Which of the junction diodes shown in figure is forward biased?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The velocity v of a particle at time t is given by\[\nu =at+\frac{b}{t+c}\], where, a, b and c are constants. The dimensions of a, b and c are
A)
\[[L],\,[LT]\,\,and\,\,[L{{T}^{-\,2}}]\]
done
clear
B)
\[[L{{T}^{-\,2}}],\,\,[L]\,\,and\,\,[T]\]
done
clear
C)
\[[{{L}^{\,2}}],\,\,[T]\,\,and\,\,[L{{T}^{-\,2}}]\]
done
clear
D)
\[[L{{T}^{-\,2}}],\,[LT]\,and\,[L]\]
done
clear
View Answer play_arrow
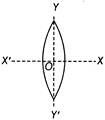
An equiconvex lens is cut into two halves along (i) XOX' and (ii) YOY' as shown in the figure. f, f', f' be the focal lengths of the complete lens, of each half in case (i), and of each half in case (ii), respectively, choose the correct statement from the following.
A)
\[\operatorname{f}'\,\,=\,\,f,\,\,f''=\,\,2f\]
done
clear
B)
\[\operatorname{f}'\,\,=\,\,2f,\,\,f''=\,\,f\]
done
clear
C)
\[\operatorname{f}'\,\,=\,\,f,\,\,f''=\,\,f\]
done
clear
D)
\[\operatorname{f}'\,\,=\,\,2f,\,\,f''=\,\,2f\]
done
clear
View Answer play_arrow
Equation of progressive wave is given by \[y=\sin 4\left[ \pi \left( \frac{t}{5}-\frac{x}{9} \right)+\frac{\pi }{6} \right]\] where y, x are in cm and t is in seconds. Then, which of the following is correct?
A)
\[\operatorname{v} = 5 cm\]
done
clear
B)
\[\lambda =18cm\]
done
clear
C)
\[a= 0.04 cm\]
done
clear
D)
\[f=50\,\,Hz\]
done
clear
View Answer play_arrow
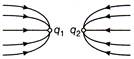
The given figure gives electric lines of force due to two charges\[{{\operatorname{q}}_{1}}\,\,and\,\,{{q}_{2}}\]. What are the sign of the two charges?
A)
\[{{\operatorname{q}}_{1}}\] is positive but \[{{q}_{2}}\] is negative
done
clear
B)
\[{{\operatorname{q}}_{1}}\] is negative but \[{{q}_{2}}\] is positive
done
clear
C)
Both are negative
done
clear
D)
Both are positive
done
clear
View Answer play_arrow
A capacitor of capacity \[{{C}_{1}}\] charged up to V volt and then connected to an uncharged capacitor of capacity \[{{C}_{2}}\] in parallel. Then, final potential difference across each will be
A)
\[\frac{{{C}_{2}}V}{{{C}_{1}}+{{C}_{2}}}\]
done
clear
B)
\[\frac{{{C}_{1}}V}{{{C}_{1}}+{{C}_{2}}}\]
done
clear
C)
\[\left( 1+\frac{{{C}_{2}}}{{{C}_{1}}} \right)\]
done
clear
D)
\[\left( 1-\frac{{{C}_{2}}}{{{C}_{1}}} \right)V\]
done
clear
View Answer play_arrow
Two similar coils of radius R are lying concentrically with their planes at right angles to each other. The current flowing in them are I and 2I, respectively. The resultant magnetic field induction at the centre will be
A)
\[\frac{\sqrt{5}{{\mu }_{0}}I}{2\,R}\]
done
clear
B)
\[\frac{3{{\mu }_{0}}I}{2\,R}\]
done
clear
C)
\[\frac{{{\mu }_{0}}I}{2\,R}\]
done
clear
D)
\[\frac{{{\mu }_{0}}I}{R}\]
done
clear
View Answer play_arrow
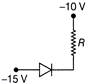
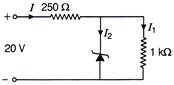
A Zener diode having breakdown voltage equal to 15 V, is used in a voltage regulator. Circuit as shown in figure, the current through the diode is
A)
5 mA
done
clear
B)
10 mA
done
clear
C)
15 mA
done
clear
D)
20 mA
done
clear
View Answer play_arrow
A 200 V input is supplied to a transformer. The output circuit draws a current of 2.0 A at 440 V. If efficiency of transformer is \[80\,%\], the current drawn by the primary winding of the transformer is
A)
3.6 A
done
clear
B)
2.8 A
done
clear
C)
2.5 A
done
clear
D)
5.0 A
done
clear
View Answer play_arrow
A vibration magnetometer is placed in magnetic meridian has a small bar magnet. The magnet executes oscillations with a time period of 2 s in earth's horizontal magnetic field of 24 microtesla. When a horizontal field of 18 microtesla is produced opposite to the earth?s field by placing a current carrying wire, the new time period of magnet will be
A)
1 s
done
clear
B)
2 s
done
clear
C)
3 s
done
clear
D)
4 s
done
clear
View Answer play_arrow
A gramophone record is revolving with an angular velocity\[\omega \]. A coin is placed at a distance r from the centre of the record. The static coefficient of friction is \[\mu \] The coin will revolve with the record, if
A)
\[r=\mu g{{\omega }^{2}}\]
done
clear
B)
\[r<\,\,\frac{{{\omega }^{2}}}{\mu g}\]
done
clear
C)
\[r\le \,\,\frac{\mu g}{{{\omega }^{2}}}\]
done
clear
D)
\[r\ge \,\,\frac{\mu g}{{{\omega }^{2}}}\]
done
clear
View Answer play_arrow
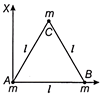
Three particles, each of mass m gram, are situated at the vertices of an equilateral triangle ABC of side l cm (as shown in figure). The moment of inertia of the system about a line AX perpendicular to AB and in plane of ABC, in \[\operatorname{gram}-c{{m}^{2}}\] units will be
A)
\[\frac{3}{4}m{{l}^{2}}\]
done
clear
B)
\[2m{{l}^{2}}\]
done
clear
C)
\[\frac{5}{4}\,m{{l}^{2}}\]
done
clear
D)
\[\frac{3}{2}\,m{{l}^{2}}\]
done
clear
View Answer play_arrow
The thermodynamic process is shown in the figure, the pressure and volume corresponding to some points in the figure are
\[{{p}_{A}}=3\times {{10}^{4}}\,Pa;\,\,{{V}_{A}}=2\times {{10}^{-\,3}}\,{{m}^{3}}\] \[{{p}_{B}}=8\times {{10}^{4}}\,Pa;\,\,{{V}_{D}}=5\times {{10}^{-\,3}}\,{{m}^{3}}\] In the process AB, 600 J of heat is added to the system and in process BC, 200 J of heat is added to the system. The change in internal energy of the system in process AC would be
A)
560 J
done
clear
B)
800 J
done
clear
C)
600 J
done
clear
D)
640 J
done
clear
View Answer play_arrow
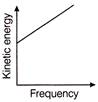
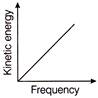
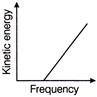
According to Einstein's photoelectric equation, the graph between the kinetic energy of photoelectrons ejected, and the frequency of incident radiation is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
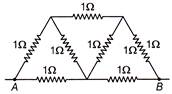
In the network shown in figure each resistance is\[1\,\Omega \]. The effective resistance between A and B is
A)
\[\frac{4}{3}\,\,\Omega \]
done
clear
B)
\[\frac{3}{2}\,\,\Omega \]
done
clear
C)
\[7\,\,\Omega \]
done
clear
D)
\[\frac{8}{7}\,\,\Omega \]
done
clear
View Answer play_arrow
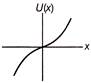
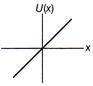
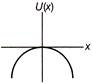
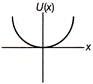
A particle is executing linear simple harmonic motion about origin\[\operatorname{x} = 0\]. Which of the group shown in figure represent the variation of the potential energy function U(x) versus x?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
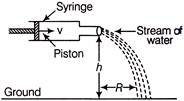
A syringe containing water is held horizontally with its nozzle at a height h above the ground as shown in figure. The cross-sectional areas of the piston and the nozzle are A and a respectively. The piston is pushed with a constant speed v. The horizontal range R of the stream of water on the ground is
A)
\[R=v\sqrt{\frac{2h}{g}}\]
done
clear
B)
\[R=v\sqrt{\frac{h}{2g}}\]
