If electronic charge e, electron mass m, speed of light in vacuum c and Planck's constant h are taken as fundamental quantities, the permeability of vacuum \[{{\mu }_{0}}\] can be expressed in units of
A)
\[\left( \frac{h}{m{{e}^{2}}} \right)\]
done
clear
B)
\[\left( \frac{hc}{m{{e}^{2}}} \right)\]
done
clear
C)
\[\left( \frac{h}{c{{e}^{2}}} \right)\]
done
clear
D)
\[\left( \frac{m{{c}^{2}}}{h{{e}^{2}}} \right)\]
done
clear
View Answer play_arrow
Three blocks of masses \[{{m}_{1}},\] \[{{m}_{2}}\] and \[{{m}_{3}}\] are connected by a massless strings, as shown, on a frictionless table. They are pulled with a force \[{{T}_{3}}=40\,N.\] If\[{{m}_{1}}=10kg,\]\[{{m}_{2}}=6kg\]and\[{{m}_{3}}=4\,kg,\] the tension \[{{T}_{2}}\] will be
A)
20 N
done
clear
B)
40 N
done
clear
C)
10 N
done
clear
D)
32 N
done
clear
View Answer play_arrow
What will happen when a 40 watt, 220 volt lamp and 100 watt, 220 volt lamp are connected in series across 40 volt supply?
A)
100 watt lamp will fuse
done
clear
B)
40 watt lamp will fuse
done
clear
C)
Both lamps will fuse
done
clear
D)
Neither lamp will fuse
done
clear
View Answer play_arrow
A pipe closed at one end produces a fundamental note of 412 Hz. It is cut into two pieces of equal length. The fundamental frequencies produced by the two pieces are
A)
206 Hz, 412 Hz
done
clear
B)
824 Hz, 1648 Hz
done
clear
C)
412 Hz, 824 Hz
done
clear
D)
206 Hz, 824 Hz
done
clear
View Answer play_arrow
Two waves whose intensities are 9 and 16 are made to interfere. The ratio of maximum and minimum intensities in the interference pattern is
A)
49 : 1
done
clear
B)
25 : 7
done
clear
C)
10 : 9
done
clear
D)
4 : 3
done
clear
View Answer play_arrow
In order to make the effective acceleration due to gravity equal to zero at the equator, the angular velocity of rotation of the earth about its axis should be (\[g=10\,\,m{{s}^{-2}}\] and radius of earth is 64000 km)
A)
\[Zero\]
done
clear
B)
\[\frac{1}{800}rad\,\,{{\sec }^{-1}}\]
done
clear
C)
\[\frac{1}{80}rad\,\,{{\sec }^{-1}}\]
done
clear
D)
\[\frac{1}{8}\,rad\,\,{{\sec }^{-1}}\]
done
clear
View Answer play_arrow
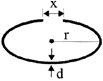
A cylindrical metal rod is shaped into a ring with a small gap as shown. On heating the system:
A)
x, r and d decrease
done
clear
B)
x and r increase, d decreases
done
clear
C)
x, r and d all increase
done
clear
D)
x and r decreased, d remains constant
done
clear
View Answer play_arrow
A coil in the shape of an equilateral triangle of side \[l\] is suspended between the pole pieces of a permanent magnet such that \[\overrightarrow{B}\] is in plane of the coil. If due to a current i in the triangle a torque\[\tau \] acts on it, the side \[l\] of the triangle is
A)
\[\frac{2}{\sqrt{3}}{{\left( \frac{\tau }{Bi} \right)}^{\frac{1}{2}}}\]
done
clear
B)
\[\frac{2}{3}\left( \frac{\tau }{Bi} \right)\]
done
clear
C)
\[2{{\left( \frac{\tau }{\sqrt{3}Bi} \right)}^{\frac{1}{2}}}\]
done
clear
D)
\[\frac{1}{\sqrt{3}}\frac{\tau }{Bi}\]
done
clear
View Answer play_arrow
A fixed mass of gas at constant pressure occupies a volume V. The gas undergoes a rise in temperature so that the root mean square velocity of its molecules is doubled. The new volume will be
A)
\[V/2\]
done
clear
B)
\[V/\sqrt{2}\]
done
clear
C)
\[2\,\,V\]
done
clear
D)
\[4\,\,V\]
done
clear
View Answer play_arrow
Energy of simple harmonic motion depends upon [Here, a = amplitude, \[\omega =\] angular velocity]
A)
\[\frac{1}{{{\omega }^{2}}}\]
done
clear
B)
\[\omega \]
done
clear
C)
\[{{a}^{2}}\]
done
clear
D)
\[\frac{1}{{{a}^{2}}}\]
done
clear
View Answer play_arrow
Let \[{{n}_{p}}\] and \[{{n}_{e}}\] be the number of holes and conduction electrons in an intrinsic semiconductor. Then
A)
\[{{n}_{p}}>{{n}_{e}}.\]
done
clear
B)
\[{{n}_{p}}={{n}_{e}}.\]
done
clear
C)
\[{{n}_{p}}<{{n}_{e}}.\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
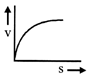
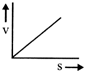
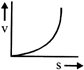
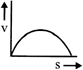
A lead shot of 1 mm diameter falls through a long column of glycerine. The variation of its velocity v with distance covered(s) is represented by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Two deuterons undergo nuclear fusion to form a Helium nucleus. Energy released in this process is: (given binding energy per nucleon for deuteron =1.1 MeV and for helium=7.0MeV)
A)
30.2 MeV
done
clear
B)
32.4 MeV
done
clear
C)
23.6 MeV
done
clear
D)
25.8 MeV
done
clear
View Answer play_arrow
In Young's expt., the distance between two slits is \[\frac{d}{3}\] and the distance between the screen and the slit is 3 D. The number of fringes in \[\frac{1}{3}m\] on the screen, formed by monochromatic light of wavelength \[3\lambda ,\] will be
A)
\[\frac{d}{9D\lambda }\]
done
clear
B)
\[\frac{d}{27D\lambda }\]
done
clear
C)
\[\frac{d}{81\,\,D\lambda }\]
done
clear
D)
\[\frac{d}{D\lambda }\]
done
clear
View Answer play_arrow
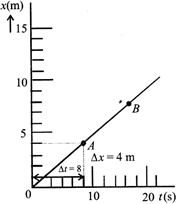
The graph of an object's motion (along the x- axis) is shown in the figure. The instantaneous velocity of the object at points A and B are \[{{v}_{A}}\]and \[{{v}_{B}}\] respectively. Then
A)
\[{{v}_{A}}={{v}_{B}}=0.5\,m/s\]
done
clear
B)
\[{{v}_{A}}=0.5\,m/s<{{v}_{B}}\]
done
clear
C)
\[{{v}_{A}}=0.5\,m/s>{{v}_{B}}\]
done
clear
D)
\[{{v}_{A}}={{v}_{B}}=2\,m/s\]
done
clear
View Answer play_arrow
When the amount of work done is 300 J and change in internal energy is 100 J, then the heat supplied is
A)
400 J
done
clear
B)
350 J
done
clear
C)
200 J
done
clear
D)
150 J
done
clear
View Answer play_arrow
If the number of turns per unit length of a coil of a solenoid is doubled, the self-inductance of the solenoid will
A)
remain unchanged
done
clear
B)
be halved
done
clear
C)
be doubled
done
clear
D)
become four times
done
clear
View Answer play_arrow
A bar magnet of length 6 cm has a magnetic moment of \[4\,J\,{{T}^{-1}}.\] Find the strength of magnetic field at a distance of 200 cm from the centre of the magnet along its equatorial line.
A)
\[4\times {{10}^{-8}}\] tesla
done
clear
B)
\[3.5\times {{10}^{-8}}\] tesla
done
clear
C)
\[5\times {{10}^{-8}}\] tesla
done
clear
D)
\[3\times {{10}^{-8}}\]tesla
done
clear
View Answer play_arrow
A gun is aimed at a horizontal target. It takes \[\frac{1}{2}s\] for the bullet to reach the target. The bullet hits the target x metre below the aim. Then, x is equal to
A)
\[\frac{9.8}{4}m\]
done
clear
B)
\[\frac{9.8}{8}m\]
done
clear
C)
\[9.8m\]
done
clear
D)
\[16.6m\]
done
clear
View Answer play_arrow
The minimum energy required to eject an electron, from the metal surface is called
A)
atomic energy
done
clear
B)
mechanical energy
done
clear
C)
electrical energy
done
clear
D)
work function
done
clear
View Answer play_arrow
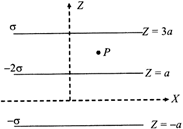
Three infinitely long charge sheets are placed as shown in figure. The electric field at point P is
A)
\[\frac{2\sigma }{{{\varepsilon }_{0}}}\hat{k}\]
done
clear
B)
\[\frac{4\sigma }{{{\varepsilon }_{0}}}\hat{k}\]
done
clear
C)
\[-\frac{2\sigma }{{{\varepsilon }_{0}}}\hat{k}\]
done
clear
D)
\[-\frac{4\sigma }{{{\varepsilon }_{0}}}\hat{k}\]
done
clear
View Answer play_arrow
If microwaves, X rays, infrared, gamma rays, ultra- violet, radio waves and visible parts of the electromagnetic spectrum are denoted by M, X, I, G, U, R and V then which of the following is the arrangement in ascending order of wavelength ?
