Calculate the negative charge contain in a cup of water:
A)
\[4.2\times {{10}^{4}}C\]
done
clear
B)
\[2.4\times {{10}^{6}}C\]
done
clear
C)
\[1.34\times {{10}^{7}}C\]
done
clear
D)
\[1.96\times {{10}^{7}}C\]
done
clear
View Answer play_arrow
An electron falls through a distance of 1.5 cm in a uniform electric field of\[2\times 104\text{ }N/C\]. Time required to fall through this distance (in sec):
A)
\[0.2\times {{10}^{9}}s\]
done
clear
B)
\[2.9\times {{10}^{-\,9}}s\]
done
clear
C)
\[1.4\times {{10}^{-\,9}}s\]
done
clear
D)
\[1.96\times {{10}^{-\,9}}s\]
done
clear
View Answer play_arrow
In a potentiometer arrangement, a cell of emf 1.25V gives a balance point at 35 cm length of the wire. If the cell is replaced by another cell and balance point shifts to 63 cm, what is the emf of second cell?
A)
2.25V
done
clear
B)
3.13V
done
clear
C)
2.9V
done
clear
D)
1.3V
done
clear
View Answer play_arrow
Storage battery of a car has an emf of 12v. If the internal resistance of the battery is \[0.4\Omega \], what is the maximum current drawn from the battery:
A)
20 A
done
clear
B)
35A
done
clear
C)
25 A
done
clear
D)
30A
done
clear
View Answer play_arrow
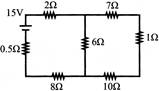
The current from the battery in circuit diagram shown is:
A)
1A
done
clear
B)
2A
done
clear
C)
1.5A
done
clear
D)
3A
done
clear
View Answer play_arrow
Two wires of same metal have the same length but their cross sections are in the ratio 3 : 1. They are joined in series. The resistance of the thicker wire is\[10\Omega \]. The total resistance of the combination will be:
A)
\[\frac{5}{2}\Omega \]
done
clear
B)
\[40\Omega \]
done
clear
C)
\[\frac{40}{3}\Omega \]
done
clear
D)
\[100\Omega \]
done
clear
View Answer play_arrow
What is the magnitude of magnetic for CE per unit length on a wire carrying a current of 8A and making an angle of \[30{}^\circ \] with the direction of a uniform magnetic field of 0.15T:
A)
0.2 N/m
done
clear
B)
0.6 N/m
done
clear
C)
0.3 N/m
done
clear
D)
0.8 N/m
done
clear
View Answer play_arrow
A 3 cm wire carrying current of 10A is placed inside a solenoid perpendicular to its axis. The \[\vec{B}\] inside the solenoid is given to be 0.27T. Magnitude of magnetic force on the wire?
A)
\[4.4\times {{10}^{-9}}N\]
done
clear
B)
\[5.6\times {{10}^{-9}}N\]
done
clear
C)
\[7.2\times {{10}^{-2}}N\]
done
clear
D)
\[8.1\times {{10}^{-2}}N\]
done
clear
View Answer play_arrow
When \[{{10}^{14}}\text{ }{{e}^{-}}\] are removed from a neutral metal sphere, the charge on the sphere becomes:
A)
\[16\mu C\]
done
clear
B)
\[-\,16\mu C\]
done
clear
C)
\[32\mu C\]
done
clear
D)
\[-\,32\mu C\]
done
clear
View Answer play_arrow
Equal charges q are placed at the four corners A, B, C and D of a square of length The magnitude of the force on the charge at B will be:
A)
\[\frac{3{{q}^{2}}}{4\pi {{\varepsilon }_{0}}{{a}^{2}}}\]
done
clear
B)
\[\frac{{{q}^{2}}}{4\pi {{\varepsilon }_{0}}{{a}^{2}}}\]
done
clear
C)
\[\left[ \frac{2\sqrt{2+1}}{2} \right]\frac{{{q}^{2}}}{4\pi {{\varepsilon }_{0}}{{a}^{2}}}\]
done
clear
D)
\[\left[ 2+\frac{1}{\sqrt{2}} \right]\frac{{{q}^{2}}}{4\pi {{\varepsilon }_{0}}{{a}^{2}}}\]
done
clear
View Answer play_arrow
The work done in turning a magnetic of magnetic moment 'm' by an angle of \[90{}^\circ \] from the meridian is 'n' times the corresponding work done to turn it through an angle of \[60{}^\circ \], where 'n' is given by:
A)
\[\frac{1}{4}\]
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[2\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
