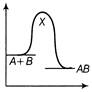
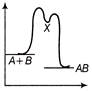
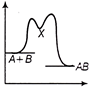
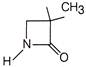
The source S of unknown frequency produces 8 beats with a source of 250 Hz and 12 beats with a source of 270 Hz. The frequency of source 5 is
A)
248 Hz
done
clear
B)
258 Hz
done
clear
C)
272 Hz
done
clear
D)
262 Hz
done
clear
View Answer play_arrow
For a coil having \[\operatorname{L} = 2 mH\], current flows through it is \[\operatorname{i}\,\,=\,\,{{t}^{2}}\,{{e}^{-\,1}}\] then, the time at which emf become zero
A)
1 s
done
clear
B)
3 s
done
clear
C)
4 s
done
clear
D)
2 s
done
clear
View Answer play_arrow
The energy required to charge a parallel plate capacitor having plate separation d and plate area of cross-section A and the uniform electric field E between plates is given by
A)
\[\frac{1}{2}\,{{\varepsilon }_{0}}{{E}^{2}}Ad\]
done
clear
B)
\[{{\varepsilon }_{0}}{{E}^{2}}Ad\]
done
clear
C)
\[\frac{1}{2}\,{{\varepsilon }_{0}}{{E}^{2}}/Ad\]
done
clear
D)
\[{{\varepsilon }_{0}}{{E}^{2}}/Ad\]
done
clear
View Answer play_arrow
To get an output \[O =1\] in the given circuit, which of the following input will be correct?
A)
1 0 0
done
clear
B)
1 0 1
done
clear
C)
1 10
done
clear
D)
0 1 0
done
clear
View Answer play_arrow
If a concave mirror of focal length \[{{f}_{1}}\] is placed at a distance of d from a convex lens of focal length\[{{f}_{2}}\]. A light ray coming from infinity is falling on this combination and returns to infinity. The distance d must equal
A)
\[-{{f}_{1}}+{{f}_{2}}\]
done
clear
B)
\[{{f}_{1}}+{{f}_{2}}\]
done
clear
C)
\[-2\,{{f}_{1}}+{{f}_{2}}\]
done
clear
D)
\[2\,{{f}_{1}}+{{f}_{2}}\]
done
clear
View Answer play_arrow
The wavelength of frequency of electromagnetic waves employed in space communication vary over a range of
A)
\[1\,\,Hz\,\,to\,\,{{10}^{11}}\,Hz\]
done
clear
B)
\[1\,\,Hz\,\,to\,\,{{10}^{4}}\,Hz\]
done
clear
C)
\[10\,\,Hz\,\,to\,\,{{10}^{7}}\,Hz\]
done
clear
D)
\[{{10}^{4}}\,\,Hz\,\,to\,\,{{10}^{11}}\,Hz\]
done
clear
View Answer play_arrow
If a monochromatic light of frequency \[6.0 \times 1{{0}^{14}}Hz\] is produced by a source and the power emitted is \[2.0 \times 1{{0}^{-}}^{3}W\], then the number of photons emitted per second by the source is
A)
\[3\,\,\times \,\,1{{0}^{10}}\]
done
clear
B)
\[5\,\,\times \,\,1{{0}^{15}}\]
done
clear
C)
\[7\,\,\times \,\,1{{0}^{12}}\]
done
clear
D)
\[9\,\,\times \,\,1{{0}^{12}}\]
done
clear
View Answer play_arrow
For the given network (figure). The equivalent resistance between points C and D is
A)
\[1\,\,\Omega \]
done
clear
B)
\[4\,\,\Omega \]
done
clear
C)
\[5\,\,\Omega \]
done
clear
D)
None of these
done
clear
View Answer play_arrow
In the following figure, the direction of induced current in figure in the coil is
A)
anit-clockwise
done
clear
B)
clockwise
done
clear
C)
zero
done
clear
D)
None of these
done
clear
View Answer play_arrow
In the network shown in figure, \[{{C}_{1}}=6\,\mu F\], F and\[C=9\,\mu F\]. The equivalent capacitance between points P and Q is
A)
\[3\,\mu F\]
done
clear
B)
\[6\,\mu F\]
done
clear
C)
\[9\,\mu F\]
done
clear
D)
\[12\,\mu F\]
done
clear
View Answer play_arrow
For the given velocity-time graph, velocity the corresponding displacement-time graph will be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Figures shows four paths for a kicked football ignoring the effects of air on the flight rank the paths according to the initial horizontal velocity component highest first.
A)
1, 2, 3, 4
done
clear
B)
2, 3, 4, 1
done
clear
C)
3, 4, 1, 2
done
clear
D)
4, 3, 2, 1
done
clear
View Answer play_arrow
Which one of the following statement is not true of de-Broglie waves?
A)
All atomic particles in motion have waves of a definite wavelength associated with them
done
clear
B)
The higher the momentum, the larger is the wavelength
done
clear
C)
The faster the particle, the shorter is the wavelength
done
clear
D)
For the same velocity, a heavier particle has a shorter wavelength
done
clear
View Answer play_arrow
If a particle of mass m and charge q is thrown at a speed u against a uniform electric field E. The distance that it will travel before coming to momentary rest is
A)
\[\frac{m{{u}^{2}}}{2qE}\]
done
clear
B)
\[\frac{mu}{qE}\]
done
clear
C)
\[\frac{qE}{m}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A thermodynamic system is taken through the cycle ABCD as shown in figure. Heat rejected by the gas during the cycle is
A)
4pV
done
clear
B)
2pV
done
clear
C)
\[\frac{pV}{2}\]
done
clear
D)
pV
done
clear
View Answer play_arrow
Sand is being dropped on a conveyer belt at the rate of M kg/s. The force necessary to keep the belt moving with a constant velocity of v m/s will be
A)
\[\frac{Mv}{2}\]
done
clear
B)
zero
done
clear
C)
2 Mv
done
clear
D)
Mv
done
clear
View Answer play_arrow
If one end of wire of length 4.5 m of radius 3 mm is fixed to a tree limb and a body weighing 100 N is tied to it then the elongation in the wire is \[\left( Y = 4.8 \times 1{{0}^{11}}N / {{m}^{2}} \right)\]
A)
\[2.22\,\,\times \,\,1{{0}^{-\,5}}\,m\]
done
clear
B)
\[3.22\,\,\times \,\,1{{0}^{-\,5}}\,m\]
done
clear
C)
\[5.5\,\,\times \,\,1{{0}^{-\,7}}\,m\]
done
clear
D)
\[9.2\,\,\times \,\,1{{0}^{-\,12}}\,m\]
done
clear
View Answer play_arrow
Sound waves travels at a speed of 350 m/s through a warm air and at 3500 m/s through gold. What will be the wavelength of a 700 Hz acoustic wave as it enters gold from warm air?