A)
R, M, I, V, U, X and G
done
clear
B)
M, R, V, X, U, G and I
done
clear
C)
G, X, U, V, I, M and R
done
clear
D)
I, M, R, U, V, X and G
done
clear
View Answer play_arrow
In an ideal gas at temperature T, the average force that a molecule applies on the walls of a closed container depends on T as \[{{T}^{q}}.\] A good estimate for q is:
A)
\[\frac{1}{2}\]
done
clear
B)
\[2\]
done
clear
C)
\[1\]
done
clear
D)
\[\frac{1}{4}\]
done
clear
View Answer play_arrow
A letter 'A' is constructed of a uniform wire with resistance \[1.0\,\Omega \] per cm, The sides of the letter are 20 cm and the cross piece in the middle is 10 cm long. The apex angle is 60. The resistance between the ends of the legs is close to:
A)
\[50.0\,\Omega \]
done
clear
B)
\[10\,\Omega \]
done
clear
C)
\[36.7\,\Omega \]
done
clear
D)
\[26.7\,\Omega \]
done
clear
View Answer play_arrow
A crane is used to lift 1000 kg of coal from a mine 100 m deep. The time taken by the crane is 1 hour. The efficiency of the crane is 80%. If \[g=10\,m{{s}^{-2}},\] then the power of the crane is
A)
\[{{10}^{4}}W\]
done
clear
B)
\[{{10}^{5}}W\]
done
clear
C)
\[\frac{{{10}^{4}}}{36\times 8}\,W\]
done
clear
D)
\[\frac{{{10}^{5}}}{36\times 8}\,W\]
done
clear
View Answer play_arrow
A steel rod of radius R = 10 mm and length L = 100 cm is stretched along its length by a force F\[=6.28\times {{10}^{4}}N.\] If the Young's modulus of steel is Y\[=2\times {{10}^{11}}N/{{m}^{2}},\] the percentage elongation in the length of the rod is
A)
0.001
done
clear
B)
0.314
done
clear
C)
2.015
done
clear
D)
1.549
done
clear
View Answer play_arrow
The sky appears blue, because
A)
blue light is scattered the most
done
clear
B)
blue light is absorbed
done
clear
C)
red light is absorbed
done
clear
D)
it is its natural colour
done
clear
View Answer play_arrow
Out of the following which one is not a possible energy for a photon to be emitted by hydrogen atom according to Bohr's atomic model?
A)
1.9 eV
done
clear
B)
11.1 eV
done
clear
C)
13.6 eV
done
clear
D)
0.65 eV
done
clear
View Answer play_arrow
A point object is 24 cm above the surface of water \[(\mu =4/3)\] in lake. A fish inside the water will observe the image to be at a point
A)
6 cm above the surface of water
done
clear
B)
6 cm below the surface of water
done
clear
C)
18 cm above the surface of water
done
clear
D)
32 cm above the surface of water
done
clear
View Answer play_arrow
A projectile is fired from the surface of the earth with a velocity of \[5\,\,m{{s}^{-1}}\] and angle \[\theta \] with the horizontal. Another projectile fired from another planet with a velocity of \[3\,\,m{{s}^{-1}}\] at the same angle follows a trajectory which is identical with the trajectory of the projectile fired from the earth. The value of the acceleration due to gravity on the planet is (in \[m{{s}^{-2}}\]) given g \[=9.8\,\,m{{s}^{2}}\]
A)
3.5
done
clear
B)
5.9
done
clear
C)
16.3
done
clear
D)
110.8
done
clear
View Answer play_arrow
The relation between B, H and I in S.I. units is
A)
\[B={{\mu }_{0}}(H+I)\]
done
clear
B)
\[B=H+4\,\pi \,I\]
done
clear
C)
\[H={{\mu }_{0}}(B+I)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If a current increases from zero to one ampere in 0.1 second in a coil of 5 mH, then the magnitude of the induced e.m.f. will be
A)
0.005 volt
done
clear
B)
0.5 volt
done
clear
C)
0.05 volt
done
clear
D)
5 volt
done
clear
View Answer play_arrow
Potentiometer measures potential more accurately because
A)
it measures potential in open circuit
done
clear
B)
it uses sensitive galvanometer for null deflection
done
clear
C)
it uses high resistance potentiometer wire
done
clear
D)
it measures potential in closed circuit
done
clear
View Answer play_arrow
When the current in an ac circuit is wattless, the phase difference between the applied voltage and circuit current will be
A)
\[45{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[180{}^\circ \]
done
clear
View Answer play_arrow
A mass of 1 kg is suspended by a thread. It is
(i) lifted with an acceleration \[4.9\,m/{{s}^{2}}\] (ii) lowered with an acceleration \[4.9\,m/{{s}^{2}}.\] The ratio of the tension is
A)
3 : 1
done
clear
B)
1 : 3
done
clear
C)
1 : 2
done
clear
D)
2 : 1
done
clear
View Answer play_arrow
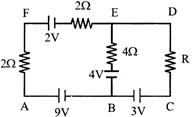
In the electric network shown, when no current flows through the \[4\Omega \] resistor in the arm EB, the potential difference between the points A and D will be:
A)
6 V
done
clear
B)
3 V
done
clear
C)
5 V
done
clear
D)
4 V
done
clear
View Answer play_arrow
If the momentum of a particle is doubled, then its de-Broglie wavelength will become
A)
two times
done
clear
B)
four times
done
clear
C)
unchanged
done
clear
D)
halftimes
done
clear
View Answer play_arrow
A capillary tube is immersed vertically in water and the height of the water column is x. When this arrangement is taken into a mine of depth d, the height of the water column is y. If R is the radius of earth, the ratio \[\frac{x}{y}\] is:
A)
\[\left( 1-\frac{d}{R} \right)\]
done
clear
B)
\[\left( 1-\frac{2d}{R} \right)\]
done
clear
C)
\[\left( \frac{R-d}{R+d} \right)\]
done
clear
D)
\[\left( \frac{R+d}{R-d} \right)\]
done
clear
View Answer play_arrow
If 'S' is stress and 'Y' is young?s modulus of material of a wire, the energy stored in the wire per unit volume is
A)
\[\frac{{{S}^{2}}}{2Y}\]
done
clear
B)
\[2{{S}^{2}}Y\]
done
clear
C)
\[\frac{S}{2Y}\]
done
clear
D)
\[\frac{2Y}{{{S}^{2}}}\]
done
clear
View Answer play_arrow
A Carnot engine is working between \[127{}^\circ C\] and \[27{}^\circ C.\] The increase in efficiency will be maximum when the temperature of
A)
the source is increased by \[50{}^\circ C\]
done
clear
B)
the sink is decreased by \[50{}^\circ C\]
done
clear
C)
source is increased by \[25{}^\circ C\] and that of sink is decreased by \[25{}^\circ C\]
done
clear
D)
both source and sink are decreased by \[25{}^\circ C\] each
done
clear
View Answer play_arrow
Current gain in common emitter configuration is more than 1 because
A)
\[{{I}_{c}}<{{I}_{b}}\]
done
clear
B)
\[{{I}_{c}}<{{I}_{e}}\]
done
clear
C)
\[{{I}_{c}}>{{I}_{e}}\]
done
clear
D)
\[{{I}_{c}}>{{I}_{b}}\]
done
clear
View Answer play_arrow
A charge of total amount Q is distributed over two concentric hollow spheres of radii r and R (R > r) such that the surface charge densities on the two spheres are equal. The electric potential at the common centre is
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{(R-r)Q}{({{R}^{2}}+{{r}^{2}})}\]
done
clear
B)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{(R+r)Q}{2({{R}^{2}}+{{r}^{2}})}\]
done
clear
C)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{(R+r)Q}{({{R}^{2}}+{{r}^{2}})}\]
done
clear
D)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{(R-r)Q}{2\,({{R}^{2}}+{{r}^{2}})}\]
done
clear
View Answer play_arrow
The magnitude of the average electric field normally present in the atmosphere just above the surface of the Earth is about 150 N/C, directed inward towards the center of the Earth. This gives the total net surface charge carried by the Earth to be: [Given\[{{\varepsilon }_{0}}=8.85\times {{10}^{-12}}{{C}^{2}}/N-{{m}^{2}},\]\[{{R}_{E}}=6.37\times {{10}^{6}}m\]]
A)
\[+670\,\,kC\]
done
clear
B)
\[-670\,\,kC\]
done
clear
C)
\[-680\,\,kC\]
done
clear
D)
\[+680\,\,kC\]
done
clear
View Answer play_arrow
A particle of mass 2 kg is moving such that at time t, its position, in meter, is given by \[\overrightarrow{r}(t)=5\hat{i}-2{{t}^{2}}\hat{j}.\] The angular momentum of the particle at t = 2s about the origin in \[kg\,\,{{m}^{-2}}{{s}^{-1}}\]
A)
\[-80\,\hat{k}\]
done
clear
B)
\[(10\hat{i}-16\hat{j})\]
done
clear
C)
\[-40\,\hat{k}\]
done
clear
D)
\[40\hat{k}\]
done
clear
View Answer play_arrow
The tuning circuit of a radio receiver has a resistance of \[50\,\Omega ,\]an inductance of 10mH and a variable capacitance C. A 1 MHz radio wave produces a potential difference of 0.1 mV. The value of capacitance to produce the resonance will be
A)
2.5 pF
done
clear
B)
5.0 pF
done
clear
C)
25 pF
done
clear
D)
50 pF
done
clear
View Answer play_arrow
Among the following pairs of ions, the lower oxidation state in aqueous solution is more stable than the other, in :
A)
\[T{{i}^{+}},\]\[T{{i}^{3+}}\]
done
clear
B)
\[C{{u}^{+}},\]\[C{{u}^{2+}}\]
done
clear
C)
\[C{{r}^{2+}},\]\[C{{r}^{3+}}\]
done
clear
D)
\[{{V}^{2+}},\]\[V{{O}^{2+}}\]
done
clear
View Answer play_arrow
The correct statement for the molecule, \[Cs{{I}_{3}}\] is:
A)
It is a covalent molecule.