A magnet of magnetic moment \[50\hat{i}A-{{m}^{2}}\]is placed along the x - axis in a magnetic field\[\vec{B}=(0.5\hat{i}+3\hat{j})T\]. The torque acting on the magnet is:
A)
\[175\hat{k}~\]
done
clear
B)
\[75\hat{k}\]
done
clear
C)
\[150\hat{k}\]
done
clear
D)
\[25\sqrt{37}\hat{k}\]
done
clear
View Answer play_arrow
A coil having 500 square loops each of side 10 cm is placed normal to a magnetic flux which increases at the rate of 1 T/sethe emf r induced is:
A)
\[0.1\,V\]
done
clear
B)
\[0.5\,V\]
done
clear
C)
\[5\,V\]
done
clear
D)
\[1\,V\]
done
clear
View Answer play_arrow
An L - R circuit has a cell of emf E, which is switched on at time t = 0. The current in the circuit after a long time will be:
A)
\[Zero\]
done
clear
B)
\[\frac{E}{R}\]
done
clear
C)
\[\frac{E}{L}\]
done
clear
D)
\[\frac{E}{\sqrt{{{L}^{2}}+{{R}^{2}}}}\]
done
clear
View Answer play_arrow
The self-inductance of solenoid of length L, area of cross section A and having N turns is
A)
\[\frac{{{\mu }_{0}}{{N}^{2}}A}{L}\]
done
clear
B)
\[\frac{{{\mu }_{0}}NA}{L}\]
done
clear
C)
\[{{\mu }_{0}}{{N}^{2}}LA\]
done
clear
D)
\[{{\mu }_{0}}NLA\]
done
clear
View Answer play_arrow
An ac supply gives 30 V rms which passes through a \[10\Omega \] resistance. The power dissipated in its is:
A)
\[90\sqrt{2}W\]
done
clear
B)
\[90W\]
done
clear
C)
\[45\,\sqrt{2}W\]
done
clear
D)
\[45\,W\]
done
clear
View Answer play_arrow
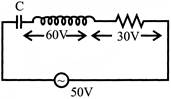
In the circuit shown in figure potential difference across capacitor will be:
A)
100V
done
clear
B)
20V
done
clear
C)
40V
done
clear
D)
60V
done
clear
View Answer play_arrow
A object of size 7.5 cm is placed in front of convex mirror of radius of curvature 25cm at a distance of 40 cm. The size of image will be
A)
2.3 cm
done
clear
B)
1.78 cm
done
clear
C)
0.8 cm
done
clear
D)
11 cm
done
clear
View Answer play_arrow
If the refractive index of a material of equilateral prism is \[\sqrt{3}\],then angle of minimum deviation of the prism is:
A)
\[30{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[75{}^\circ \]
done
clear
View Answer play_arrow
Which of following is conserved when light waves interfere?
A)
Intensity
done
clear
B)
Energy
done
clear
C)
Amplitude
done
clear
D)
Momentum
done
clear
View Answer play_arrow
It the de-broglie wavelength for a proton and for \[\alpha \]- particles are equal then the ratio of their velocities will be in the ratio:
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
4 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
The de Broglie wavelength of an electron having 80 eV of energy is nearly:
A)
\[140\overset{o}{\mathop{A}}\,\]
done
clear
B)
\[0.14\overset{o}{\mathop{A~}}\,\]
done
clear
C)
\[14\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[1.4\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
A radioactive isotope X with half-life \[1.5\times {{10}^{9}}\,\] yr decays into a stable nucleus Y. A rock sample contains both elements X and Y in the ratio 1:15. The age of the rock is:
A)
\[3\times {{10}^{9}}\,\,yr\]
done
clear
B)
\[6\times {{10}^{9}}\,\,yr\]
done
clear
C)
\[4.5\times {{10}^{9}}\,\,yr\]
done
clear
D)
\[9\times {{10}^{9}}\,\,yr\]
done
clear
View Answer play_arrow
A body is projected at an angle of \[30{}^\circ \] with the horizontal with momentum P. At its highest point the magnitude of the momentum is:
A)
\[\frac{\sqrt{3}P}{2}\]
done
clear
B)
\[\frac{2P}{\sqrt{3}}\]
done
