question_answer 1) An electron moves through a small distance in a uniform electric field. The magnitude of electric field is \[2\times {{10}^{4}}N{{C}^{-1}}.\]Now, if the direction of field is reversed keeping the magnitude same and a proton moves through the same distance, then which of the following options is correct?
A)
The time of fall will be more in case of electron
done
clear
B)
The time of fall will be more in case of proton
done
clear
C)
The time of fall will be same in both cases
done
clear
D)
The time of fall will be independent of charge
done
clear
View Answer play_arrow
question_answer 2) The length of the wire 15 doubled, Its conductance will
A)
remain unchanged
done
clear
B)
be halved
done
clear
C)
be doubled
done
clear
D)
be quadrupled
done
clear
View Answer play_arrow
question_answer 3) The path of an electron in a uniform magnetic field may be
A)
circular but not helical
done
clear
B)
helical but not circular
done
clear
C)
neither helical nor circular
done
clear
D)
either helical or circular
done
clear
View Answer play_arrow
question_answer 4) A magnetic needle is placed on a cork floating in a still lake in the northern hemisphere. Does the needle together with the cork move towards the north of the lake?
A)
Yes
done
clear
B)
No
done
clear
C)
May or may not move
done
clear
D)
Nothing can be said
done
clear
View Answer play_arrow
question_answer 5) Use of eddy currents is done in the following except
A)
moving coil galvanometer
done
clear
B)
electric brakes
done
clear
C)
induction motor
done
clear
D)
dynamo
done
clear
View Answer play_arrow
question_answer 6) When air is replaced by a dielectric medium of constant K. The maximum force of attraction between two charges separated by a distance
A)
increases K-1 times
done
clear
B)
increases K times
done
clear
C)
decreases K times
done
clear
D)
remains constant
done
clear
View Answer play_arrow
question_answer 7) If the tension on a wire is removed at once, then
A)
it will break
done
clear
B)
its temperature wilt reduce
done
clear
C)
there will be no change in its temperature
done
clear
D)
its temperature increases
done
clear
View Answer play_arrow
question_answer 8) A capillary tube is attached horizontally to a constant head arrangement. If the radius of the capillary tube is increased by 10%, then the rate of flow of liquid will change nearly by
A)
+10 %
done
clear
B)
+46 %
done
clear
C)
-10 %
done
clear
D)
-40 %
done
clear
View Answer play_arrow
question_answer 9) Two equal drops of water are falling through air with a steady velocity v. If the drops coalesced what will be the new velocity?
A)
\[{{\left( 2 \right)}^{\frac{1}{3}}}v\]
done
clear
B)
\[{{\left( 2 \right)}^{\frac{3}{2}}}v\]
done
clear
C)
\[{{\left( 2 \right)}^{\frac{2}{3}}}v\]
done
clear
D)
\[{{\left( 2 \right)}^{\frac{1}{4}}}v\]
done
clear
View Answer play_arrow
question_answer 10) A good lubricant should have
A)
high viscosity
done
clear
B)
low viscosity
done
clear
C)
moderate viscosity
done
clear
D)
high density
done
clear
View Answer play_arrow
question_answer 11) Two friends A and B are waiting for another friend for tea. A took the tea in a cup and mixed the cold milk and then waits. B took the tea in the cup and then mixed the cold milk when the friend comes. Then the tea will be hotter in the cup of
A)
A
done
clear
B)
B
done
clear
C)
tea will be equally hot in both cups
done
clear
D)
friend's cup
done
clear
View Answer play_arrow
question_answer 12) The internal energy of an ideal gas increases during an isothermal process when the gas Is
A)
expanded by adding more molecules to It
done
clear
B)
expanded by adding more heat to it
done
clear
C)
expanded against zero pressure
done
clear
D)
compressed by doing work on it
done
clear
View Answer play_arrow
question_answer 13) At what temperature, the mean kinetic energy of C > 2 will be the same as that of Ha molecules at \[- 73{}^\circ C\]?
A)
\[{{127}^{0}}C\]
done
clear
B)
\[527{}^\circ C\]
done
clear
C)
\[-73{}^\circ C\]
done
clear
D)
\[-137{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 14) The fundamental frequency of a closed pipe is 220 Hz. If - of the pipe is filled with water, the frequency of the first overtone of the pipe now is
A)
220 Hz
done
clear
B)
440 Hz
done
clear
C)
880 Hz
done
clear
D)
1760 Hz
done
clear
View Answer play_arrow
question_answer 15) The intensity of sound increases at night due to
A)
increase in density of air
done
clear
B)
decrease in density of air
done
clear
C)
low temperature
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 16) If the energy of a hydrogen atom in nth orbit is En, then energy in the nth orbit of a singly ionized helium atom will be
A)
\[4{{E}_{n}}\]
done
clear
B)
\[{{E}_{n}}/4\]
done
clear
C)
\[2{{E}_{n}}\]
done
clear
D)
\[{{E}_{n}}/2\]
done
clear
View Answer play_arrow
question_answer 17) The fission of \[^{235}U\]can be triggered by the absorption of a slow neutron by a nucleus. Similarly a slow proton can also be used. This statement is
A)
correct
done
clear
B)
wrong
done
clear
C)
information is insufficient
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 18) Would there be any advantage to add n-type or n - type impurities to copper?
A)
Yes
done
clear
B)
No
done
clear
C)
Maybe
done
clear
D)
Information is insufficient
done
clear
View Answer play_arrow
question_answer 19) In semiconductor the concentrations of electrons and holes are \[8\times {{10}^{18}}{{m}^{3}}\]and \[5\times {{10}^{18}}/{{m}^{3}}\]respectively. If the mobilities of electrons and holes are 2.3 m2 /Vs it and 0.01 m2 /Vs respectively, then semiconductor is
A)
n-type and its resistivity is 0.34 \[\Omega \]-m
done
clear
B)
p-type and its resistivity is 0.034 \[\Omega \]-m
done
clear
C)
n-type and its resistivity is 0.034 \[\Omega \]-m
done
clear
D)
p-type and its resistivity is 3.4 \[\Omega \]-m
done
clear
View Answer play_arrow
question_answer 20) When a sample of solid lithium is placed in a flask of hydrogen gas then following reaction happened \[{{^{1}}_{1}}H{{+}_{3}}L{{i}^{7}}{{\xrightarrow[{}]{{}}}_{2}}H{{e}^{4}}{{+}_{2}}H{{e}^{4}}\] This statement is
A)
true
done
clear
B)
false
done
clear
C)
may be true at a particular pressure
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 21) A capacitor of 20 \[\mu \]F charged upto 500 V is connected in parallel with another capacitor of 10 \[\mu \]F which is charged upto 200V. The common potential is
A)
250V
done
clear
B)
300V
done
clear
C)
400V
done
clear
D)
600V
done
clear
View Answer play_arrow
question_answer 22) The slope of the velocity- time graph for retarded motion is
A)
positive
done
clear
B)
negative
done
clear
C)
zero
done
clear
D)
can be + ve , - ve or zero
done
clear
View Answer play_arrow
question_answer 23) A man throws balls with the same speed vertically upwards one after the other at an interval of 2s. What should be the speed of the throw so that more than two balls are in the sly at any time? (Given g = 9.8 m/s2)
A)
Any speed less than 19.6 m/s
done
clear
B)
Only with speed 19.6 m/s
done
clear
C)
More than 19.6 m/s
done
clear
D)
At least 9.8 m/s
done
clear
View Answer play_arrow
question_answer 24) The driver of a car travelling at velocity v suddenly sees a broad wall in front of him at a distance d. He should
A)
brake sharply
done
clear
B)
turn sharply
done
clear
C)
(a) and (b) both
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 25) A 60 kg man stands on a spring scale in the lift. At some instant he finds, scale reading has changed from 60 kg to 50 kg for a while and then comes back to the original mark. What should we conclude?