done
clear
B)
It contains \[C{{s}^{+}}\] and \[I_{^{3}}^{-}\] ions.
done
clear
C)
It contains \[C{{s}^{3+}}\] and \[{{I}^{-}}\] ions.
done
clear
D)
It contains \[C{{s}^{+}},\] \[{{I}^{-}}\]and lattice \[{{I}_{2}}\] molecule.
done
clear
View Answer play_arrow
The optically inactive compound from the following is:
A)
2 - Chloropropanal
done
clear
B)
2 - Chlorobutane
done
clear
C)
2 - Chloropentane
done
clear
D)
2 - Chloro - 2-methylbutane
done
clear
View Answer play_arrow
Which of the following statements, about the advantage of roasting of sulphide ore before reduction is not true?
A)
The\[\Delta G_{f}^{o}\] of the sulphide is greater than those for \[C{{S}_{2}}\] and \[{{H}_{2}}S.\]
done
clear
B)
The \[\Delta G_{f}^{o}\] is negative for roasting of sulphide ore to oxide.
done
clear
C)
Roasting of the sulphide to the oxide is thermodynamically feasible.
done
clear
D)
Carbon and hydrogen are suitable reducing agents for metal sulphides.
done
clear
View Answer play_arrow
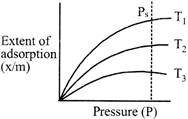
For the graph below, select correct order of temperature?
A)
\[{{T}_{1}}>{{T}_{2}}>{{T}_{3}}\]
done
clear
B)
\[{{T}_{2}}>{{T}_{3}}>{{T}_{1}}\]
done
clear
C)
\[{{T}_{3}}>{{T}_{2}}>{{T}_{1}}\]
done
clear
D)
\[{{T}_{1}}={{T}_{2}}={{T}_{3}}\]
done
clear
View Answer play_arrow
The correct order of increasing \[{{[H_{3}^{2}{{O}^{+}}]}^{3}}\] in the following aqueous solutions is
A)
\[0.01\,M\,{{H}_{2}}S<0.01\,M\,{{H}_{2}}S{{O}_{4}}<0.01\,M\,NaCl<0.01\,M\,NaN{{O}_{2}}\]
done
clear
B)
\[0.01\,\,M\,\,NaCl<0.01M\,\,NaN{{O}_{2}}<0.01\,\,M\,{{H}_{2}}S<0.01M\,{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[0.01\,\,M\,\,NaN{{O}_{2}}<0.01M\,\,NaCl<0.01\,\,M\,{{H}_{2}}S<0.01M\,{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[0.01\,\,M\,\,{{H}_{2}}S<0.01M\,\,NaN{{O}_{2}}<0.01\,\,M\,NaCl<0.01M\,{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
Pollution in large cities can be checked only by:
A)
Shifting of factories out of the residential area.
done
clear
B)
Less use of insecticides.
done
clear
C)
Proper disposal of organic wastes, sewage and industrial effluents.
done
clear
D)
All the above.
done
clear
View Answer play_arrow
All form ideal solution except
A)
\[{{C}_{6}}{{H}_{6}}\] and \[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] and \[{{C}_{2}}{{H}_{5}}I\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}Cl\] and \[{{C}_{6}}{{H}_{5}}Br\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}\] and \[{{C}_{2}}{{H}_{5}}OH.\]
done
clear
View Answer play_arrow
Which of the following metal ions will form complexes with the same magnetic moment and geometry irrespective of the nature of ligands?
A)
\[N{{i}^{2+}}\]
done
clear
B)
\[F{{e}^{2+}}\]
done
clear
C)
\[C{{u}^{2+}}\]
done
clear
D)
\[C{{o}^{2+}}\]
done
clear
View Answer play_arrow
The hybridization of atomic orbitals of nitrogen in \[NO_{2}^{+},\]\[NO_{2}^{-}\]and \[NH_{4}^{+}\] are
A)
\[s{{p}^{2}},\] \[s{{p}^{3}}\] and \[s{{p}^{2}}\] respectively
done
clear
B)
\[sp,\] \[s{{p}^{2}}\] and \[s{{p}^{3}}\] respectively
done
clear
C)
\[s{{p}^{2}},\] \[sp\] and \[s{{p}^{3}}\] respectively
done
clear
D)
\[s{{p}^{2}},\] \[s{{p}^{3}}\] and \[sp\] respectively
done
clear
View Answer play_arrow
Enthalpy and entropy change for a chemical reaction are \[-2.5\times {{10}^{3}}\]cals and 7.4 cals \[{{\deg }^{-1}}\] respectively. At \[25{}^\circ C\] the reaction is:
A)
Reversible
done
clear
B)
Irreversible
done
clear
C)
Spontaneous
done
clear
D)
Non spontaneous
done
clear
View Answer play_arrow
Which of the following has correct increasing basic strength?
A)
\[MgO<BeO<CaO<BaO\]
done
clear
B)
\[BeO<MgO<CaO<BaO\]
done
clear
C)
\[BaO<CaO<MgO<BeO\]
done
clear
D)
\[CaO<BaO<BeO<MgO\]
done
clear
View Answer play_arrow
One litre of 1 M \[CuS{{O}_{4}}\] solution is electrolysed. After passing 2 F of electricity, molarity of \[CuS{{O}_{4}}\]solution will be
A)
\[\frac{M}{2}\]
done
clear
B)
\[\frac{M}{4}\]
done
clear
C)
\[M\]
done
clear
D)
\[0\]
done
clear
View Answer play_arrow
Which of the following is commercially known as oxone?
A)
\[N{{a}_{2}}{{O}_{2}}+HCl\]
done
clear
B)
\[N{{a}_{2}}O+HCl\]
done
clear
C)
\[N{{a}_{2}}{{O}_{2}}+N{{a}_{2}}O\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Solid \[Ba{{(N{{O}_{3}})}_{2}}\] is gradually dissolved in a \[1.0\times {{10}^{-4}}\,\,M\,\,N{{a}_{2}}C{{O}_{3}}\] solution. At which concentration of \[B{{a}^{2+}},\] precipitate of \[BaC{{O}_{3}}\] begins to form? \[({{K}_{sp}}\,for\,BaC{{O}_{3}}=5.1\times {{10}^{-9}})\]
A)
\[5.1\times {{10}^{-5}}M\]
done
clear
B)
\[7.1\times {{10}^{-8}}M\]
done
clear
C)
\[4.1\times {{10}^{-5}}M\]
done
clear
D)
\[8.1\times {{10}^{-7}}M\]
done
clear
View Answer play_arrow
Which is not correct regarding the adsorption of a gas on surface of solid?