A)
increases by a factor 20
done
clear
B)
decreases of a factor 20
done
clear
C)
increases by a factor 10
done
clear
D)
decreases by a factor 10
done
clear
View Answer play_arrow
If two electrical devices, whose resistances are in the ratio of \[1: 2\] and connected in series, the power dissipated in them has the ratio of
A)
\[1:1\]
done
clear
B)
\[1:4\]
done
clear
C)
\[2:1\]
done
clear
D)
\[1:2\]
done
clear
View Answer play_arrow
If force F is given by relation \[F=M\sqrt{x}+N{{t}^{2}}\], where x is distance and t is time, then the dimensions of M/N will be
A)
\[[{{L}^{2}}{{T}^{2}}]\]
done
clear
B)
\[[{{L}^{1/2}}\,{{T}^{2}}]\]
done
clear
C)
\[[{{L}^{-1/2}}\,{{T}^{2}}]\]
done
clear
D)
\[[ML{{T}^{-\,2}}]\]
done
clear
View Answer play_arrow
An artificial satellite moving in a circular orbit around the earth has a total \[\left( kinetic + potential \right)\] energy potential energy is
A)
\[-\,{{E}_{0}}\]
done
clear
B)
\[1.5\,\,{{E}_{0}}\]
done
clear
C)
\[2\,\,{{E}_{0}}\]
done
clear
D)
\[{{E}_{0}}\]
done
clear
View Answer play_arrow
A disc is rolling (without slipping) on a horizontal surface. C is its centre and Q and P are two points equidistant from C. Let \[{{\nu }_{p}},\,\,{{\nu }_{Q}}\,and\,{{\nu }_{C}}\] be the magnitudes of velocities of points P, Q and C respectively, then
A)
\[{{v}_{Q}}> {{V}_{C}} > {{V}_{P}}\]
done
clear
B)
\[{{v}_{Q}}< {{V}_{C}} < {{V}_{P}}\]
done
clear
C)
\[{{v}_{Q}}= {{v}_{P}}, \,{{v}_{C}}= \frac{1}{2}{{v}_{P}}\]
done
clear
D)
\[{{\operatorname{v}}_{Q}}< {{V}_{C}} > {{V}_{P}}\]
done
clear
View Answer play_arrow
A thin spherical shell of radius R has charge Q spread uniformly over its surface. Which of the following graphs, most closely represents the electric field E (r) produced by the shell in the range \[0\le r<\infty \], where r is the distance from the centre of the shell?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A bullet losses \[19 %\] of its kinetic energy when passes through an obstacle. The percentage change in its speed is
A)
\[\operatorname{increase} by 10%\]
done
clear
B)
\[\operatorname{decrease} by 10%\]
done
clear
C)
\[\operatorname{increase} by 20%\]
done
clear
D)
\[\operatorname{decrease} by 20 %\]
done
clear
View Answer play_arrow
The spring constant of a spring K, when it is divided into n equal parts, then the spring constant of one piece is
A)
K/n
done
clear
B)
n K
done
clear
C)
\[\frac{nK}{(n+1)}\]
done
clear
D)
\[\frac{(n+1)\,K}{n}\]
done
clear
View Answer play_arrow
If mass-energy equivalence is taken into account, when water is cooled to form ice, the mass of water should
A)
increase
done
clear
B)
remain unchanged
done
clear
C)
decrease
done
clear
D)
first increase then decrease
done
clear
View Answer play_arrow
The nuclei \[_{7}^{14}N\,\,and\,\,_{6}^{13}C\] can be described as
A)
isobars
done
clear
B)
isotones
done
clear
C)
isotopes of carbon
done
clear
D)
isotopes of nitrogen
done
clear
View Answer play_arrow
For amplification by a triode, the signal to be amplified is given to
A)
the grid
done
clear
B)
the cathode
done
clear
C)
the glass envelope
done
clear
D)
the anode
done
clear
View Answer play_arrow
In the phenomenon of electric discharge through gases at low pressure, the coloured glow in the tube appears as a result of
A)
excitation of electron in atoms
done
clear
B)
collision between the atom of gas
done
clear
C)
collision between the charged particles emitted from the cathode and the atoms of the gas
done
clear
D)
collision between different electron of the atoms of the gas
done
clear
View Answer play_arrow
A thin rod of length L and mass M is bent at the middle point O at an angle \[60{}^\circ \]. The moment of inertia of the rod about an axis passing through O and perpendicular to the plane of the rod will be
A)
\[\frac{M{{L}^{2}}}{6}\]
done
clear
B)
\[\frac{M{{L}^{2}}}{12}\]
done
clear
C)
\[\frac{M{{L}^{2}}}{24}\]
done
clear
D)
\[\frac{M{{L}^{2}}}{32}\]
done
clear
View Answer play_arrow
If 3 bulbs P, Q and R are connected as shown in figure. What changes occur in brightness of the bulbs, when the switch is closed?
A)
Brightness of P increases but that of R decreases.
done
clear
B)
Brightness of P remains the same but that of R decreases.
done
clear
C)
Brightness of both P and R decreases.
done
clear
D)
Brightness of P increase but that of R remains same.
done
clear
View Answer play_arrow
In the following arrangement as shown in figure, the magnetic field at point O is given by
A)
\[B=\frac{{{\mu }_{0}}}{4\pi }\frac{i}{a}\,(sin\,{{\theta }_{1}}+sin\,{{\theta }_{2}})\]
done
clear
B)
\[B=\frac{{{\mu }_{0}}}{4\pi }\frac{i}{a}\,(sin\,{{\phi }_{1}}+sin\,{{\phi }_{2}})\]
done
clear
C)
Both of [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
In following figure, four curves A, B, C and D are given. The curves are
A)
isothermal for A and D while adiabatic for B and C
done
clear
B)
adiabatic for A and C while isothermal for B and D
done
clear
C)
isothermal for A and 6 which adiabatic for C and D
done
clear
D)
isothermal for A and C while adiabatic for B and D
done
clear
View Answer play_arrow
From the given figure describing the photoelectric effect it may be inferred that
A)
A and 8 both have the same threshold frequency
done
clear
B)
Maximum kinetic energy for both metals depend linearly on frequency both A
done
clear
C)
Stopping potential are different for A and B for some change in frequency
done
clear
D)
B is better photosensiuve material than A
done
clear
View Answer play_arrow
A stone is dropped into a well which is H metre deep, then the time after which the splash of sound is heard (let speed of sound in air is v)
A)
\[\sqrt{\frac{2\,H}{g}}\]
done
clear
B)
\[\sqrt{\frac{2\,H}{g}}+\frac{2\,H}{v}\]
done
clear
C)
\[\sqrt{\frac{2\,H}{g}}+\frac{H}{v}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
For the given system as shown in figure, the pulley are light and frictionless. The tension in the string is
A)
\[2\,\,Mg\,\,sin\,\theta \]
done
clear
B)
\[\frac{1}{2}\,\,Mg\,\,sin\,\theta \]
done
clear
C)
\[\frac{2}{3}\,Mg \sin \,\theta \]
done
clear
D)
\[\frac{3}{2}\,Mg \sin \,\theta \]
done
clear
View Answer play_arrow
A particle of mass \[\operatorname{m}= 5\] units is moving with a uniform speed \[\operatorname{v} =3\,\sqrt{2}\] m in the XOY plane along the\[\operatorname{y}=x+4\]. What will be the magnitude of the angular momentum about the origin?
A)
0 units
done
clear
B)
30 units
done
clear
C)
60 units
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two balls of masses \[{{\operatorname{M}}_{1}}\,\,and\,\,{{M}_{2}}\] are placed on a horizontal frictionless table connected by a spring as shown in figure, how mass \[{{M}_{2}}\] is pulled to the right with a force F? If the acceleration of mass \[{{\operatorname{M}}_{1}}\] is a, then acceleration of mass \[{{M}_{2}}\] will be
A)
\[\frac{F}{{{M}_{1}}}\]
done
clear
B)
\[\frac{F-{{M}_{1}}\,a}{{{M}_{2}}}\]
done
clear
C)
\[\frac{F}{{{M}_{2}}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A block is attached to the three identical springs as shown in figure. Each spring has spring constant k and mass of block is M. If spring 1 is compressed by a small distance x, then time period of oscillation of block is?
A)
\[T=2\pi \sqrt{\frac{M}{k}}\]
done
clear
B)
\[T=2\pi \sqrt{\frac{M}{2\,k}}\]
done
clear
C)
\[T=2\pi \sqrt{\frac{2\,M}{k}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The figure shows variation of electric potential V as a function of x. Arrange the four region in descending order according to the magnitude of n-component of the electric field E within them?
A)
\[{{\operatorname{E}}_{2}}={{E}_{4}}>{{E}_{1}}={{E}_{3}}\]
done
clear
B)
\[{{\operatorname{E}}_{1}}={{E}_{2}}>{{E}_{3}}>{{E}_{4}}\]
done
clear
C)
\[{{\operatorname{E}}_{4}} >{{E}_{1}}= {{E}_{3}} > {{E}_{2}}~\]
done
clear
D)
\[{{\operatorname{E}}_{1}} > {{E}_{2}} > {{E}_{3}} > {{E}_{4}}\]
done
clear
View Answer play_arrow
If a parallel plate capacitor with air as dielectric has capacitance C. A slab of dielectric constant k and having same thickness as the separation between the plates is introduced so as to fill one-fourth of the capacitor as shown in figure, then new capacitance will be
A)
\[(4+k)\frac{C}{4}\]
done
clear
B)
\[(3+k)\frac{C}{4}\]
done
clear
C)
\[\frac{kC}{2}\]
done
clear
D)
\[\frac{kC}{4}\]
done
clear
View Answer play_arrow
In the given distribution, what is the value off?
