name of sanjeevani is
A)
Selaginella chrysocaulos
done
clear
B)
Selaginella bryopteris
done
clear
C)
Selaginella chrysorhizos
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 147) The embryonic membrane involved in the formation of placenta in female is
A)
yolk sac
done
clear
B)
allantosis
done
clear
C)
amnion
done
clear
D)
chorion
done
clear
View Answer play_arrow
question_answer 148) Elbow joint is
A)
ball and socket
done
clear
B)
hinge joint
done
clear
C)
pivot joint
done
clear
D)
saddle joint
done
clear
View Answer play_arrow
question_answer 149) Agar- agar is obtained from
A)
fungi
done
clear
B)
algae
done
clear
C)
bacteria
done
clear
D)
lichens
done
clear
View Answer play_arrow
question_answer 150) Condom "Nirodh" formed by
A)
Hindustan Liver Ltd
done
clear
B)
Hindustan Latex Ltd
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 151) Spider web is formed by a fluid secreted by its
A)
abdominal gland
done
clear
B)
salivary gland
done
clear
C)
cephalothorax
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 152) Cellulose is a polymer of
A)
\[\text{ }\!\!\alpha\!\!\text{ -}\]glucose
done
clear
B)
\[\text{ }\!\!\alpha\!\!\text{ -}\]fructose
done
clear
C)
\[\text{ }\!\!\beta\!\!\text{ -}\]glucose
done
clear
D)
\[\text{ }\!\!\beta\!\!\text{ -}\]fructose
done
clear
View Answer play_arrow
question_answer 153) The point in eye of mammals from which optic nerves and blood vessels leaves the eye ball is
A)
yellow spot
done
clear
B)
blind spot
done
clear
C)
pars optica
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 154) Which one is a neem product used as insect repellent?
A)
Azadirachtin
done
clear
B)
Rotenon
done
clear
C)
Parathione
done
clear
D)
Endrin
done
clear
View Answer play_arrow
question_answer 155) Disease associated with secretion of toxin is
A)
tetanus
done
clear
B)
TB
done
clear
C)
food poisoning
done
clear
D)
AIDS
done
clear
View Answer play_arrow
question_answer 156) Cork tissue arises from
A)
periderm
done
clear
B)
phellogen
done
clear
C)
pelloderm
done
clear
D)
phellum
done
clear
View Answer play_arrow
question_answer 157) An autoimmune disease is
A)
AIDS
done
clear
B)
haemophilia
done
clear
C)
allergy
done
clear
D)
myasthenia gravis
done
clear
View Answer play_arrow
question_answer 158) Cerebral hemispheres of rat are connected by
A)
corpus luteum
done
clear
B)
corpus callosum
done
clear
C)
corpus albicans
done
clear
D)
corpus spongiosum
done
clear
View Answer play_arrow
question_answer 159) The polysaccharide portion of a proteoglycan present in the matrix of cartilage is known as
A)
ossein
done
clear
B)
cartilin
done
clear
C)
casein
done
clear
D)
chondroitin
done
clear
View Answer play_arrow
question_answer 160) The activity of sucdnate dehydrogenase is inhibited by
A)
pyruvate
done
clear
B)
glycolate
done
clear
C)
melonate
done
clear
D)
phosphoglycerate
done
clear
View Answer play_arrow
question_answer 161) The nucleolus is the site of formation of
A)
spindle fibres
done
clear
B)
chromosomes
done
clear
C)
ribosomes
done
clear
D)
peroxysomes
done
clear
View Answer play_arrow
question_answer 162) Critical state of disease is called
A)
acme
done
clear
B)
acromion
done
clear
C)
acholin
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 163) The total number of cells in the human body is at least
A)
\[100\times {{10}^{-12}}\]
done
clear
B)
\[{{10}^{6}}\]
done
clear
C)
\[{{10}^{9}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 164) Thread like protoplasmic projection on the free surface of absorptive cells (such as intestinal cells) are called
A)
plasmodesmata
done
clear
B)
microfilaments
done
clear
C)
cilia
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 165) Deposition of uric acid crystals within the synovial joint causes