A)
On increasing temperature, adsorption increases contineuously
done
clear
B)
Enthalpy and entropy changes are -ve
done
clear
C)
Adsorption is more for some specific substance
done
clear
D)
This Phenomenon is reversible
done
clear
View Answer play_arrow
The unusual properties of water in the condensed phase (liquid and solid states) are due to the
A)
presence of hydrogen and covalent bonding, between the water molecules.
done
clear
B)
presence of covalent bonding between the water molecules.
done
clear
C)
presence of extensive hydrogen bonding between water molecules.
done
clear
D)
presence of ionic bonding.
done
clear
View Answer play_arrow
Match the organic compounds in column-I with the Lassaigne's test results in column-II appropriately:
Column-I Column-II [A] Aniline (i) Red colour with \[FeC{{l}_{3}}\] [B] Benzene sulfonic acid (ii) Violet colour with sodium [C] Thiourea (iii) Blue colour with hot and acidic solution of \[FeS{{O}_{4}}\]
A)
A\[\to \](ii); B\[\to \](iii); C\[\to \](i)
done
clear
B)
A\[\to \](iii); B\[\to \](i); C\[\to \](ii)
done
clear
C)
A\[\to \](iii); B\[\to \](ii); C\[\to \](i)
done
clear
D)
A\[\to \](ii); B\[\to \](i); C\[\to \](iii)
done
clear
View Answer play_arrow
A and B in the following reactions are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
On adding 0.1 M solution each of \[[A{{g}^{+}}],\]\[[B{{a}^{2+}}],\]\[[C{{a}^{2+}}]\] in a \[NaS{{O}_{4}}\] solution, species first precipitated is \[[\]\[{{K}_{sp}}\,\,BaS{{O}_{4}}={{10}^{-11}},\]\[{{K}_{sp}}\,\,CaS{{O}_{4}}={{10}^{-6}},\]\[{{K}_{sp}}A{{g}_{2}}S{{O}_{4}}={{10}^{-5}}]\]
A)
\[A{{g}_{2}}S{{O}_{4}}\]
done
clear
B)
\[BaS{{O}_{4}}\]
done
clear
C)
\[CaS{{O}_{4}}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which one of the following statements concerning lanthanide elements is false?
A)
Lanthanides are separated from one another by ion exchange method.
done
clear
B)
The ionic radii of trivalent lanthanides steadily increase with increase in atomic number.
done
clear
C)
All lanthanides are highly dense metals.
done
clear
D)
Most characteristic oxidation state of lanthanides is+3.
done
clear
View Answer play_arrow
The fraction of total volume occupied by the atoms present in a simple cube is
A)
\[\frac{\pi }{3\sqrt{2}}\]
done
clear
B)
\[\frac{\pi }{4\sqrt{2}}\]
done
clear
C)
\[\frac{\pi }{4}\]
done
clear
D)
\[\frac{\pi }{6}\]
done
clear
View Answer play_arrow
The hypothetical complex chlorodiaquo-triammine cobalt (III) chloride can be represented as
A)
\[[CoCl{{(N{{H}_{3}})}_{3}}\,{{({{H}_{2}}O)}_{2}}]C{{l}_{2}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{3}}\,({{H}_{2}}O)C{{l}_{3}}]\]
done
clear
C)
\[[Co{{(N{{H}_{2}})}_{3}}\,{{({{H}_{2}}O)}_{2}}Cl]\]
done
clear
D)
\[[Co{{(N{{H}_{3}})}_{3}}\,{{({{H}_{2}}O)}_{3}}]C{{l}_{3}}\]
done
clear
View Answer play_arrow
Which of the following may be considered to be an organometallic compound?
A)
Nickel tetracarbonyl
done
clear
B)
Chlorophyll
done
clear
C)
\[{{K}_{3}}\,[Fe\,{{({{C}_{2}}{{O}_{4}})}_{3}}]\]
done
clear
D)
\[[Co{{(en)}_{3}}]C{{l}_{3}}\]
done
clear
View Answer play_arrow
If \[{{K}_{1}}\] and \[{{K}_{2}}\] are respective equilibrium constants for the two reactions \[Xe{{F}_{6}}(g)+{{H}_{2}}O\,(g)XeO{{F}_{4}}(g)+2HF\,(g)\] \[Xe{{O}_{4}}(g)+Xe{{F}_{6}}\,(g)XeO{{F}_{4}}(g)+Xe{{O}_{3}}{{F}_{2}}\,(g)\] the equilibrium constant for the reaction \[Xe{{O}_{4}}(g)+2HF\,(g)Xe{{O}_{3}}{{F}_{2}}(g)+{{H}_{2}}O\,(g)\] will be
A)
\[\frac{{{K}_{1}}}{K_{2}^{2}}\]
done
clear
B)
\[{{K}_{1}}.{{K}_{2}}\]
done
clear
C)
\[\frac{{{K}_{1}}}{{{K}_{2}}}\]
done
clear
D)
\[\frac{{{K}_{2}}}{{{K}_{1}}}\]
done
clear
View Answer play_arrow
For the compounds \[C{{H}_{3}}Cl,\]\[C{{H}_{3}}Br,\]\[C{{H}_{3}}I\]and \[C{{H}_{3}}F,\]the correct order of increasing C-halogen bond length is:
A)
\[C{{H}_{3}}F<C{{H}_{3}}Cl<C{{H}_{3}}Br<C{{H}_{3}}I\]
done
clear
B)
\[C{{H}_{3}}F<C{{H}_{3}}Br<C{{H}_{3}}Cl<C{{H}_{3}}I\]
done
clear
C)
\[C{{H}_{3}}F<C{{H}_{3}}I<C{{H}_{3}}Br<C{{H}_{3}}Cl\]
done
clear
D)
\[C{{H}_{3}}Cl<C{{H}_{3}}Br<C{{H}_{3}}F<C{{H}_{3}}I\]
done
clear
View Answer play_arrow
In the following reaction 'A ' is
A)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}CHO\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}C{{H}_{2}}OH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}CHO\]
done
clear
View Answer play_arrow
The standard electrode potentials of four elements A, B, C and D are \[-3.05,\]\[-1.66,\]\[-0.40\] and \[+0.80.\] The highest chemical reactivity will be exhibited by:
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Which of the following statements are correct?
(i) Cationic detergents have germicidal properties. (ii) Bacteria can degrade the detergents containing highly branched chains. (iii) Some synthetic detergents can give foam even in ice cold water. (iv) Synthetic detergents are not soaps.
A)
(i), (ii) and (iii)
done
clear
B)
(i), (iii) and (iv)
done
clear
C)
(ii), (iii) and (iv)
done
clear
D)
(iii) and (iv)
done
clear
View Answer play_arrow
Which of the following statements is incorrect regarding benzyl chloride?
A)
It gives white precipitate with alcoholic \[AgN{{O}_{3}}\]
done
clear
B)
It is an aromatic compound with substitution in the side chain
done
clear
C)
It undergoes nucleophilic substitution reaction
done
clear
D)
It is less reactive than vinyl chloride
done
clear
View Answer play_arrow
One mole of an ideal gas is allowed to expand reversibly and adiabatically from a temperature of \[27{}^\circ C.\] If the work done during the process is 3 kJ, then final temperature of the gas is \[({{C}_{v}}=20J/K)\]
A)
100 K
done
clear
B)
150 K
done
clear
C)
195 K
done
clear
D)
255 K
done
clear
View Answer play_arrow
A compound X, of boron reacts with \[N{{H}_{3}}\] on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating \[B{{F}_{3}}\]with lithium aluminium hydride. The compounds X and Y are represented by the formulas.