A)
8 A
done
clear
B)
0
done
clear
C)
2 A
done
clear
D)
5 A
done
clear
View Answer play_arrow
If minimum angle of deviation for a glass prism is equal to its refracting angle. The refractive index of glass is then the angle of prism is
A)
\[2\text{ }si{{n}^{-}}^{1}\,\left( \frac{3}{4} \right)\]
done
clear
B)
\[si{{n}^{-}}^{1}\,\left( \frac{3}{4} \right)\]
done
clear
C)
\[{{\cos }^{-}}^{1}\,\left( \frac{3}{2} \right)\]
done
clear
D)
\[2\,\,{{\cos }^{-}}^{1}\,\left( \frac{3}{4} \right)\]
done
clear
View Answer play_arrow
If a body falls in air, the resistance of air depends to a great extent on the shape of body. Three different shapes are given. Identify the combination of air resistances which truly represents the physical situation? (Cross sectional areas are the same)
A)
\[1<2<3\]
done
clear
B)
\[2<3<1\]
done
clear
C)
\[3<2<1\]
done
clear
D)
\[3<1<2\]
done
clear
View Answer play_arrow
A small sphere carrying a charge q is having in between two parallel plates by a string of length L. Time period of pendulum is \[{{T}_{0}}\]. When parallel plates are charged, the time period changes to T, the ratio of \[\operatorname{T} / {{T}_{0}}\] is equal to
A)
\[{{\left( \frac{g+\frac{qE}{m}}{g} \right)}^{1/2}}\]
done
clear
B)
\[{{\left( \frac{g}{g+\frac{qE}{m}} \right)}^{3/2}}\]
done
clear
C)
\[{{\left( \frac{g}{g+\frac{qE}{m}} \right)}^{1/2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Arrange following in correct sequence of basic strength
A)
\[\operatorname{IV}>I>III>II\]
done
clear
B)
\[III>I>II>IV\]
done
clear
C)
\[II > I > III > IV\]
done
clear
D)
\[\operatorname{I} > III > II > IV\]
done
clear
View Answer play_arrow
Strength of acid increases with the attachment of group showing-J effect and decreases with the attachment of group showing +1 effect. Which of the following is correct sequence of basic strength in aqueous solution?
A)
\[C{{H}_{3}}N{{H}_{2}}<{{(C{{H}_{3}})}_{2}}NH<{{(C{{H}_{3}})}_{3}}N\]
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH<C{{H}_{3}}N{{H}_{2}}<{{(C{{H}_{3}})}_{3}}N\]
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N<{{(C{{H}_{3}})}_{2}}NH<C{{H}_{3}}N{{H}_{2}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}N<C{{H}_{3}}N{{H}_{2}}<{{(C{{H}_{3}})}_{2}}NH\]
done
clear
View Answer play_arrow
IUPAC name of
A)
1, 2-dimethyl cyclobutane and 2, 3-dimethyl butene
done
clear
B)
1, 2-dimethyl cyclobut 1-ene and 3, 4-dimethyl cyclo-but-1-ene
done
clear
C)
2, 3-dimethyl cyclobutene and 2, 3-dimethyl cyclobutane
done
clear
D)
2, 3-dimethyl butene and 1, 2-dimethylcyclobut-1-ene
done
clear
View Answer play_arrow
Phosphorous in any organic compound can be estimated by the formula \[% of P=wt. of M{{g}_{2}} {{P}_{2}}{{O}_{7}}\,\,\frac{formed\,\,\times \,62}{wt.\,\,of\,\,compound}\times 100\,\,\times 222\]\[% of P=wt. of M{{g}_{2}} {{P}_{2}}{{O}_{7}}\,\,\frac{formed\,\,\times \,62}{wt.\,\,of\,\,compound}\times 100\,\,\times 222\] This formula is related to
A)
Carius method
done
clear
B)
Lassaigne's method
done
clear
C)
Kjeldahl's method
done
clear
D)
Duma's method
done
clear
View Answer play_arrow
reacts with \[{{B}_{2}}{{H}_{6}}\] in presence of \[{{\operatorname{H}}_{2}}{{O}_{2}}\] produces
A)
done
clear
B)
done
clear
C)
done
clear
D)
\[{{\operatorname{RCH}}_{2}}CHO\]
done
clear
View Answer play_arrow
When benzene is heated with chlorine in the presence of sunlight, it forms
A)
BHC
done
clear
B)
cyclopropane
done
clear
C)
p-dichlorobenzene
done
clear
D)
None of these
done
clear
View Answer play_arrow
The product formed when toluene is heated in light with \[C{{l}_{2}}\] and in absence of halogen carrier is
A)
chlorobenzene
done
clear
B)
gammexane
done
clear
C)
benzotrichloride
done
clear
D)
DDT
done
clear
View Answer play_arrow
In \[{{\operatorname{IF}}_{7}}\] molecule, central atom has all the bonded electrons i.e., no lone pair is present on the central atom. Structure of \[{{\operatorname{IF}}_{7}}\] is
A)
pentagonal bipyramidal
done
clear
B)
octahedral
done
clear
C)
square pyramidal
done
clear
D)
octahedral monopyramidal
done
clear
View Answer play_arrow
On dissolving moderate amount of sodium metal in liquid \[{{\operatorname{NH}}_{3}}\] at low temperature, which of the following does not occur?
A)
\[{{\operatorname{Na}}^{+}}\] ions are formed in the solution
done
clear
B)
Blue coloured solution is obtained
done
clear
C)
Liquid \[N{{H}_{3}}\] becomes good conductor of electricity
done
clear
D)
Liquid \[N{{H}_{3}}\] remains diamagnetic
done
clear
View Answer play_arrow
Most of the compounds of transition metals are coloured in solid state or solutions. The transition metals which have either completely filled \[d-orbitals\] or completely empty \[d-orbitals\] are colourless. The colour of transition metal is due to
A)
presence of unpaired d-electrons
done
clear
B)
small size
done
clear
C)
metallic nature
done
clear
D)
All of these
done
clear
View Answer play_arrow
Electron gain enthalpy increases across the period and decreases down the group. Which of the following order of electron gain enthalpy is correct?
A)
\[F < Cl < Br < l\]
done
clear
B)
\[F < Cl > Br > l\]
done
clear
C)
\[I > Br > F < Cl~\]
done
clear
D)
\[\operatorname{I} < Cl < Br > F\]
done
clear
View Answer play_arrow
In which of the following pairs both the completes show optical isomerism?
A)
\[cis-\,\,{{\left[ Cr{{({{C}_{2}}{{O}_{4}})}_{2}}C{{l}_{2}}^{\,} \right]}^{3}}^{-},\,\,cis-\left[ Co{{(N{{H}_{3}})}_{4}}Cl \right]\]
done
clear
B)
\[\left[ PtCl\,(dien) \right]Cl,\,\,{{[NiC{{l}_{2}}B{{r}_{2}}]}^{\,2\,-}}\]
done
clear
C)
\[[CO(N{{O}_{3}}){{(N{{N}_{3}})}_{3}}],\,\,cis\,\,-[Pt{{(en)}_{2}}]C{{l}_{2}}]\]
done
clear
D)
\[\left[ Co{{\left( en \right)}_{3}} \right]C{{l}_{3}}, \,cis\,-\,\,\left[ Co{{\left( en \right)}_{2}}C{{l}_{2}} \right]Cl\]
done
clear
View Answer play_arrow
\[Xe{{F}_{\text{6}}}\] is colourless crystalline solid. It undergoes hydrolysis in water. The final product obtained is an explosive solid. This explosive solid will be
A)
\[\operatorname{Xe}{{O}_{3}}\]
done
clear
B)
XeO
done
clear
C)
\[\operatorname{Xe}{{O}_{2}}\]
done
clear
D)
Xe
done
clear
View Answer play_arrow
The number of spectral lines that can be possible when electrons in 7th shell in different hydrogen atoms return to the second shell is
A)
12
done
clear
B)
15
done
clear
C)
14
done
clear
D)
10
done
clear
View Answer play_arrow
Principal quantum number defines the principal shell in which electron is revolving around the nucleus and Azimuthal quantum number described the name of the subshell and the shape of orbitals. The values of principal and Azimuthal quantum number for 23rd electron present in iron atom are.
A)
3 and 2
done
clear
B)
3 and 1
done
clear
C)
4 and 1
done
clear
D)
4 and 0
done
clear
View Answer play_arrow
A compound A is obtained by reaction between first member of 1st group element and lightest element of periodic table, then correct statement about compound A is
A)
A is acidic in nature
done
clear
B)
A is basic in nature
done
clear
C)
A is amphoteric in nature
done
clear
D)
A turns blue litmus paper red
done
clear
View Answer play_arrow
Potassium superoxide is the inorganic compound with the formula \[K{{O}_{2}}\]. It is a yellow solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide ion. Which of the following statement is incorrect regarding potassium superoxide?