done
clear
C)
\[R=\frac{av}{A}\sqrt{\frac{2h}{g}}\]
done
clear
D)
\[R=\frac{Av}{a}\sqrt{\frac{2h}{g}}\]
done
clear
View Answer play_arrow
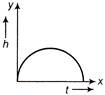
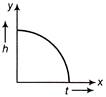
Which of the following is the altitude-time graph (figure) for a projectile thrown horizontally form the top of the tower?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A body is projected at an angle \[\theta \] with the horizontal. When it is at highest point, the ratio of the potential and kinetic energies of the body is
A)
\[\tan \,\theta \]
done
clear
B)
\[{{\tan }^{2}}\,\theta \]
done
clear
C)
\[\frac{2u}{g\,\sin \,\theta }\]
done
clear
D)
\[\frac{u}{g\,\sin \,\theta }\]
done
clear
View Answer play_arrow
The power of a biconvex lens is 10 dioptre and the radius of curvature of each surface is 10 cm. Then, the refractive index of the material of the lens is
A)
\[\frac{3}{2}\]
done
clear
B)
\[\frac{4}{3}\]
done
clear
C)
\[\frac{9}{8}\]
done
clear
D)
\[\frac{5}{3}\]
done
clear
View Answer play_arrow
Two beams of light of intensity \[{{I}_{1}}\,\,and\,\,{{I}_{2}}\] interfere to give an interference pattern. If the ratio of maximum intensity to minimum intensity is \[\frac{25}{9}\]then \[{{I}_{1}}\,/\,\,{{I}_{2}}\] is
A)
5/3
done
clear
B)
4
done
clear
C)
81/625
done
clear
D)
16
done
clear
View Answer play_arrow
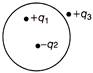
Figure shows a spherical Gaussian surface and a charge distribution. When calculating the flux of electric field through the Gaussian surface, the electric field will be due to
A)
\[+\,{{q}_{3}} alone\]
done
clear
B)
\[+\,{{q}_{1}} and\,\,+{{q}_{3}}\]
done
clear
C)
\[+\,{{q}_{1}}+{{q}_{3}}\,and\,\,-{{q}_{2}}\]
done
clear
D)
\[+\,{{q}_{1}}\,\,and\,\,-{{q}_{2}}\]
done
clear
View Answer play_arrow
The magnifying power of objective of a compound microscope is 5. If the magnifying power of microscope is 30, then magnifying power of eyepiece will be
A)
0.17
done
clear
B)
6
done
clear
C)
3
done
clear
D)
25
done
clear
View Answer play_arrow
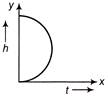
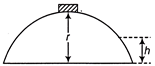
A small body of mass m slides down from the top of a hemisphere of radius r (figure). The surface of block and hemisphere are frictionless. The height at which the body loose contact with the surface of sphere is
A)
\[\frac{3}{2}\,r\]
done
clear
B)
\[\frac{2}{3}\,r\]
done
clear
C)
\[\frac{1}{2}\,g{{t}^{2}}\]
done
clear
D)
\[\frac{{{v}^{2}}}{2g}\]
done
clear
View Answer play_arrow
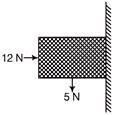
A block of weight 5 N is pushed against vertical wall by a force 12 N. The coefficient of friction between the wall and block is 0.6. The magnitude of the force exerted by the wall on the block is
A)
12 N
done
clear
B)
5 N
done
clear
C)
7.2 N
done
clear
D)
13 N
done
clear
View Answer play_arrow
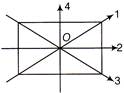
The moment of inertia of a thin square plate ABCD (figure) of uniform thickness about an axis passing through the centre of and perpendicular to the plane of plate is
A)
\[{{l}_{1}}+{{l}_{3}}\]
done
clear
B)
\[{{l}_{2}}+{{l}_{3}}+{{l}_{4}}\]
done
clear
C)
\[{{l}_{2}}+{{l}_{3}}\]
done
clear
D)
\[{{l}_{1}}+{{l}_{2}}+{{l}_{3}}+{{l}_{4}}\]
done
clear
View Answer play_arrow
The work done in turning a magnet, moment M by an angle of \[90{}^\circ \] from the meridian is n times the corresponding work done to turn it through an angle of \[60{}^\circ \].
A)
\[n=\frac{1}{2}\]
done
clear
B)
\[\operatorname{n} = 2\]
done
clear
C)
\[\operatorname{n} = \frac{1}{4}\]
done
clear
D)
\[\operatorname{n}=1\]
done
clear
View Answer play_arrow
A student A does the capillary rise experiment at equator and gets the surface tension value\[{{T}_{A}}\]. Another student does the same experiment in same conditions at poles and gets the surface tension value\[{{T}_{B}}\].
A)
\[{{T}_{A}}>{{T}_{B}}\]
done
clear
B)
\[{{T}_{A}}<{{T}_{B}}\]
done
clear
C)
\[{{T}_{A}}={{T}_{B}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
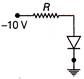
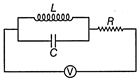
In the circuit of given figure, the current following through resistance R at resonance is
A)
zero
done
clear
B)
\[\operatorname{V}/R\]
done
clear
C)
\[\frac{\operatorname{V}}{\sqrt{{{R}^{2}}+{{\omega }^{2}}{{L}^{2}}}}\]
done
clear
D)
\[\frac{\operatorname{V}}{\sqrt{{{R}^{2}}+\,\,(1/\omega {{L}^{2}})}}\]
done
clear
View Answer play_arrow
Two particles A and B are projected with same speed so that the ratio of their maximum height reached is\[3 :1\]. If the speed of A is doubled without altering other parameters, the ratio of the horizontal ranges obtained by A and B is
A)
\[1:1\]
done
clear
B)
\[2:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[3:2\]
done
clear
View Answer play_arrow
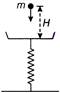
Particle each of mass m are falling into weighing pan from height H at a rate of n particles per second. Find reading of weighing pan after time T, if collision of particle with weighing pan is perfectly inelastic. (Neglect oscillation of weighing pan).
A)
\[\operatorname{mgH} + mgnT\]
done
clear
B)
\[mn\,(gT\,+\,\,\sqrt{2\,\,gH})\]
done
clear
C)
\[\operatorname{mng}\,\sqrt{2\,gH}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Choose the incorrect option.
A)
If electric field has zero value at a certain point the electric potential has to be zero at that point
done
clear
B)
If electric potential has zero value at a certain point then electric field has to be zero at that point
done
clear
C)
If electric field at certain point is non-zero then electric potential has to be non-zero at that point
done
clear
D)
None of these are correct
done
clear
View Answer play_arrow
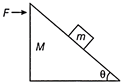
Find the force F to be applied on M, so that m remains stationary with respect to M?
A)
\[\left( M+m \right)g\]
done
clear
B)
\[\left( M + m \right) g\,sin\,\theta \]
done
clear
C)
\[\left( M+m \right)g\,cos\,\theta \]
done
clear
D)
\[\left( M+m \right)g\,\tan \,\theta \]
done
clear
View Answer play_arrow
Two short magnets have equal pole strengths but one is twice as long at the other. The shorter magnet is placed 20 cm in tan A position from the compass needle, the longer magnet must be placed on the other side of the magnetometer for no deflection at a distance equal to
A)
\[20\times {{\left( 2 \right)}^{1/3}}\,cm\]
done
clear
B)
\[20\times {{\left( 2 \right)}^{2/3}}\,cm\]
done
clear
C)
20 cm
done
clear
D)
\[20\times \left( 2 \right)\,\,cm\]
done
clear
View Answer play_arrow
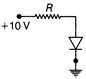
The potential difference between A and B in the following figure is
A)
12 V
done
clear
B)
24 V
done
clear
C)
48 V
done
clear
D)
None of these
done
clear
View Answer play_arrow
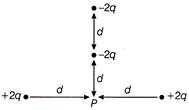
In figure shown what will be the net potential at point P due to four point charges?