A)
\[{{B}_{2}}{{H}_{6}},\]\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\]
done
clear
B)
\[{{B}_{2}}{{O}_{3}},\]\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\]
done
clear
C)
\[B{{F}_{3}},\]\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\]
done
clear
D)
\[{{B}_{3}}{{N}_{3}}{{H}_{6}},\]\[{{B}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
Acetylenic hydrogens are acidic because
A)
Sigma electron density of C - H bond in acetylene is nearer to carbon, which has 50% s-character.
done
clear
B)
Acetylene has only open hydrogen in each carbon.
done
clear
C)
Acetylene contains least number of hydrogens among the possible hydrocarbons having two carbons.
done
clear
D)
Acetylene belongs to the class of alkynes with molecular formula, \[{{C}_{n}}{{H}_{2n-2}}.\]
done
clear
View Answer play_arrow
\[{{H}_{2}}S(g)\xrightarrow{{}}H{{S}^{-}}(g)+{{H}^{+}}(g),\]\[\Delta H{}^\circ ={{x}_{1}}\] \[\Delta H_{f}^{{}^\circ }[{{H}_{2}}S(g)={{x}_{2}},\]\[\Delta H_{f}^{{}^\circ }[H(g)]={{x}_{3}}\] hence, \[\Delta H_{f}^{{}^\circ }{{[HS]}^{-}}\] is
A)
\[{{x}_{1}}+{{x}_{2}}-{{x}_{3}}\]
done
clear
B)
\[{{x}_{3}}-{{x}_{1}}-{{x}_{2}}\]
done
clear
C)
\[{{x}_{1}}-{{x}_{2}}-{{x}_{3}}\]
done
clear
D)
\[{{x}_{3}}-{{x}_{1}}-{{x}_{2}}\]
done
clear
View Answer play_arrow
In qualitative analysis, the metals of Group I can be separated from other ions by precipitating them as chloride salts. A solution initially contains \[A{{g}^{+}}\] and \[P{{b}^{2+}}\] at a concentration of 0.10 M. Aqueous HCl is added to this solution until the \[C{{l}^{-}}\]concentration is 0.10 M. What will the concentrations of \[A{{g}^{+}}\] and \[P{{b}^{2+}}\] be at equilibrium? \[({{K}_{sp}}\,for\,AgCl=1.8\times {{10}^{-10}},\]\[{{K}_{sp}}\,for\,PbC{{l}_{2}}=1.7\times {{10}^{-5}})\]
A)
\[[A{{g}^{+}}]=1.8\times {{10}^{-7}}M;\] \[[P{{b}^{2}}^{+}]=1.7\times {{10}^{-6}}M\]
done
clear
B)
\[[A{{g}^{+}}]=1.8\times {{10}^{-11}}M;\] \[[P{{b}^{2}}^{+}]=8.5\times {{10}^{-5}}M\]
done
clear
C)
\[[A{{g}^{+}}]=1.8\times {{10}^{-9}}M;\] \[[P{{b}^{2}}^{+}]=1.7\times {{10}^{-3}}M\]
done
clear
D)
\[[A{{g}^{+}}]=1.8\times {{10}^{-11}}M;\] \[[P{{b}^{2}}^{+}]=8.5\times {{10}^{-4}}M\]
done
clear
View Answer play_arrow
Carbon and CO gas are used to reduce which of the following pairs of metal oxides for extraction of metals?
A)
\[FeO,\]\[SnO\]
done
clear
B)
\[SnO,\]\[ZnO\]
done
clear
C)
\[BaO,\]\[N{{a}_{2}}{{O}_{2}}\]
done
clear
D)
\[FeO,\]\[ZnO\]
done
clear
View Answer play_arrow
Aqueous solution of group 2 is precipitated by adding \[N{{a}_{2}}C{{O}_{3}},\] then this precipitate is tested on flame, no light in visible region is observed, this element can be
A)
Ba
done
clear
B)
Mg
done
clear
C)
Ca
done
clear
D)
Sr
done
clear
View Answer play_arrow
Identify the correct order of electron affinity for \[{{O}^{-}},\]\[O,\]\[{{F}^{-}}\]and \[N{{a}^{+}}.\]
A)
\[N{{a}^{+}}<{{F}^{-}}<{{O}^{-}}<O\]
done
clear
B)
\[O<{{O}^{-}}<{{F}^{-}}<N{{a}^{+}}\]
done
clear
C)
\[{{O}^{-}}<O<<{{F}^{-}}<N{{a}^{+}}\]
done
clear
D)
\[{{F}^{-}}<N{{a}^{+}}<{{O}^{-}}<O\]
done
clear
View Answer play_arrow
A reaction at 1 bar is non-spontaneous at low temperature but becomes spontaneous at high temperature. Identify the correct statement about the reaction among the following:
A)
\[\Delta H\] is negative while \[\Delta S\] is positive.
done
clear
B)
Both \[\Delta H\] and \[\Delta S\] are negative.
done
clear
C)
\[\Delta H\] is positive while \[\Delta S\] is negative.
done
clear
D)
Both \[\Delta H\]and \[\Delta S\] are positive.
done
clear
View Answer play_arrow
Which of the following can not be isoelectronic?
A)
Two different cations
done
clear
B)
Two different anions
done
clear
C)
Cation and anion
done
clear
D)
Two different atoms
done
clear
View Answer play_arrow
When \[C{{H}_{3}}Cl\] and \[AlC{{l}_{3}}\] are used in Friedel-Crafts reaction, the electrophile is
A)
\[C{{l}^{+}}\]
done
clear
B)
\[AlC{{l}_{4}}^{-}\]
done
clear
C)
\[C{{H}_{3}}^{+}\]
done
clear
D)
\[AlC{{l}_{2}}^{+}\]
done
clear
View Answer play_arrow
The rate law for the reaction below is given by the expression k [A][B] A + B \[\to \] Product If the concentration of B is increased from 0.1 to 0.3 mole, keeping the value of A at 0.1 mole, the rate constant will be:
A)
3k
done
clear
B)
9k
done
clear
C)
k/3
done
clear
D)
k
done
clear
View Answer play_arrow
The alkaline earth metals Ba, Sr, Ca and Mg may be arranged in the order of their decreasing first ionisation potential as
A)
Mg, Ca, Sr, Ba
done
clear
B)
Ca, Sr, Ba, Mg
done
clear
C)
Sr, Ba, Mg, Ca
done
clear
D)
Ba, Mg, Ca, Sr
done
clear
View Answer play_arrow
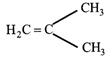
The monomer of the polymer;
A)
done
clear
B)
\[C{{H}_{3}}CH=CHC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}CH=C{{H}_{2}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}}C=C{{(C{{H}_{3}})}_{2}}\]
done
clear
View Answer play_arrow
Two electrolytic cells, one containing acidified ferrous chloride and another acidified ferric chloride, are connected in series. The ratio of iron deposited at cathodes in the two cells will be;
A)
3 : 1
done
clear
B)
2 : 1
done
clear
C)
1 : 1
done
clear
D)
3 : 2
done
clear
View Answer play_arrow
The living organisms can be unexceptionally distinguished from the non-living things on the basis of their ability for
A)
interaction with the environment and progressive evolution
done
clear
B)
reproduction
done
clear
C)
growth and movement
done
clear
D)
responsiveness to touch.
done
clear
View Answer play_arrow
The largest flower found is known as
A)
Rafflesia
done
clear
B)
Tecoma
done
clear
C)
Musa
done
clear
D)
Cauliflower
done
clear
View Answer play_arrow
Pulses belong to the family
A)
fabaceae
done
clear
B)
asteraceae
done
clear
C)
poaceae
done
clear
D)
solanaceae
done
clear
View Answer play_arrow
Which of the following are protozoans?
A)
diatoms, flagellates, amoebas, and ciliates
done
clear
B)
sporozoa, flagellates, amoebas, and ciliates
done
clear
C)
amoebas, actinomycetes, ciliates, and flagellates
done
clear
D)
flagellates, ciliates, cyanobacteria, and apicomplexans
done
clear
View Answer play_arrow
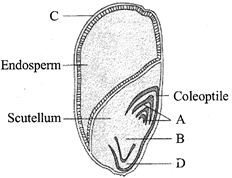
In a cereal grain the single cotyledon of embryo is represented by
A)
scutellum
done
clear
B)
prophyll
done
clear
C)
coleoptile
done
clear
D)
coleorrhiza
done
clear
View Answer play_arrow
A plant tissue when stained showed the presence of hemicellulose and pectin in cells wall of its cells. The tissue is called
A)
collenchyma
done
clear
B)
sclerenchyma
done
clear
C)
xylem
done
clear
D)
meristem
done
clear
View Answer play_arrow
Consider the following statements regarding the major pigments and stored food in the different groups of algae and choose the correct option.
(i) In Chlorophyceae, the stored food material is starch and the major pigments are chlorophyll a and d. (ii) In Phaeophyceae, laminarin is the stored food and major pigments are chlorophyll a and b. (iii) In Rhodophyceae, floridean starch is the stored food and the major pigments are chlorophyll a, d and phycoerythrin.
A)
(i) is correct, but (ii) and (iii) are wrong.
done
clear
B)
(i) and (ii) are correct, but (iii) is wrong.
done
clear
C)
(i) and (iii) are correct, but (ii) is wrong.
done
clear
D)
(iii) is correct, but (i) and (ii) are wrong.
done
clear
View Answer play_arrow
Chromosome duplication without nuclear division refers to
A)
Meiosis
done
clear
B)
Mitosis
done
clear
C)
Androgenesis
done
clear
D)
Endomitosis
done
clear
View Answer play_arrow
During which stages (or prophase 1 substages) of meiosis do you expect to find the bivalents and DNA replication respectively?
