A mixture of gas consist of 2 moles of oxygen and 4 moles of argon at temperature T. If all vibrational modes as neglected, then the total energy of system is
A)
3RT
done
clear
B)
9RT
done
clear
C)
11RT
done
clear
D)
None of these
done
clear
View Answer play_arrow
The threshold wavelength for a metal having work function\[{{w}_{0}}\text{ }is\text{ }{{\lambda }_{0}}\]. What is the threshold wavelength for a metal whose work function is\[\frac{{{w}_{0}}}{2}\]?
A)
\[4{{\lambda }_{0}}\]
done
clear
B)
\[2{{\lambda }_{0}}\]
done
clear
C)
\[\frac{{{\lambda }_{0}}}{2}\]
done
clear
D)
\[\frac{{{\lambda }_{0}}}{4}\]
done
clear
View Answer play_arrow
The angular speed \[\left( \omega \right)\] of an electron revolving in nth Bohr orbit and corresponding principal quantum number (n) are related as
A)
\[\omega \propto \frac{1}{{{n}^{3}}}\]
done
clear
B)
\[\omega \propto \frac{1}{{{n}^{2}}}\]
done
clear
C)
\[\omega \propto {{n}^{2}}\]
done
clear
D)
\[\omega \propto {{n}^{3}}\]
done
clear
View Answer play_arrow
A uniform steel rod of \[2.5 m{{m}^{2}}\] cross-sectional area is heated through\[40{}^\circ C\]. The force exerted on it is \[({{\alpha }_{steel}}\,=\,\,1.2\times {{10}^{-\,5}}\,/{}^\circ C,\,\,y\,\,=\,\,2\times 1{{0}^{11}}\,N/{{m}^{2}})\]
A)
480 N
done
clear
B)
240 N
done
clear
C)
140 N
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two particles on a wave having wavelength 2m are at the distance of 5m and 9 m respectively from origin. The phase difference between particle is
A)
\[\pi \]
done
clear
B)
\[2\pi \]
done
clear
C)
\[\frac{\pi }{2}\]
done
clear
D)
\[4\pi \]
done
clear
View Answer play_arrow
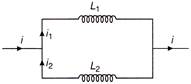
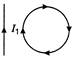
Two inductors \[{{\operatorname{L}}_{1}}\,\,and\,\,{{L}_{2}}\] are connected in parallel and a time varying current flows as shown in figure. Then the ratio of currents \[{{i}_{1}}/{{i}_{2}}\] at any time t is
A)
\[\frac{{{L}_{1}}}{{{L}_{2}}}\]
done
clear
B)
\[\frac{{{L}_{2}}}{{{L}_{1}}}\]
done
clear
C)
\[\frac{{{L}_{1}}{{L}_{2}}}{{{({{L}_{1}}+{{L}_{2}})}^{2}}}\]
done
clear
D)
\[\frac{{{({{L}_{1}}{{L}_{2}})}^{2}}}{{{(L_{1}^{2}+L_{2}^{2})}^{2}}}\]
done
clear
View Answer play_arrow
In Young's double slit experiment, the separation between slits is halved and the distance between slits and tin screen is doubled. The fringe width is
A)
unchanged
done
clear
B)
halved
done
clear
C)
doubled
done
clear
D)
quadrupled
done
clear
View Answer play_arrow
Damping force on an oscillator is directly proportionally to the velocity, then units of the constant a proportionality are
A)
kg s
done
clear
B)
\[kg\text{ }{{s}^{-\,1}}\]
done
clear
C)
\[kg\text{ m}{{s}^{-\,2}}\]
done
clear
D)
\[kg\text{ m}{{s}^{-\,1}}\]
done
clear
View Answer play_arrow
A wave equation is given by \[y=4\,\sin \,\left[ \pi \left( \frac{t}{5}-\frac{x}{9}+\frac{1}{6} \right) \right]\] Where x is in cm and t in second. Which of the following is true?
A)
\[\lambda =1.8 cm\]
done
clear
B)
\[\operatorname{v}=4\,\,m{{s}^{-}}^{1}\]
done
clear
C)
\[\operatorname{a} = 0.4 m\]
done
clear
D)
\[f=50\,\,Hz\]
done
clear
View Answer play_arrow
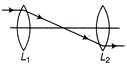
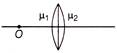
In figure shown, there are two convex lenses \[{{L}_{1}}\,\,and\,\,{{L}_{2}}\] having focal length \[{{F}_{1}}\,\,and\,\,{{F}_{2}}\] respectively. The distance between \[{{L}_{1}}\,\,and\,\,{{L}_{2}}\] will be
A)
\[{{\operatorname{F}}_{1}}\,\,+\,\,{{F}_{2}}\]
done
clear
B)
\[{{\operatorname{F}}_{1}}\,\,-\,\,{{F}_{2}}\]
done
clear
C)
\[{{\operatorname{F}}_{1}}\]
done
clear
D)
\[{{F}_{2}}\]
done
clear
View Answer play_arrow
The radioactivity of a sample is \[{{I}_{1}}\] at a time \[{{t}_{1}}\] and \[{{I}_{2}}\] at a time\[{{t}_{2}}\]. If the half-life of the sample is \[{{\tau }_{1/2}}\] then the number of nuclei that have disintegrated in the time \[{{t}_{2}}-{{t}_{1}}\] is proportional to
A)
\[{{l}_{1}}{{t}_{2}}-{{l}_{2}}{{t}_{1}}\]
done
clear
B)
\[{{l}_{1}}-{{l}_{2}}\]
done
clear
C)
\[\frac{{{l}_{1}}-{{l}_{2}}}{{{\tau }_{1/2}}}\]
done
clear
D)
\[\left( {{l}_{1}}-{{l}_{2}} \right)\,{{\tau }_{1/2}}\]
done
clear
View Answer play_arrow
Two radioactive materials \[{{x}_{1}}\,\,and\,\,{{x}_{2}}\] have decay constants\[5\lambda \,\,and\,\,\lambda \], respectively. If initially they have the same number of nuclei, then the ratio of the number of nuclei of \[{{x}_{1}}\] to that of \[{{x}_{2}}\] will be after a time.
A)
\[\lambda \]
done
clear
B)
\[\frac{1}{2}\lambda \]
done
clear
C)
\[\frac{1}{4}\,\lambda \]
done
clear
D)
\[\frac{e}{\lambda }\,\]
done
clear
View Answer play_arrow
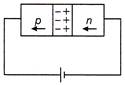
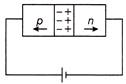
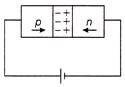
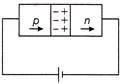
In the case of forward biasing of a p-n junction diode, which one of the following figures correctly depicts the direction of conventional current?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
How many images are formed by the lens shown, if an object is kept on its axis as shown below?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following in case of spherical mirror depend on whether the rays are paraxiol or not?