A)
osteoarthritis
done
clear
B)
rheumatoid arthritis
done
clear
C)
gout
done
clear
D)
paralysis
done
clear
View Answer play_arrow
question_answer 166) Dahlia and asparagus possess
A)
stilt roots
done
clear
B)
fusiform roots
done
clear
C)
tuberous roots
done
clear
D)
fasciculated roots
done
clear
View Answer play_arrow
question_answer 167) The distance between two base pairs in DNA is
A)
34 \[{{A}^{o}}\]
done
clear
B)
3.4 \[{{A}^{o}}\]
done
clear
C)
0.34 \[{{A}^{o}}\]
done
clear
D)
20 \[{{A}^{o}}\]
done
clear
View Answer play_arrow
question_answer 168) Five gram moles of glucose on complete oxidation releases
A)
3430 kcal of energy
done
clear
B)
343 kcal of energy
done
clear
C)
2020 kcal of energy
done
clear
D)
430 kcal of energy
done
clear
View Answer play_arrow
question_answer 169) Hybridoma is a biotechnique which involves fusion of
A)
B-cell with T-cell
done
clear
B)
T-cell with spleen cell
done
clear
C)
spleen cell with myeloma cell
done
clear
D)
myeloma cell with B-cell
done
clear
View Answer play_arrow
question_answer 170) The quickest method of plant breeding is
A)
introduction
done
clear
B)
selection
done
clear
C)
hybridization
done
clear
D)
mutation breeding
done
clear
View Answer play_arrow
question_answer 171) X- chromosomes of female in a sex- linked inheritance case can be passed on to
A)
only female progeny
done
clear
B)
only male progeny
done
clear
C)
only in grand daughter
done
clear
D)
male and female progeny
done
clear
View Answer play_arrow
question_answer 172) Spadix is an inflorescene found only in
A)
monocots
done
clear
B)
dicots
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 173) Botryoidal tissue is found in
A)
Hirudinea
done
clear
B)
Polychaeta
done
clear
C)
Oligochaeta
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 174) Pollination by bats is called
A)
anemophily
done
clear
B)
hydrophily
done
clear
C)
ornithophily
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 175) Schufmers granules are found in
A)
sporozoit
done
clear
B)
trophozoite
done
clear
C)
merozoite
done
clear
D)
schizont
done
clear
View Answer play_arrow
question_answer 176) Which zone of a lake has photosynthetic organsim?
A)
Profundal zone
done
clear
B)
Littoral zone
done
clear
C)
Limmetic zone
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 177) Interferon- \[\beta \] is also termed as
A)
immune interferon
done
clear
B)
fibroblast interferon
done
clear
C)
leucocyte interferon
done
clear
D)
anti-immune interferon
done
clear
View Answer play_arrow
question_answer 178) Terai forest is
A)
tropical forest
done
clear
B)
coniferous forest
done
clear
C)
deciduous forest
done
clear
D)
temperate deciduous forest
done
clear
View Answer play_arrow
question_answer 179) Waggle dance in honeybees tells about
A)
direction of food source
done
clear
B)
distance of food source
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 180) Which of the following has the highest pH?
A)
Gastric juice
done
clear
B)
Bile
done
clear
C)
Pancreatic juice
done
clear
D)
Secretions of the intestinal glands
done
clear
View Answer play_arrow
question_answer 181) A coordinated unit which controls the genetic expression is known as
A)
cistron
done
clear
B)
recon
done
clear
C)
operon
done
clear
D)
muton
done
clear
View Answer play_arrow
question_answer 182) Ultrasound is produced by
A)
lead bead
done
clear
B)
lead zirconate
done
clear
C)
lithium chloride
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 183) The pioneer country in the production of fuel-alcohol is
A)
Saudi Arabia
done
clear
B)
Iran, Iraq
done
clear
C)
Brazil
done
clear
D)
Japan
done
clear
View Answer play_arrow
question_answer 184) Who stated that human population grows geometrically?