A)
\[\frac{q}{4\pi {{\varepsilon }_{0}}d}\]
done
clear
B)
\[\frac{-2q}{4\pi {{\varepsilon }_{0}}d}\]
done
clear
C)
\[\frac{q}{4\pi {{\varepsilon }_{0}}d}\]
done
clear
D)
\[\frac{-q}{4\pi {{\varepsilon }_{0}}d}\]
done
clear
View Answer play_arrow
The length of a metal wire is \[{{L}_{1}}\] when the tension in it is \[{{T}_{1}}\,and\,\,{{L}_{2}}\] when tension in it is \[{{T}_{2}}\]. The original length of the wire is
A)
\[\frac{\sqrt{{{T}_{1}}{{T}_{2}}}}{{{L}_{1}}+{{L}_{2}}}\]
done
clear
B)
\[\frac{{{T}_{1}}{{L}_{1}}\,-\,\,{{T}_{2}}{{L}_{2}}}{{{T}_{1}}+{{T}_{2}}}\]
done
clear
C)
\[\frac{{{T}_{2}}{{L}_{1}}\,-\,\,{{T}_{1}}{{L}_{2}}}{{{T}_{2}}-{{T}_{1}}}\]
done
clear
D)
\[\frac{{{T}_{1}}{{L}_{1}}\,-\,\,{{T}_{2}}{{L}_{2}}}{{{T}_{1}}+{{T}_{2}}}\]
done
clear
View Answer play_arrow
A magnetic flux \[\phi =at(\tau -t)\] through a stationary loop with a resistance R varies during the time interval\[\tau \]. The amount of heat generated in the loop during that time is (neglect inductance of loop)
A)
\[\frac{3\,R\,{{\tau }^{3}}}{{{a}^{2}}}\]
done
clear
B)
\[\frac{{{a}^{3}}}{3\,R{{\tau }^{2}}}\]
done
clear
C)
\[\frac{3R\,a\tau }{4}\]
done
clear
D)
\[\frac{{{a}^{2}}{{\tau }^{2}}}{3R}\]
done
clear
View Answer play_arrow
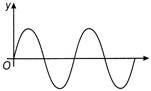
If the displacement time graph of a particle executing SHM is as shown in figure
The corresponding force-time graph of the particle is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A boy of mass 50 kg is standing at one end on a boat of length 25 m and mass 200 kg. If he starts running and when he reaches the other end he has velocity 2 m/s with respect to the boat. The final velocity of the boat is (in m/s).
A)
\[\frac{2}{3}\]
done
clear
B)
\[\frac{2}{5}\]
done
clear
C)
\[\frac{8}{9}\]
done
clear
D)
\[\frac{5}{7}\]
done
clear
View Answer play_arrow
A man of mass m is standing on a structure made up of pulley, strings and platform as shown in figure. Find the force exerted by the person on the rope, so that the system \[\left( person + structure \right)\] remains in equilibrium.
A)
\[\left( M' +M+m \right)g\]
done
clear
B)
\[\frac{\left( M' +M+m \right)g}{g}\]
done
clear
C)
\[\frac{\left( M' +M+m \right)g}{2}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A non-conducting infinitely long plane sheet has given a charge in such a way that \[{{Q}_{1}}\] appears on one side and \[{{Q}_{2}}\] appears on other side. The face area of plate is A, then electric at points 1 and 2 is
A)
\[\frac{{{Q}_{1}}-{{Q}_{2}}}{2\,{{\varepsilon }_{0}}A},\,\,\frac{{{Q}_{1}}-{{Q}_{2}}}{2\,{{\varepsilon }_{0}}A}\]
done
clear
B)
\[\frac{{{Q}_{1}}+{{Q}_{2}}}{2\,{{\varepsilon }_{0}}A},\,\,\frac{{{Q}_{1}}+{{Q}_{2}}}{2\,{{\varepsilon }_{0}}A}\]
done
clear
C)
\[\frac{{{Q}_{1}}+{{Q}_{2}}}{2\,{{\varepsilon }_{0}}A},\,\,\frac{{{Q}_{1}}-{{Q}_{2}}}{2\,{{\varepsilon }_{0}}A}\]
done
clear
D)
\[\frac{{{Q}_{1}}+{{Q}_{2}}}{{{\varepsilon }_{0}}A},\,\,\frac{{{Q}_{1}}-{{Q}_{2}}}{{{\varepsilon }_{0}}A}\]
done
clear
View Answer play_arrow
A particle is moving in X-Y plane, when position vectors are given as a function of time \[x= 2.0\,m\,-\,\left( 0.25 m/{{s}^{2}} \right){{t}^{2}}\] and \[\operatorname{y}= \left( 1.0 m/s \right)t\,+\,\,(0.025m/{{s}^{2}}){{t}^{2}}\] Find the unit vector along particle velocity at\[\operatorname{t}= 2s\].
A)
\[\frac{\hat{i}-2.2\,\hat{j}}{2.42}\]
done
clear
B)
\[\frac{-\,\hat{i}+1.3\,\hat{j}}{1.64}\]
done
clear
C)
\[\frac{\hat{j}+\,\hat{k}}{\sqrt{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Consider a region bounded by conical surface as shown in figure
In this region, E is in vertical upward direction. Find flux crossing this surface.
A)
\[E\pi {{r}^{2}}\,sin \theta \]
done
clear
B)
\[\frac{E}{3}\pi r/\,sin \theta \]
done
clear
C)
\[E\pi {{r}^{2}}\]
done
clear
D)
\[\frac{E}{3}\pi {{r}^{2}}/\,cos \theta \]
done
clear
View Answer play_arrow
In the measurement of resistance of a wire using Ohm?s law, the plot between V and I drawn as shown
A)
0.909
done
clear
B)
0.896
done
clear
C)
1
done
clear
D)
None of these
done
clear
View Answer play_arrow
If the concentration of glucose \[({{C}_{6}}{{H}_{12}}{{O}_{6}})\] in blood is\[0.9 g {{L}^{-\,1}}\]. What will be the molarity of glucose in blood?
A)
5 M
done
clear
B)
50 M
done
clear
C)
0.005 M
done
clear
D)
0.5 M
done
clear
View Answer play_arrow
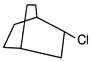
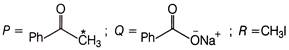
The product P in following reaction is
A)
2-phenyl propanoic acid
done
clear
B)
benzoic acid
done
clear
C)
2-phenyl ethanoic acid
done
clear
D)
phenyl methanoic acid
done
clear
View Answer play_arrow
Under standard condition, what is the potential for the reaction? \[\operatorname{Sn}(s)+2F{{e}^{3\,+}}(aq)\,\,\xrightarrow{{}}\,\,2F{{e}^{2\,+}}(aq)+S{{n}^{2\,+}}(aq)\]\[E{{{}^\circ }_{F{{e}^{3\,+}}/F{{e}^{2\,+}}}}\,\,=\,\,0.77\,V\]
A)
- 0.91 V
done
clear
B)
+0.91 V
done
clear
C)
- 0.41 V
done
clear
D)
+0.41 V
done
clear
View Answer play_arrow
Bithional is added to soap because it function as a/an
A)
hardener
done
clear
B)
softner
done
clear
C)
dryer
done
clear
D)
antiseptic
done
clear
View Answer play_arrow
Which forces are involved in holding the drugs to the active site of enzymes? I. Ionic bonding II. Hydrogen bonding III. van der Waal?s interaction IV. Dipole-Dipole interaction Choose the correct option.
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and IV
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
Which of the following statement is not characteristic of free radical chain reaction?
A)
It gives major product derived from most stable free radical
done
clear
B)
It is usually sensitive to change in solvent polarity
done
clear
C)
It proceeds in three main steps like initiation, propagation and termination
done
clear
D)
It may be initiated by UV light
done
clear
View Answer play_arrow
Give the decreasing order of reactivities of the following monomers towards anionic addition polymerisation. I. \[\operatorname{Me}-CH=C{{H}_{2}}\] II. \[\operatorname{Ph}\,-\,\,CH=C{{H}_{2}}\] III. \[{{\operatorname{CH}}_{2}}=CH\,-CN\] IV. \[{{\operatorname{CH}}_{2}} = CC{{l}_{2}}\] V. \[{{\operatorname{CH}}_{2}} = C{{F}_{2}}\] VI. \[{{F}_{2}}C=C{{F}_{\text{2}}}\] The correct order is
A)
\[\operatorname{VI}>III>V>IV>II\]
done
clear
B)
\[\operatorname{VI}>V>III>IV>II>I\]
done
clear
C)
\[\operatorname{VI}>IV>V>III>II>I\]
done
clear
D)
\[\operatorname{VI}>V>IV>II>I>III\]
done
clear
View Answer play_arrow
Which of the reagent among four alternative choice will lead the given conversion?
A)
\[{{\operatorname{H}}_{2}}O, \,{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[Cr{{O}_{3}},\,\,{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
m C P B A
done
clear
View Answer play_arrow
In which of the following case, the forms of amino acid and pH is not correctly matched?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following reagent is used to convert?
A)
Zn-Hg in HCl
done
clear
B)
\[\operatorname{P}{{H}_{3}}\,\,P=\,\,C{{H}_{2}}\]
done
clear
C)
Both a and b
done
clear
D)
None of these
done
clear
View Answer play_arrow
The reduction of
is done by Wolf-Kishner reduction. The reagent used in Wolf-Kishner reduction is
A)
\[{{\operatorname{NH}}_{2}}-N{{H}_{2}} in presence of base\]
done
clear
B)
Zn?Hg in dil HCl
done
clear
C)
\[{{\operatorname{PH}}_{3}}\,\,P\,=\,\,C{{H}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Methyl amine react with \[HN{{O}_{2}}\]to form I. \[{{\operatorname{CH}}_{3}}ONO\] II. \[{{\operatorname{CH}}_{3}}OC{{H}_{3}}\] III. \[{{\operatorname{CH}}_{3}}OH\] IV. \[{{\operatorname{CH}}_{3}}CHO\] Which of the above is formed. Choose the correct option.