A)
The lift was in constant motion upwards
done
clear
B)
The lift was in constant modal downwards
done
clear
C)
The lift while in constant motion upwards, is stopped suddenly
done
clear
D)
The lift while in constant motto downwards, is suddenly stopped
done
clear
View Answer play_arrow
question_answer 26) The limiting friction is
A)
always greater than the dynamic friction
done
clear
B)
always less than the dynamic friction
done
clear
C)
equal to the dynamic friction
done
clear
D)
sometimes greater and sometimes less than the dynamic friction
done
clear
View Answer play_arrow
question_answer 27) A free \[\alpha \]-particle and a free proton, which are separated by a distance of 10-10 m are released. The KE of \[\alpha \]-particle when at infinite separation is
A)
\[46\times {{10}^{-19}}J\]
done
clear
B)
\[23\times {{10}^{-19}}J\]
done
clear
C)
\[36.8\times {{10}^{-19}}J\]
done
clear
D)
\[9.2\times {{10}^{-19}}J\]
done
clear
View Answer play_arrow
question_answer 28) The modon of planets in the solar system is an example of conservation of
A)
mass
done
clear
B)
momentum
done
clear
C)
angular momentum
done
clear
D)
kinetic energy
done
clear
View Answer play_arrow
question_answer 29) The ratio of the dimensions of Planck's constant and that of the moment of inertia is the dimension of
A)
frequency
done
clear
B)
velocity
done
clear
C)
angular momentum
done
clear
D)
time
done
clear
View Answer play_arrow
question_answer 30) The time period of a simple pendulum in a stationary train is T. The time period of a mass attached to a spring is also T. The train accelerates at the rate 5 m/s. If the new time periods of the pendulum and spring be Tp and T, respectively, then
A)
Tp = T1
done
clear
B)
Tp > Ts
done
clear
C)
Tp < Ts
done
clear
D)
cannot be predicted
done
clear
View Answer play_arrow
question_answer 31) A particle starting from the origin (0, 0) moves in a straight line in the (x, y) plane. Its coordinates at a later time are (-/3, 3). The path of the particle makes with the x-axis an angle of
A)
\[30{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[0{}^\circ \]
done
clear
View Answer play_arrow
question_answer 32) Two satellites of earth, S1 and S2 are moving in the same orbit. The mass of S1 is four times the mass of S2. Which one of the following statements is true?
A)
The time period of S1 is four times that of S2
done
clear
B)
The potential energies of earth and satellite in the two cases are equal
done
clear
C)
S1 and S2 are moving with the same speed
done
clear
D)
The kinetic energies of the two satellites are equal
done
clear
View Answer play_arrow
question_answer 33) If the length of tube is less and cannot accommodate the maximum rise of liquid then
A)
liquid will form fountain
done
clear
B)
liquid will not rise
done
clear
C)
the meniscus will adjust itself so that the water does not spill
done
clear
D)
none of the above
done
clear
View Answer play_arrow
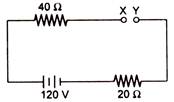
question_answer 34)
In the circuit shown in the figure the potential difference between X and Y will be
A)
(a) zero
done
clear
B)
20 V
done
clear
C)
60 V
done
clear
D)
120 V
done
clear
View Answer play_arrow
question_answer 35) In which of the following systems will the radius of the first orbit (n = 1) be minimum?
A)
Deuterium atom
done
clear
B)
Hydrogen atom
done
clear
C)
Doubly ionized lithium
done
clear
D)
Singly ionized helium
done
clear
View Answer play_arrow
question_answer 36) In hydrogen atom spectrum, frequency of \[2.7\times {{10}^{15}}Hz\]of electromagnetic wave is emitted when transmission takes place from to 1. If it moves from 3 to 1, the frequency emitted will be
A)
\[3.2\times {{10}^{15}}Hz\]
done
clear
B)
\[32\times {{10}^{15}}Hz\]
done
clear
C)
\[1.6\times {{10}^{15}}Hz\]
done
clear
D)
\[16\times {{10}^{15}}Hz\]
done
clear
View Answer play_arrow
question_answer 37) In case of a forced vibration, the resonance wave becomes very sharp when the
A)
applied periodic force is small
done
clear
B)
quality factor is small
done
clear
C)
damping force is small
done
clear
D)
restoring force is small
done
clear
View Answer play_arrow
question_answer 38) Total internal reflection takes place
A)
when a ray moves from denser to rarer and incident angle is greater than critical angle
done
clear
B)
when a ray moves from rarer to denser and incident angle is less than critical angle
done
clear
C)
when a ray moves from rarer to denser and incident angle is equal to critical angle
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 39) The density of newly discovered planet is twice that of earth. The acceleration due to gravity at the surface of the planet is equal to that at the surface of the earth. If the radius of the earth is R, the radius of the planet would be
A)
2 R
done
clear
B)
4 R
done
clear
C)
\[\frac{1}{4}\] R
done
clear
D)
\[\frac{1}{2}\] R
done
clear
View Answer play_arrow
question_answer 40) In a p- n junction photo cell, the value of the photo-electro motive force produced by monochromatic light is proportional to
A)
the barrier voltage at the p- n junction
done
clear
B)
the intensity of the light falling on the cell
done
clear
C)
the frequency of the light falling on the cell
done
clear
D)
the voltage applied at the p- n junction
done
clear
View Answer play_arrow
question_answer 41) A child is swinging a swing. Minimum and maximum heights of swing from earth's surface are 0.75 m and 2 m respectively. The maximum velocity of this swing is
A)
5 m/s
done
clear
B)
10 m/s
done
clear
C)
15 m/s
done
clear
D)
20 in/s
done
clear
View Answer play_arrow
question_answer 42) Ozone layer in atmosphere is useful, because it
A)
stops ultraviolet radiation
done
clear
B)
absorbs polluent gases
done
clear
C)
stops green house effect
done
clear
D)
stops increase in temperature of atmosphere
done
clear
View Answer play_arrow
question_answer 43) In which type of material the magnetic susceptibility does not depend on temperature?