A)
Radius of curvature
done
clear
B)
Pole
done
clear
C)
Focus
done
clear
D)
Principal axis
done
clear
View Answer play_arrow
If in a wire of Young?s modulus Y, longitudinal strain X is produced then the potential energy stored in its unit volume will be
A)
\[0.5 Y{{X}^{2}}\]
done
clear
B)
\[0.5 {{Y}^{2}}X\]
done
clear
C)
\[2 Y{{X}^{2}}\]
done
clear
D)
\[{{\operatorname{YX}}^{2}}\]
done
clear
View Answer play_arrow
A wire is suspended vertically from one of its ends is stretched by attaching a weight of 200 N to the lower end. The weight stretches the wire by 1 mm. Then the elastic energy stored in wire is
A)
0.2 J
done
clear
B)
10 J
done
clear
C)
20 J
done
clear
D)
0.1 J
done
clear
View Answer play_arrow
If a car of mass 1000 kg negotiates a banked curve of radius 90 m on a irictioluess load. If the angle of banking is \[45{}^\circ \], then speed of car is
A)
10 m/s
done
clear
B)
5 m/s
done
clear
C)
20 m/s
done
clear
D)
30 m/s
done
clear
View Answer play_arrow
A hollow cylinder has a charge q within it. If \[\phi \] is the electric flux in unit of voltmeter associated with the curved surface B, the flux linked with the plane surface A in unit of voltmeter will be
A)
\[\frac{1}{2}\left( \frac{q}{{{\varepsilon }_{0}}}-\phi \right)\]
done
clear
B)
\[\frac{q}{2\,{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{\phi }{3}\]
done
clear
D)
\[\frac{q}{\,{{\varepsilon }_{0}}}-\phi \]
done
clear
View Answer play_arrow
The thermo emf-E in volts of a certain thermocouple is found to vary with temperature difference \[\theta \] in \[{}^\circ C\] between the two junctions according to the relation \[E= 30 \theta \,\,- \frac{{{\theta }^{2}}}{15}\] then the neutral temperature of thermocouple is
A)
\[400{}^\circ C\]
done
clear
B)
\[225{}^\circ C\]
done
clear
C)
\[450{}^\circ C\]
done
clear
D)
\[30{}^\circ C\]
done
clear
View Answer play_arrow
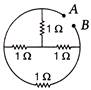
The equivalent resistance between A and B as shown in figure is
A)
\[0.2\text{ }\Omega \]
done
clear
B)
\[0.4\text{ }\Omega \]
done
clear
C)
\[0.6\text{ }\Omega \]
done
clear
D)
None of these
done
clear
View Answer play_arrow
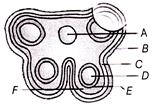
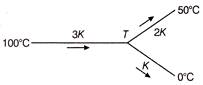
Three rods of same dimensions have thermal conductivities 3K, 2K and K they are arranged as shown below
What will be the temperature T of the junction?
A)
\[\frac{200}{3}{}^\circ C\]
done
clear
B)
\[\frac{100}{3}{}^\circ C\]
done
clear
C)
\[75\,{}^\circ C\]
done
clear
D)
\[\frac{50}{3}\,{}^\circ C\]
done
clear
View Answer play_arrow
If 8 dipoles of charges of magnitude \[\pm \,e\] are placed inside the cube. The electric flux coming out of cube will be
A)
\[16\,e/{{E}_{0}}\]
done
clear
B)
\[8\,e/{{E}_{0}}\]
done
clear
C)
\[75\,{}^\circ C\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A ladder is leaned against a smooth wall and it is allowed to slip on a frictionless floor. Which figure represents the track of its centre of mass?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The coordinates of a moving particle at any time t are given by \[\operatorname{x}=\alpha {{t}^{3}}\,\,and\,\,y=\beta {{t}^{3}}\]. The speed of the particle at time t is given by
A)
\[3t\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
B)
\[3{{t}^{2}}\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
C)
\[{{t}^{2}}\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
D)
\[\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
View Answer play_arrow
If a solenoid of magnetic moment \[0.2\,A{{m}^{2}}\] in a uniform magnetic field 0.16 T experiences a torque 0.016 Nm, then angle made by a solenoid axis with magnetic field is
A)
\[30{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[90{}^\circ \]
done
clear
View Answer play_arrow
The maximum percentage error in the determination of \[\operatorname{g}(g= 4{{\pi }^{2}}l/{{t}^{2}})\], when l and t are measured with \[\pm 2%\] and \[\pm 3\,%\] errors is
A)
\[2\,%\]
done
clear
B)
\[6\,%\]
done
clear
C)
\[8\,%\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A hollow metal sphere of radius 0.01 m is charged such that potential on its surface is 80 V. The potential at centre sphere is
A)
8 V
done
clear
B)
80 V
done
clear
C)
zero
done
clear
D)
800 V
done
clear
View Answer play_arrow
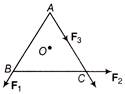
ABC is an equilateral triangle with O as its centre. \[{{F}_{1}},\,\,{{F}_{2}}\,\,and\,\,{{F}_{3}}\] represents three forces acting along the sides AB, BC and AC respectively. If total torque about O is zero, then the magnitude of \[{{F}_{3}}\] is
A)
\[\frac{{{F}_{1}}\,\,and\,\,{{F}_{2}}}{2}\]
done
clear
B)
\[{{F}_{1}}\,\,-\,\,{{F}_{2}}\]
done
clear
C)
\[{{F}_{1}}\,\,+\,\,{{F}_{2}}\]
done
clear
D)
\[2({{F}_{1}}\,\,+\,\,{{F}_{2}})\]
done
clear
View Answer play_arrow
If a plane is flying horizontally at 100 m/s at a height of 1000 m releases a bomb from it, then velocity with which the bomb hits the ground is
A)
72.1 m/s
done
clear
B)
172.1 m/s
done
clear
C)
197.2 m/s
done
clear
D)
None of these
done
clear
View Answer play_arrow
In the given figure the loop is fixed but straight wire can move. The straight wire will
A)
remain stationary
done
clear
B)
move towards the loop
done
clear
C)
move away from the loop
done
clear
D)
rotates about the axis
done
clear
View Answer play_arrow
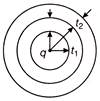
Figure shows three spherical and equipotential surfaces A, B and C round a point charge q. The potential difference\[{{V}_{A}}-{{V}_{B}}={{V}_{B}}-{{V}_{C}}\]. If \[{{t}_{1}}\,\,and\,\,{{t}_{2}}\] be the distances between them then
A)
\[{{t}_{1}}\,\,=\,\,{{t}_{2}}\]
done
clear
B)
\[{{t}_{1}}\,\,>\,\,{{t}_{2}}\]
done
clear
C)
\[{{t}_{1}}\,\,<\,\,{{t}_{2}}\]
done
clear
D)
\[{{t}_{1}}\,\,\le \,\,{{t}_{2}}\]
done
clear
View Answer play_arrow
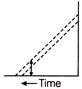
Relative velocity of geostationary satellite with respect to the spinning motion of earth is
A)
0 m/s
done
clear
B)
6 m/s
done
clear
C)
12 m/s
done
clear
D)
14 m/s
done
clear
View Answer play_arrow
Monochromatic light of wavelength 4500 A falls on slit width a. In diffraction pattern secondary maxima deviates through\[30{}^\circ \]. The slit width is
A)
1800 A
done
clear
B)
13500 A
done
clear
C)
22500 A
done
clear
D)
None of these
done
clear
View Answer play_arrow
When light is incident on a transparent medium at a polarising angle, then the angle between the reflected and refracted ray is
A)
\[30{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[90{}^\circ \]
done
clear
View Answer play_arrow
An engine has a efficiency of 1/6. When the temperature of sink is reduced by \[62{}^\circ C\], its efficiency is doubled, then the temperature of source is
A)
\[62{}^\circ C\]
done
clear
B)
\[99{}^\circ C\]
done
clear
C)
\[37{}^\circ C\]
done
clear
D)
\[124{}^\circ C\]
done
clear
View Answer play_arrow
A charged particle is released from rest in a region of steady and uniform electric and magnetic fields, which are parallel to each other. The particle will move in a
A)
circle
done
clear
B)
straight line
done
clear
C)
helix
done
clear
D)
cycloid
done
clear
View Answer play_arrow
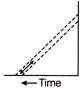
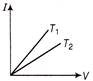
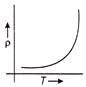
If the current-voltage graphs for a given metallic wire at two different temperatures \[{{T}_{1}}\,\,and\,\,{{T}_{2}}\] are shown in figure, then
A)
\[{{\operatorname{T}}_{1}}>{{T}_{2}}\]
done
clear
B)
\[{{\operatorname{T}}_{2}}>{{T}_{1}}\]
done
clear
C)
\[{{\operatorname{T}}_{1}}={{T}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