A)
I and II
done
clear
B)
I and III
done
clear
C)
I and IV
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
C is
A)
2, 4, 6-trinitro bromobenzene
done
clear
B)
2, 4, 6-tribromo nitrobenzene
done
clear
C)
2, 4, 6-tribromo aniline
done
clear
D)
2, 4, 5-tribromo aniline
done
clear
View Answer play_arrow
In
, electrophilic substitution occurs at
A)
ortho/para at first ring
done
clear
B)
mete at first ring
done
clear
C)
ortho/para at second ring
done
clear
D)
meta at second ring
done
clear
View Answer play_arrow
Essential oils are steam volatile organic compounds, responsible for the fragnance of flowers. At room temperature, these are generally insoluble in water but are miscible with water-vapour in vapour phase. The best method for the extraction of these oils from the flower is
A)
crystallisation
done
clear
B)
distillation
done
clear
C)
steam distillation
done
clear
D)
distillation under reduced pressure
done
clear
View Answer play_arrow
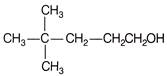
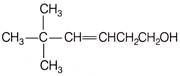
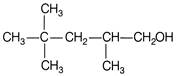
Which of the following are Newmann projections of meso-2,3-butanediol are
A b
c d Choose the correct option.
A)
B, D
done
clear
B)
A, B
done
clear
C)
C, D
done
clear
D)
B, C
done
clear
View Answer play_arrow
Which will not undergo \[{{S}_{N}}1\] reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Among the following statements about control of particulate pollution is incorrect.
A)
Cyclone collector removes fine particles in the diameter range 5-20 microns.
done
clear
B)
Wet scrubbers are used to wash away all types of particulates
done
clear
C)
In electrostatic precipitator, the particulates are made to acquire positive charge which are then attracted by the negative electrode and removed.
done
clear
D)
Gravity settling chamber removes larger particles from the air.
done
clear
View Answer play_arrow
Number of \[\sigma -bond\] present in cyclometaphosphoric acid is
A)
13
done
clear
B)
15
done
clear
C)
14
done
clear
D)
17
done
clear
View Answer play_arrow
The atomic radius of a chemical element is a measure of the size of its atoms, usually the mean or typical distance from the nucleus to the boundary of the surrounding cloud of electrons.
A)
\[C{{l}^{+}}<C{{l}^{-}}<Cl\]
done
clear
B)
\[C{{l}^{+}}<Cl<C{{l}^{-}}\]
done
clear
C)
\[C{{l}^{-}}<Cl<C{{l}^{+}}\]
done
clear
D)
\[Cl<C{{l}^{+}}<C{{l}^{-}}\]
done
clear
View Answer play_arrow
On mixing 300 g of \[25\,%\] solution and 400 g of \[40\,%\] solution by mass, a new solution is obtained. Calculate the mass percentage of solute in the new solution
A)
66.43
done
clear
B)
19.24
done
clear
C)
33.57
done
clear
D)
87.23
done
clear
View Answer play_arrow
The decomposition of \[{{\operatorname{NH}}_{3}}\] on Pt surface is zero order reaction. Calculate the rate of production of \[{{\operatorname{N}}_{2}}\,\,and\,\,{{H}_{2}}\] if \[\operatorname{k}=2.5\,\times 1{{0}^{-}}^{4}\,mo{{l}^{-1}}\,{{s}^{-}}^{1}\]?
A)
\[1.25 \times 1{{0}^{-}}^{4}\,and 3.75 \times 1{{0}^{-\,4}} respectively\]
done
clear
B)
\[3.75 \times 1{{0}^{-}}^{4}\,and 1.25 \times 1{{0}^{-\,4}} respectively\]
done
clear
C)
\[1.25 \times 1{{0}^{-}}^{3}\,and 3.75 \times 1{{0}^{-\,3}} respectively\]
done
clear
D)
\[3.75 \times 1{{0}^{-}}^{3}\,and 1.25 \times 1{{0}^{-\,3}} respectively\]
done
clear
View Answer play_arrow
According to Werner's theory coordination compounds shows two types of valency called primary and secondary valencies. Secondary valency of \[{{\left[ CO{{({{H}_{2}}O)}_{4}} \right]}^{2\,+}}\] is
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
In the following reaction sequence, the structure of P, Q and R are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Quantum numbers describe values of conserved quantities in the dynamics of quantum system. Orientation of an atomic orbital is governed by
A)
magnetic quantum number
done
clear
B)
principal quantum number
done
clear
C)
azimuthal quantum number
done
clear
D)
spin quantum number
done
clear
View Answer play_arrow
Helium atom is two times heavier than hydrogen molecule. At 298 K, the average kinetic energy of helium atom is
A)
two times that of hydrogen molecule
done
clear
B)
same as that of hydrogen molecule
done
clear
C)
four times that of hydrogen molecule
done
clear
D)
half that of hydrogen molecule
done
clear
View Answer play_arrow
Among following reactions, which one will produce dihydrogen?
A)
\[{{H}_{2}}{{S}_{4}}{{O}_{8}} + {{H}_{2}}O\]
done
clear
B)
\[\operatorname{BaO} + HCl\]
done
clear
C)
\[\operatorname{Mg}\,\,+\,\,{{H}_{2}}O\]
done
clear
D)
\[{{\operatorname{Na}}_{2}}{{O}_{2}}+\,\,2HCl\]
done
clear
View Answer play_arrow
Arrange the given complex ions in ascending order of increasing crystal field splitting energy\[({{\Delta }_{0}})\]. [a] \[{{\left[ Cr{{\left( Cl \right)}_{6}} \right]}^{\,3\,-}}\] [b] \[{{\left[ (Cr{{\left( CN \right)}_{6}} \right]}^{\,3\,-}}\] [c] \[{{\left[ Cr{{\left( N{{H}_{3}} \right)}_{6}} \right]}^{\,3\,+}}\]
A)
\[\operatorname{A}<B<C\]
done
clear
B)
\[A<C<B\]
done
clear
C)
\[\operatorname{B}<C<A\]
done
clear
D)
\[\operatorname{C}<B<A\]
done
clear
View Answer play_arrow
Among the following four compounds, which one is used for fast drying of neutral gases?
A)
Phosphorous pentaoxide
done
clear
B)
Sodium phosphite
done
clear
C)
Anhydrous calcium chloride
done
clear
D)
Sodium sulphate
done
clear
View Answer play_arrow
Which of the following statement is not true regarding silicones?