A)
Diamagnetic
done
clear
B)
Paramagnetic
done
clear
C)
Ferromagnetic
done
clear
D)
Ferrite
done
clear
View Answer play_arrow
question_answer 44) In compound X(n, \[\alpha \]) \[\to \] \[_{3}L{{i}^{7}},\], the element X is
A)
2He4
done
clear
B)
5B10
done
clear
C)
5B9
done
clear
D)
4Be11
done
clear
View Answer play_arrow
question_answer 45) A car runs at a constant speed on a circular track of radius 100 m, taking 62.8 s for every circular lap- The average velocity and average speed for each circular lap respectively is
A)
0, 0
done
clear
B)
0, 10 m/s
done
clear
C)
10 m/s, 10 m/s
done
clear
D)
10 m/s, 0
done
clear
View Answer play_arrow
question_answer 46) A convex lens is dipped in a liquid whose refractive index is equal to the refractive index of the lens. Then its focal length will
A)
become small, but non- zero
done
clear
B)
remain unchanged
done
clear
C)
become zero
done
clear
D)
become infinite
done
clear
View Answer play_arrow
question_answer 47) A stationary particle explodes into two particles of masses m1 and m2 which move in opposite directions with velocities v1 and v2. The ratio of their kinetic energies E1 /E2 is
A)
1
done
clear
B)
\[{{m}_{1}}{{v}_{2}}/{{m}_{2}}{{v}_{1}}\]
done
clear
C)
\[{{m}_{2}}/{{m}_{1}}\]
done
clear
D)
\[{{m}_{1}}/{{m}_{2}}\]
done
clear
View Answer play_arrow
question_answer 48) Diameter of human eye lens is 2 mm. What will be the minimum distance between two points to resolve them, which are situated at a distance of 50 m from eye? The wavelength of light is 5000 A.
A)
2.32 m
done
clear
B)
4.28 mm
done
clear
C)
1.25 cm
done
clear
D)
12.48 cm
done
clear
View Answer play_arrow
question_answer 49) A sample of gas expands from volume V1 to V2. The amount of work done by the gas is greatest when the expansion is
A)
adiabatic
done
clear
B)
isobaric
done
clear
C)
isothermal
done
clear
D)
equal in all above cases
done
clear
View Answer play_arrow
question_answer 50) The degrees of freedom of a molecule of a triatomic gas are
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 51) If the wavelength of photon is \[2.2\times {{10}^{-11}}m,h=6.6\times {{10}^{-34}}J-s,\] then momentum of photon is
A)
\[3\times {{10}^{-23}}kg\,m{{s}^{-1}}\]
done
clear
B)
\[3.33\times {{10}^{22}}\,kg\,m{{s}^{-1}}\]
done
clear
C)
\[1.452\times {{10}^{-44}}\,kg\,m{{s}^{-1}}\]
done
clear
D)
\[6.89\times {{10}^{43}}\,kg\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 52) In a given atom no two electrons can have the same values for all the four quantum numbers. This is called
A)
Hund's rule
done
clear
B)
Aufbau's principle
done
clear
C)
Uncertainty principle
done
clear
D)
Fault's exclusion principle
done
clear
View Answer play_arrow
question_answer 53) Which of the following particle is emitted in the reaction? \[_{13}A{{l}^{27}}{{+}_{2}}H{{e}^{4}}{{\xrightarrow{{}}}_{14}}{{P}^{30}}+...\]
A)
\[_{0}{{n}^{1}}\]
done
clear
B)
\[_{-1}{{e}^{0}}\]
done
clear
C)
\[_{1}{{H}^{1}}\]
done
clear
D)
\[_{1}{{H}^{2}}\]
done
clear
View Answer play_arrow
question_answer 54) For reaction,\[2NOCl(g)2NO(g)+C{{l}_{2}}(g),\]\[{{K}_{c}}\]at \[427{{\,}^{o}}C\]is \[3\times {{10}^{-6}}L\,mo{{l}^{-1}}.\] The value of \[{{K}_{p}}\]is nearly
A)
\[7.5\times {{10}^{-5}}\]
done
clear
B)
\[2.5\times {{10}^{-5}}\]
done
clear
C)
\[2.5\times {{10}^{-4}}\]
done
clear
D)
\[1.72\times {{10}^{-4}}\]
done
clear
View Answer play_arrow
question_answer 55) What is the order of a reaction which has a rate expression \[rate=k{{(A)}^{3/2}}{{(B)}^{-1}}\]?
A)
3/2
done
clear
B)
1/2
done
clear
C)
0
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 56) Which compound is highest covalent?
A)
LiCI
done
clear
B)
LiF
done
clear
C)
LiBr
done
clear
D)
Lil
done
clear
View Answer play_arrow
question_answer 57) Shape of \[\text{Xe}{{\text{F}}_{\text{4}}}\]molecule is
A)
linear
done
clear
B)
pyramidal
done
clear
C)
tetrahedral
done
clear
D)
square planar
done
clear
View Answer play_arrow
question_answer 58) An aqueous solution of glucose is 10% in strength. The volume in which 1 g mole of it is dissolved will be
A)
18 L
done
clear
B)
9L
done
clear
C)
0.9 L
done
clear
D)
1.8 L
done
clear
View Answer play_arrow
question_answer 59) Which one of the following elements has the highest ionisation energy?
A)
\[[Ne]3{{s}^{2}}3{{p}^{1}}\]
done
clear
B)
\[[Ne]3{{s}^{2}},3{{p}^{2}}\]
done
clear
C)
\[[Ne]3{{s}^{2}}3{{p}^{3}}\]
done
clear
D)
\[[Ar]3{{d}^{10}}\,4{{s}^{2}}4{{p}^{2}}\]
done
clear
View Answer play_arrow
question_answer 60) Important ore of zinc is
A)
calamine
done
clear
B)
cryolite
done
clear
C)
cassiterite
done
clear
D)
malachite
done
clear
View Answer play_arrow
question_answer 61) A metal which is refined by polling is
A)
sodium
done
clear
B)
blister copper
done
clear
C)
zinc
done
clear
D)
silver
done
clear
View Answer play_arrow
question_answer 62) Nessler's reagent is
A)
\[KHg{{I}_{4}}\]
done
clear
B)
\[{{K}_{2}}Hg{{I}_{4}}+N{{H}_{4}}OH\]
done
clear
C)
\[[Ne]3{{s}^{2}}3{{p}^{3}}\]
done
clear
D)
\[KHg{{I}_{4}}+N{{H}_{4}}OH\]
done
clear
View Answer play_arrow
question_answer 63) A brown ring appears in the test for
A)
nitrate
done
clear
B)
nitrite
done
clear
C)
bromide
done
clear
D)
iron
done
clear
View Answer play_arrow
question_answer 64) Potassium is kept in
A)
alcohol
done
clear
B)
water
done
clear
C)
kerosene
done
clear
D)
liquid ammonia
done
clear
View Answer play_arrow
question_answer 65) Glauber salt is
A)
\[MgS{{O}_{4}}.{{H}_{2}}O\]
done
clear
B)
\[CuS{{O}_{4}}.5{{H}_{2}}O\]
done
clear
C)
\[FeS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
D)
\[N{{a}_{2}}S{{O}_{4}}.10{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 66) The liquid field metal expanding on solidification is
A)
Ga
done
clear
B)
Al
done
clear
C)
Zn
done
clear
D)
Cu
done
clear
View Answer play_arrow
question_answer 67) Which of the following oxides of nitrogen is the anhydride of nitrous acid?