A)
Pachytene and interphase (between two meiotic divisions)
done
clear
B)
Pachytene and interphase (just prior to prophase I)
done
clear
C)
Pachytene and S phase (of interphase just prior to prophase I)
done
clear
D)
Zygotene and S phase (of interphase prior to prophase I)
done
clear
View Answer play_arrow
Sieve tubes are suited for translocation of food because they possess
A)
bordered pits.
done
clear
B)
no ends walls.
done
clear
C)
broader lumen and perforated cross walls.
done
clear
D)
no protoplasm.
done
clear
View Answer play_arrow
Identified A, B, C and D of a seed.
A)
A - Plumule; B - Radicle; C - Pericarp; D-Coleorhiza
done
clear
B)
A - Radicle; B - Plumule; C - Pericarp; D-Coleorhiza
done
clear
C)
A - Radicle; B - Plumule; C - Coleorhiza; D-Pericarp
done
clear
D)
A- Radicle; B - Coleorhiza; C - Plumule; D-Pericarp
done
clear
View Answer play_arrow
One of the most resistant biological material is
A)
lignin
done
clear
B)
hemicellulose
done
clear
C)
lignocellulose
done
clear
D)
sporopollenin
done
clear
View Answer play_arrow
Nuclear power stations even with adequate radiation safety measures generate
A)
thermal pollution of water bodies.
done
clear
B)
thermal pollution of soil.
done
clear
C)
noise polllution.
done
clear
D)
all of the above
done
clear
View Answer play_arrow
Apopulation of genetically identical individuals, obtained from asexual reproduction is
A)
Callus
done
clear
B)
Clone
done
clear
C)
Deme
done
clear
D)
Aggregate
done
clear
View Answer play_arrow
Which of the following floral parts forms pericarp after fertilization?
A)
Nucellus
done
clear
B)
Outer integument
done
clear
C)
Ovary wall
done
clear
D)
loner integument
done
clear
View Answer play_arrow
In which one of the following habitats does the diurnal temperature of soil surface vary most?
A)
Shrub land
done
clear
B)
Forest
done
clear
C)
Desert
done
clear
D)
Grassland
done
clear
View Answer play_arrow
In increasing order of organizational complexity, which one of the following is the correct sequence?
A)
Population, species, community, ecosystem
done
clear
B)
Population, variety, species, ecosystem
done
clear
C)
Population, ecosystem, species, community
done
clear
D)
Species, variety, ecosystem, community
done
clear
View Answer play_arrow
When secondary growth is initiated in dicot stem what will happen first?
A)
The cells of cambium divide periclinally to form xylem mother cells
done
clear
B)
Inter tascicular cabium join with intrafascicular cambium
done
clear
C)
Parenchymatous cells present between vascular bundle become meristematic
done
clear
D)
Pith get obliterated
done
clear
View Answer play_arrow
Osmosis means movement of
A)
solute from lower concentration to higher concentration.
done
clear
B)
solute from higher concentration to lower concentration.
done
clear
C)
solvent from lower concentration of solution to higher concentration of solution.
done
clear
D)
solvent from higher concentration of solution to lower concentration of solution.
done
clear
View Answer play_arrow
If the pressure potential is \[+0.16\] megapascals (mPa) and the osmotic potential is \[-0.24\] megapascals, then the water potential would be
A)
\[+0.4\,\,mPa\]
done
clear
B)
\[+0.08\,\,mPa\]
done
clear
C)
\[-0.08\,\,mPa\]
done
clear
D)
\[+0.16\,\,mPa.\]
done
clear
View Answer play_arrow
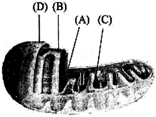
The figure below shows the structure of a mitochondrion with its four parts labelled [A], [B], [C] and [D].
Select the part correctly matched with its function.
A)
Part [D]: Outer membrane - gives rise to inner membrane by splitting
done
clear
B)
Part [B]: Inner membrane - forms infoldings called cristae
done
clear
C)
Part [C]: Cristae - possess single circular DNA molecule and ribosomes
done
clear
D)
Part [A]: Matrix - major site for respiratory chain enzymes
done
clear
View Answer play_arrow
Which of the following is correct set of micronutrient for plants?
A)
Mg, Si, Fe, Cu, Ca
done
clear
B)
Cu, Fe, Zn, B, Mn
done
clear
C)
Mg, Fe, Zn, B, Mn
done
clear
D)
Mo, Zn, Cl, Mg, Ca
done
clear
View Answer play_arrow
During \[N{{a}^{+}}-{{K}^{+}}\] pump
A)
\[3N{{a}^{+}}\] and \[2{{K}^{+}}\] are transported
done
clear
B)
\[1N{{a}^{+}}\] and \[2{{K}^{+}}\] are transported
done
clear
C)
\[3N{{a}^{+}}\] and \[3{{K}^{+}}\] are transported
done
clear
D)
Depends on requirement of cell
done
clear
View Answer play_arrow
The technique of obtaining large number of plantlets by tissue culture method is called
A)
Plantlet culture
done
clear
B)
Organ culture
done
clear
C)
Micropropagation
done
clear
D)
Macropropagation
done
clear
View Answer play_arrow
Which of the following is a recessive autosomal disorder
A)
Cat-cry syndrome
done
clear
B)
Haemophilia
done
clear
C)
Huntington's chorea
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which one of the following pairs, is not correctly matched?
A)
Gibberellic acid - Leaf fall
done
clear
B)
Cytokinin - Cell wall elongation
done
clear
C)
IAA - Cell wall elongation
done
clear
D)
Abscissic acid - Stomatal closure.
done
clear
View Answer play_arrow
Calculate the percentage of pureline individuals from a cross between AaBbCcDdee \[\times \] aabbccddee
A)
50%
done
clear
B)
25%
done
clear
C)
12.5%
done
clear
D)
6.25%
done
clear
View Answer play_arrow
Mutations can be induced with
A)
infrared radiations
done
clear
B)
IAA
done
clear
C)
ethylene
done
clear
D)
gamma radiations
done
clear
View Answer play_arrow
Which of following methods was not used by Mendel in his study of the genetics of the garden pea?
A)
Maintenance of true-breeding lines
done
clear
B)
Cross-pollination
done
clear
C)
Microscopy
done
clear
D)
Production of hybrid plants
done
clear
View Answer play_arrow
Pneumatophores are characteristic of plants growing in
A)
saline soils
done
clear
B)
sandy soils
done
clear
C)
marshy places and salt lakes
done
clear
D)
dryland regions
done
clear
View Answer play_arrow
What is antisense technology?
A)
A cell displaying a foreign antigen used for synthesis of antigens
done
clear
B)
Production of somaclonal variants in tissue cultures
done
clear
C)
When a piece of RNA that is complementary in sequence is used to stop expression of a specific gene
done
clear
D)
RNA polymerase producing DNA
done
clear
View Answer play_arrow
The reaction rate of salivary amylase on starch decreases as the concentration of chloride ions is reduced. Which of the following describes the role of the chloride ions?
A)
allosteric inhibitors
done
clear
B)
co-enzymes
done
clear
C)
co-factors
done
clear
D)
competitive inhibitors
done
clear
View Answer play_arrow
The enzymes hexokinase which catalyses glucose to glucose-6-phosphate in glycolysis is inhibited by glucose-6-phosphate. This is an example of
A)
competitive inhibition
done
clear
B)
non-competitive inhibition
done
clear
C)
feedback allosteric inhibition
done
clear
D)
positive feedback
done
clear
View Answer play_arrow
Apical dominance in plants is caused by
A)
high concentration of auxins in the terminal bud.
done
clear
B)
high concentration of gibberellins in the apical bud.
done
clear
C)
high concentration of auxins in the lateral bud.
done
clear
D)
absence of auxins and gibberellins in apical bud.
done
clear
View Answer play_arrow
Primary precursor of IAA is
A)
phenylalanine
done
clear
B)
tyrosine
done
clear
C)
tryptophan
done
clear
D)
leucine
done
clear
View Answer play_arrow
Which of the following statements are correct?
(i) Magnesium competes with iron and manganese for uptake and with iron for binding with enzymes. (ii) Magnesium inhibit calcium translocation in shoot apex. (iii) Excess of manganese may induce deficiencies of iron, magnesium and calcium. (iv) Symptoms of manganese toxicity may actually be the deficiency symptoms of iron, magnesium and calcium.