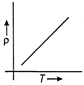
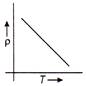
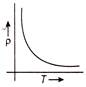
The temperature (T) dependence of resistivity (p) of a semiconductor is represented by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If a ball is thrown vertically upwards with a speed of 19.6 m/s from the top of a tower returns to earth in 6 seconds, then the height is tower is
A)
38.8 m
done
clear
B)
58.8 m
done
clear
C)
98.8 m
done
clear
D)
None of these
done
clear
View Answer play_arrow
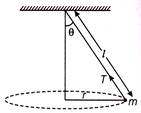
A string of length I fixed at one end carries a mass m at the other end. The string makes \[\frac{2}{\pi }\,rev/s\] around the horizontal axis through the fixed end as shown in the figure, the tension in the string is
A)
16 ml
done
clear
B)
4 ml
done
clear
C)
8 ml
done
clear
D)
2 ml
done
clear
View Answer play_arrow
A spherical planet has a mass \[{{M}_{z}}\] and diameter\[{{D}_{z}}\]. A particle of mass m falling freely near the surface of the planet, then it will experience on acceleration due to gravity equal to
A)
\[\frac{G{{M}_{z}}}{D_{z}^{2}}\]
done
clear
B)
\[\frac{4G{{M}_{z}}}{D_{z}^{2}}\]
done
clear
C)
\[\frac{G{{M}_{z}}}{D_{z}^{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If a solid cylinder of mass 3 kg is rolling on a horizontal surface with velocity 4 m/s. It collides with a horizontal spring of force constant 200 N/m. The maximum compression produced in the spring will be
A)
0.6 m
done
clear
B)
0.5 m
done
clear
C)
0. 7 m
done
clear
D)
0.2 m
done
clear
View Answer play_arrow
If a particle has initial velocity \[\left( 2\hat{i} +3\hat{j} \right)\] and acceleration\[\left( 0.3\hat{i} +0.2\hat{j} \right)\]. The magnitude of velocity after 10 seconds will be
A)
5 units
done
clear
B)
9 units
done
clear
C)
\[5\sqrt{2}\,\,units\]
done
clear
D)
\[9\sqrt{2}\,\,units\]
done
clear
View Answer play_arrow
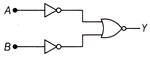
The logic gate represented by the following combination of logic gate is
A)
OR
done
clear
B)
NAND
done
clear
C)
AND
done
clear
D)
NOR
done
clear
View Answer play_arrow
50 mL each of \[1\,\,M\,\,C{{H}_{3}}COOH\] and 0.50 M NaOH are mixed\[({{K}_{a}}\,\,for\,\,C{{H}_{3}}COOH\,\,=\,\,1.8\times 1{{0}^{-\,5}})\]. The pH of the mixture is
A)
6.27
done
clear
B)
2.67
done
clear
C)
1.67
done
clear
D)
4.74
done
clear
View Answer play_arrow
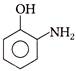
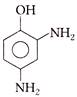
Chlorobenzene reacts with chlorine in the presence of Anhyd. \[FeC{{l}_{3}}\] gives ortho and para dichlorobenzene. Which of the following order is correct regarding dipole moment of o, m and p dichloro benzene?
A)
\[\operatorname{o}>p>m\]
done
clear
B)
\[\operatorname{p}>o>m\]
done
clear
C)
\[\operatorname{m} > o > p\]
done
clear
D)
\[\operatorname{o} > m > p\]
done
clear
View Answer play_arrow
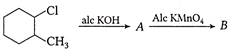
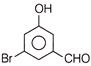
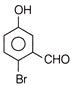
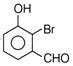
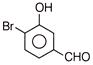
A compound obtained by the hydrolysis of the substance A, on reduction forms 2-hexanol. Hence the substance A is
A)
3, 3-dichlorohexane
done
clear
B)
2, 3-dichlorohexane
done
clear
C)
2, 2-dichlorohexane
done
clear
D)
1, 1-dichlorohexane
done
clear
View Answer play_arrow
A metal is said to be diamagnetic if it does not have any unpaired electron. Among the following, which one is diamagnetic?
A)
\[C{{o}^{2+}}\]
done
clear
B)
\[C{{u}^{2\text{+}}}\]
done
clear
C)
\[M{{n}^{2\text{+}}}\]
done
clear
D)
\[S{{c}^{3\text{+}}}\]
done
clear
View Answer play_arrow
Metals are generally hard, opaque, shiny and good conductor of heat and electricity. Which of the following elements has highest density?
A)
Os
done
clear
B)
Ir
done
clear
C)
Fe
done
clear
D)
Ru
done
clear
View Answer play_arrow
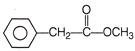
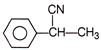
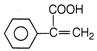
Paracetamol is classified as a mild analgesic. It is commonly used for the relief of headaches and other minor aches and pains and is a major ingredients in numerous cold and flu remedies. Paracetamol can be produced by acetylation of
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Match List I with List II and select the correct answer using the codes given below the lists.
List I List II I. Iodoform A. Anasthetic II. Methyl salicylate B. Antiseptic III. Diethyl ether C. Insecticide IV. Hexachloroxyclohexane D. Detergent E. Pain Balm
Codes
A)
I-B, II-E, III-C, IV-D
done
clear
B)
I-D, II-B, III-A, IV-C
done
clear
C)
I-B, II-E, III-A, IV-C
done
clear
D)
I-C, II-A, III-D, IV-B
done
clear
View Answer play_arrow
Consider following statements I. Cationic polymerisation occurs in monomers with electron donating substituents. II. Anionic polymerisation occurs in monomers with electron withdrawing substituents. III. Head to head chain growth polymerisation occurs in polystyrene. Select the correct statements.
A)
I, II
done
clear
B)
I, III
done
clear
C)
II. III
done
clear
D)
I, II, III
done
clear
View Answer play_arrow
A DNA nucleotide chain has AGCTTCGA sequence. The nucleotide sequence of other chain would be
A)
TCGAAGCT
done
clear
B)
GCTAAGCT
done
clear
C)
TAGCATAT
done
clear
D)
GATCCTAG
done
clear
View Answer play_arrow
Owing to the presence of a double bond in each repeat unit, natural rubber is susceptible to vulcanization and sensitive to ozone cracking. When rubber is heated with sulphur, it produces
A)
galvanised rubber
done
clear
B)
vulcanised rubber
done
clear
C)
bessemerised rubber
done
clear
D)
sulphonised rubber
done
clear
View Answer play_arrow
What will be the final product of following reaction?
A)
done
clear
B)
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Formalin is a colourless solution in water that is mostly used as a preservative. It is also used as a disinfactant and an antibacterial. It is
A)
\[\operatorname{a} solution of 40 % HCHO and 60 % C{{H}_{3}}CHO\]
done
clear
B)
\[40\,%\] aqueous solution of HCHO
done
clear
C)
solution of fructose
done
clear
D)
None of these
done
clear
View Answer play_arrow
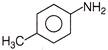
Which of the following reaction will not produce an amine?
A)
done
clear
B)
\[C{{H}_{3}}CH=C{{H}_{2}}\,\,\xrightarrow{NaN{{H}_{2}}}\]
done
clear
C)
\[2C{{H}_{3}}C{{H}_{2}}\,-\,Cl\,\,\,\,\xrightarrow{N{{H}_{3}}}\]
done
clear
D)
done
clear
View Answer play_arrow
Propyl nitrile may be converted into butyl amine by using reduction. Which of the following reagent can do this?
A)
Sn/HCl
done
clear
B)
\[\operatorname{L}iAI{{H}_{4}}\]
done
clear
C)
cone. HCl
done
clear
D)
All of these
done
clear
View Answer play_arrow
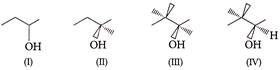
Arrange these substrate in case of dehydration.
A)
\[\operatorname{I} < II < III < IV\]
done
clear
B)
\[\operatorname{I}II < IV< II < I~\]
done
clear
C)
\[\operatorname{I} < II < IV < III\]
done
clear
D)
\[\operatorname{II} < III < IV < I\]
done
clear
View Answer play_arrow
A common nitrate test is known as brown ring test. An organic compound which give possible brown ring test and chiral is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
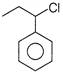
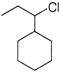
Which of the following will undergo fastest \[{{S}_{N}}1\] reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The number of reducing agents are used to reduce double bond into single bond. Which of the following will lead the conversion of cyclohexene to cyclohexane?