A)
NO
done
clear
B)
\[{{N}_{2}}{{O}_{3}}\]
done
clear
C)
\[{{N}_{2}}{{O}_{4}}\]
done
clear
D)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 68) Which one below is a pseudohalide?
A)
\[C{{N}^{-}}\]
done
clear
B)
\[ICl\]
done
clear
C)
\[I{{F}_{5}}\]
done
clear
D)
\[I_{3}^{-}\]
done
clear
View Answer play_arrow
question_answer 69) Which of the following gas mixture is used by the divers inside the sea?
A)
\[{{O}_{2}}+He\]
done
clear
B)
\[{{O}_{2}}+{{X}_{e}}\]
done
clear
C)
\[{{O}_{2}}+Ar\]
done
clear
D)
\[{{O}_{2}}+{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 70) In which of the following metallic bond is strongest?
A)
Fe
done
clear
B)
Sc
done
clear
C)
V
done
clear
D)
Cr
done
clear
View Answer play_arrow
question_answer 71) Acidified potassium permanganate solution is decolourised by
A)
bleaching powder
done
clear
B)
white vitripl
done
clear
C)
Monies salt
done
clear
D)
microcosmic salt
done
clear
View Answer play_arrow
question_answer 72) The pressure and temperature of \[4\text{ }d{{m}^{3}}\] of carbon dioxide gas are doubled. Then volume of carbon dioxide gas would be
A)
\[2\,d{{m}^{3}}\]
done
clear
B)
\[3\,d{{m}^{3}}\]
done
clear
C)
\[4\,d{{m}^{3}}\]
done
clear
D)
\[8\,d{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 73) An fee unit cell of aluminium contains the equivalent of how many atoms?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 74) Hess law deals with
A)
changes in heat of reaction
done
clear
B)
rate of reaction
done
clear
C)
equilibrium constant
done
clear
D)
influence of pressure on volume of gas
done
clear
View Answer play_arrow
question_answer 75) By adding 20 mL 0.1 N HCl to 20 mL 0.001 N KOH, the pH of the obtained solution will be
A)
2
done
clear
B)
1.3
done
clear
C)
0
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 76) Hydrolysis of sodium acetate will give
A)
acidic solution
done
clear
B)
basic solution
done
clear
C)
neutral solution
done
clear
D)
normal solution
done
clear
View Answer play_arrow
question_answer 77) Which of the following is not a non- electrolyte?
A)
Acetic acid
done
clear
B)
Glucose
done
clear
C)
Ethanol
done
clear
D)
Urea
done
clear
View Answer play_arrow
question_answer 78) When lead storage battery is charged
A)
lead dioxide dissolves
done
clear
B)
sulphuric acid is regenerated
done
clear
C)
the lead electrode becomes coated with lead sulphate
done
clear
D)
the amount of sulphuric acid decrease
done
clear
View Answer play_arrow
question_answer 79) The oxidation state of S in \[{{H}_{2}}{{S}_{2}}{{O}_{8}}\]is
A)
+2
done
clear
B)
+4
done
clear
C)
+ 6
done
clear
D)
+7
done
clear
View Answer play_arrow
question_answer 80) The equation for Freundlich adsorption isotherm is
A)
\[\frac{x}{m}=k{{p}^{1/n}}\]
done
clear
B)
\[x=mk{{p}^{1/n}}\]
done
clear
C)
\[\frac{x}{m}=k{{p}^{-n}}\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 81) Example of intrinsic colloid is
A)
glue
done
clear
B)
sulphur
done
clear
C)
Fe
done
clear
D)
\[A{{s}_{2}}{{S}_{3}}\]
done
clear
View Answer play_arrow
question_answer 82) Which of the following free radicals is the most stable?
A)
Primary
done
clear
B)
Methyl
done
clear
C)
Secondary
done
clear
D)
Tertiary
done
clear
View Answer play_arrow
question_answer 83) Which of the following will have a most isomer also?
A)
2, 3-dichloropentane
done
clear
B)
2, 3-dichlorobutane
done
clear
C)
2-chlorobutane
done
clear
D)
2-hydroxy propanoic acid
done
clear
View Answer play_arrow
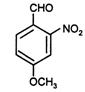
question_answer 84)
The correct IUPAC name of the compound
A)
4 methoxy-2- nitrobenzaldehyde
done
clear
B)
4- formyl -3 nitro anisole
done
clear
C)
4- methoxy -6- nitro benzladehyde
done
clear
D)
2- formyi -5- methoxy nitrobenzene
done
clear
View Answer play_arrow
question_answer 85) Which of the following has highest knocking property?
A)
Aromatic hydrocarbons
done
clear
B)
Olefins
done
clear
C)
Branched chain paraffins
done
clear
D)
Straight chain paraffins
done
clear
View Answer play_arrow
question_answer 86) Which of the following is the most stable alkene?
A)
\[{{R}_{2}}C=C{{R}_{2}}\]
done
clear
B)
\[RCH=CHR\]
done
clear
C)
\[RCH=C{{H}_{2}}\]
done
clear
D)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 87) In which alkyl halide, \[{{S}_{N}}2\]mechanism is favoured maximum
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CHCl\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}C-Cl\]
done
clear
View Answer play_arrow
question_answer 88)
In the following reaction 'A is
A)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}CHO\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}OH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}CHO\]
done
clear
View Answer play_arrow
question_answer 89) From Williamson's synthesis preparation of which of the following is possible?
A)
Only symmetrical ethers
done
clear
B)
Only asymmetrical ethers
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 90)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 91) Dry heating of calcium acetate gives
A)
acetaldehyde
done
clear
B)
ethane
done
clear
C)
acetic acid
done
clear
D)
acetone
done
clear
View Answer play_arrow
question_answer 92) Formaldehyde reacts with ammonia to give urotropine is
A)
\[{{(C{{H}_{2}})}_{6}}{{N}_{4}}\]
done
clear
B)
\[{{(C{{H}_{2}})}_{4}}{{N}_{3}}\]
done
clear
C)
\[{{(C{{H}_{2}})}_{6}}{{N}_{6}}\]
done
clear
D)
\[{{(C{{H}_{2}})}_{3}}{{N}_{3}}\]
done
clear
View Answer play_arrow
question_answer 93) The reagent which does not give acid chloride on treating with a carboxylic acid is
A)
\[PC{{l}_{5}}\]
done
clear
B)
\[C{{l}_{2}}\]
done
clear
C)
\[SOC{{l}_{2}}\]
done
clear
D)
\[PC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 94) Aniline react with acetaldehyde to form
A)
Schiffs base
done
clear
B)
carbyl amine
done
clear
C)
immine
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 95) Three dimensional molecules with cross links are formed in the case of a
A)
thermoplastic
done
clear
B)
thermosetting plastic
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 96) The reagent which forms crystalline osazone derivative when reacted with glucose, is
A)
Fehling solution
done
clear
B)
phenyl hydrazine
done
clear
C)
Benedict solution
done
clear
D)
hydroxyl amine
done
clear
View Answer play_arrow
question_answer 97) Insulin is
A)
an amino acid
done
clear
B)
a protein
done
clear
C)
a carbohydrate
done
clear
D)
a lipid
done
clear
View Answer play_arrow
question_answer 98) Which of the following polymer has ester linkage?