A)
Oxidation state of oxygen in \[K{{O}_{2}}\] is -1/2
done
clear
B)
Oxidation state of potassium is + 1
done
clear
C)
It is used in preparation of breathing mask
done
clear
D)
It is a reducing agent
done
clear
View Answer play_arrow
Werner's theory was the first successful theory, which explained the properties of complexes. According to Werner's theory the primary and secondary valence of \[[Co\,C{{l}_{3}} {{\left( N{{H}_{3}} \right)}_{3}}C{{l}_{3}}\] is
A)
3 and 3 respectively
done
clear
B)
6 and 6 respectively
done
clear
C)
3 and 6 respectively
done
clear
D)
6 and 3 respectively
done
clear
View Answer play_arrow
Boron on reaction with \[{{\operatorname{CO}}_{2}}\,\,and\,\,{{H}_{2}}O\] produces boron hydride as a major constituent the other byproduct produced in these reactions are
A)
\[{{\operatorname{H}}_{2}}O and C\]
done
clear
B)
\[{{\operatorname{N}}_{2}} and C{{O}_{2}}\]
done
clear
C)
\[{{\operatorname{H}}_{2}} and C\]
done
clear
D)
\[{{\operatorname{H}}_{2}}O and C{{O}_{2}}\]
done
clear
View Answer play_arrow
\[{{\operatorname{NaBH}}_{4}}\] on reaction with \[{{I}_{2}}\] produces a compound X, X on reaction with oxygen produces an anhydride. X on reaction with \[N{{H}_{3}}\] forms an adduct. X is
A)
\[{{B}_{2}}{{H}_{6}}\]
done
clear
B)
LiH
done
clear
C)
\[B{{H}_{4}}\]
done
clear
D)
\[IB{{H}_{2}}\]
done
clear
View Answer play_arrow
Equilibrium constant of a chemical reaction is 1.5 and rate constant of forward reaction is\[7.5\,\,\times \,\,1{{0}^{-\,4}}\]. Then, rate constant of backward reaction will be
A)
\[1.125\,\,\times \,1{{0}^{-\,3}}\]
done
clear
B)
\[2.225\,\,\times \,1{{0}^{-\,3}}\]
done
clear
C)
\[3.335\,\,\times \,1{{0}^{-\,5}}\]
done
clear
D)
\[1.125\,\,\times \,1{{0}^{-\,1}}\]
done
clear
View Answer play_arrow
The pH of the solution is defined as the negative logarithm of the concentration of hydrogen ions which it contains. pH of \[0.365 g{{L}^{-\,1}}\,HCl\] solution will be
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
0.1
done
clear
View Answer play_arrow
When copper metal is dipped in a solution of \[HN{{O}_{3}}\], it produces copper nitrate and some gaseous molecule, the change in oxidation number of nitrogen is
A)
+2
done
clear
B)
+1
done
clear
C)
+4
done
clear
D)
0
done
clear
View Answer play_arrow
What amount of current will be required to evolve 20 g of chlorine in 6 h from HCl solution?
A)
10 A
done
clear
B)
15 A
done
clear
C)
5 A
done
clear
D)
2.45 A
done
clear
View Answer play_arrow
Which of the following is not the assumption of kinetic theory of gases?
A)
The actual volume of the gaseous molecules is negligible as compared to the total volume of the gas
done
clear
B)
Molecules are perfectly elastic
done
clear
C)
The critical temperature is the measure of the kinetic energy of the molecule
done
clear
D)
The effect of gravity on motion of molecules is negligible
done
clear
View Answer play_arrow
A solid is made of two elements P and Q. Atoms P are in ccp arrangement and atoms Q occupy all the octahedral voids and half of the tetrahedral voids, then the simplest formula of the compound is
A)
\[PQ{{\,}_{2}}\]
done
clear
B)
\[{{P}_{2}}Q\]
done
clear
C)
PQ
done
clear
D)
\[{{P}_{2}}{{Q}_{2}}\]
done
clear
View Answer play_arrow
The space occupied in different packing?s is called packing fraction. \[\operatorname{Packing} fraction =\,\,\frac{total\,\,volume\,\,of\,\,sphere}{volume\,\,of\,\,the\,\,unit\,\,cell}\] Fraction of total volume occupied by atoms in a simple cubic unit cell is
A)
\[\frac{\pi }{2}\]
done
clear
B)
\[\frac{\sqrt{3\,\pi }}{8}\]
done
clear
C)
\[\frac{\sqrt{2\,\pi }}{6}\]
done
clear
D)
\[\frac{\pi }{6}\]
done
clear
View Answer play_arrow
Spontaneity of a process is decided by the value of\[\Delta \,A\]. A negative value of \[\Delta \,G\] shows a spontaneous process. Since, the value of \[\Delta \,G\] depends upon \[\Delta S and \Delta H\], these two also play an important role for deciding spontaneity of a process. The condition for spontaneity of any chemical reaction, if
A)
\[T\Delta S =\Delta H\] and both \[\Delta H and \Delta S\] are positive
done
clear
B)
\[T\Delta S >\Delta H\] and both \[\Delta H and \Delta S\] are positive
done
clear
C)
\[T\Delta S <\Delta H\] and both \[\Delta H and \Delta S\] are positive
done
clear
D)
\[T\Delta S >\Delta H\] and \[\Delta H\] is positive and \[\Delta S\] is negative
done
clear
View Answer play_arrow
A container has hydrogen and oxygen mixture in ratio of \[1 : 1\] by weight, then
A)
internal energy of the mixture decreases
done
clear
B)
internal energy of the mixture increases
done
clear
C)
entropy of the mixture increases
done
clear
D)
entropy of the mixture decreases
done
clear
View Answer play_arrow
Arrhenius proposed a quantitative relationship between rate constant and temperature as \[\operatorname{k}=\,\,A{{e}^{-{{E}_{a}}/\,RT}}\] or In \[k=\ln \,\,A-{{E}_{a}}/RT\]. What will be the slope of graph when ln k is plotted versus 1/T?
A)
\[\frac{{{E}_{a}}}{R}\]
done
clear
B)
\[-\frac{{{E}_{a}}}{R}\]
done
clear
C)
\[\frac{1}{T}\]
done
clear
D)
ln A
done
clear
View Answer play_arrow
Freundlich gave the relationship between the gas adsorbed by solid adsorbent surface and pressure of the gas at a particular temperature. Which of the following equation represent Freundlich adsorption isotherm?
A)
\[\frac{x}{m}=k{{p}^{1\,/\,n}}\]
done
clear
B)
\[\frac{x}{m}=\frac{k}{p}\]
done
clear
C)
\[\frac{x}{m}=kp\]
done
clear
D)
\[\frac{x}{m}=k{{p}^{n}}\]
done
clear
View Answer play_arrow
An exothermic chemical reaction occurs in two steps as follows I. \[A+B\,\,\to \,\,X\,(fast)\] II. \[X\,\to \,\,AB\,(slow)\] The progress of the reaction can be best represented by
A)
done
clear
B)
done
clear
C)
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following plots represents the behaviour of an ideal binary liquid solution?
A)
Plot of \[{{p}_{total}}\,\,vs\,\,{{Y}_{A}}\] (mole-fraction of A in vapour phase) is linear
done
clear
B)
Plot of \[{{p}_{total}}\,\,vs\,\,{{Y}_{B}}\]
done
clear
C)
Plot of \[1/{{p}_{total}}\,\,vs\,\,{{Y}_{A}}\]
done
clear
D)
Plot of \[1/{{p}_{total}}\,\,vs\,\,{{Y}_{B}}\] is non-linear
done
clear
View Answer play_arrow
A compound obtained by burning of an element belongs to 2nd group and 3rd period when passes through red litmus paper it turns blue the equivalent weight of compound is
A)
58
done
clear
B)
29
done
clear
C)
116
done
clear
D)
14.5
done
clear
View Answer play_arrow
What will be the formation of \[C{{O}_{2}}\] is? \[\operatorname{C}+{{O}_{2}}\,\xrightarrow{{}}\,\,C{{O}_{2}}~~~~~\Delta H\,\,=\,\,X\] \[\operatorname{CO}+\frac{1}{2}{{O}_{2}}\,\xrightarrow{{}}\,\,C{{O}_{2}}~~~~~\Delta H\,\,=\,\,Y\]
A)
\[\operatorname{X}\,-Y\]
done
clear
B)
\[Y-2X\]
done
clear
C)
\[X+Y\]
done
clear
D)
\[2\,X-Y\]
done
clear
View Answer play_arrow
Which of the following is not correctly matched? Column I Column II
A)
\[Xe{{O}_{3}}\] tetrahedral
done
clear
B)
\[Xe{{F}_{4}}\] square planar
done
clear
C)
\[Xe{{F}_{2}}\] linear
done
clear
D)
\[Xe{{O}_{\text{2}}}{{\text{F}}_{\text{2}}}\] square planar
done
clear
View Answer play_arrow
How many carbon in 2 methyl pentan-3-one is \[{{\operatorname{sp}}^{2}}\] hybridised?