A)
Silicones have high thermal stability
done
clear
B)
Silicones are chemically inert
done
clear
C)
Substituted silicones or hydrolysis give cyclic trimer of silicones
done
clear
D)
Trisubstituted silicones on hydrolysis give cyclic trimer of silicones
done
clear
View Answer play_arrow
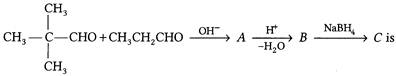
What will be the final product C in the reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
What will be the value of equilibrium constant for the reaction? \[\operatorname{AgCl}(s)\,\,\xrightarrow{{}}\,\,A{{g}^{+}}\,(aq)+C{{l}^{-}}\,(aq)\]\[E{{{}^\circ }_{AgCl\,(s)/Ag(s),\,C{{l}^{-}}}}\,\,=\,\,0.22\,V\]and \[E{{{}^\circ }_{[A{{g}^{+}}\left( aq \right)/Ag\left( s \right)]}}\,\,=\,\,0.80\,V\]
A)
- 22.57
done
clear
B)
\[1.6\,\,\times \,\,1{{0}^{-}}^{10}\]
done
clear
C)
\[1.6\,\,\times \,\,1{{0}^{10}}\]
done
clear
D)
\[1.6\,\,\times \,\,1{{0}^{9}}\]
done
clear
View Answer play_arrow
Solubility of AgCl in water, 0.01 M \[CaC{{l}_{2}}\], 0.01 M NaCl and 0.05 M \[AgN{{O}_{3}}\] are \[{{S}_{1}},\,\,{{S}_{2}},\,\,{{S}_{3}}\,and\,{{S}_{4}}\] respectively, then
A)
\[{{\operatorname{S}}_{1}}<{{S}_{2}} <{{S}_{3}} <{{S}_{4}}\]
done
clear
B)
\[{{\operatorname{S}}_{1}} >{{S}_{3}} >{{S}_{2}} >{{S}_{4}}\]
done
clear
C)
\[{{\operatorname{S}}_{1}} >{{S}_{2}}={{S}_{3}} >{{S}_{4}}\]
done
clear
D)
\[{{\operatorname{S}}_{1}} >{{S}_{3}} >{{S}_{4}} >{{S}_{2}}\]
done
clear
View Answer play_arrow
\[N{{H}_{2}}COON{{H}_{4}}(s)\,\,\,2N{{H}_{3}}(g)+C{{O}_{2}}(g)\] equilibrium constant of given reaction is\[2.9\,\,\times \,\,1{{0}^{-}}^{5}\,at{{m}^{3}}\]. The total pressure of gases in equilibrium with \[{{\operatorname{NH}}_{3}}COON{{H}_{4}}(s)\,\,at\,\,30{}^\circ C\] is
A)
0.0194 atm
done
clear
B)
0.194 atm
done
clear
C)
5.82 atm
done
clear
D)
0.0582 atm \[\underset{1-x}{\mathop{{{\operatorname{NH}}_{2}}COON{{H}_{4}}(s)}}\,\,\,\,\,2N{{H}_{\underset{2x}{\mathop{3}}\,}}\,(g)\,+\,\,C{{O}_{\underset{x}{\mathop{2}}\,}} (g)\] \[{{K}_{p}}={{({{\rho }_{N{{H}_{3}}}})}^{2}}\times {{\rho }_{C{{O}_{2}}}}\] \[2.9\,\,\times \,\,1{{0}^{-}}^{5}={{\left( 2x \right)}^{2}}\times x\] \[2.9\,\,\times \,\,1{{0}^{-}}^{5}=4{{x}^{3}}\] \[\operatorname{x}= 0.0194 atm\] \[\operatorname{Total} pressure = 2x +x=3x = 3\,\,\times \,\, 0.0194\] \[= 0.0582 atm\]
done
clear
View Answer play_arrow
Value of equilibrium constant for following reaction is 0.50 at\[400{}^\circ C\]. What is the value of \[{{K}_{p}}\] at\[400{}^\circ C\]. When the unit of pressure is in atmosphere and unit of concentration is in\[m{{L}^{-1}}\]. Chemical reaction is \[{{N}_{2}}(g)+3{{H}_{2}}(g)\,\,\,\,2N{{H}_{3}}(g)\]
A)
\[1.64\,\times \,\,1{{0}^{-}}^{3}\]
done
clear
B)
\[1.64\times 1{{0}^{4}}\]
done
clear
C)
\[1.64\times 1{{0}^{-\,4}}\]
done
clear
D)
\[164\times 1{{0}^{-\,4}}\]
done
clear
View Answer play_arrow
Calculate the value of \[E_{cell}^{{}^\circ }\] if standard reduction potential of \[F{{e}^{2\,+}}\] /Fe and \[S{{n}^{2\,+}}\]/Sn electrodes are - 0.44 V and - 0.14 V respectively. The cell reaction is\[{{\operatorname{Fe}}^{2\,+}}+\,\,Sn\,\,\xrightarrow{{}}\,\,Fe+S{{n}^{2\,+}}\]
A)
0.42 V
done
clear
B)
- 0.42 V
done
clear
C)
- 0.30 V
done
clear
D)
-1.10 V
done
clear
View Answer play_arrow
Ratio of rate of diffusion of He and \[C{{H}_{4}}\] under identical condition of pressure and temperature is
A)
4
done
clear
B)
2
done
clear
C)
1
done
clear
D)
0.5
done
clear
View Answer play_arrow
Which of the following represent correct order of packing fraction?
A)
\[FCC < BCC < Simple cubic\]
done
clear
B)
\[FCC < BCC > Simple cubic\]
done
clear
C)
\[FCC > Simple cubic < BCC\]
done
clear
D)
\[BCC < FCC < Simple cubic\]
done
clear
View Answer play_arrow
Which of the following is correct reason for conductor like property of graphite?
A)
Due to presence of cations
done
clear
B)
Due to presence of anions
done
clear
C)
Due to presence of free electrons
done
clear
D)
Due to presence of lone pair of electrons
done
clear
View Answer play_arrow
A chemical reaction occuring in open vessel is observed by student. He was observing the reaction from 3 h. He has not notice any change. Suddenly his friend comes and increases the temperature. He suddenly get the desired product. What will be the value of Gibbs free energy change of the reaction during those 3 h?.
A)
> 0
done
clear
B)
< 0
done
clear
C)
= 0
done
clear
D)
\[\ne \,\,0\]
done
clear
View Answer play_arrow
Which of the following statement is true regarding heat of combustion of a substance?
A)
It is always a positive quantity
done
clear
B)
It is always a negative quantity
done
clear
C)
It is equal to heat of formation
done
clear
D)
Nothing can be said with considering any chemical reaction
done
clear
View Answer play_arrow
Among the following compounds those cannot be prepared by Gabriel method from their corresponding halides of tosylates are I. 1 - butylamine; II. Neopentylamine III. Diethylamine IV. p - toluidine V. m - nitro aniline VI. Vinylamine VII. p - nitro aniline VIII. o -nitro aniline IX. Allyl amine Choose the correct option.
A)
I, II, VI, VII. VIII
done
clear
B)
I, II, III, IV, V, VI
done
clear
C)
III, IV, V, VI, IX
done
clear
D)
All of these
done
clear
View Answer play_arrow
The half-life of a reaction is halved as the initial concentration of the reactant is double [d] The order of reaction is
A)
0.5
done
clear
B)
1
done
clear
C)
2
done
clear
D)
0
done
clear
View Answer play_arrow
Coagulation is the process by which blood forms clots. Which of the following has highest coagulating power?
A)
Phosphate ion
done
clear
B)
Sulphate ion
done
clear
C)
Chloride ion
done
clear
D)
Hydride ion
done
clear
View Answer play_arrow
The free energy change for the following reactions are given below \[{{C}_{2}}{{H}_{2}}(g)+\frac{5}{2}{{O}_{2}}(g)\xrightarrow{{}}\,\,2C{{O}_{2}}(g)+{{H}_{2}}O(l)\]\[\Delta G{}^\circ =-\,394\,J\] \[C(s)+{{O}_{2}}(g))\,\,\xrightarrow{{}}\,\,C{{O}_{2}}(g);\]\[\Delta G{}^\circ =-1234\,J\] \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}\,\,{{H}_{2}}O\,(l)\] \[\Delta G{}^\circ =-237\,kJ\] Calculate the standard free energy change for the reaction? \[{{\operatorname{H}}_{2}}\left( g \right)+2C\left( s \right) \xrightarrow{{}}\,\,{{C}_{2}}{{H}_{2}}\left( g \right)\]
A)
- 2259 kJ
done
clear
B)
209 kJ
done
clear
C)
+2259 kJ
done
clear
D)
- 209 KJ
done
clear
View Answer play_arrow
Which of the following is not a characteristic feature of spermatocytogenesis?
A)
It is the second phase of spermatid formation
done
clear
B)
It produces mature haploid spermatozoa
done
clear
C)
It involves DNA replication
done
clear
D)
It involves addition of cytoplasm to cells
done
clear
View Answer play_arrow
When normal cell has lost the usual control over its division differentiation and apoptosis it becomes cancerous cell. Cancerous tumours cells are known as
A)
lymphocytes
done
clear
B)
hybridomas
done
clear
C)
myelomas
done
clear
D)
monoclonal cells
done
clear
View Answer play_arrow
Consider the following statements and choose the correct option given below. I. When photon of light energy falls on chlorophyll molecule, one of the electrons pair from ground or singlet state passes into higher energy level called excited singlet state. II. The chloroplast in \[{{C}_{3}}\] leaves are dimorphic. The chloroplasts of bundle sheath cells contain starch grains but lack grana. The mesophyll cells on the other hand, contain normal type of chloroplasts.
A)
Statement I is correct, II is incorrect
done
clear
B)
Statement I is incorrect, II is correct
done
clear
C)
Statement I and II are correct
done
clear
D)
Statements I and II are incorrect
done
clear
View Answer play_arrow
A mutation that leads to a successful evolution, must occur in
A)
somatoplasm DNA
done
clear
B)
germplasm DNA
done
clear
C)
somatic RNA
done
clear
D)
plasma proteins
done
clear
View Answer play_arrow
Project Tiger was been initiated to protect Tigers in India by providing safe place to tigers. For this purpose tiger reserves have been formed. Meloghat National Park in also one of them.
A)
Andra Pradesh
done
clear
B)
Madhya Pradesh
done
clear
C)
Maharashtra
done
clear
D)
Uttar Pradesh
done
clear
View Answer play_arrow
Which of the following organism has been categorised under the category of extinct?
A)
Cheetah
done
clear
B)
Gharial
done
clear
C)
Dodo
done
clear
D)
Indian Rhinoceros
done
clear
View Answer play_arrow
Bryophytes are primitive plants having primitive body structure. They are also known as mosses. Mosses are indicators of
A)
water pollution
done
clear
B)
soil pollution
done
clear
C)
radiation pollution
done
clear
D)
air pollution
done
clear
View Answer play_arrow
Which among the following ammo acid is coded by only one codon?