A)
Nylon-66
done
clear
B)
PVC
done
clear
C)
Terylene
done
clear
D)
SBR
done
clear
View Answer play_arrow
question_answer 99) Chloramine-T is a
A)
disinfectant
done
clear
B)
antiseptic
done
clear
C)
analgesic
done
clear
D)
antipyretic
done
clear
View Answer play_arrow
question_answer 100) Which one is disperse dye?
A)
Congo red
done
clear
B)
Alizarin
done
clear
C)
Celliton
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 101) Darwin?s finches provide excellent evidence in favour of organic evolution. These are related to which of the following evidences?
A)
Embryology
done
clear
B)
Palaeontology (or fossils)
done
clear
C)
Anatomy
done
clear
D)
Biogeography(or geographic distribution)
done
clear
View Answer play_arrow
question_answer 102) Relative Biological Effectiveness (RBE) usually refers to the damages caused by :
A)
low temperature
done
clear
B)
high temperature
done
clear
C)
radiation
done
clear
D)
pollution
done
clear
View Answer play_arrow
question_answer 103) Lysosomes are the reservoirs (store houses) of
A)
hydrolytic enzymes
done
clear
B)
secretory glycoproteins
done
clear
C)
RNA and protein
done
clear
D)
fats (or sugars or ATP)
done
clear
View Answer play_arrow
question_answer 104) The greatest biomass of autotrophs in the world?s oceans is that of
A)
benthic brown algae, coastal red algae and daphnids
done
clear
B)
benthic diatoms and marine viruses
done
clear
C)
sea grasses and slime molds
done
clear
D)
free-floating micro-algae, cyanobacteria and nanoplankton
done
clear
View Answer play_arrow
question_answer 105) For yielding one molecule of glucose the Calvin cycle turns
A)
two times
done
clear
B)
four times
done
clear
C)
six times
done
clear
D)
eight times
done
clear
View Answer play_arrow
question_answer 106) Industrial production of ethanol from starch is brought about by a certain species of
A)
Azotobacter
done
clear
B)
Lactobacillus
done
clear
C)
Saccharomyces
done
clear
D)
Penicillium
done
clear
View Answer play_arrow
question_answer 107) In most of the fungi, the food material is stored in the form of
A)
starch
done
clear
B)
glucose
done
clear
C)
sucrose
done
clear
D)
glycogen
done
clear
View Answer play_arrow
question_answer 108) Feed back inhibition of an enzymatic reaction is caused by
A)
substrate
done
clear
B)
enzyme
done
clear
C)
end product
done
clear
D)
rise in temperature
done
clear
View Answer play_arrow
question_answer 109) Geocarpic fruits are produced by :
A)
cannot
done
clear
B)
onion
done
clear
C)
ground nut
done
clear
D)
watermelon
done
clear
View Answer play_arrow
question_answer 110) The endangered largest living lemur Idriidri is inhabitant of
A)
Madagascar
done
clear
B)
Mauritius
done
clear
C)
Sri Lanka
done
clear
D)
India
done
clear
View Answer play_arrow
question_answer 111) Which one of the following is not a vestigial part in human?
A)
Coccyx
done
clear
B)
Finger nails
done
clear
C)
Third molar of each side in each jaw
done
clear
D)
Segmental muscles of abdomen
done
clear
View Answer play_arrow
question_answer 112) Small proteins produced by vertebrate cells naturally in response to viral infections and which inhibit multiplication of viruses are called
A)
immunoglobulin?s
done
clear
B)
inter ferons
done
clear
C)
antitoxins
done
clear
D)
lipoproteins
done
clear
View Answer play_arrow
question_answer 113) Black rust of wheat is caused by a member species of the genus
A)
Mucor
done
clear
B)
Rhizopus
done
clear
C)
Aspergillus
done
clear
D)
Puccinia
done
clear
View Answer play_arrow
question_answer 114) An institution where valuable plant material-likely to become irretrievably lost in the wild or in cultivation is preserved in a viable condition is known as
A)
genome
done
clear
B)
gene library
done
clear
C)
gene bank
done
clear
D)
herbarium
done
clear
View Answer play_arrow
question_answer 115) Among mammals, a significant role in the digestion of milk is played by
A)
rennin
done
clear
B)
invertase
done
clear
C)
amylase
done
clear
D)
intestinal bacteria
done
clear
View Answer play_arrow
question_answer 116) Which one of the following characters studied by Mendel in garden pea was found to be dominant ?
A)
Green seed colour
done
clear
B)
Terminal flower position
done
clear
C)
Green pod colour
done
clear
D)
Wrinkled seed
done
clear
View Answer play_arrow
question_answer 117) The replication of DNA is a pre- requisite for a eukaryotic cell to undergo division. During the cell cycle the DNA replicates in
A)
S phase
done
clear
B)
\[{{G}_{1}}\] phase
done
clear
C)
\[{{G}_{2}}\]phase
done
clear
D)
M phase
done
clear
View Answer play_arrow
question_answer 118) Which one of the following is correctly matched pair of the given secretion and its primary role in human physiology?
A)
Seburtise - Sexual attraction
done
clear
B)
Sweat - Thermoregulation
done
clear
C)
Saliva - Tasting food
done
clear
D)
Tears - Excretion of salts
done
clear
View Answer play_arrow
question_answer 119) The function of copper T is to prevent
A)
fertilization
done
clear
B)
egg maturation
done
clear
C)
ovulation
done
clear
D)
implantation of blastocyst
done
clear
View Answer play_arrow
question_answer 120) Plasmids are suitable vectors for gene cloning because
A)
these are small circular DNA molecules which can integrate with host chromosomal DNA
done
clear
B)
these are small circular DNA molecules with their own replication origin site
done
clear
C)
these can shuttle between prokaryotic and eukaryotic cells
done
clear
D)
these often carry antibiotic resistance genes
done
clear
View Answer play_arrow
question_answer 121) As per geological time scale, hominids evolved during
A)
Miocene
done
clear
B)
Pliocene
done
clear
C)
Pleistocene
done
clear
D)
Oligocene The family Hominids includes humans of today. These are the most intelligent of the hominoids. They are distinguished from the other families of hominoids in that they are bipedal i.e, they walk upright on two legs. Hominids appeared during Pliocene but modern human (Homo sapiens) are believed to have arisen about 150.000 years back during Pleistocene.
done
clear
View Answer play_arrow
question_answer 122) Which one of the following correctly matches a Sexually Transmitted Disease (STD) with its pathogen?