A)
Wilkinson's catalyst
done
clear
B)
\[\operatorname{Li}/N{{H}_{3}}\,(l)\]
done
clear
C)
\[{{\operatorname{H}}_{2}}/Ni\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
A drop of a solution \[\left( volume =0.05 mL \right)\] contains\[6.0\times 1{{0}^{-}}^{7}\,mol of\,\,{{H}^{+}}\]. If the rate of disappearence from the drop? \[6.0 \times 1{{0}^{5}}mol {{L}^{-}}^{1}\,s\], how long will it take for \[{{H}^{+}}\] to disappear from the drop?
A)
\[8.0\,\times {{10}^{8}}s\]
done
clear
B)
\[2.0\,\,\times \,\,1{{0}^{-}}^{8}s\]
done
clear
C)
\[6.0\,\,\times \,\,1{{0}^{-}}^{6}s\]
done
clear
D)
\[2.0\,\,\times \,\,1{{0}^{-}}^{2}s\]
done
clear
View Answer play_arrow
When two or more species have same structures are called isostructural species. \[S{{F}^{\text{4}}}\] is isostructural with
A)
\[{{\operatorname{TeF}}_{4}}\]
done
clear
B)
\[C{{H}_{4}}\]
done
clear
C)
\[SnC{{l}_{4}}\]
done
clear
D)
\[{{\operatorname{XeOF}}_{4}}\]
done
clear
View Answer play_arrow
When a compound has \[\operatorname{M}-C\] bond called organometallic compound. Which of the following is not an organometallic compound?
A)
Tristriphenyl phosphine rhodium chloride
done
clear
B)
Ziesse's salt
done
clear
C)
Ferrocene
done
clear
D)
Sodium cyanide
done
clear
View Answer play_arrow
When a substance or element easily reduced, it is said to be oxidising agent. Which of the following represent correct order of oxidising power?
A)
\[{{\operatorname{F}}_{2}}<C{{l}_{2}}<B{{r}_{2}}<{{I}_{2}}\]
done
clear
B)
\[{{I}_{2}}<{{F}_{2}} <C{{l}_{2}}< B{{r}_{2}}\]
done
clear
C)
\[{{\operatorname{Br}}_{2}}< {{I}_{2}} < {{F}_{2}} < C{{l}_{2}}\]
done
clear
D)
\[{{\operatorname{I}}_{2}} < B{{r}_{2}} < C{{l}_{2}}<{{F}_{2}}\]
done
clear
View Answer play_arrow
Which of the following compound is not a sandwich compound? I. Ferrorcene II. Uranocene III. Nickelocene IV. Dibenzene chromium
A)
I, II and III
done
clear
B)
I, II and IV
done
clear
C)
All of these
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[\left[ Co{{\left( N{{H}_{3}} \right)}_{4}}{{\left( N{{O}_{2}} \right)}_{2}} \right]\,Cl\] exhibits
A)
ionisation isomerism, geometrical isomerism and optical isomerism
done
clear
B)
linkage isomerism, geometrical isomerism and optical isomerism
done
clear
C)
linkage isomerism. ionisation isomerism and optical isomerism
done
clear
D)
linkage isomerism, ionisation isomerism and geometrical isomerism
done
clear
View Answer play_arrow
Select correct statements and choose the correct code out of the following. I. \[{{\operatorname{CH}}_{3}} -C\,-C{{H}_{3}}\] angle in \[{{(C{{H}_{3}})}_{2}}C\,\,=\,\,C{{H}_{2}}\] is smaller and the \[{{\operatorname{CH}}_{3}} -\,\,C=C{{H}_{2}}\]; angle is larger than trigonal \[120{}^\circ \]. II. \[O-I-F\] angle is less than \[90{}^\circ \left( 89{}^\circ \right) in IOF_{4}^{\Theta }\]. III. \[SeOC{{l}_{2}}, Cl\,\,-Se\,\,-\,\,Cl\] angle is less than the \[\operatorname{Cl}\,-Se-O\] angle. IV. \[\operatorname{POC}{{l}_{3}}\] is tetrahedral with a double bond between P and O, there is no lone pair on central atom.
A)
I and II are correct
done
clear
B)
II and III are correct
done
clear
C)
I, II and III are correct
done
clear
D)
All are correct
done
clear
View Answer play_arrow
Van-Arkel method of purification of metals involves converting the metals to a
A)
volatile stable compound
done
clear
B)
non-volatile stable compound
done
clear
C)
volatile unstable compound
done
clear
D)
None of these
done
clear
View Answer play_arrow
Element A burns in nitrogen to given an ionic compound B. Compound B reacts with water to given C and D. AB solution of C becomes 'Milky' on bubbling carbon dioxide. The element A is
A)
Na
done
clear
B)
Mg
done
clear
C)
Ca
done
clear
D)
Be
done
clear
View Answer play_arrow
The product of the following reaction
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A certain current liberates 0.500 g of \[{{H}_{2}}\] in 2.00 hr. How many gram of oxygen can be liberated by the same current in the same time?
A)
0.500 g
done
clear
B)
8.00 g
done
clear
C)
4.00 g
done
clear
D)
16.00 g
done
clear
View Answer play_arrow
The product X in the reaction,
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Carbon sub-oxide \[\left( {{C}_{3}}{{O}_{2}} \right)\] has recently been shown as a component of the atmosphere of Venus. Which of the following formulation represents the correct ground state Lewis structure for carbon suboxide?
A)
\[:O:C::C:C:O:\]
done
clear
B)
\[:O::C::C:C::O:\]
done
clear
C)
\[:\overset{.\,\,.}{\mathop{O}}\,::C::C::C::\overset{.\,\,.\,}{\mathop{O}}\,:\]
done
clear
D)
\[:O:C:C:C:O:\]
done
clear
View Answer play_arrow
Which graph represents auto catalysis?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If the degree of ionisation of water will be \[1.8 \times 1{{0}^{-}}^{9}\] at 298 K, its ionisation constant will be
A)
\[1.8\,\,\times \,\,1{{0}^{-}}^{16}\]
done
clear
B)
\[1\,\,\times \,\,1{{0}^{-}}^{14}\]
done
clear
C)
\[1\,\,\times \,\,1{{0}^{-}}^{16}\]
done
clear
D)
\[1.67\,\,\times \,\,1{{0}^{-}}^{14}\]
done
clear
View Answer play_arrow
Active mass of 32 g of HI in a 4 L flask would be
A)
1/4
done
clear
B)
1/8
done
clear
C)
1/16
done
clear
D)
2/18
done
clear
View Answer play_arrow
Calculate the value of \[\Delta G\] of a cell reaction \[\operatorname{AgCl}\,\,+\,\,{{e}^{-}} \xrightarrow{{}}\, Ag\,\,+\,\,C{{l}^{\Theta }}\] If standard emf of a cell is 0.220 V
A)
21.20kJ
done
clear
B)
-21.20 kJ
done
clear
C)
-2.12 kJ
done
clear
D)
+2.12 kJ
done
clear
View Answer play_arrow
What will be the percentage of iron (III) present in Fey 930?
A)
\[84.95 %\]
done
clear
B)
\[45.64%\]
done
clear
C)
\[15.05 %~\]
done
clear
D)
\[44.36\,%\]
done
clear
View Answer play_arrow
On the basis of different possible values of \[\alpha ,\,\,\beta \,\,and\,\,\gamma \]and y and different axial distances or edge lengths a, b and c following seven types of three dimensional primitive cells or crystal systems. Which of the following represent triclinic crystal?