A)
Glycine
done
clear
B)
Valine
done
clear
C)
Tryptophan
done
clear
D)
Isoleucine
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Annelida 1. Pseudocoelomata B. Arthropoda 2. True coelom C. Nemathelminthes 3. Acoelomata D. Platyhelminthes 4.
A)
A-1, B-2, C-3, D-2
done
clear
B)
A-2, B-2, C-1, D-3
done
clear
C)
A-2, B-3, C-2, D-1
done
clear
D)
A-3, B-1, C-2, D-2
done
clear
View Answer play_arrow
Various technologies have been developed to study DNA and other biomolecules. DNA footprinting is used for identification of
A)
tRNA
done
clear
B)
protein binding sites in DNA
done
clear
C)
mRNA
done
clear
D)
rRNA
done
clear
View Answer play_arrow
Immunity is a state of protection from foreign molecules. It has two branches, i.e. Innate and adaptive immunity. Specificity, diversity, memory are characteristics of
A)
innate immunity
done
clear
B)
non-specific immunity
done
clear
C)
acquired immunity
done
clear
D)
external defence
done
clear
View Answer play_arrow
The loop of Henie, proximal convoluted tubule and distal convoluted tubule are regins of
A)
nephron
done
clear
B)
renal tubule
done
clear
C)
urinary bladder
done
clear
D)
ureter
done
clear
View Answer play_arrow
Which of the following carries glucose from digestive system to liver?
A)
Pulmonary vein
done
clear
B)
Hepatic portal vein
done
clear
C)
Hepatic artery
done
clear
D)
None of these
done
clear
View Answer play_arrow
Assertion [A] Second infection of the same pathogen is eliminated quickly. Reason [R] Every new encounter with same antigen results in a rapid proliferation of memory cells.
A)
Both A and R are true and R is correct explanation of
done
clear
B)
Both A and R are true, but R is not the correct explanation of
done
clear
C)
A is true, but R is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
Plants generally do not show movement, but movement is being shown by plant part, the stimulus for geotropism in root is perceived by
A)
root cap
done
clear
B)
quiescent centre
done
clear
C)
root tip
done
clear
D)
None of these
done
clear
View Answer play_arrow
Archaeopteryx is a link between reptiles and birds. It feature of both which of the following is a Reptilian feature of Archaeopteryx
A)
U-shaped furcula
done
clear
B)
presence of beak
done
clear
C)
feathers with barbules
done
clear
D)
presence of abdominal ribs
done
clear
View Answer play_arrow
Nucleosome model is used to explain the convened used form of DNA The size of Nucleosome is at which level
A)
2 nm
done
clear
B)
11 nm
done
clear
C)
30 nm
done
clear
D)
300 nm
done
clear
View Answer play_arrow
DDT residues are rapidly passed through food chain causing biomagniflcation because DDT is
A)
lipo soluble
done
clear
B)
moderately toxic
done
clear
C)
non-toxic to aquatic animals
done
clear
D)
water soluble
done
clear
View Answer play_arrow
Crasulacean Acid metabolism is found is which of the following plants?
A)
\[{{C}_{3}} plants\]
done
clear
B)
64 plants
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Several association have been found in two organism for the purpose of either benefit or exploitation. Mycorrhiza in which a fungus and a root are involved is an example of
A)
Symbiosis
done
clear
B)
Protocooperation
done
clear
C)
Comrriensalism
done
clear
D)
Ammensalism
done
clear
View Answer play_arrow
Where the fertilisation in human female occurs?
A)
Ovary
done
clear
B)
Uterus
done
clear
C)
Vagina
done
clear
D)
Fallopian tube
done
clear
View Answer play_arrow
Indian Government has initiated various steps to protect plants and animals of our country. Wildlife protection act was enacted by Indian Government in
A)
2001
done
clear
B)
1972
done
clear
C)
1985
done
clear
D)
1952
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Septal nephridia 1. Phallomere B. Genitalia 2. Mosaic vision C. Open circulatory system 3. Earthworm D. Compound eye 4. Cockroach
A)
A-2, B-4, C-1, D-3
done
clear
B)
A-3, B-1, C-4, D-2
done
clear
C)
A-3, B-4, C-2, D-1
done
clear
D)
A-1, B-2, C-3, D-4
done
clear
View Answer play_arrow
Development of animals may be direct or indirect. Indirect development include larval stage. Rhabditiform larva is the first stage larva of
A)
Wuchereria
done
clear
B)
Ascaris
done
clear
C)
Ringworm
done
clear
D)
Plasmodium
done
clear
View Answer play_arrow
Sella turcica is associated with
A)
pons
done
clear
B)
cerebrum
done
clear
C)
hippocampus
done
clear
D)
pituitary
done
clear
View Answer play_arrow
Which one of the statement is given below is not correct?
A)
Drosophyllum is an aquatic free floating rootless insectivorous plant, which possesses leaves similar to those of Dionaea
done
clear
B)
Aulosira fertilissima is an important nitrogen fixer rice fields
done
clear
C)
Arceuthobium is total parasite that grows on a number of forest trees including Pinus and Juniper
done
clear
D)
An important function of zinc is synthesis of auxin
done
clear
View Answer play_arrow
Restriction endonuclease are used is various purpose in biotechnology RFLP stands for
A)
restricted fragment length polymorphism
done
clear
B)
reduced filter length polymorphism
done
clear
C)
random filtered length polymorphic
done
clear
D)
random filtered length polymorphism
done
clear
View Answer play_arrow
In cockraoch, mushroom gland is located in
A)
2-5 segment
done
clear
B)
7-8 segment
done
clear
C)
3-5 segment
done
clear
D)
6-7 segment
done
clear
View Answer play_arrow
Which component of neuron is associated with enzyme formation required for synthesis of neurotransmitters?
A)
Neurofibrils
done
clear
B)
Schwann?s cells
done
clear
C)
Axon hillock
done
clear
D)
Nissl?s granules
done
clear
View Answer play_arrow
Where a foreign gene from trans species is inserted in the genome of a plant the plant becomes transgenic plant. Bacterium used for the production of transgenic plants is
A)
Clostridium septicum
done
clear
B)
Agrobacterium tumefaciens
done
clear
C)
Xanthomonas citri
done
clear
D)
Escherichia coli
done
clear
View Answer play_arrow
Ecosystem has different trophic level which are interrelated to each other. This relation can be represented by a pyramid. Who occupies the tip of an ecological pyramid?
A)
Herbivores
done
clear
B)
Carnivores
done
clear
C)
Producers
done
clear
D)
None of these
done
clear
View Answer play_arrow
Okazaki fragments are the DNA fragment produced.
A)
produce due to radiation
done
clear
B)
on the leading strand
done
clear
C)
on the lagging strand
done
clear
D)
required for initiation of DNA synthesis
done
clear
View Answer play_arrow
Choose the correct statement.
A)
Sub-order - Lemuroidea tarsier
done
clear
B)
Anthropoidea - Monkey, apes and man
done
clear
C)
Hominidae - Gorilla and gibbon
done
clear
D)
Tarsioidea - Lemurs and lorises
done
clear
View Answer play_arrow
The study of improvement of human functioning and well-being by improvement of living condition.
A)
euphenics
done
clear
B)
euthenics
done
clear
C)
eugenics
done
clear
D)
None of these
done
clear
View Answer play_arrow
Consider the following statements about micronutrients. I. A chemical element required by plants in relatively small quantity is known as micronutrient. II. They are typically found in cofactors and coenzymes. III. They include copper, zinc, molybdenum, manganese, cobalt and boron. IV. Boron is required for synthesis of mitotic spindle. Which of the statement given above are correct?
A)
I, II and III
done
clear
B)
I, III and IV
done
clear
C)
II, III and IV
done
clear
D)
I, II and IV
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Paratype 1. Duplicate of holotype B. Syntype 2. Original nomenclature missing C. Isotype 3. Describe along holotype D. Neotype 4. Specimen cited by author when no holotype is present
A)
A-4, B-2, C-1, D-3
done
clear
B)
A-3, B-1, C-4, D-2
done
clear
C)
A-3, B-4, C-1, D-2
done
clear
D)
A-2, B-4, C-1, D-3
done
clear
View Answer play_arrow
Public participation for having the degration of forest have been seen many times. Chipko movement was also part of it. Who is known for the Chipko Movement?