A)
AIDS ? Bacillus anthracis
done
clear
B)
Syphilis ? Treponemapallidum
done
clear
C)
Urethritis ? Entamoebagingivalis
done
clear
D)
Gonorrhoea ? Leishmaniadonovani
done
clear
View Answer play_arrow
question_answer 123) ATP is a
A)
nucleotide
done
clear
B)
nucleosome
done
clear
C)
purine base
done
clear
D)
nucleoside
done
clear
View Answer play_arrow
question_answer 124) Removal of apical (terminal) bud of a flowering plant (or pruning of a flowering plant) leads to
A)
formation of new apical buds.
done
clear
B)
formation of adventitious roots on the cut side
done
clear
C)
early flowering(or stopping of floral growth)
done
clear
D)
promotion of lateral branches
done
clear
View Answer play_arrow
question_answer 125) Coconut milk (coconut water) is widely used in tissue culture because it contains
A)
auxins
done
clear
B)
ethylene
done
clear
C)
cytokinin
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
question_answer 126) A research scholar once collected certain alga and found that its cells contained chlorophyll a, chlorophyll d and phycoerythrin. The alga must belong to
A)
Chlorophyceae
done
clear
B)
Rhodopyceae
done
clear
C)
Bacillariophyceae
done
clear
D)
Cyanophyceae
done
clear
View Answer play_arrow
question_answer 127) In a given plant, red colour (R) of fruits is dominant over white fruit (r); and tallness (T) is dominant over dwarfness (t). If a plant with genotype RRTt, is crossed with a plant of genotype rrtt what will be the percentage of tall plants with red fruits in the next generation?
A)
100%
done
clear
B)
25%
done
clear
C)
50%
done
clear
D)
75%
done
clear
View Answer play_arrow
question_answer 128) In an animal cell, protein synthesis takes place
A)
onlyon the ribosomes protein in the cytosol
done
clear
B)
only on the ribosomes attached to nuclear envelope and endoplasmic reticulum
done
clear
C)
on ribosomes present in the nucleolus as well as in cytoplasm
done
clear
D)
on ribosomes present in the cytosol as well as in the mitochondria
done
clear
View Answer play_arrow
question_answer 129) Anticodon is present on
A)
rRNA
done
clear
B)
fRNA
done
clear
C)
mtDNA
done
clear
D)
mRNA
done
clear
View Answer play_arrow
question_answer 130) Blastopore is the opening to the exterior of
A)
coelom
done
clear
B)
coelenteron
done
clear
C)
archentron
done
clear
D)
blastocoel
done
clear
View Answer play_arrow
question_answer 131) Fixation of one molecule of \[C{{O}_{2}}\]through Calvin cycle requires
A)
3 ATP and 3 \[\text{NADP}{{\text{H}}_{\text{2}}}\]molecules
done
clear
B)
3 ATP and 2 \[\text{NADP}{{\text{H}}_{\text{2}}}\]molecules
done
clear
C)
2 ATP and 1 \[\text{NADP}{{\text{H}}_{\text{2}}}\]molecule
done
clear
D)
1 ATP and 2 \[\text{NADP}{{\text{H}}_{\text{2}}}\]molecules
done
clear
View Answer play_arrow
question_answer 132) Movement of ions or molecules against the electrochemical gradient is called
A)
diffusion
done
clear
B)
pinocytosis
done
clear
C)
Brownian movement
done
clear
D)
active transport
done
clear
View Answer play_arrow
question_answer 133) The enteronephricnephridia of earthworms are mainly concerned with
A)
digestion
done
clear
B)
respiration
done
clear
C)
osmoregulation
done
clear
D)
excretion of nitrogenous wastes
done
clear
View Answer play_arrow
question_answer 134) Which one of the following is a skull bone?
A)
Coracoid
done
clear
B)
Arytaenoid
done
clear
C)
Atlas
done
clear
D)
Pterygoid
done
clear
View Answer play_arrow
question_answer 135) Which method out of the following renders the seed coat permeable to water so that embryo expansion is not physically retarded?
A)
Stratification
done
clear
B)
Denudation
done
clear
C)
Vernalisation
done
clear
D)
Scarification
done
clear
View Answer play_arrow
question_answer 136) Many scientists consider viruses as living entities because these
A)
respire
done
clear
B)
can cause diseases
done
clear
C)
reproduce (inside host)
done
clear
D)
respond to tough environment
done
clear
View Answer play_arrow
question_answer 137) Drosophila Hies with XXY genotype are females, but human beings with such genotype are abnormal males. It shows that
A)
Y-chromosome is essential for sex determination in Drosophila
done
clear
B)
Y-chromosome is female determination in Drosophila
done
clear
C)
Y-chromosome is male determining in human beings
done
clear
D)
Y-chromosomehas no roleinsexdetermination either in Drosophila or in human beings
done
clear
View Answer play_arrow
question_answer 138) Which one pair of parents out of the following is most likely get a child who would suffer from hemolytic disease of the new- born
A)
\[\text{R}{{\text{h}}^{+}}\]mother and\[R{{h}^{-}}\]father
done
clear
B)
\[R{{h}^{-}}\]mother and\[R{{h}^{-}}\]father
done
clear
C)
\[\text{R}{{\text{h}}^{+}}\]mother and\[\text{R}{{\text{h}}^{+}}\]father
done
clear
D)
\[R{{h}^{-}}\]mother and\[\text{R}{{\text{h}}^{+}}\]father
done
clear
View Answer play_arrow
question_answer 139) What is correct regarding leucocytes?
A)
These can squeeze out through (can cross)the capillary walls
done
clear
B)
These are enucleate
done
clear
C)
Sudden fall in their number indicates cancer
done
clear
D)
These are produced in thymus
done
clear
View Answer play_arrow
question_answer 140) RNA and DNA are similar in
A)
having similar sugars
done
clear
B)
having similar pyrimidine bases
done
clear
C)
being capable to replicate
done
clear
D)
being polymers of nucleotides
done
clear
View Answer play_arrow
question_answer 141) The length of one turn of the helix in a B- form DNA is approximately
A)
0.34 nm
done
clear
B)
20 nm
done
clear
C)
2 nm
done
clear
D)
3.4 nm
done
clear
View Answer play_arrow
question_answer 142) An action potential in the nerve fibre is produced when positive and negative charges on the outside and the inside of the axon membrane are reversed because
A)
more potassium ions enter the axon as compared to sodium ions leaving it
done
clear
B)
more sodium ions enter the axon as compared to potassium ions leaving it
done
clear
C)
all potassium ions leave the axon
done
clear
D)
all sodium ions enter the axon
done
clear
View Answer play_arrow
question_answer 143) Melanocyte Stimulating Hormone (MSH) is produced by
A)
anterior pituitary
done
clear
B)
posterior pituitary
done
clear
C)
parsintermedia of pituitary
done
clear
D)
parathyroid
done
clear
View Answer play_arrow
question_answer 144) Which of the following is not a characteristic feature of all the chordates?