A)
2
done
clear
B)
1
done
clear
C)
0
done
clear
D)
3
done
clear
View Answer play_arrow
Most of the organic compounds are generally used in the formation of drugs. \[1.1%\] solution of phenol is used as
A)
antiseptic
done
clear
B)
antipyretic
done
clear
C)
disinfectant
done
clear
D)
tranquilizer
done
clear
View Answer play_arrow
CFCs, HCFCs and haloes destroy the earth's protective ozone layer, which shields the earth from harmful ultraviolet rays generated from the SUN. Why CFC are widely used in air conditioners?
A)
Because CFC are highly reactive
done
clear
B)
It is highly inflammable
done
clear
C)
It is non-reactive
done
clear
D)
All of these are true
done
clear
View Answer play_arrow
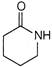
Nylon 4 can be prepared by polymerisation of
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Aldo hexoses are six carbon sugars with four chirality centres and an aldehyde carbonyle group. There are 16 possible stereo isomeric aldo hexoses. D-glucose and D-manmose are X of each other X is
A)
epimer
done
clear
B)
anomer
done
clear
C)
enantiomer
done
clear
D)
diasteromer
done
clear
View Answer play_arrow
In the below reaction, B is
A)
caproic acid
done
clear
B)
caproic anhydride
done
clear
C)
caprolactum
done
clear
D)
cyclohexanone oxime
done
clear
View Answer play_arrow
Which of the following chemical reactions will produce phenol?
A)
\[Aniline\,\,\xrightarrow[0-5{}^\circ \,\,C]{HN{{O}_{2}}}\,\,A\,\,\xrightarrow[{}]{{{H}_{3}}P{{O}_{4}}}\,B\]
done
clear
B)
\[Aniline\,\,\xrightarrow[0-5{}^\circ \,\,C]{HN{{O}_{2}}}\,\,A\,\,\xrightarrow[{}]{{{H}_{2}}O\,\,(hot)}\,B\]
done
clear
C)
\[Benzene\,\,\xrightarrow[{}]{FeC{{l}_{3}}}\,\,A\,\,\xrightarrow[{}]{alc.\,KOH}\,B\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
Primary amine on reaction with a mixture of chloroform and ale. KOH produced isocyanides while phenol on reaction with a mixture of chloroform and aq KOH produces
A)
salicylic acid
done
clear
B)
benzole acid
done
clear
C)
benzaldehyde
done
clear
D)
salicylaldehyde
done
clear
View Answer play_arrow
\[LiAl{{H}_{4}}\] is a strong reducing agent and reduces most of the functional groups to alcohol. The product obtained after reduction of dimethyl carbinol with \[LiAl{{H}_{4}}\], it produces
A)
propan-2-ol
done
clear
B)
propan-1-ol.
done
clear
C)
pentan-2-ol
done
clear
D)
propanoic acid
done
clear
View Answer play_arrow
Assertion [A] Blood is a connective tissue with fluid matrix. Reason [R] Haemopoiesis is the process of formation of blood.
A)
Both A and R are true and R is the connect explanation of A
done
clear
B)
Both A and R are true, but R is not the connect explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
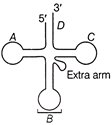
Label the parts in the given diagram with the following set of codes,
A)
A-D-loop; B-Anticodon arm; \[C-T\Psi C\] arm; D-CCA acceptor arm
done
clear
B)
\[A-C\Psi C\] Loop; B-Amino acid arm; C-Acceptor arm; D-Anticodon arm
done
clear
C)
A-D-loop; \[B-T\Psi C\] arm; C-Acceptor arm; D-Anticodon arm
done
clear
D)
A-Anticodon arm; B-Amino acid arm; \[C-C\Psi C\] arm; D-Acceptor arm
done
clear
View Answer play_arrow
Touch receptors present in the skin are known as
A)
Parisian corpuscles
done
clear
B)
Meissner's corpuscles
done
clear
C)
Krause's end bulbs
done
clear
D)
Organs of Ruffian
done
clear
View Answer play_arrow
Select the incorrect statement from the following options.
A)
Nucleic acid - Genetic material
done
clear
B)
Proteins - Hetero polymers of amino acids
done
clear
C)
Ribozymes - Protein with catalytic activity
done
clear
D)
Collagen - Most abundant protein in animal world
done
clear
View Answer play_arrow
Bone and cartilage can be distinguished by the presence of one of the following structure
A)
lacuna
done
clear
B)
myofibril
done
clear
C)
Haversian canal
done
clear
D)
chromatophores
done
clear
View Answer play_arrow
Which of the following disease(s) may lead to Iron Deficiency Anaemia (IDA). Peptic ulcer Inflammatory Bowel Disease (IBD) Hypochlorhydria Pregnancy
A)
I and III
done
clear
B)
All may leads to IDA
done
clear
C)
Only IV
done
clear
D)
I, II and IV
done
clear
View Answer play_arrow
When osmotic potential is either zero or negative and pressure potential is positive, then the water potential will be
A)
always negative
done
clear
B)
always positive
done
clear
C)
sometimes negative and sometimes positive
done
clear
D)
do not show any specificity
done
clear
View Answer play_arrow
Mesangial cells are specialised smooth muscle cells that function to regulate blood flow in the blood vessels. They are found in which organ of the body?
A)
Testis
done
clear
B)
Brain
done
clear
C)
Kidney
done
clear
D)
Ovary
done
clear
View Answer play_arrow
Match the following columns.
Column I (Structure of Earthworm) Column II (Its location) A. Pairs of testis 1. 10th and 11th segment B. Pairs of seminal vesicles 2. 17th and 19th segment C. Pair of ovary 3. 13th segment D. Accessory gland 4. 11th and 12th segment
A)
A-1, B-4, C-3, D-2
done
clear
B)
A-2, B-4, C-3, D-1
done
clear
C)
A-4, B-1, C-3, D-2
done
clear
D)
A-2, B-3, C-1, D-4
done
clear
View Answer play_arrow
The biochemical or morphological change of meristematic daughter cell to become a permanent cell is called
A)
differentiation
done
clear
B)
dedifferentiation
done
clear
C)
dedifferentiation
done
clear
D)
differentiation
done
clear
View Answer play_arrow
Which one of the following is an exclusive character of living beings?
A)
Isolated metabolic reactions occurring in vitro.
done
clear
B)
Increase in the body mass from inside only
done
clear
C)
Perception of happening of events in the environment and their memory
done
clear
D)
Increase in mass by accumulation of material both on surface as well as internally
done
clear
View Answer play_arrow
Match the following columns.
Column I (Organism) Column II (Characteristics) A. Euglena 1. Cuasing haemoglobinuric fever in cattle B. Noctiluca 2. Connecting link between plants and animals C. Babesia 3. Free living marine D. Triconympha 4. Symbiotism in termite?s get
A)
A-4, B-3, C-1, D-2
done
clear
B)
A-2, B-4, C-3, D-1
done
clear
C)
A-2, B-3, C-1, D-4
done
clear
D)
A-3, B-4, C-2, D-1
done
clear
View Answer play_arrow
CFC is an organic compound that contains only carbon, chlorine and fluorine, produced as a volatile derivative of methane and ethane. They are the main culprit for ozone depletions. Which of the following pledged to phase out CFC?s?
A)
Riodeclaration
done
clear
B)
Helsinki declaration
done
clear
C)
Kyoto Protocol
done
clear
D)
Earth Summit
done
clear
View Answer play_arrow
Which of the following statement is not correct?
A)
Double fertilisation is characteristic feature of angiosperms. It was discovered by SG Naw as chin
done
clear
B)
In adventive embryos, embroys develop directly from the nucellus
done
clear
C)
Milky water of coconut is called liquid endosperm
done
clear
D)
In anatropous ovule, the micropyle, funicle and chalaza lie in a straight line
done
clear
View Answer play_arrow
You have hired a driver, which is 55 year old. During the interaction before joing. He had informed you very honestly that he is a diabetic patient. Which of the following suggestion you will give him before going on a drive. I. Take a double dose of insulin, so that he does not develop any problem during driving II. Always carry a identification card with full detail III. Take regular snacks or meal during long journey. IV. Drink only 500 mL of water in 24 hours to avoid any hypoglycemic attack
A)
I and III
done
clear
B)
III and IV
done
clear
C)
I and II
done
clear
D)
II and III
done
clear
View Answer play_arrow
Cichlid fish became extinct due to a alien species in the lake Victoria. Find out the name of that alien species from the following options.