A)
\[a=b=c,\,\,\alpha =\beta =\gamma =90{}^\circ \]
done
clear
B)
\[a\ne b=c,\,\,\alpha \ne \beta =\gamma =90{}^\circ \]
done
clear
C)
\[a\ne b\ne c,\,\,\alpha \ne \beta \ne \gamma =90{}^\circ \]
done
clear
D)
\[a\ne b\ne c,\,\,\alpha =\beta =\gamma \ne 90{}^\circ \]
done
clear
View Answer play_arrow
For the following reaction \[{{H}_{2}}\,(g)+{{I}_{2}}\,\,(g)\,\,\rightleftharpoons \,\,2HI\,\,(g)\] If value of equilibrium constant is 50 at \[25{}^\circ C\]. Then, the value of \[\Delta G\] will be
A)
- 6.964 kJ
done
clear
B)
- 4.964 kJ
done
clear
C)
- 9.693 kJ
done
clear
D)
- 6.496 kJ
done
clear
View Answer play_arrow
Carnot in 1824 gave an imaginary reversible cycle which demonstrates the maximum conversion of heat into work. In a cyclic process, net work done by a system is equal to
A)
0
done
clear
B)
\[\Delta U\]
done
clear
C)
\[\Delta H\]
done
clear
D)
q
done
clear
View Answer play_arrow
Molarity (M) of aqueous solution of acetic acid having density 1.12 g/mL and molality 5.0 m is
A)
4.31
done
clear
B)
4.13
done
clear
C)
8.62
done
clear
D)
5.0
done
clear
View Answer play_arrow
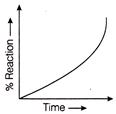
On the basis of nature of force existing between adsorbate and adsorbent adsorption can be of the following two types 1. Physiosorption 2. Chemisorption. Which of the following is incorrect regarding physisorption?
A)
Adsorption on solid is reversible
done
clear
B)
Adsorption is spontaneous
done
clear
C)
Adsorption increases with increase in temperature
done
clear
D)
Both AH and AS are negative
done
clear
View Answer play_arrow
A first order reaction is half complete in 45 min. How long does it need for 99.9% of the reaction to be completed?
A)
20 h
done
clear
B)
10 h
done
clear
C)
7.5 h
done
clear
D)
5 h
done
clear
View Answer play_arrow
The properties of a dilute solution which depends only upon the number of particles present in a solution are called colligative properites. Which of the following is not a colligative property?
A)
Osmotic pressure
done
clear
B)
Relative lowering of vapour pressure
done
clear
C)
Elevation in boiling, point
done
clear
D)
Vapour pressure
done
clear
View Answer play_arrow
A mixture of ethanol and \[CC{{l}_{4}}\] contain \[30\,%\]\[{{\operatorname{CCl}}_{4}}\] by weight. Mole fraction of \[CC{{l}_{4}}\] in the mixture is
A)
0.99
done
clear
B)
0.11
done
clear
C)
0.19
done
clear
D)
0.81
done
clear
View Answer play_arrow
Major product of the following reaction is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Assertion [A] Activity of an enzyme is pH dependent. Reason [R] Change in pH affects the solubility of the enzyme in water.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
Which among the following is an auto-immune disease?
A)
Hashimoto's disease
done
clear
B)
Addison's disease
done
clear
C)
Adrenal virilism
done
clear
D)
My xo edema
done
clear
View Answer play_arrow
An individual had been diagnosed with a Salmonella infection, which he acquired, while on vacation. She had been treated with antibiotic and the diarrhoea she was experiencing, had resolved. One day later, he developed a chest pain in the centre of chest. The important point was that the person had never any problem with his cardiovascular system. Even at that time his BP and pulse rate is normal, but ECG showed diffuse PR depression in the anterolateral leads. Most probable diagnosis of above history is
A)
angina pectoris
done
clear
B)
pericarditis
done
clear
C)
relation between Salmonella infection and chest pain could not be established
done
clear
D)
typhoid
done
clear
View Answer play_arrow
Which of the following is an example of adaptive radiation?
A)
Wings of insect, bird, bat
done
clear
B)
Tadpole larva of Herdmania
done
clear
C)
Darwin's finches of Galapagos
done
clear
D)
Egg laying mammals
done
clear
View Answer play_arrow
Which of the following elements is/are trace elements or microelements for human? I. Zinc II. Manganese III. Molybdenum IV. Calcium
A)
All of these
done
clear
B)
I, II and IV
done
clear
C)
I, III and IV
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Which of the following low temperature treatment of flowering plants that reduces the growing phase of plant and induces early flowering is termed as?
A)
Geotropism
done
clear
B)
Senescence
done
clear
C)
Vernalisation
done
clear
D)
Epinasty
done
clear
View Answer play_arrow
Choose the correct statements. I. Abiogenesis means spontaneous generation. II. Theory of biogenesis was given by Redi. III. Spallanzani stated that air carried microorganisms. IV. Cosmozoic theory of life was given by Louis Pasteur.
A)
I, II and IV
done
clear
B)
Ill, II and I
done
clear
C)
IV, II and III
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
World Summit of Sustainable Development 2002 was held in which of the following country?
A)
Rio, Brazil
done
clear
B)
Sydney, Australia
done
clear
C)
Johansburg, South Africa
done
clear
D)
Amsterdam, Netherlands
done
clear
View Answer play_arrow
Which of the following national park is first to established in India for conserving wildlife?
A)
Gir National Park, Gujarat
done
clear
B)
Periyar National Park, Kerala
done
clear
C)
Jim Corbett National Park, Uttarakhand
done
clear
D)
Kaziranga National Park, Asom
done
clear
View Answer play_arrow
'XX' is a small crystals of calcium carbonate in the saccule and utricle of the ear. In the given statement term 'XX' has been used for one of the following.
A)
Fenestra rotunda
done
clear
B)
Malleus
done
clear
C)
Otoconia
done
clear
D)
Utricles
done
clear
View Answer play_arrow
Choose the correct statement. I. pH of human blood is 7.4. II. \[C{{a}^{+}}\] is essential for muscle contraction. III. Nerve cells do not divide because they do not have nucleus. IV. Calcium carbonate is maximum in bones.
A)
I and III
done
clear
B)
I and II
done
clear
C)
Ill and IV
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
In the case of Drosophila, which of the following chromosome complement represents intersex?
A)
\[\operatorname{XY}+3A\]
done
clear
B)
\[\operatorname{XX}+3A\]
done
clear
C)
\[\operatorname{XO}+2A\]
done
clear
D)
\[\operatorname{XXX} + 3A\]
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Dysuria 1. Output 0 to 100 mL/day B. Oliguria 2. WBC s or pus in urine C. Pyuria 3. Painful urination D. Ketonuria 4. Presence of acetone bodies in urine
A)
A- 1 B-2 C-3 D-4
done
clear
B)
A-3 B- 5 C-2 D-4
done
clear
C)
A- 5 B-2 C-1 D-3
done
clear
D)
A-4 B-3 C-5 D-2
done
clear
View Answer play_arrow
Which of the following statement is true for amount of DNA in G 3-phase of cell cycle? It is
A)
equal to amount of DNA is \[{{G}_{1}}\]
done
clear
B)
double the amount of DNA is \[{{G}_{1}}\]
done
clear
C)
half the amount of DNA in \[{{G}_{1}}\]
done
clear
D)
four times the amount of DNA in \[{{G}_{1}}\]
done
clear
View Answer play_arrow
Paracetamol is the most common medicine used during the febrile condition. It is excreted by following process
Identify the location of reactions A, B, C and D of above process
A)
A-Liver B-Liver C-Liver D-Kidney
done
clear
B)
A-Stomach B-Kidney C-Liver D-Kidney
done
clear
C)
A-Kidney B-Lung C-Kidney D-Liver
done
clear
D)
A-Kidney B-Muscle C-Liver D-Kidney
done
clear
View Answer play_arrow
Recombination DNA technology can be used to produce medically useful protein. Which of the following can be produced by using this technology?
A)
Somatostatin
done
clear
B)
Insulin
done
clear
C)
Interferon
done
clear
D)
All of these
done
clear
View Answer play_arrow
Study the following pairs carefully and identify the pair which is not correctly matched.