A)
Presence of coelom
done
clear
B)
Pharyngeal gill clefts in the early embryonic stages
done
clear
C)
A diaphragm that separates thorax from abdomen
done
clear
D)
Dorsal nerve cord
done
clear
View Answer play_arrow
question_answer 145) A common scent producing gland among mammals is
A)
anal gland
done
clear
B)
prostate gland
done
clear
C)
adrenal gland
done
clear
D)
Bartholin?s gland
done
clear
View Answer play_arrow
question_answer 146) Double fertilization (or triple fusion) leading to initiation of endosperm in angiosperms, requires
A)
fusion of 4 or more polar nuclei and the second male gamete only
done
clear
B)
fusion of 2 polar nuclei and second male gamete only
done
clear
C)
fusion of one polar nucleus and second male gamete only
done
clear
D)
all the above type of fusions in different types of angiosperms
done
clear
View Answer play_arrow
question_answer 147) A person suffering from the deficiency of the visual pigment rhodopsin is advised to take more of
A)
radish and potato
done
clear
B)
apple and grapes
done
clear
C)
carrot and ripe papaya
done
clear
D)
guava and ripe banana
done
clear
View Answer play_arrow
question_answer 148) Eight nucleate embryo sacs are
A)
always bisporic
done
clear
B)
always tetrasporic
done
clear
C)
always monosporic
done
clear
D)
sometimes monosporic, sometines bisporic and sometimes tetrasporic
done
clear
View Answer play_arrow
question_answer 149) Occurrence of endemic species in South America and Australia is due to
A)
extinction of these species from other regions
done
clear
B)
continental separation
done
clear
C)
absence of terrestrial route to these places
done
clear
D)
retrogressive evolution
done
clear
View Answer play_arrow
question_answer 150) Anemophilous type of pollination is found in
A)
Salvia
done
clear
B)
Bottle brush
done
clear
C)
Vallisneria
done
clear
D)
Coconut
done
clear
View Answer play_arrow
question_answer 151) Who was the first Indian ruler to face the Mahmud Ghaznavi?
A)
Prithviraj Chauhan
done
clear
B)
King Jaipal
done
clear
C)
Jaichand
done
clear
D)
Firdausi
done
clear
View Answer play_arrow
question_answer 152) The scientific study of birds is called
A)
Ethnology
done
clear
B)
Ornithology
done
clear
C)
Anthropology
done
clear
D)
Onchology
done
clear
View Answer play_arrow
question_answer 153) Which of the following taxes is not a source of revenue for the Union Government?
A)
Income tax
done
clear
B)
Corporation tax
done
clear
C)
Land revenue
done
clear
D)
Customs duties
done
clear
View Answer play_arrow
question_answer 154) The finance commission is appointed by the
A)
President
done
clear
B)
Prime Minister
done
clear
C)
Council of Ministers
done
clear
D)
Parliament
done
clear
View Answer play_arrow
question_answer 155) The famous Big Bang Theory is based on which of the following principles?
A)
Zeoman?s effect
done
clear
B)
Doppler Effect
done
clear
C)
De Broglie Theorem
done
clear
D)
Principle of Thermodynamics
done
clear
View Answer play_arrow
question_answer 156) Red blood corpuscles are formed in
A)
liver
done
clear
B)
bone marrow
done
clear
C)
kidneys
done
clear
D)
blood vessels
done
clear
View Answer play_arrow
question_answer 157) Yuan is the currency of
A)
China
done
clear
B)
Indonesia
done
clear
C)
Thailand
done
clear
D)
Japan
done
clear
View Answer play_arrow
question_answer 158) Agha Khan cup is associated with
A)
Hockey
done
clear
B)
Football
done
clear
C)
Cricket
done
clear
D)
Badminton
done
clear
View Answer play_arrow
question_answer 159) Nobel Prizes are distributed anunally at
A)
New York
done
clear
B)
German
done
clear
C)
Stockholm
done
clear
D)
Rome
done
clear
View Answer play_arrow
question_answer 160) Which of the following is not the main organ of the United Nations Organisation?
A)
General Assembly
done
clear
B)
Trusteeship Council
done
clear
C)
Security Council
done
clear
D)
World Health Organisation
done
clear
View Answer play_arrow
question_answer 161) Pick the odd one out.
A)
Sun
done
clear
B)
Moon
done
clear
C)
Mars
done
clear
D)
Earth
done
clear
View Answer play_arrow
question_answer 162) Pick the odd one out.
A)
Swim
done
clear
B)
Run
done
clear
C)
Dance
done
clear
D)
Anticipate
done
clear
View Answer play_arrow
question_answer 163) Fill in the blanks. 3, 6 , 6 , 12 ,9.....12
A)
18
done
clear
B)
8
done
clear
C)
22
done
clear
D)
17
done
clear
View Answer play_arrow
question_answer 164) Fill in the blanks. U, O, I, ? , A
A)
E
done
clear
B)
C
done
clear
C)
S
done
clear
D)
G
done
clear
View Answer play_arrow
question_answer 165) Fill in the blanks. D-4, F-6, H-8, ?, L-12
A)
J-10
done
clear
B)
K -12
done
clear
C)
M -14
done
clear
D)
L- 10
done
clear
View Answer play_arrow
question_answer 166) Who is the professor of Psychology?
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 167) Who is the professor of Philosophy?
A)
D
done
clear
B)
B
done
clear
C)
C
done
clear
D)
A
done
clear
View Answer play_arrow
question_answer 168) Who is the wife of E?
A)
C
done
clear
B)
D
done
clear
C)
A
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 169) Who is the professor of Economics?
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
E
done
clear
View Answer play_arrow
question_answer 170) Find the value of\[\sqrt{\frac{0.081\times 0.484}{0.0064\times 6.25}}\].
A)
0.81
done
clear
B)
0.99
done
clear
C)
64
done
clear
D)
25
done
clear
View Answer play_arrow
question_answer 171) The average of first 10 even numbers is
A)
10
done
clear
B)
12.5
done
clear
C)
9
done
clear
D)
11
done
clear
View Answer play_arrow
question_answer 172) 25% of x is same as 40% of y. The ratio x : y is
A)
8 : 5
done
clear
B)
5 :3
done
clear
C)
3 : 8
done
clear
D)
5 :8
done
clear
View Answer play_arrow
question_answer 173) A and B can finisha piece of work in16 days while A alone can do the same work in 24 day sin how many days can B alone complete the work ?
A)
56 days
done
clear
B)
48 days
done
clear
C)
36 days
done
clear
D)
164 days
done
clear
View Answer play_arrow
question_answer 174) If ?-? means ?x?, ?x? means ?+?, ?+? means and ?+? means then \[\text{4}0\times \text{12}+\text{3}-\text{6}+\text{6}0=?\]
A)
482.9
done
clear
B)
16
done
clear
C)
44
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 175) The diameter of a copper sphere is 6 cm. It is melted to form a wire of uniform cross-section. If the length of the wire is 36 cm then find the radius of the wire.