A)
(i), (ii) and (iii)
done
clear
B)
(i) and (ii)
done
clear
C)
(iii) and (iv)
done
clear
D)
(ii), (iii) and (iv)
done
clear
View Answer play_arrow
Which one of the following structural formulae of two organic compounds is correctly identified along with its related function? A.
B.
A)
B: Adenine \[-\] a nucleotide that makes up nucleic acids
done
clear
B)
A: Triglyceride \[-\] major source of energy
done
clear
C)
B: Uracil \[-\] a component of DNA
done
clear
D)
A: Lecithin \[-\] a component of cell mealyane
done
clear
View Answer play_arrow
Silencing of mRNA has been used in producing transgenic plants resistant to:
A)
bollworms
done
clear
B)
nematodes
done
clear
C)
white rusts
done
clear
D)
bacterial blights
done
clear
View Answer play_arrow
Expression vectors are different from other vectors because they
A)
contain drug resistance markers.
done
clear
B)
contain telomeres.
done
clear
C)
contain regulatory regions that permit the cloned DNA to produce a gene product
done
clear
D)
contain DNA origins
done
clear
View Answer play_arrow
Continuous addition of sugars in 'fed batch' fermentation is done to:
A)
produce methane
done
clear
B)
obtain antibiotics
done
clear
C)
purify enzymes
done
clear
D)
degrade sewage
done
clear
View Answer play_arrow
Cell elongation in intern odal regions of the green plants takes place due to
A)
indole acetic acid
done
clear
B)
cytokinins
done
clear
C)
gibberellins
done
clear
D)
ethylene
done
clear
View Answer play_arrow
The 'Earth Summit' held at Rio de Janerio in 1992 resulted into
A)
Compilation of Red list
done
clear
B)
Establishment of Biosphere Reserves
done
clear
C)
Convention on Biodiversity
done
clear
D)
Development of Hot Spots of Biodiversity
done
clear
View Answer play_arrow
Tobacco plants resistant to a nematode have been developed by the introduction of DNA that produced (in the host cells)
A)
both sense and anti-sense RNA
done
clear
B)
a particular hormone
done
clear
C)
an antifeedant
done
clear
D)
a toxic protein
done
clear
View Answer play_arrow
Chloramphenicol and erythromycin (broad spectrum antibiotics) are produced by
A)
Streptomyces
done
clear
B)
Nitrobacter
done
clear
C)
Rhizobium
done
clear
D)
Penicillium
done
clear
View Answer play_arrow
For biogas production besides dung an extensive use of which weed is recommended in our country-
A)
Mangifera indica
done
clear
B)
Hydrilla
done
clear
C)
Eicchornia crassipes
done
clear
D)
Solanum
done
clear
View Answer play_arrow
Which one of the following statements about Mycoplasma is wrong?
A)
They are pieomorphic.
done
clear
B)
They are sensitive to penicillin.
done
clear
C)
They cause diseases in plants.
done
clear
D)
They are also called (Pleuro pneumonia like organisms) PPLO.
done
clear
View Answer play_arrow
Housefly possesses
A)
two pairs of wings
done
clear
B)
one pair of wings
done
clear
C)
three pairs of wings
done
clear
D)
four pair of wings
done
clear
View Answer play_arrow
An example of a holocrine gland is
A)
A sweat gland
done
clear
B)
A salivary gland
done
clear
C)
A pancreatic gland
done
clear
D)
A sebaceous gland
done
clear
View Answer play_arrow
Major inorganic component of vertebrate bone is
A)
calcium carbonate
done
clear
B)
calcium phosphate
done
clear
C)
sodium hydroxide
done
clear
D)
potassium hydroxide
done
clear
View Answer play_arrow
Which of the following is a large middle layer consisting of main uterine gland as well as blood vessels?
A)
Stratum granulosum
done
clear
B)
Stratum lucidium
done
clear
C)
Stratum comeum
done
clear
D)
Stratum spongiosum
done
clear
View Answer play_arrow
The largest corpuscles of human blood are
A)
Neutrophils
done
clear
B)
Monocyte
done
clear
C)
Lymphocyte
done
clear
D)
Eosinophils
done
clear
View Answer play_arrow
A hormone which will increase blood sugar while increasing cardiac output is
A)
norepinephrine
done
clear
B)
acetylcholine
done
clear
C)
antidiuretic hormone
done
clear
D)
insulin
done
clear
View Answer play_arrow
Which one of the following statements is correct with respect to salt water balance inside the body of living organisms?
A)
When water is not available, camels do not produce urine but store urea in tissues.
done
clear
B)
Salmon fish excretes lot of stored salt through gill membrane in fresh water.
done
clear
C)
Paramecium discharges concentrated salt solution by contractile vacuoles.
done
clear
D)
The body fluids of fresh water animals are generally hypotonic to surrounding water.
done
clear
View Answer play_arrow
The given figure shows the digestive system of cockroach with few structures marked as A, B, C and D.
Identify structures A to D.
A)
A-Gizzard, B-Crop- C-Hepatic caecae, D- Malpighian tubules
done
clear
B)
A-Crop, B-Gizzard, C-Hepatic caecae, D- Malpighian tubules
done
clear
C)
A- Crop, B-Gizzard, C-Malpighian tubules, D-Hepatic caecae
done
clear
D)
A- Gizzard, B-Crop, C-Malpighian tubules, D-Hepatic caecae
done
clear
View Answer play_arrow
Tying up or removing a small part of fallopian duct is called
A)
Vasectomy
done
clear
B)
Ductus arteriosus
done
clear
C)
Archidectomy
done
clear
D)
Tubectomy
done
clear
View Answer play_arrow
The technique called Gamete Intrafallopian Transfer (GIFT) is recommended for those females:
A)
who cannot produce an ovum
done
clear
B)
who cannot retain the foetus inside uterus.
done
clear
C)
whose cervical canal is too narrow to allow passage for the sperms
done
clear
D)
who cannot provide suitable environment for fertilization
done
clear
View Answer play_arrow
Fertilization occurs in, human, rabbit and other placental mammals in-
A)
Ovary
done
clear
B)
Uterus
done
clear
C)
Fallopian tubes
done
clear
D)
Vagina
done
clear
View Answer play_arrow
Which of the following statement(s) is/are correct regarding class aves?
(i) The forelimbs are modified into wings and the hindlimbs generally have scales and are modified for walking, swimming or clasping the tree branches. (ii) Heart is completely four-chambered. (iii) They are warm- blooded (homoiothermous) animals i.e., they are able to maintain a constant body temperature. (iv) They are oviparous and development is direct.
A)
Both (i) and (iii)
done
clear
B)
Both (i) and (iv)
done
clear
C)
(i), (ii) and (iii)
done
clear
D)
All of these
done
clear
View Answer play_arrow
Birth control pills check ovulation in female by inhibiting the secretion of-
A)
follicle stimulating hormone
done
clear
B)
luteinizing hormone
done
clear
C)
Both [a] and [b]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
If you examine the reproductive system of a normal human female before she undergoes puberty, which of the following would you not expect to find?
A)
Corpus luteum
done
clear
B)
Ovaries
done
clear
C)
Primary oocytes
done
clear
D)
Oviducts
done
clear
View Answer play_arrow
What happens during fertilisation in humans after many sperms reach close to the ovum?
A)
Cells of corona radiata trap all the sperms except one
done
clear
B)
Only two sperms nearest the ovum penetrate zona pellucida
done
clear
C)
Secretions of acrosome helps one Sperm enter cytoplasm of ovum through zona pellucida
done
clear
D)
All sperms except the one nearest to the ovum lose their tails
done
clear
View Answer play_arrow
Coronary artery disease (CAD) is often referred to as
A)
Heart failure
done
clear
B)
Cardiac arrest
done
clear
C)
Atherosclerosis
done
clear
D)
Thrombosis
done
clear
View Answer play_arrow
Antidiuretic hormone
A)
Secretion is determined by plasma osmolarity
done
clear
B)
Increases permeability of renal collecting duct cells to water
done
clear
C)
Is secreted by nerve cells with their cell bodies in hypothalamus
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which one of the following is a matching pair of a substrate and its particular digestive enzyme?
A)
Starch \[-\] amylase
done
clear
B)
Lactose \[-\] rennin
done
clear
C)
Maltose \[-\] steapsin
done
clear
D)
Casein \[-\] chymotrypsin
done
clear
View Answer play_arrow
Which one of the following pairs of the cells with their secretion is correctly matched?