A)
Nile perch
done
clear
B)
African catfish
done
clear
C)
Morels
done
clear
D)
Water hyacinth
done
clear
View Answer play_arrow
As per the fluid mosaic theory/model of cell membrane, lipid is also present in the membrane select the correct organelles in which it synthesised
A)
smooth endoplasmic reticulum
done
clear
B)
Golgi apparatus
done
clear
C)
mitochondria
done
clear
D)
lysosome
done
clear
View Answer play_arrow
Label the given diagram with options given below.
A)
A-Epicotyl, B-Hypocotyl, C-Cotyledons
done
clear
B)
A-Hypocotyl, B-Epicotyl, C-Cotyledons
done
clear
C)
A-Hypocotyl, B-Cotyledons, C-Epicotyl
done
clear
D)
A-Epicotyl, B-Cotyledons, C-Hypocotyl
done
clear
View Answer play_arrow
Biosystematics is
A)
the identification and arrangement of different organisms on the basis of their morphological characters
done
clear
B)
the systematic arrangement of different organisms on the basis of their ecological niche
done
clear
C)
the analysis of the geographical distribution of population or organisms and than systematic organisation of the population/organisms
done
clear
D)
the systematic arrangement of organisms on the basis of their evolutionary, genetic and biochemical framework to assess the taxonomic relationships of organisms or populations
done
clear
View Answer play_arrow
Statement [A] Fertilised eggs of honeybee develop into drone and unfertilised eggs develop into female. Statement [B] Female develops par then genetically.
A)
Both statements are true and B is the correct explanation of A.
done
clear
B)
Both statements are true, but B is not the correct explanation of A.
done
clear
C)
Both A are B false
done
clear
D)
A is false, but B is true
done
clear
View Answer play_arrow
All below are the symptoms of one of the following condition Urtirearia Hypotension Wheeze Flushing Angioedema of lips
A)
Mitral stenosis
done
clear
B)
Aortic stenosis
done
clear
C)
Acute asthma
done
clear
D)
Anaphylaxis
done
clear
View Answer play_arrow
These processes are compulsory for the complete development of the male gametophyte from pollen mother cell
A)
two meiotic cell division
done
clear
B)
one mitotic cell division and two meiotic cell division
done
clear
C)
one meiotic cell division and two mitotic cell division
done
clear
D)
two meiotic cell division and one mitotic cell division
done
clear
View Answer play_arrow
Which one of following term is used for local population, adapted genetically to its particular environment
A)
biotic community
done
clear
B)
demes
done
clear
C)
biome
done
clear
D)
ecotype
done
clear
View Answer play_arrow
Human eyes have lot of glands, which are prone to infection. Infection of which modified gland of eyelids causes style or external hordeolum?
A)
Gland of Moll
done
clear
B)
Meibomian gland
done
clear
C)
Gland of Zeis
done
clear
D)
Lacrimal gland
done
clear
View Answer play_arrow
The Respiratory Quotient (RQ) of \[{{C}_{4}}{{H}_{6}}{{O}_{6}}\], is
A)
1.33 (more than unity)
done
clear
B)
0.7 (less than unity)
done
clear
C)
zero
done
clear
D)
\[\infty \,\,(infinite)\]
done
clear
View Answer play_arrow
Identify the correct relationship with reference to water potential of a plant cell.
A)
\[{{\Psi }_{w}}={{\Psi }_{m}}+{{\Psi }_{s}}+{{\Psi }_{p}}\]
done
clear
B)
\[{{\Psi }_{w}}={{\Psi }_{m}}-{{\Psi }_{s}}-{{\Psi }_{p}}\]
done
clear
C)
\[{{\Psi }_{w}}={{\Psi }_{m}}-{{\Psi }_{s}}+{{\Psi }_{p}}\]
done
clear
D)
\[{{\Psi }_{w}}={{\Psi }_{m}}+{{\Psi }_{s}}-{{\Psi }_{p}}\]
done
clear
View Answer play_arrow
Symptoms alcoholic withdrawal are
A)
tranquillizers
done
clear
B)
sedatives
done
clear
C)
opiate
done
clear
D)
stimulants
done
clear
View Answer play_arrow
Which among the following represent central dogma?
A)
\[\operatorname{RNA} \to DNA\,\to \,\,Protein\]
done
clear
B)
\[\Pr otein \to RNA\,\to \,\,DNA\]
done
clear
C)
\[\operatorname{DNA}\,\,\rightleftharpoons \,\,RNA\,\,\to \,\,Protein\]
done
clear
D)
\[\operatorname{DNA}\,\to Protein\,\,\to \,\,RNA\]
done
clear
View Answer play_arrow
Match the followings columns.
Column I Column II A. Food storing tissue 1. Grapes B. Parthenocarpic fruit 2. Mango C. Membranous seed out 3. Maize D. Single seeded fruit developing from monocarpellary superior ovary 4. Litchi Endosperm
A)
A-1, B-2, C-4, D-5
done
clear
B)
A-5, B-1, C-3, D-2
done
clear
C)
A-2, B-4, C-3, D-1
done
clear
D)
A-5, B-2, C-4, D-1
done
clear
View Answer play_arrow
A 12 year old boy, whose body mass index BMI is 18.5, starts complaining of stomach-ache on every Monday morning. His doctor has not found any organic cause despite of all investigations. What may be the cause of this abdominal pain
A)
Psychogenic
done
clear
B)
Pseudo-obstruction
done
clear
C)
Haem orrhoids
done
clear
D)
Hirsch rung?s disease
done
clear
View Answer play_arrow
Match the following columns.
Column I (Classes of Enzymes) Column II (Example) A. Transferases 1. Histadine decarboxylases B. Lyases 2. Amulase C. Hydrolases 3. Glutamate pyruvate transaminase D. Ligases 4. Pyruvate carboxylase
A)
A-4, B-2, C-3, D-1
done
clear
B)
A-3, B-1, C-2, D-4
done
clear
C)
A-1, B-2, C-3, D-4
done
clear
D)
A-4, B-1, C-3, D-2
done
clear
View Answer play_arrow
Which of the following enzyme is called regulatory enzyme of glycolysis?
A)
Phosphoglucoisomerase
done
clear
B)
Phosphofructokinase
done
clear
C)
Phosphotriose isomerase
done
clear
D)
Mutase
done
clear
View Answer play_arrow
A individual has low blood potassium and hypertension. Blood investigations show that has blood aldosterone x very high. He might be suffering from
A)
Addison's disease
done
clear
B)
cretinism
done
clear
C)
gigantism
done
clear
D)
Conn's disease
done
clear
View Answer play_arrow
Which of the following statement(s) is/are true I. Salivary gland chromosomes or polytene chromosome have been seen in dipteran insects II. Lampbrush chromosomes have been observed in primary oocyte nuclei of vertebrate only III. Lampbrush chromosomes have been observed in primary oocyte nuclei of vertebrates as well as inventebrates. IV. Kinetochore is an another name for satellites chromosomes.
A)
I and IV
done
clear
B)
I, II, III and IV
done
clear
C)
II and III
done
clear
D)
I and III
done
clear
View Answer play_arrow
Identify the correct sequence of gene in an operon.
A)
Promoter, operator, regulator, structural
done
clear
B)
Structural, regulator, operator, promoter
done
clear
C)
Regulator, promoter, operator, structural
done
clear
D)
Operator, structural, regulator, promoter
done
clear
View Answer play_arrow
Select the correct statement from the given options regarding homologous organs?
A)
Similar in origin but dissimilar in functions
done
clear
B)
Similar in origin with similar/dissimilar function
done
clear
C)
Dissimilar in origin and dissimilar in structures
done
clear
D)
Similar in origin but dissimilar in functions
done
clear
View Answer play_arrow
Which is not used for detection of cancer of internal organs?
A)
Radiography
done
clear
B)
Computed tomography
done
clear
C)
Magnetic resonance imaging
done
clear
D)
Enzyme linked immune sorbent assay
done
clear
View Answer play_arrow
What happens during stomatal opening
A)
Malate enter into the guard cell
done
clear
B)
\[{{H}^{+}}\] extruding from guard cells
done
clear
C)
\[{{K}^{+}}\] enters into the guard cell
done
clear
D)
All of these
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Azoic 1. Era of ancient life B. Proterozoic 2. Era of no life C. Palaeozoic 3. Era of medieval life D. Mesozoic 4. Era of early life
Codes
A)
A-1, B-3, C-4, D-2
done
clear
B)
A-2, B-4, C-1, D-3
done
clear
C)
A-1, B-2, C-3, D-4
done
clear
D)
A-4, B-3, C-2, D-1
done
clear
View Answer play_arrow
Hassal's bodies which area potent source of the cytokine TS2P, are found in one of the following organ
A)
liver
done
clear
B)
thymus
done
clear
C)
adrenal
done
clear
D)
thyroid
done
clear
View Answer play_arrow
Being a student of biology, your one of the relative called you and informed that he has a chest pain which is peripheral, extracted by deep breathing or coughing. Which of the following system is most likely seems to be involved?