A)
Malthus
done
clear
B)
Darwin
done
clear
C)
Cannon
done
clear
D)
Lamarck
done
clear
View Answer play_arrow
question_answer 185) A disease caused by eating fish contaminated by industrial waste containing mercury compounds is known as
A)
Brights disease
done
clear
B)
Minamata disease
done
clear
C)
Hashimotos disease
done
clear
D)
Osteosclerosis
done
clear
View Answer play_arrow
question_answer 186) A cybrid is hybrid carrying
A)
genomes and cytoplasms of two different plants
done
clear
B)
cytoplasms of two different plants cytoplasms of two different plants but
done
clear
C)
genome of one plant
done
clear
D)
genomes of two different plants
done
clear
View Answer play_arrow
question_answer 187) The "witches broom" is caused by a
A)
virus
done
clear
B)
mycoplasma
done
clear
C)
bacterium
done
clear
D)
fungus
done
clear
View Answer play_arrow
question_answer 188) Leaf in young condition in fern is called
A)
scale leaf
done
clear
B)
sporophyll
done
clear
C)
circinate ptyris
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 189) What is common in photosynthesis and respiration?
A)
Light energy
done
clear
B)
\[NADP{{H}_{2}}\]
done
clear
C)
Cytochromes
done
clear
D)
\[NAD{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 190) The disease caused by virus which is 42 nm in size and contians double stranded DNA is
A)
Hepatitis A
done
clear
B)
AIDS
done
clear
C)
Hepatitis B
done
clear
D)
Leprosy
done
clear
View Answer play_arrow
question_answer 191) Smell of urine is due to the
A)
urochrome
done
clear
B)
urinode
done
clear
C)
urea
done
clear
D)
melanin
done
clear
View Answer play_arrow
question_answer 192) Hormone which is responsible for contraction of uterus is
A)
vasopressin
done
clear
B)
oxytocin
done
clear
C)
thyrotropin
done
clear
D)
gonadotropin
done
clear
View Answer play_arrow
question_answer 193) The existence of two types of leaves in the same plant is called
A)
phyllody
done
clear
B)
phylloclade
done
clear
C)
heterophylly
done
clear
D)
heterosis
done
clear
View Answer play_arrow
question_answer 194) Sertoli cells are involved in
A)
excretion
done
clear
B)
nutrition of sperms
done
clear
C)
respiration
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 195) The name given to the fossil hominid of Shivalik hills in India was
A)
Ramapithecus
done
clear
B)
Sinanthropus
done
clear
C)
Pithecanthropus
done
clear
D)
Australopithecus
done
clear
View Answer play_arrow
question_answer 196) Empty glumes are
A)
petals
done
clear
B)
bracts
done
clear
C)
anthers
done
clear
D)
carpels
done
clear
View Answer play_arrow
question_answer 197) Hexose monophosphate pathway takes place in
A)
ER
done
clear
B)
cristae
done
clear
C)
cytoplasm
done
clear
D)
mitochondrial matrix
done
clear
View Answer play_arrow
question_answer 198) CFCL is situated at
A)
Delhi
done
clear
B)
Faridabad
done
clear
C)
Mumbai
done
clear
D)
Amritsar
done
clear
View Answer play_arrow
question_answer 199) Congression is a phenomenon of
A)
movement of sister chromatids towards the poles
done
clear
B)
pairing of homologous chromosomes
done
clear
C)
separation of paired chromosomes
done
clear
D)
bringing the chromosomes on equator of spindle apparatus
done
clear
View Answer play_arrow
question_answer 200) Kappa particles are
A)
protozoans parasite whose multipli- cation is controlled by host metabolites
done
clear
B)
viral particles capable of self perpetuation in host cytoplasm
done
clear
C)
endosymbiont representing gram negative bacteria species
done
clear
D)
sub microscopic granules formed by the folding of naked DNA
done
clear
View Answer play_arrow