A)
Oxyntic cells - A secretion with pH between 2.0 and 3.0.
done
clear
B)
Alpha cells of Islets of Langerhans - Secretion that decreases blood sugar level.
done
clear
C)
Kupffer cells - A digestive enzyme that hydrolysis nucleic acids.
done
clear
D)
Sebaceous glands - A digestive enzyme that hydrolysis nucleic acids
done
clear
View Answer play_arrow
Bulk of carbon dioxide \[(C{{O}_{2}})\] released from body tissues into the blood is present as
A)
bicarbonate in blood plasma and RBCs
done
clear
B)
free \[C{{O}_{2}}\] in blood plasma
done
clear
C)
25% carbamino-haemoglobin and 75% as bicarbonate
done
clear
D)
carbamino-haemoglobin in RBCs
done
clear
View Answer play_arrow
'Bundle of His' is a part of which one of the following organs in humans?
A)
Brain
done
clear
B)
Heart
done
clear
C)
Kidney
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
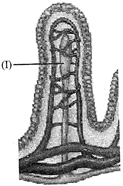
The given figure shows a section of small intestinal mucosa showing villi. What is the function of structure marked as I in the given figure?
A)
To absorb amino acids.
done
clear
B)
To carry blood.
done
clear
C)
To transport fat
done
clear
D)
To transport glucose
done
clear
View Answer play_arrow
Ban-body in mammals represents
A)
all the heterochromatin in female cells
done
clear
B)
Y-chromosomes in somatic cells of male
done
clear
C)
all heterochromatin in male and female cells
done
clear
D)
one of the two X-chromosomes in somatic cells of females
done
clear
View Answer play_arrow
Genetic drift is change of
A)
gene frequency in same generation
done
clear
B)
appearance of recessive genes
done
clear
C)
gene frequency from one generation to next
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Two different species cannot live for long duration in the same niche or habitat. This law is
A)
Alien's law
done
clear
B)
Cause's hypothesis
done
clear
C)
Dollo's rule
done
clear
D)
Weisman's theory
done
clear
View Answer play_arrow
Which one of the following statements is correct in relation to honey bees?
A)
Apis indica is the largest wild honey bee in India
done
clear
B)
Honey is predominantly sucrose and arabinose
done
clear
C)
Beewax is a waste product of honey bees
done
clear
D)
Communication in honey bees was discovered by Karl Von Frisch
done
clear
View Answer play_arrow
Select the incorrect statement from the following:
A)
Galactosemia is an inborn error of metabolism
done
clear
B)
Small population size results in random genetic drift in a population
done
clear
C)
Baldness is a sex -limited trait
done
clear
D)
Linkage is an exception to the principle of independent assortment in heredity
done
clear
View Answer play_arrow
Biometric genetics deals with:
A)
the biochemical explanations of various genetical phenomena
done
clear
B)
the effect of environment on genetic set up organisms
done
clear
C)
the genetical radiations on the living organisms
done
clear
D)
the inheritance of quantitative traits
done
clear
View Answer play_arrow
In which part of the body honey bee keep the nectar for sometime?
A)
Crop
done
clear
B)
Mouth
done
clear
C)
Gizzard
done
clear
D)
Pollen basket
done
clear
View Answer play_arrow
Identify the molecules [A] and [B] shown below and select the right option giving their source and use.
[A] [B]
Options
A)
Molecule ? [A] Cocaine Source - Erythroxylum coca Use - Accelerates the transport of dopamine
done
clear
B)
Molecule ? [B] Heroin Source - Cannabis sativa Use - Depressant and slows down body functions
done
clear
C)
Molecule ? [B] Cannabinoid Source - Atropa belladona Use - Produces hallucinations
done
clear
D)
Molecule ? [A] Morphine Source - Papaver somniferum Use - Sedative and pain killer
done
clear
View Answer play_arrow
The technique used for estimation of minute amounts of hormones and drugs is called
A)
electrophoresis
done
clear
B)
electroencephalogram
done
clear
C)
fractionation
done
clear
D)
radioimmunoassay
done
clear
View Answer play_arrow
Muscle A and muscle B are the same size, but muscle A is capable of much finer control than muscle B. Which one of the following is likely to be true of muscle A?
A)
It contains fewer motor units than muscle B.
done
clear
B)
It has larger sarcomeres than muscle B.
done
clear
C)
It is controlled by more neurons than muscle B.
done
clear
D)
It is controlled by fewer neurons than muscle B.
done
clear
View Answer play_arrow
During oxygen transport the oxyhaemoglobin at the tissue level liberates oxygen to the cells because in tissue
A)
\[{{O}_{2}}\] concentration is high and \[C{{O}_{2}}\] is low
done
clear
B)
\[{{O}_{2}}\] concentration is low and \[C{{O}_{2}}\] is high
done
clear
C)
\[{{O}_{2}}\] tension is high and \[C{{O}_{2}}\] tension is low
done
clear
D)
\[{{O}_{2}}\] tension is low and \[C{{O}_{2}}\] tension is high
done
clear
View Answer play_arrow
Select the correct statement regarding the specific disorder of muscular or skeletal system :
A)
Myasthenia gravis - Autoimmune disorder which inhibits sliding of myosin filaments.
done
clear
B)
Gout - inflammation of joints due to extra deposition of calcium.
done
clear
C)
Muscular dystrophy - age related shortening of muscles.
done
clear
D)
Osteoporosis - decrease in bone mass and higher chances of fractures with advancing age.
done
clear
View Answer play_arrow
Which one of the following pairs is incorrectly matched?
A)
Glucagon - Beta cells (source)
done
clear
B)
Somatostatin - Delta cells (source)
done
clear
C)
Corpus luteum - Progesterone (secretion)
done
clear
D)
Insulin - Diabetes mellitus (disease)
done
clear
View Answer play_arrow
Melanocyte stimulating hormone (MSH) is produced by
A)
parathyroid
done
clear
B)
pars intermedia of pituitary
done
clear
C)
anterior pituitary
done
clear
D)
posterior pituitary
done
clear
View Answer play_arrow
Cyclosporine is used as :
A)
Allergic eczema
done
clear
B)
Immunosuppressant
done
clear
C)
Prophylactic for viruses
done
clear
D)
Prophylactic for marasmus
done
clear
View Answer play_arrow
A major component of gobar gas is ?
A)
Butane
done
clear
B)
Ammonia
done
clear
C)
Methane
done
clear
D)
Ethane
done
clear
View Answer play_arrow
Sour taste of vinegar is due to
A)
lactic acid
done
clear
B)
acetic acid
done
clear
C)
butyric acid
done
clear
D)
fumaric acid
done
clear
View Answer play_arrow
Which of the following do sticky ends andnucleic acid probes have in common?
A)
They both are used as gene vectors in genetic engineering.
done
clear
B)
They both involve complementary base pairing.
done
clear
C)
They both are parts of RNA molecules.
done
clear
D)
They both are produced by the action of restriction enzymes.
done
clear
View Answer play_arrow
Which of the following statements regarding the multiple histocompatibility complex (MHC) is true?
A)
An individual produces only one specific MHC glycoproteins.
done
clear
B)
MHC glycoproteins normally are found only on lymphocytes and macrophages.
done
clear
C)
RBC blood groups are based on MHC antigens
done
clear
D)
Antigens are presented to T-cells on MHC glycoproteins.
done
clear
View Answer play_arrow
A person likely to develop tetanus is immunized by administering
A)
preformed antibodies
done
clear
B)
wide spectrum antibiotics
done
clear
C)
weakened germs
done
clear
D)
dead germs
done
clear
View Answer play_arrow
cDNA probes are copied from the messenger RNA molecules with the help of-
A)
Restriction enzymes
done
clear
B)
Reverse transcriptase
done
clear
C)
DNA polymerase
done
clear
D)
Adenosine deaminase
done
clear
View Answer play_arrow
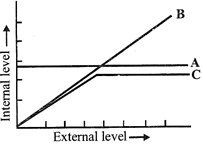
The figure given below is a diagrammatic representation of response of organisms to abiotic factors. What do A, B and C represent respectively?
A)
A - Conformer B - Regulator C - Partial regulator
done
clear
B)
A - Regulator B - Partial regulator C - Conformer
done
clear
C)
A - Partial regulator B - Regulator C - Conformer
done
clear
D)
A - Regulator B - Conformer C - Partial regulator
done
clear
View Answer play_arrow
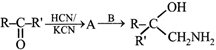
 Select the part correctly matched with its function.
Select the part correctly matched with its function. 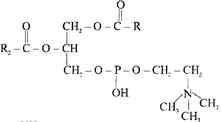 B.
B. 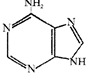
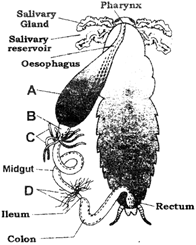 Identify structures A to D.
Identify structures A to D.