A)
Tendon - connective tissue
done
clear
B)
Smooth muscle - involuntary
done
clear
C)
Myosin - contractile protein
done
clear
D)
Troponin - fibrous protein
done
clear
View Answer play_arrow
Filariasis causing Nematoda in India is
A)
Taeniasolium
done
clear
B)
Treponema pallidum
done
clear
C)
Ancyclostoma duodenale
done
clear
D)
Brugia malayi
done
clear
View Answer play_arrow
Statement [A] HUBS Statement A HBOT can be used for wound healing Statement [B] Oxygen is a critical factor in wound healing
A)
Both statements are true and statement B is the correct explanation of statement A.
done
clear
B)
Both statements are true, but not linked
done
clear
C)
Statement B is true, but A is false
done
clear
D)
Both statements are false
done
clear
View Answer play_arrow
Which of the following statement(s) is/are true for the structure of ear? I. Malleus is attached to tympanic membrane. II. Anvil is attached to oval window. III. Malleus is attached to oval window. IV. Stapes is attached to oval window.
A)
None of the above are true
done
clear
B)
I and III only
done
clear
C)
I and IV
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Give the name of the drug, which is produced using biotechnology and is used for the treatment of cancer.
A)
Insulin
done
clear
B)
TSH
done
clear
C)
HGH
done
clear
D)
Interferon
done
clear
View Answer play_arrow
Colchine is extracted from which of the following flora?
A)
Rice
done
clear
B)
Wheat
done
clear
C)
Corn
done
clear
D)
Barley
done
clear
View Answer play_arrow
'Decarboxylation' refers to the removal of a \[C{{O}_{2}}\] molecule during a reaction. One of the following reaction involves decarboxylation. Identify that reaction
A)
Electron transport system
done
clear
B)
Lactic acid fermentation
done
clear
C)
Alcohol fermentation
done
clear
D)
Glycolysis
done
clear
View Answer play_arrow
Your friend Sangita has a history of mild to moderate phenylketonuria (PKU). She is very anxious regarding her progeny and approached you (as you are a biology student). What would be your advice to her?
A)
Never ever go for conception
done
clear
B)
Assure her that phenylalanine is not a teratogenic, so she should go for pregnancy
done
clear
C)
Tell her that phenylalanine is teratogenic, but she can have a normal baby with slight precaution
done
clear
D)
Both [b] and [c]
done
clear
View Answer play_arrow
Which of the following carbon provides the attachment point of base to form a nucleoside in pentose sugar?
A)
\[{{C}_{1}}\]
done
clear
B)
\[{{C}_{3}}\]
done
clear
C)
\[{{C}_{2}}\]
done
clear
D)
\[{{C}_{4}}\]
done
clear
View Answer play_arrow
Many care painting have been discovered in recent centuries. One of the following fossil man bellied to make these painting is
A)
Java ape man
done
clear
B)
Australopithecus
done
clear
C)
Cro-magnon man
done
clear
D)
Peking man
done
clear
View Answer play_arrow
Which of the following statement is not correct?
A)
Adrenal cortex develops from the ectoderm
done
clear
B)
Adrenal medulla is not enclosed by a fibrous capsule
done
clear
C)
Glucocorticoids are secreted by zona fasciculata of adrenal cortex
done
clear
D)
Aldosterone is a mineralocorticoids
done
clear
View Answer play_arrow
The sclerenchyma of the hypodermis in the Pinus needle helps in one of the following function
A)
increasing the absorptive surface of the cell
done
clear
B)
reducing transpiration
done
clear
C)
mechanical support
done
clear
D)
photosynthesis
done
clear
View Answer play_arrow
Identify the correct statement among following regarding the growth in plants
A)
Growth does not involve increase in parts
done
clear
B)
Growth occurs during definite periods
done
clear
C)
Growth is indeterminate
done
clear
D)
Growing regions are absent
done
clear
View Answer play_arrow
The baby given/shown in the adjacent figure is suffering from
A)
Charoot-Marie tooth disease
done
clear
B)
Tay-Sachs disease
done
clear
C)
Prader-Willi syndrome
done
clear
D)
Down?s syndrome
done
clear
View Answer play_arrow
The mutation theory was given by Hugo de Vries, while working on one of the following organised
A)
Althea rosea
done
clear
B)
Pisum sativum
done
clear
C)
Oenothera lamarckiana
done
clear
D)
Drosophila melanogaster
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Periyar National Park 1. Tamil Nadu B. Kalkad Sanctuary 2. Gujarat C. Dhangadhra Sanctuary 3. Kerala D. Rajaji National Park 4. Chandigarh
A)
A-2, B-4, C-3, D-1
done
clear
B)
A-1, B-3, C-4, D-2
done
clear
C)
A-3, B-1, C-2, D-4
done
clear
D)
A-4, B-2, C-1, D-3
done
clear
View Answer play_arrow
Consider the following statements. I. Montreal Protocol is a landmark international agreement to protect the stratospheric ozone and to limit the product and use of ozone-depleting substances. II. Earth summit adopted the recommendations of convention on climate change for reducing greenhouse gases. Which of the statement given above is/are correct?
A)
Only I
done
clear
B)
Only II
done
clear
C)
I and II
done
clear
D)
None of these
done
clear
View Answer play_arrow
Identify the A, B, C and D of ETS for I. Matrix of mitochondria II. Intermembrane space III. Inner membrane - Mitochondrial
A)
A-1, B-3, C-2, D-\[Cyt.-c\]
done
clear
B)
A-2, B-1, C-3, D-\[UQ{{H}_{2}}\]
done
clear
C)
A-3, B-2, C-1, D-\[UQ{{H}_{2}}\]
done
clear
D)
A-3, B-2, C-1, D-\[Cyt.-c\]
done
clear
View Answer play_arrow
Choose the correct statement (s). I. Cnidarians exhibit polymorphism. II. Hydrozoa is distinguished from other coelenterates by interstitial cells. III. Coelenterates differ from other animals in having nematocysts. IV. Polyphase is absent in Hydra.
A)
I and IV
done
clear
B)
II and III
done
clear
C)
I and III
done
clear
D)
IV and II
done
clear
View Answer play_arrow
Urea is produced by urea cycle and is transported via one of the following mode.
A)
CSF
done
clear
B)
RBC
done
clear
C)
Blood plasma
done
clear
D)
WBC
done
clear
View Answer play_arrow
Which of the following is an allergic reaction?
A)
Ulcerative colitis
done
clear
B)
Typhoid
done
clear
C)
Hay fever
done
clear
D)
Encephalitis
done
clear
View Answer play_arrow
If a strand of DNA has 0.1 mm length, what will be the ratio of length to diameter?
A)
500
done
clear
B)
50
done
clear
C)
50,000
done
clear
D)
5000
done
clear
View Answer play_arrow
Which one of the following component can function as carrier in active absorption?
A)
Ferredoxin and cytochrome
done
clear
B)
Cytochrome and lecithin
done
clear
C)
Lecithin
done
clear
D)
Plastoqu in one
done
clear
View Answer play_arrow
Match the following with their importance.
Column I Column II A. Endonucleases 1. Fermentation B. Streptokinase 2. Softening of bread and meat C. Zymase 3. Consistency and flavour to cheese D. Protease 4. Breaking DNA at specific sites
Codes
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-4, B-5, C-1, D-2
done
clear
C)
A-2, B-4, C-3, D-1
done
clear
D)
A-5, B-2, C-4, D-2
done
clear
View Answer play_arrow
In 'obstetric and gynecology' the term 'nulliparous' is frequently use It refers to women having
A)
uterus absent
done
clear
B)
no children
done
clear
C)
irregular or absent menstrual cycle
done
clear
D)
ovary absent
done
clear
View Answer play_arrow
In the given urea cycle identify the markings, if I Arginosuccinate \[{{\operatorname{H}}_{2}}O\] in III \[N{{H}_{3}}\] IV L-Ornithine
A)
A-III, B-II, C-IV, D-I
done
clear
B)
A-IV, B-I, C-II, D-IV
done
clear
C)
A-III, B-I, C-II, D-IV
done
clear
D)
A-IV, B-II, C-I, D-III
done
clear
View Answer play_arrow
What is common in Bryophyllum, Sansevieria and Kalanchoe?