A)
Respiratory system
done
clear
B)
Cardiac system (cardio-vascular system)
done
clear
C)
Neural system
done
clear
D)
Sensory system
done
clear
View Answer play_arrow
RJFLP, RAPD, VNTR and STR are examples of
A)
enzymes
done
clear
B)
molecular marker
done
clear
C)
plasmids
done
clear
D)
antigens
done
clear
View Answer play_arrow
Assertion [A] Neo-Lamarckism is the modified form of Lamarckism. Reason [R] Evidences in favour of inheritance of acquired characters support Neo-Lamarckism.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
When one molecule of glucose enters into glycolysis two molecules of pyruvic acid is formed then pyruvic acid enter into the Krebs' cycle. How many carbon dioxide molecules are formed when one glucose molecule passes through glycolyis and Krebs' cycle?
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
Auxin cause apical dominance this hormone is mainly produces in the apical part of short the reason for apical dominance by auxin.
A)
Auxin in apical part of shoot have the ability to bans part the cytokinin from root in the apical part of shoot
done
clear
B)
Cytokinin is not transported to apical part that is why auxin cause apical dominance
done
clear
C)
Both [a] and [b].
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Choose the correct statement.
I. LSD - Ergot
II. Mescaline - Peyote cactus
III. Marijuana - Angel dust
IV. Ganja - Cannabis sativa
V. Opium - Synthetic drug
A)
I, II and V
done
clear
B)
II, III and IV
done
clear
C)
I, II and IV
done
clear
D)
None of these
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Biosynthesis of IAA 1. Zinc B. Protein synthesis 2. Manganese C. Hydrolysis of pyrophosphate 3. Nitrogen D. Photolysis of water 4. Phosphate
A)
A-1, B-4, C-2, D-3
done
clear
B)
A-4, B-2, C-1, D-3
done
clear
C)
A-1, B-3, C-4, D-2
done
clear
D)
A-3, B-4, C-2, D-1
done
clear
View Answer play_arrow
A hormone is a class of regulatory biochemical produced in particular parts of organism by specific cells, glands and/or tissues. Identify the systemic hormone from the following options.
A)
Oxytocin
done
clear
B)
Soma to statin
done
clear
C)
Melatonin
done
clear
D)
Soma to troth in
done
clear
View Answer play_arrow
Which of the following statement is incorrect?
A)
Ethidium bromide stained DNA can be seen in light
done
clear
B)
DNA cannot be seen in visible light
done
clear
C)
Ethidium bromide stained DNA can be seen when exposed to UV-light
done
clear
D)
DNA cannot be seen in visible light even if stained
done
clear
View Answer play_arrow
Identify the types of birth control device based on the features given below. I. Requires the erection of penis II. Can cause excess menstrual bleeding and pain. III. Relatively unreliable and sometimes messy.
A)
I-Diaphragm II-Cervical cap III-Implant
done
clear
B)
I-Levonorgestral II-Hormonal pill III-Norplant
done
clear
C)
I-Condom II-IUD III-Vaginal suppositories
done
clear
D)
I-Cervical cap II-Oral contraceptive III-Condom
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Lymph 1. Wharton?s jelly B. Blood 2. Adipose cells C. Mucoid tissue 3. Less glucose concentration D. Reticular tissue 4. Middle man
A)
A-1, B-4, C-3, D-2
done
clear
B)
A-2, B-4, C-3, D-1
done
clear
C)
A-4, B-1, C-3, D-2
done
clear
D)
A-2, B-3, C-1, D-4
done
clear
View Answer play_arrow
Match the following columns.
Phase Duration in hours Vicia faba Mouse L-cells Human Hela cells A. \[{{G}_{1}}\] 1. 12 3-4 3 B. S 2. 12 12 12 C.\[{{G}_{2}}\] 3. 1 1 1 D. M 4. 6 10 10
A)
A-2, B-4, C-2, D-3
done
clear
B)
A-3, B-2, C-1, D-4
done
clear
C)
A-4, B-1, C-3, D-2
done
clear
D)
A-1, B-2, C-3, D-4
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Allogamy 1. Fusion of gametes B. Homogamy 2. Cross-pollination C. Syngamy 3. Male and female parts of a flower mature simultaneously
A)
A-3, B-1, C-2
done
clear
B)
A-2, B-3, C-1
done
clear
C)
A-3, B-2, C-1
done
clear
D)
A-1, B-2, C-3
done
clear
View Answer play_arrow
Assertion [A] Plasmodium is an endoparasite. Reason [R] Female mosquito is an example of temporary parasite.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
A is false but R is true
done
clear
View Answer play_arrow
Identify the incorrect statement regarding oogenesis.
A)
It has long casting periods
done
clear
B)
It forms one functional and three non-functional cells
done
clear
C)
Its growth phase is very short
done
clear
D)
The division of primary oocyte is not equal
done
clear
View Answer play_arrow
Which one of the statement given below is not correct?
A)
The new name of dark reaction is carbon assimilation, in which \[C{{O}_{2}}\] gas reduces into carbohydrates glucose
done
clear
B)
Photolysis means splitting of water molecules to release oxygen in photosystem-l
done
clear
C)
A chlorophyll molecule has a tadpole like structure with phytol head and porphyria tail
done
clear
D)
ATP is energy rich compound where maximum energy is present in terminal pyrophosphate bonds
done
clear
View Answer play_arrow
Which of the following is not a vestigial organ in man?
A)
Nail
done
clear
B)
Muscles of ear pinna
done
clear
C)
Wisdom teeth
done
clear
D)
Tail vertebra
done
clear
View Answer play_arrow
Match the followings columns.
Column I Column II A. Calcium 1. Required for synthesis of mitotic spindle B. Chlorine 2. Required to active respiratory enzyme. C. Manganese 3. Essential for constitution of nucleic acid D. Phosphorous 4. Required for ionic balance
A)
A-1, B-3, C-2, D-4
done
clear
B)
A-1, B-4, C-2, D-3
done
clear
C)
A-4, B-2, C-3, D-1
done
clear
D)
A-2, B-3, C-4, D-1
done
clear
View Answer play_arrow
Consider the following statements. I. In non-endospermic seeds, food is stored in seed coat. II. Caryopsis is one seeded dry indehiscent fruit developed from a monocarpellary, unilocular and superior ovary. III. Hesperidium is a modification berry. Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Which of the following is a missing link?
A)
Periapts
done
clear
B)
Neopilina
done
clear
C)
Ichthyostega
done
clear
D)
Protopterus
done
clear
View Answer play_arrow
Which hormone of pancreas inhibits the release of pancreatic juice?
A)
Insulin
done
clear
B)
Glucagon
done
clear
C)
Somato statin
done
clear
D)
Pancreatic polypeptide
done
clear
View Answer play_arrow
Select the correct statement regarding active ion uptake, which differentiate it from secondary transport
A)
ions are active
done
clear
B)
ions move freely
done
clear
C)
energy is expended
done
clear
D)
ions move passively
done
clear
View Answer play_arrow
Which of the following combination of drugs has a dramatic rapid increase in sedative effect?
A)
Alcohol + Antihistamines
done
clear
B)
Alcohol + Benzodiazepines
done
clear
C)
Alcohol 4- Aspirin
done
clear
D)
Amphetamine + Insulin
done
clear
View Answer play_arrow
Which one of the statement given below is not correct?
A)
Kreb?s cycle is a common pathway of oxidative breakdown of carbohydrates, fatty acids and amino acids
done
clear
B)
The energy present in one gram carbohydrate in 4.4 Real or 18.4 kJ
done
clear
C)
When protein are used as respiratory substrate respiration is called protoplasmic respiration
done
clear
D)
The energy present in one gram of fat is 4.8 k cal or 20 kJ
done
clear
View Answer play_arrow
Genetically modified foods contain
A)
same number of genes as conventional crop
done
clear
B)
same number of genes as hybrid crop
done
clear
C)
one of two additional genes
done
clear
D)
hundreds of additional genes
done
clear
View Answer play_arrow
During photorespiration, the conversion of phosphoglycolate to glycolate takes place in which cell organelle.
A)
Mitochondria
done
clear
B)
Peroxisome
done
clear
C)
Chloroplast
done
clear
D)
Glyoxysome
done
clear
View Answer play_arrow
Which of the following statements is correct for Cro-magnon man?