A)
Amrita Devi
done
clear
B)
Vijay Bahugana
done
clear
C)
Sunder Lal Bahuguna
done
clear
D)
Bishnois
done
clear
View Answer play_arrow
Foods from GMFs are superior to conventional foods. Above statement is
A)
Yes, they offer substantial health advantage
done
clear
B)
Yes, they are some health advantage
done
clear
C)
No, they are neither better nor worse
done
clear
D)
No, they are slightly less healthful.
done
clear
View Answer play_arrow
Airlayering or pot layering is employed in plants with
A)
thick non-flexible branches which cannot be easily bent
done
clear
B)
thin and flexible branches which can be easily bent
done
clear
C)
thick and flexible branches which can be easily bent
done
clear
D)
thin and non-flexible branches which cannot be easily bent
done
clear
View Answer play_arrow
In a living cell various biomolecules are found in different composition the average composition of proteins is
A)
\[70-90%\]
done
clear
B)
\[10-15%\]
done
clear
C)
\[20-25%\]
done
clear
D)
\[50-60%\]
done
clear
View Answer play_arrow
Biotechnology is used is various sectors such as medicine, foods, agriculture; etc. The main technique involved in agricultural biotechnology is called
A)
DNA replication
done
clear
B)
plant breeding
done
clear
C)
Both [a] and [b]
done
clear
D)
tissue culture
done
clear
View Answer play_arrow
DNA fingerprinting is a very useful biotechnology. It uses which of the following concept.
A)
RNA
done
clear
B)
Protein
done
clear
C)
VNTRs
done
clear
D)
None of these
done
clear
View Answer play_arrow
The modern man has been evolved from a series of several ancestors such as Modern man differs from the apes in
A)
protruding eyes
done
clear
B)
spore body hair
done
clear
C)
shorter arms than legs
done
clear
D)
communication by vocal means
done
clear
View Answer play_arrow
Sex-limited and sex-linked genes are located on
A)
autosomes
done
clear
B)
X-chromosome
done
clear
C)
Y-chromosome
done
clear
D)
Both [b] and [c]
done
clear
View Answer play_arrow
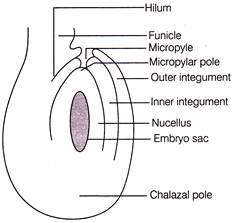
Given below is the diagrammatic sketch of a certain type of angiospermic ovule. Identify the type of ovule.
A)
Orthotropous
done
clear
B)
Anatropous
done
clear
C)
Amphitropous
done
clear
D)
Circinotropous
done
clear
View Answer play_arrow
In sickle-cell haemoglobin, the glutamali amino acid present in the 6th position of beta chain of Hb is converted into which of the following amino acid
A)
valine
done
clear
B)
alanine
done
clear
C)
Leucine
done
clear
D)
glycine
done
clear
View Answer play_arrow
Which of the following is an example of embryonic connective tissue?
A)
Wolman's jelly
done
clear
B)
Wharton's jelly
done
clear
C)
Wright's jelly
done
clear
D)
Wrigley's jelly
done
clear
View Answer play_arrow
Which of the following isotopes is used for proving semiconservative nature of DNA replication?
A)
\[{{\operatorname{C}}^{14}}\,and\,\,{{P}^{31}}\]
done
clear
B)
\[{{\operatorname{N}}^{14}}\,and\,\,{{N}^{15}}\]
done
clear
C)
\[{{\operatorname{N}}^{14}}\,and\,\,{{C}^{14}}\]
done
clear
D)
\[{{\operatorname{N}}^{14}}\,and\,\,{{P}^{31}}\]
done
clear
View Answer play_arrow
Match the following columns.
Column I (Apparatus) Column II (Uses) A. Parometer 1. Measuring the rate of transpiration B. Ganong?s photometer 2. For measuring root pressure C. Manometer 3. Measuring the area of the stomatal openings of a leaf
A)
A-1, B-3, C-2
done
clear
B)
A-3, B-1, C-2
done
clear
C)
A-1, B-2, C-3
done
clear
D)
A-2, B-3, C-1
done
clear
View Answer play_arrow
The organisms belonging to kingdom - Protista shows which of the following features? I. Unicellular and eukaryote II. Multicellular and eukaryote III. Unicellular and prokaryote IV. Autotroph or hetrotroph Select the correct option.
A)
I, III and IV
done
clear
B)
I and IV
done
clear
C)
Only IV
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Assertion [A] Rhoeo leaves contain anthocyanin pigments in epidermal cells. Reason [R] Anthocyanin is a photosynthetic pigment.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but Reason is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
Identify the correct statement. I. The pentose sugar in nucleotide occur in two types. II. Adenine and guanine are the pyremidines. III. Purines are 9-membered double rings. IV. A nucleotide is basic in nature.
A)
I, II and III
done
clear
B)
IV III and I
done
clear
C)
I and III
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Chlamydomonas, Volvox, Ulothrix, Spirogyra and Chara are the examples of class
A)
Chlorophyceae
done
clear
B)
Phaeophyceae
done
clear
C)
Rhodophyceae
done
clear
D)
Cyanophyceae
done
clear
View Answer play_arrow
In cockroach, corpora cardiaca is closely associated with
A)
heart
done
clear
B)
cloaca
done
clear
C)
kidney
done
clear
D)
oesophagus
done
clear
View Answer play_arrow
I. Geitonogamy is the transfer of pollen grains from the anther to the stigma of another flower of the same plant. II. Xenogamy is the transfer of pollen grains from anther to the stigma of a different plant.
A)
Statement I is true, II is false
done
clear
B)
Statement I is false, II is true
done
clear
C)
Both I and II statement are true
done
clear
D)
Both I and II statement are false
done
clear
View Answer play_arrow
Genetic recombination is the proces of exchange of chromosomal part between two chromosomes. Genetic recombination between homologus chromosome is facilitated by
A)
movement of centromere
done
clear
B)
movement of centriole
done
clear
C)
formation of synaptonemal complex
done
clear
D)
packing of chromatin
done
clear
View Answer play_arrow
The function of innermost layer of pollen sac, named tapetum is
A)
protective
done
clear
B)
nutritive
done
clear
C)
dehiscence
done
clear
D)
mechanical
done
clear
View Answer play_arrow
Which of the following is incorrect?
A)
Starfish - Radial symmetry
done
clear
B)
Prawn - Haemoceal
done
clear
C)
Sycon - Canal system
done
clear
D)
Ascaris - Flame cells
done
clear
View Answer play_arrow
Which of the following is affected by wavelength of illumination in optical microscope?
A)
Resolution
done
clear
B)
Clarity
done
clear
C)
Contrast
done
clear
D)
Magnification
done
clear
View Answer play_arrow
Appearance of male characters in female occurs in
A)
Aldosteronism
done
clear
B)
Adrenal virilism
done
clear
C)
Acromegaly
done
clear
D)
Addison?s disease
done
clear
View Answer play_arrow
Protein synthesis stops at the time of occurrence of terminator codon. Which of the following is a termination codon?
A)
AUG
done
clear
B)
GAU
done
clear
C)
UAG
done
clear
D)
GAA
done
clear
View Answer play_arrow
Consider the following statements. I. Endodermis is the last layer of cortex and is single layerd. It is characterised by presence of Casparian strips. II. Cotton is chiefly composed of epidermal hair of seed chemically, it is cellulose. III. The vascular bundles containing cambium are said to closed but if no cambium develops, they are referred as open. Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Statement I Agrobacterium tumefaciens is the causing agent of crown gall disease of dicots. Statement II Agrobacterium tumefaciens cause infections by entering the plant through wounds and injuries.
A)
Statement I is correct and II is incorrect
done
clear
B)
Statement II is correct and I is incorrect
done
clear
C)
Both Statements I and II are correct
done
clear
D)
Both Statements I and II are incorrect
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Diabetes mellitus 1. Pancreatic disorder B. Diabetes insipidus 2. Osteoporosis C. Adrenal virilism 3. Hypothalamic disorder D. Hyperparathyroidism 4. Sexcorticoids
A)
A-1, B-3, C-4, D-2
done
clear
B)
A-1, B-2, C-4, D-3
done
clear
C)
A-4, B-2, C-1, D-3
done
clear
D)
A-2, B-4, C-3, D-1
done
clear
View Answer play_arrow
Boron is an essential microelement or trace element, which is required for
A)
pollen grain germination
done
clear
B)
good growth of pollen tube
done
clear
C)
fertilisation
done
clear
D)
All of these
done
clear
View Answer play_arrow
Industrial melanism is an example of
A)
drug resistance
done
clear
B)
darkening of skin due to industries
done
clear
C)
Protective resemblances with the surroundings
done
clear
D)
Defensive adaptation of skin against UV-radiation.
done
clear
View Answer play_arrow
Choose the correct statements. I. Homolecithal eggs- Yolk uniformly distributed II. Telolecithal eggs - Yolk is very large III. Centrolecithal eggs - Yolk is localised in nucleus IV. Meiolecithal eggs - Yolk is concentrated in vegetal half
A)
I and IV
done
clear
B)
II and III
done
clear
C)
I and III
done
clear
D)
All of these
done
clear
View Answer play_arrow
Consider the following pairs.