A)
0.5 cm
done
clear
B)
1 m
done
clear
C)
1.5 cm
done
clear
D)
3.14 cm
done
clear
View Answer play_arrow
question_answer 176) In each of the following questions, choose the correct antonym for the word given in capital letters. AMICABLE
A)
Cunning
done
clear
B)
Shy
done
clear
C)
Hostile
done
clear
D)
Crazy
done
clear
View Answer play_arrow
question_answer 177) In each of the following questions, choose the correct antonym for the word given in capital letters. CLARITY
A)
Exaggeration
done
clear
B)
C and our
done
clear
C)
Confusion
done
clear
D)
Reserve
done
clear
View Answer play_arrow
question_answer 178) In each of the following questions, choose the correct antonym for the word given in capital letters. OPULENT
A)
Wealthy
done
clear
B)
Poor
done
clear
C)
Sumptuous
done
clear
D)
Drooping
done
clear
View Answer play_arrow
question_answer 179) In each of the following questions, choose the correct antonym for the word given in capital letters. SANCTIFY
A)
Dedicate
done
clear
B)
Patronise
done
clear
C)
Venerate
done
clear
D)
Pollute
done
clear
View Answer play_arrow
question_answer 180) In each of the following questions, choose the correct synonym for the word given in capital letters. JOVIAL
A)
Merry
done
clear
B)
Funny
done
clear
C)
Smart
done
clear
D)
Triumphant
done
clear
View Answer play_arrow
question_answer 181) In each of the following questions, choose the correct synonym for the word given in capital letters. EXCERPT
A)
Extract
done
clear
B)
Invitation
done
clear
C)
Debate
done
clear
D)
Speech
done
clear
View Answer play_arrow
question_answer 182) In each of the following questions, choose the correct synonym for the word given in capital letters. ERADICATE
A)
Complicate
done
clear
B)
Indicate
done
clear
C)
Dedicate
done
clear
D)
Eliminate
done
clear
View Answer play_arrow
question_answer 183) In each of the following questions, choose the correct synonym for the word given in capital letters. OSTENTATIOUS
A)
Noisy
done
clear
B)
Talkative
done
clear
C)
Showy
done
clear
D)
Wealthy
done
clear
View Answer play_arrow
question_answer 184) In each of the following questions, choose the alternative which can be substituted for the given words/sentence. Study of mankind
A)
Pathology
done
clear
B)
Physiology
done
clear
C)
Philology
done
clear
D)
Anthropology
done
clear
View Answer play_arrow
question_answer 185) In each of the following questions, choose the alternative which can be substituted for the given words/sentence. Intentional destruction of racial groups
A)
Homicide
done
clear
B)
Genocide
done
clear
C)
Fratricide
done
clear
D)
Regicide
done
clear
View Answer play_arrow
question_answer 186) In each of the following questions, choose the alternative which can be substituted for the given words/sentence. One who is interested in the welfare of women
A)
Feminine
done
clear
B)
Feminist
done
clear
C)
Effeminate
done
clear
D)
Flamboyant
done
clear
View Answer play_arrow
question_answer 187) In each of the following questions, choose the alternative which can be substituted for the given words/sentence. Study of statistics of births, deaths, diseases to show the state of community
A)
Geography
done
clear
B)
Anthropology
done
clear
C)
Topography
done
clear
D)
Demography
done
clear
View Answer play_arrow
question_answer 188) In each of the following questions, choose the most suitable alternative to fill in the blank. The summit meeting provided him the much .....shot in the arm.
A)
required
done
clear
B)
desired
done
clear
C)
needed
done
clear
D)
urgent
done
clear
View Answer play_arrow
question_answer 189) In each of the following questions, choose the most suitable alternative to fill in the blank. If you drink too much, it will .....your judgment.
A)
obstruct
done
clear
B)
impede
done
clear
C)
impair
done
clear
D)
hinder
done
clear
View Answer play_arrow
question_answer 190) In each of the following questions, choose the most suitable alternative to fill in the blank. The government should provide attractive tax .....to create the market for quality goods.
A)
controls
done
clear
B)
incentives
done
clear
C)
revenues
done
clear
D)
structures
done
clear
View Answer play_arrow
question_answer 191) In each of the following questions, choose the most suitable alternative to fill in the blank. Each cause conditions a .....effect and there can be no cause without effect.
A)
specific
done
clear
B)
relevant
done
clear
C)
requisite
done
clear
D)
proper
done
clear
View Answer play_arrow
question_answer 192) In each of the following questions, re-arrange the given four parts P, Q, R and S to make a complete and meaningful sentence. (P) The management having agreed (Q) the workers called off the strike (R) to increase their wages (S) and returned to work
A)
PQRS
done
clear
B)
PRQS
done
clear
C)
RSQP
done
clear
D)
SPQR
done
clear
View Answer play_arrow
question_answer 193) In each of the following questions, re-arrange the given four parts P, Q, R and S to make a complete and meaningful sentence. The Bible, (P) has in many respects (Q) the sacred book of all Christians (R) among all the books of the world (S) a unique character and position
A)
QPSR
done
clear
B)
QRPS
done
clear
C)
RPQS
done
clear
D)
RQPS
done
clear
View Answer play_arrow
question_answer 194) In each of the following questions, re-arrange the given four parts P, Q, R and S to make a complete and meaningful sentence. The ultimate hope (P) will force the nations (Q) that the destructive nature of weapons (R) to give up war (S) has not been fulfilled
A)
PQRS
done
clear
B)
PRQS
done
clear
C)
QPRS
done
clear
D)
RSQP
done
clear
View Answer play_arrow
question_answer 195) In each of the following questions, re-arrange the given four parts P, Q, R and S to make a complete and meaningful sentence. (P) deserve all honour in society (Q) in doing their job well (R) men of conscience who take pride (S) whatever its nature
A)
PRSQ
done
clear
B)
QRPS
done
clear
C)
RQSP
done
clear
D)
SPQR
done
clear
View Answer play_arrow
question_answer 196) According to the passage, a journalist?s work includes
A)
writing
done
clear
B)
news gathering
done
clear
C)
sorting out the news
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 197) What, according to the passage, is the common characteristic of all journalists?
A)
They all write for the newspapers
done
clear
B)
They all have to first probe into the news they gather
done
clear
C)
They all make an adventure to find a worthy news
done
clear
D)
They all can forecast future news
done
clear
View Answer play_arrow
question_answer 198) What is most important for a journalist?
A)
He must be able to interpret the new correctly
done
clear
B)
He must know how to sort out the news
done
clear
C)
He must be able to write effectively
done
clear
D)
He must know how to gather the right news
done
clear
View Answer play_arrow
question_answer 199) What does the passage say about successful journalists?
A)
They reject a major portion of the gathered news
done
clear
B)
They can use a relatively small portion of the news
done
clear
C)
Their news come from unexpected sources
done
clear
D)
They present advance news
done
clear
View Answer play_arrow
question_answer 200) What is the basic requisite for exclusive and exhaustive coverage of newspapers?
A)
Forecast of tomorrow?s news from today?s news
done
clear
B)
Collecting the news of a ?real scoop?
done
clear
C)
Selection of a small portion of the gathered news
done
clear
D)
Effective style of writing
done
clear
View Answer play_arrow
 is
is