A)
Advanced stage of man?s evolution than Homo erectus
done
clear
B)
Predecessor of Homo neanderthalensis
done
clear
C)
Direct ancestor of man
done
clear
D)
Lived during last ice age
done
clear
View Answer play_arrow
Steps of relaxation are I. Discharge of neuron II. Release of \[C{{a}^{+}}\] from troponin III. Generation of end plate potential IV. Cessation of interaction between actin and myosin.
A)
I, II, III and IV
done
clear
B)
II and IV
done
clear
C)
I, II and III
done
clear
D)
I, III and IV
done
clear
View Answer play_arrow
Clotting is a process via which flow of blood from the wound stops naturally. A step of clotting is given bellows identify the correct catalyst \[Fibrinogen\,\to \,\,Fibrin\]
A)
prothrombium
done
clear
B)
thrombium
done
clear
C)
thrombin
done
clear
D)
thromboplastin
done
clear
View Answer play_arrow
Which of the following is incorrect about eutrophication?
A)
Found in all ecosystems
done
clear
B)
Leads to organic loading
done
clear
C)
Bloom formation is eutrophic water
done
clear
D)
Enrichment with plant nutrients
done
clear
View Answer play_arrow
Two chromatin fibre per chromosome is present in
A)
\[{{G}_{1}}\]
done
clear
B)
\[{{G}_{2}}\]
done
clear
C)
S
done
clear
D)
Telophase
done
clear
View Answer play_arrow
Wobble pairing takes place
A)
under unusual condition between homologous chromosomes of a somatic cell causing somatic crossing over
done
clear
B)
in some cases between the third base of a codon and that of an anticodon
done
clear
C)
in those rare instances where unequal crossing over takes place for the lack of segment to segment pairing
done
clear
D)
in radiation included base deletion from one strand of a DNA molecule so that the complementary counter-part of the other strand exhibits m is pairing
done
clear
View Answer play_arrow
Which of the following statement is true for hetero chromatinisation. It takes place
A)
during fertilisation
done
clear
B)
early in the gestational development
done
clear
C)
immediately before birth
done
clear
D)
prior to the con set of puberty
done
clear
View Answer play_arrow
Genetically modified plants are superior to parent plants in many ways. Identify the correct effect of GMFs on human gene.
A)
it causes damage to human gene
done
clear
B)
It has no effect on human gene
done
clear
C)
it causes change in number of human gene
done
clear
D)
it modifies human gene
done
clear
View Answer play_arrow
Match tile following columns.
Column I Column II A. Apple 1. Perianth B. Walnuts 2. Juicy testa C. Pomegranate 3. Cotyledons D. Mulberry 4. Fleshy thalamus
A)
A-4, B-3, C-2, D-1
done
clear
B)
A-3, B-2, C-1, D-4
done
clear
C)
A-4, B-2, C-1, D-3
done
clear
D)
A-2, B-1, C-3, D-4
done
clear
View Answer play_arrow
Due to imbibition, the seed coat breaks because, kernels swell to higher degree because
A)
kernel is made up of cellulose
done
clear
B)
kernel is made up of proteins, lipid and starch
done
clear
C)
depends on nature of medium
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Ozone depletion of stratosphere shall results in lot of side effects to atmosphere one of them is that it
A)
causes forest fires
done
clear
B)
increased incidence of skin cancer.
done
clear
C)
causes blood cancer
done
clear
D)
None of these
done
clear
View Answer play_arrow
Extinction of passenger pigeon (Ictopistus migratorius) is a result of one of the following cause.
A)
absence of food
done
clear
B)
presence of predators
done
clear
C)
over exploitation by humans
done
clear
D)
bird flu
done
clear
View Answer play_arrow
Stellosis sea cow (Hydrodamalis gigas) was a large herbivorous marine mammals. It was the largest number of the order-Sirenia. It was discovered in
A)
Africa
done
clear
B)
Russia
done
clear
C)
Mauritius
done
clear
D)
Australia
done
clear
View Answer play_arrow
Which one of the following human genes has the longest stretch of DNA\[\left( \tilde{\ } 24 Mb \right)\]?
A)
Globin gene
done
clear
B)
Histone gene
done
clear
C)
Dystrophin gene
done
clear
D)
RNA gene
done
clear
View Answer play_arrow
Which of the following is true about Lamarckism? I. It was proposed by Jean Baptiste de Lamarck. II. It is based on use and disuse of organs, III. Principle of natural selection. IV. Inheritance of acquired characters.
A)
I, II and III
done
clear
B)
IV, III and II
done
clear
C)
I, IV and II
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
Mental retardation in man associated with sex chromosomal abnormality is usually due to
A)
moderate increase in Y complement
done
clear
B)
large increase in Y complement
done
clear
C)
reduction in X complement
done
clear
D)
increase in X complement
done
clear
View Answer play_arrow
Identify the correct statement regarding pBR322. It is a
A)
bacteriophage
done
clear
B)
yeast
done
clear
C)
plasmid
done
clear
D)
parasite
done
clear
View Answer play_arrow
The first step in dark reaction of photosynthesis is
A)
formation of ATP
done
clear
B)
excitation of chlorophyll molecule
done
clear
C)
photolysis of water
done
clear
D)
attachment of carbon dioxide to a pentose sugar
done
clear
View Answer play_arrow
Which of the following is helpful in studying sex-linked characters?
A)
Back cross
done
clear
B)
Test cross
done
clear
C)
Reciprocal cross
done
clear
D)
Trihybrid cross
done
clear
View Answer play_arrow
Which scientist pair determined the given structure of DNA?
A)
Brenner and Nirenberg
done
clear
B)
Jacob and Monod
done
clear
C)
Watson and Crick
done
clear
D)
Signer and Nicolson
done
clear
View Answer play_arrow
Which one of the following statements correctly describes allosteric enzymes?
A)
Not usually controlled by feedback inhibition
done
clear
B)
Regulatory site may be catalytic site
done
clear
C)
Michaelis-Menten kinetics describe their activity
done
clear
D)
Effectors may enhance or inhibit substrate binding
done
clear
View Answer play_arrow
An ideal vector must have which among the following. I. An origin of replication. II. Single cloning site. III. Have a gene encoding transcription. IV. Have a gene encoding resistance to antibiotic. Choose the correct combination.
A)
I and IV
done
clear
B)
III and IV
done
clear
C)
II and IV
done
clear
D)
I and II
done
clear
View Answer play_arrow
Haemophilic man marries a normal woman. Their offsprings will be all
A)
haemophilia
done
clear
B)
boys haemophilia
done
clear
C)
girls
done
clear
D)
normal
done
clear
View Answer play_arrow
Assertion [A] In human eye, cornea absorbs UV-B radiation and a high dose of UV-B causes inflammation of cornea. Reason [R] Such exposure may permanently damage the cornea.
A)
Both A and R are true and R is the correct explanation of A.
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
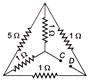
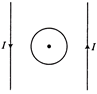
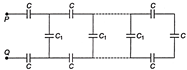
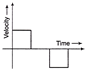
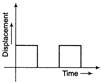

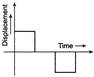
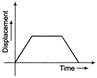
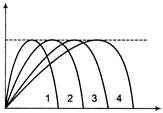
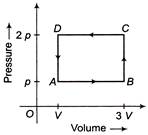
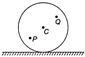
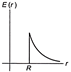
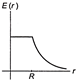
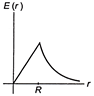
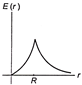
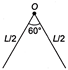
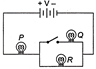
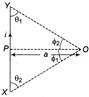
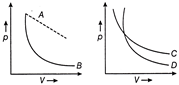
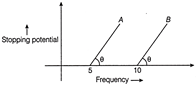
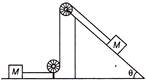
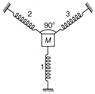
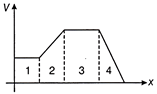
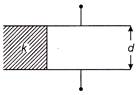
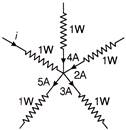
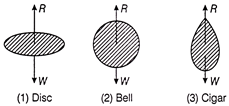
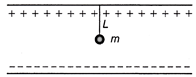
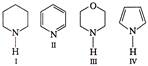
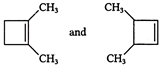
 reacts with \[{{B}_{2}}{{H}_{6}}\] in presence of \[{{\operatorname{H}}_{2}}{{O}_{2}}\] produces
reacts with \[{{B}_{2}}{{H}_{6}}\] in presence of \[{{\operatorname{H}}_{2}}{{O}_{2}}\] produces