I. Ribosomes Protein synthesis II. Okazaki fragment RNA synthesis III. Ligase DNA synthesis
Which of the above pairs is/are correctly matched?
A)
Only I
done
clear
B)
II and III
done
clear
C)
I and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Neuron transmits the nerve impulse by the process of decolonisation and repolarisation across its cell membrane- Refractory period of a nerve is characterised by
A)
generation of action potential
done
clear
B)
zero transmission of an impulse
done
clear
C)
complete excitability of a nerve
done
clear
D)
depolarisation of neural membrane
done
clear
View Answer play_arrow
In a woody dicotyledonous tree, which of the following parts, will mainly consist of primary tissues?
A)
Stem root and leaves
done
clear
B)
Flowers, fruits and leave
done
clear
C)
Shoot tip and root tips
done
clear
D)
Whole tree body
done
clear
View Answer play_arrow
Blood is made up of plasma and formed element. FE are cellular portion of blood conain WBC, RBC, platelet, etc. The ratio of WBC of RBC is
A)
\[1 : 100\]
done
clear
B)
\[1 : 1000\]
done
clear
C)
\[1 :10\]
done
clear
D)
\[1 :10000\]
done
clear
View Answer play_arrow
Which of the following is not an attribute of population growth?
A)
Natality
done
clear
B)
Mortality
done
clear
C)
Emigration
done
clear
D)
None of these
done
clear
View Answer play_arrow
rII locus in phage \[{{T}_{4}}\] is one of the loci affecting
A)
rapid lysis of K-bacterium
done
clear
B)
rapid lysis of B-bacterium
done
clear
C)
protein caot synthesis
done
clear
D)
attachment of phage to the bacterium
done
clear
View Answer play_arrow
Skeleton system is made up of axial and appendages skeleton. Axial skeleton is made up of skull, verteberal column, sternum and ribs. Skull consists of......... bones. Fill up the blank.
A)
30
done
clear
B)
20
done
clear
C)
29
done
clear
D)
19
done
clear
View Answer play_arrow
Which of the following statement is incorrect about cardiac muscle fibres?
A)
They are immune to fatigue
done
clear
B)
They are not supplied with autonomic nervous system
done
clear
C)
They have a rich blood supply
done
clear
D)
Property of contraction even when they are isolated from body temporarily
done
clear
View Answer play_arrow
Which one of the following statements is not correct?
A)
P-proteins are components found in large amounts in phloem companion cells
done
clear
B)
Water molecules remain attached to one another by a strong mutual force of attraction called cohesion force
done
clear
C)
Root hair absorbs water from soil through the process of osmosis
done
clear
D)
Water is absorbed by germination seeds through the process of imbibition and help in rupturing of seed coat
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Unaltered fossils 1. Altered fossils B. Petrified fossils 2. Soft mud C. Casts 3. Faecal pellets in sediments D. Prints 4. Original soft part of animals 5. Moulds of hardened and fossilized mud
A)
A-3, B-2, C-5, D-1
done
clear
B)
A-2, B-5, C-4, D-3
done
clear
C)
A-4, B-1, C-5, D-1
done
clear
D)
A-1, B-2, C-3, D-4
done
clear
View Answer play_arrow
Which one of the statement given below is not correct?
A)
Cyanides, azides and carbon monoxide work as enzyme inhibitors, thus the rate of respiration decrease
done
clear
B)
In apple, gaseous hormone ethylene is produced which helps in ripening of banana to convert it yellow
done
clear
C)
Water is not essential for aerobic respiration
done
clear
D)
Oxygen is essential for aerobic respiration it is the ultimate acceptor of electrons in electron transport system
done
clear
View Answer play_arrow
The first uracil nucleotides 'UUU' codon was deciphered by
A)
Khorana
done
clear
B)
Nirenberg
done
clear
C)
Boyer
done
clear
D)
Dr lan Wilmut
done
clear
View Answer play_arrow
Stomach cancer is a type of
A)
sarcoma
done
clear
B)
carcinoma
done
clear
C)
osteoma
done
clear
D)
lipomas
done
clear
View Answer play_arrow
Which of the following is non-condensed area of the chromosome?
A)
Telomere
done
clear
B)
Primary constriction
done
clear
C)
Euchromation
done
clear
D)
Chromosome puff
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Symport 1. Two types of molecules move in opposite direction B. Aritiport 2. Molecule moves across a membrane independent C. Uniport 3. Two types of molecules cross the membrane in same direction
A)
A-3, B-2, C-1
done
clear
B)
A-3, B-1, C-2
done
clear
C)
A-1, B-2, C-3
done
clear
D)
A-2, B-3, C-1
done
clear
View Answer play_arrow
Which of the following is likely to have the maximum concentration of DDI in body?
A)
Eel
done
clear
B)
Sea gull
done
clear
C)
Phytoplankton
done
clear
D)
Zooplankton
done
clear
View Answer play_arrow
Which of the following hormone is released by posterior pituitary in humans?
A)
Prolactin
done
clear
B)
Oestradiol
done
clear
C)
Oxytocin
done
clear
D)
Human placental lactogen
done
clear
View Answer play_arrow
Which of the following is not a port of plasma membrane?
A)
Oligosccharides
done
clear
B)
Fally acid
done
clear
C)
Cholesterol
done
clear
D)
None of these
done
clear
View Answer play_arrow
Cell cycle is regulated by cyclin cdK complexes. Which of the following is a cell cycle cyclin cdK complex?
A)
\[{{P}_{53}}\]
done
clear
B)
Cyclin D-cdK 4/6
done
clear
C)
Rb
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[{{P}_{53}}\] is called as gaurdian of the genome. It is associated with which of the following?
A)
Tumour suppression
done
clear
B)
Post translational activity
done
clear
C)
Programmed cell death
done
clear
D)
Transcription
done
clear
View Answer play_arrow
Which of the following amino acids with pKa closest to physiological pH?
A)
Serine
done
clear
B)
Proline
done
clear
C)
Histidin
done
clear
D)
Theornin
done
clear
View Answer play_arrow
Which of the following favours Z DNA confirmations?
A)
2?OH steric Hinderence
done
clear
B)
Anti G and \[{{C}_{2}}\] endo sugar
done
clear
C)
Syn G and \[{{C}_{3}}\] sugar
done
clear
D)
None of these
done
clear
View Answer play_arrow
Glycolysis occurs in which of the following part of the call?
A)
Cytoplasm
done
clear
B)
Chloroplast
done
clear
C)
Mitochondria
done
clear
D)
Peroxisome
done
clear
View Answer play_arrow
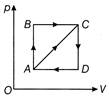 \[{{p}_{A}}=3\times {{10}^{4}}\,Pa;\,\,{{V}_{A}}=2\times {{10}^{-\,3}}\,{{m}^{3}}\] \[{{p}_{B}}=8\times {{10}^{4}}\,Pa;\,\,{{V}_{D}}=5\times {{10}^{-\,3}}\,{{m}^{3}}\] In the process AB, 600 J of heat is added to the system and in process BC, 200 J of heat is added to the system. The change in internal energy of the system in process AC would be
\[{{p}_{A}}=3\times {{10}^{4}}\,Pa;\,\,{{V}_{A}}=2\times {{10}^{-\,3}}\,{{m}^{3}}\] \[{{p}_{B}}=8\times {{10}^{4}}\,Pa;\,\,{{V}_{D}}=5\times {{10}^{-\,3}}\,{{m}^{3}}\] In the process AB, 600 J of heat is added to the system and in process BC, 200 J of heat is added to the system. The change in internal energy of the system in process AC would be 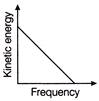
 The corresponding force-time graph of the particle is
The corresponding force-time graph of the particle is 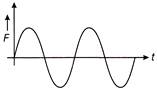
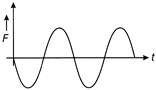
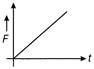
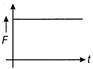
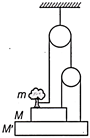
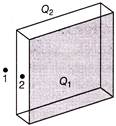
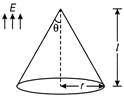 In this region, E is in vertical upward direction. Find flux crossing this surface.
In this region, E is in vertical upward direction. Find flux crossing this surface. 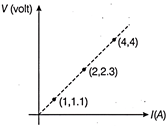

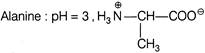
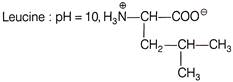
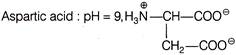
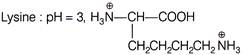
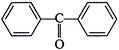 , electrophilic substitution occurs at
, electrophilic substitution occurs at 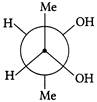
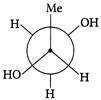 A b
A b 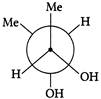
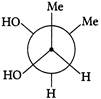 c d Choose the correct option.
c d Choose the correct option.