A)
All are members of family-Rubiaceae
done
clear
B)
All reproduce only sexually
done
clear
C)
All reproduce by leaf cutting vegetatively
done
clear
D)
All lack heterospory
done
clear
View Answer play_arrow
Tertiary structure of protein does not contain one of the following structure?
A)
di-sulphide bonds
done
clear
B)
van der Waal's bonds
done
clear
C)
hydrogen bonds
done
clear
D)
salt linkage
done
clear
View Answer play_arrow
Choose the correct option.
A)
When a fruit develops from the inflorescence, it is composite.
done
clear
B)
Mesocarp is the edible part in apple.
done
clear
C)
Hypanthium is a special type of inflorescence found in Euphorbia sp.
done
clear
D)
Inflorescence in Musa paradisiaca is a raceme.
done
clear
View Answer play_arrow
What is true about Oparin-Haldane theory? I. Coacervates were non-living structures that led to the formation of first living cells. II. Oxygen was not present in primitive atmosphere. III. Life originated in water. IV. UV rays and lightening are the possible early sources of energy.
A)
All except I
done
clear
B)
I, II and III
done
clear
C)
III, IV and I
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
Which of the following is not a physiological effect of ABA?
A)
Inhibition of growth
done
clear
B)
Tropic response
done
clear
C)
Role in water stress
done
clear
D)
Abscission
done
clear
View Answer play_arrow
Different types of cleavage found in different organisms but the cleavage which found in human egg is
A)
superficial
done
clear
B)
discoidal
done
clear
C)
radial
done
clear
D)
holoblastic
done
clear
View Answer play_arrow
Transfer of pollen grains from the anther to the stigma of another flower of the same plant is called
A)
Xenogeny
done
clear
B)
Geitonogamy
done
clear
C)
Autogamy
done
clear
D)
Misogamy
done
clear
View Answer play_arrow
Great increase in the production of food grains is a result of Green Revolution due to
A)
use of pesticides
done
clear
B)
use of agrochemicals
done
clear
C)
high-yielding varieties
done
clear
D)
All of these
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Volvox. sponges 1. Biradial symmetry B. Hydra, star fish 2. Spherical summetry C. Sea anemones 3. Bilateral symmetry D. Vertebrates 4. Radial symmetry
A)
A-1, B-2, C-4, D-3
done
clear
B)
A-2, B-4, C-1, D-3
done
clear
C)
A-1, B-2, C-3, D-4
done
clear
D)
A-3, B-1, C-4, D-2
done
clear
View Answer play_arrow
Protein \[\alpha -1\] antitrypsin is used fot the treatment of the disease
A)
cancer
done
clear
B)
emphysema
done
clear
C)
rheumatoid arthritis
done
clear
D)
hepatitis B
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Allopatric speciation a new niche 1. Population enters a new niche B. Sympatric speciation 2. Rapid and abrupt mode of species formation C. Parapatric speciation 3. Small segment of the original population becomes isolated reproductively D. Quantum speciation 4. Part of population becomes geographically isolated
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-4, B-3, C-1, D-2
done
clear
C)
A-3, B-1, C-4, D-2
done
clear
D)
A-2, B-4, C-3, D-1
done
clear
View Answer play_arrow
A cell increase in volume if the external medium is
A)
hypertonic
done
clear
B)
hypotonic
done
clear
C)
isotonic
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
Identify the causative agent of syphilis
A)
Mycobacterium leprae
done
clear
B)
Leishmania donovani
done
clear
C)
Bacillus conjugal is
done
clear
D)
Treponema pallidum
done
clear
View Answer play_arrow
Hepatitis-B, lyme diseases vaccines, which are prepared through recombinant DNA technology are called
A)
first generation vaccines
done
clear
B)
second generation vaccines
done
clear
C)
third generation vaccines
done
clear
D)
None of these
done
clear
View Answer play_arrow
There are lots of intermediates which play vital roles in immune system, interleukin is one of them. Identify the origin of interleukin
A)
mitochondria
done
clear
B)
ribosome
done
clear
C)
ER
done
clear
D)
epithelial cells
done
clear
View Answer play_arrow
What is the cotyledon of maize embryo called?
A)
Coleoptile
done
clear
B)
Scutellum
done
clear
C)
Plumule
done
clear
D)
Coleomiza
done
clear
View Answer play_arrow
Among the following, which microorganism is photoautotrophic in nature
A)
Ferro bacillus
done
clear
B)
Beggiotoa
done
clear
C)
Nitro so monas and Nitro sococcus
done
clear
D)
Green-sulphur bacteria and purple bacteria
done
clear
View Answer play_arrow
Which of the following is not true about proteins?
A)
20 amino acids are used in synthesis of all proteins
done
clear
B)
Proteins are hetero polymers of amino acids
done
clear
C)
Cellulose is most abundant in biosphere
done
clear
D)
Proteins contain peptide bonds
done
clear
View Answer play_arrow
Calculate the number of Barr body in female having the genotype of \[2A+XXXX\]
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following material is/are used in determining the age of a fossil? (1) Radioactive carbon (2) Uranium (3) Argon (4) Potassium
A)
(2) and (3)
done
clear
B)
(1) and (3)
done
clear
C)
all are used
done
clear
D)
(1) and (4)
done
clear
View Answer play_arrow
Which of the following area is popularly known as they lungs of Earth
A)
Tropics
done
clear
B)
Amazon Rainforest
done
clear
C)
Alpines
done
clear
D)
Himalayas
done
clear
View Answer play_arrow
Which of the following is not an unconditioned reflex?
A)
Corneal reflex
done
clear
B)
Knee jerk
done
clear
C)
Breast feeding
done
clear
D)
Riding a bicycle
done
clear
View Answer play_arrow
Nissi granules are found in nerve cell and identified as one of the following structure
A)
ribosome
done
clear
B)
rough ER
done
clear
C)
mitochondria
done
clear
D)
lysosome
done
clear
View Answer play_arrow
I. water hyacinth and water lily, the flowers emerge above the level of water and are pollinated by insects or wind. II. In Vallisneria, the female flower reach the surface of water by the long stalk and the male flowers or pollen grains are released on to the surface of water. They are carried passively by water currents.
A)
Statement I is true, II is false
done
clear
B)
Statement I is false, II is true
done
clear
C)
Both Statement I and Statement II are true
done
clear
D)
Both Statement I and Statement II are false
done
clear
View Answer play_arrow
How does frogs differ from humans?
A)
Frogs possess hepatic portal system
done
clear
B)
Frogs have nucleated acid blood RBCs
done
clear
C)
Frogs have thyroid gland
done
clear
D)
Frogs contain paired cerebral hemispheres
done
clear
View Answer play_arrow
Which of the following is responsible for amplification of signal in hormone action?
A)
\[{{P}_{1}}{{P}_{2}}\]
done
clear
B)
Insulin
done
clear
C)
\[I{{P}_{3}}\]
done
clear
D)
Sodium
done
clear
View Answer play_arrow
Which of the following bird lack on oil gland? I. Parrot II. Owl III. Kiwi IV. Ostrich
A)
I and II
done
clear
B)
IV and I
done
clear
C)
II and III
done
clear
D)
None of these
done
clear
View Answer play_arrow
Consider the following statements. I. Any deficiency of \[{{O}_{2}}\] stops the respiration of roots and causes accumulation of \[C{{O}_{2}}\] thus the protoplasm becomes viscous and the permeability of plasma membrane decreases. II. The absorption of water still occurs when concentration of outer soil water is more than root hair cell sap. III. Adhesion is the attraction between the molecules of the same substances. Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Following is the diagram of TS of anther. Identify the parts labelled as A, B, C, D, E and F.
A)
A-Tapetum B-epidermis C-endothecium , D-Tapetum E-Sporogenous tissue F-Middle layers
done
clear
B)
A-Connective B-epidermis C-endothecium D-Sporogenous tissue E-Tapetum F-Middle layers
done
clear
C)
A-Tapetum B-epidermis C-endodermis D-Middle layers E-Tapetum F-Sporogenous tissue
done
clear
D)
A-Connective B-epidermis C-endodermis D-Sporogenous tissue E-Middle layers F-Tapetum
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Starch 1. N-acetyl glucosamine B. Glycogen 2. Fibrous homopolysaccharide C. Chitin 3. Mucilages D. Cellulose 4. Storage polysaccharide of plants
A)
A-3, B-5, C-2, D-4
done
clear
B)
A-2, B-2, C-5, D-3
done
clear
C)
A-4, B-5, C-1, D-2
done
clear
D)
A-4, B-5, C-1, D-2
done
clear
View Answer play_arrow
Instrument used for the study of arrangement of nucleotide in DNA is
A)
centrifuge
done
clear
B)
electron microscope
done
clear
C)
X ray crystallography
done
clear
D)
photo chromatography
done
clear
View Answer play_arrow
Egg activation during fertilisation is related to
A)
secreting hyaluronidase enzymes
done
clear
B)
preventing polyspermy
done
clear
C)
dissolving egg envelop
done
clear
D)
pH change of egg cytoplasm
done
clear
View Answer play_arrow
Choose the incorrect statement.
A)
Annelida - Enter o coelomate
done
clear
B)
Platyhelminthes - Acoelomate
done
clear
C)
Nemat helminthes - Pseudo coelomate
done
clear
D)
Arthropoda - Schizocoelomate
done
clear
View Answer play_arrow
Which of the following reptile is a Living fossil?
A)
Peripatus
done
clear
B)
Sphenodon
done
clear
C)
Archaeopteryx
done
clear
D)
Latimeria
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Green sulphur bacteria 1. Rhodospirillum B. Purple sulphur bacteria 2. Chlorobium C. Purple non-sulphur bacteria 3. Chromatium
A)
A-2, B-1, C-3
done
clear
B)
A-2, B-3, C-1
done
clear
C)
A-3, B-1, C-2
done
clear
D)
A-1, B-2, C-3
done
clear
View Answer play_arrow
Select the incorrect statement from the following.
A)
Galactosemia is an inborn error of metabolism
done
clear
B)
Small population size results in random genetic drift in a population
done
clear
C)
Baldness is a sex-limited trait
done
clear
D)
Linkage is an exception to the principle of independent assortment in heredity
done
clear
View Answer play_arrow
Which one of the statement given below is not correct?
A)
In the photosynthesis process P-II absorbs energy at or just below 680 mm
done
clear
B)
The pigment that is present in the pigment system I is \[{{P}_{683}}\]
done
clear
C)
P-II receives electrons from photolytic dissociation of water
done
clear
D)
P-I lies on the outer surface of thylakoid
done
clear
View Answer play_arrow
Among the following which microorganism respire anaerobically under normal conditions.
A)
Bacillus subtilis
done
clear
B)
Rho do pseudomonas
done
clear
C)
Archaebacteria
done
clear
D)
Thermus aquaticus
done
clear
View Answer play_arrow
Which among the following remains constant for given species?
A)
\[\operatorname{A}+C/T+G\]
done
clear
B)
\[\operatorname{G}+C/A+T~\]
done
clear
C)
\[\operatorname{A}+T/G+C\]
done
clear
D)
\[\operatorname{T}+C/G+A\]
done
clear
View Answer play_arrow
Identify the correct statement(s). I. Bony fishes use pectoral, pelvic, dorsal, anal and caudal fins in swimming. II. In torpedo, electric organs are used for defence. III. Gills and scales are characteristic of fishes. IV. Chondrichthyes possess bony endoskeleton.
A)
I and IV
done
clear
B)
IV and III
done
clear
C)
I and II
done
clear
D)
III and II
done
clear
View Answer play_arrow
Which one of the following statement is not correct?
A)
Hydrilla plant is used in an experiment commonly performed in laboratory in demonstrate evolution of \[{{O}_{2}}\] in photosynthesis
done
clear
B)
Cytochrome proteins serving as electron carriers in photosynthesis and other oxidative reduction reactions.
done
clear
C)
In angiosperms, synthesis of chlorophyll occurs in presence of light.
done
clear
D)
The plants growing in dark show yellowing in leaves and elongated internodes, this condition is called as dark effect.
done
clear
View Answer play_arrow
Which among the following type of DNA is present in bacterium E.coli?
A)
Double-stranded straight
done
clear
B)
Single-stranded straight
done
clear
C)
Double-stranded circular
done
clear
D)
Single-stranded circular
done
clear
View Answer play_arrow
Which animal is the symbol of World Wildlife Fund?
A)
Tiger
done
clear
B)
Giant Panda
done
clear
C)
Hornbill
done
clear
D)
White Bear
done
clear
View Answer play_arrow
A human male produces sperms with the genotypes AB, Ab, aB, and ab pertaining to two diallelic characters equal proportions. What is the corresponding genotype of this person?
A)
AaBB
done
clear
B)
AABb
done
clear
C)
AABB
done
clear
D)
AaBb
done
clear
View Answer play_arrow
Which one of the statements given below is not correct?
A)
The leaves of \[{{C}_{4}}\] plants have Kranz anatomy
done
clear
B)
In \[{{C}_{4}}-plants\], carbon dioxide is picked up by phosphoenol pyruvate and the reaction being catalysed by PEP carboxylase
done
clear
C)
Light reaction of photosynthesis occurs inside stroma
done
clear
D)
Sunken stomata are usually found in CAM plants
done
clear
View Answer play_arrow
Production of bio products from microbes involve the use of
A)
thermo cycler
done
clear
B)
fermenter
done
clear
C)
autoclave
done
clear
D)
micro projective
done
clear
View Answer play_arrow
The genetic defect-adenosine deaminase (ADA) deficiency may be cured permanently by
A)
administering adenosine deaminase activators
done
clear
B)
introducing bone marrow cells producing ADA into cells at early embryonic stages
done
clear
C)
enzyme replacement therapy
done
clear
D)
periodic infusion of genetically engineered lymphocytes having functional ADA CDNA
done
clear
View Answer play_arrow
In DNA of E. coli has \[20\,%\] bases of guanine. What will be fraction of thymine?
A)
30
done
clear
B)
3
done
clear
C)
15
done
clear
D)
5
done
clear
View Answer play_arrow
 What will be the temperature T of the junction?
What will be the temperature T of the junction? 
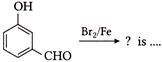
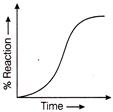
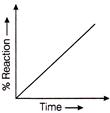
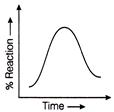
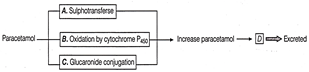 Identify the location of reactions A, B, C and D of above process
Identify the location of reactions A, B, C and D of above process 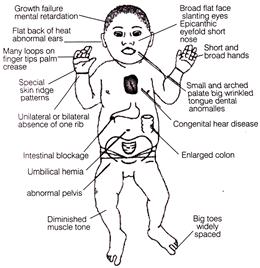
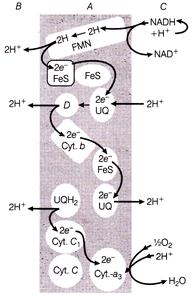 Identify the A, B, C and D of ETS for I. Matrix of mitochondria II. Intermembrane space III. Inner membrane - Mitochondrial
Identify the A, B, C and D of ETS for I. Matrix of mitochondria II. Intermembrane space III. Inner membrane - Mitochondrial 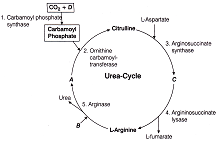 In the given urea cycle identify the markings, if I Arginosuccinate \[{{\operatorname{H}}_{2}}O\] in III \[N{{H}_{3}}\] IV L-Ornithine
In the given urea cycle identify the markings, if I Arginosuccinate \[{{\operatorname{H}}_{2}}O\] in III \[N{{H}_{3}}\] IV L-Ornithine