clear
C)
\[P\]
done
clear
D)
\[\frac{P}{3}\]
done
clear
View Answer play_arrow
A body projected with velocity u at projection angle \[\theta \] has horizontal Range R. For the same velocity and projection angle its range on the moon surface will be:
A)
\[36R\]
done
clear
B)
\[\frac{R}{36}\]
done
clear
C)
\[\frac{R}{16}\]
done
clear
D)
\[6R\]
done
clear
View Answer play_arrow
Two stones having different masses \[{{m}_{1}}\] and \[{{m}_{2}}\] are projected at an angle \[\alpha \] and \[(90-\alpha )\]with same speed from same point. The ratio of their maximum heights is:
A)
\[1:1\]
done
clear
B)
\[1:tan\alpha \]
done
clear
C)
\[tan\alpha :1\]
done
clear
D)
\[ta{{n}^{2}}\alpha :1\]
done
clear
View Answer play_arrow
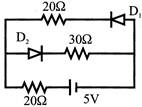
The Boolean equation for the circuit given in figure is:
A)
\[Y=\underline{\underline{A+\overline{B}}}\]
done
clear
B)
\[Y=\overline{\underline{A}+B}\]
done
clear
C)
\[Y=A+B\]
done
clear
D)
\[Y=A+B\]
done
clear
View Answer play_arrow
The current in the circuit will be:
A)
\[\frac{5}{40}A\]
done
clear
B)
\[\frac{5}{50}A\]
done
clear
C)
\[\frac{5}{20}A\]
done
clear
D)
\[\frac{5}{10}A\]
done
clear
View Answer play_arrow
Displacement of particles in a string stretched in the x-direction is represented by y. Among the following expressions for y, those describing wave motion are:
A)
\[Cos\,kx\,Sin\,\omega t\]
done
clear
B)
\[Cos\,(kx\,+\omega t)\]
done
clear
C)
\[{{k}^{2}}{{x}^{2}}-{{\omega }^{2}}{{t}^{2}}\]
done
clear
D)
\[Cos\,({{k}^{2}}{{x}^{2}}-{{\omega }^{2}}{{t}^{2}})\]
done
clear
View Answer play_arrow
A transverse wave is described by the equation\[Y={{Y}_{0}}\,\sin \,2\pi \left( ft\frac{x}{\lambda } \right)\] . The maximum particle velocity is equal to four times the wave velocity if:
A)
\[\lambda =\frac{\pi {{Y}_{0}}}{4}\]
done
clear
B)
\[\lambda =\pi {{Y}_{0}}\]
done
clear
C)
\[\lambda =\frac{\pi {{Y}_{0}}}{2}\]
done
clear
D)
\[\lambda =2\pi {{Y}_{0}}\]
done
clear
View Answer play_arrow
A tuning fork of frequency 80 Hz produced 10 beats per sec when sounded with a vibrating sonometer string. What must have been the frequency of string if slight increase in tension produces fever beats per sec than before:
A)
460 Hz
done
clear
B)
470 Hz
done
clear
C)
480 Hz
done
clear
D)
490 Hz
done
clear
View Answer play_arrow
A stone released with zero velocity from the top of the tower reaches the ground in 4 seThe height of the tower is about:
A)
40m
done
clear
B)
20m
done
clear
C)
160m
done
clear
D)
80m
done
clear
View Answer play_arrow
A ray of light passes through an equilateral prism\[(\mu =1.5)\]. The angle of deviation is
A)
\[45{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[30{}^\circ \]
done
clear
D)
\[37{}^\circ \]
done
clear
View Answer play_arrow
A block of wood floats in water in a vessel 1/10 of its volume exposeSufficient oil of density \[0.8\text{ }g/c{{m}^{3}}\]is poured into the vessel till the block is completely submergeWhat percentage of its volume in the water now:
A)
80%
done
clear
B)
60%
done
clear
C)
75%
done
clear
D)
50%
done
clear
View Answer play_arrow
A ray of light incident normally on one face of a prism \[(\mu =2)\] undergoes grazing emergence. The angle of prism is:
A)
\[30{}^\circ \]
done
clear
B)
\[50{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[60{}^\circ \]
done
clear
View Answer play_arrow
1 kg of ice at \[0{}^\circ C\]is mixed with 1 kg of water at\[80{}^\circ C\]. The final temperature of the mixture is \[[given\,{{S}_{W}}4200J/kg\,K,{{L}_{ice}}336KJ/kg]\]:
A)
\[50{}^\circ \]
done
clear
B)
\[0{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[40{}^\circ \]
done
clear
View Answer play_arrow
64 small drops of mercury, each of radius r and charge q coalesce to form a big drop. The ratio of the surface density of charge of each small drop with that of big drop is;
A)
64 : 1
done
clear
B)
1 : 64
done
clear
C)
1 : 4
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
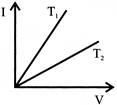
The current - voltage graph for a given metallic conductor at two different temperature \[{{T}_{1}}\] and\[{{T}_{2}}\] are as shown in the figure. Choose the correct inference:
A)
\[{{T}_{2}}>{{T}_{1}}\]
done
clear
B)
\[{{T}_{2}}<{{T}_{1}}\]
done
clear
C)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
D)
\[None\]
done
clear
View Answer play_arrow
A body is projected vertically upwards from the surface of a planet of radius R with a velocity equal to half the escape velocity for that planet. The maximum height attained by the body is:
A)
\[\frac{R}{2}\]
done
clear
B)
\[\frac{R}{3}\]
done
clear
C)
\[\frac{R}{4}\]
done
clear
D)
\[\frac{R}{5}\]
done
clear
View Answer play_arrow
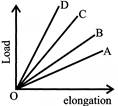
Load versus elongation graph for four wires of the same materials is shown in the figure. The thinnest wire is represented by the line:
A)
OD
done
clear
B)
OC
done
clear
C)
OB
done
clear
D)
OA
done
clear
View Answer play_arrow
The material used for permanent magnet has
A)
Low retentivity, high coercivity
done
clear
B)
High retentivity, low coercivity
done
clear
C)
High retentivity, high coercivity
done
clear
D)
Low retentivity, low coercivity
done
clear
View Answer play_arrow
If \[{{r}_{1}}\] and \[{{r}_{2}}\] are the radii of the atomic nuclei of mass numbers 64 and 125 respectively then the ratio \[{{r}_{1}}/{{r}_{2}}\] is:
A)
\[\frac{64}{125}\]
done
clear
B)
\[\frac{8}{5}\]
done
clear
C)
\[\frac{5}{4}\]
done
clear
D)
\[\frac{4}{5}\]
done
clear
View Answer play_arrow
A stone is fastened to one end of a string and is whirled in a vertical circle of radius R. Find the minimum speed the stone can have at the highest point of the circle:
A)
\[\sqrt{gR}\]
done
clear
B)
\[\sqrt{\frac{2R}{g}}\]
done
clear
C)
\[\sqrt{\frac{R}{g}}\]
done
clear
D)
\[\sqrt{2Rg}\]
done
clear
View Answer play_arrow
A cylinder of fixed capacity (of 44.8 Ltr) contains 2 moles of He gas at STP. What is the amount of heat needed to raise the gas temperature by \[20{}^\circ C\text{ }\!\![\!\!\text{ }R=8.31\text{ }J\text{ }mo{{l}^{-1}}\text{ }{{K}^{-1}}]\]:
A)
966 J
done
clear
B)
498 J
done
clear
C)
831 J
done
clear
D)
374 J
done
clear
View Answer play_arrow
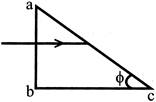
An incident ray of light is incident perpendicular to the face ab of a prism \[(\mu =1.5)\] as shown below. Find the largest value of the angle \[\phi \] so that the incident light is totally reflected back at the face ac of the prism:
A)
\[Co{{s}^{-1}}\left( \frac{1}{2} \right)\]
done
clear
B)
\[Co{{s}^{-1}}\left( \frac{3}{2} \right)\]
done
clear
C)
\[Co{{s}^{-1}}\left( \frac{2}{3} \right)\]
done
clear
D)
\[Co{{s}^{-1}}\left( \frac{1}{3} \right)\]
done
clear
View Answer play_arrow
19.7 kg of gold was recovered from a smuggler. How many atoms of gold were recovered ?(Au = 197)
A)
\[100\]
done
clear
B)
\[6.023\times 1023\]
done
clear
C)
\[6.023\times 1024\]
done
clear
D)
\[6.023\times 1025\]
done
clear
View Answer play_arrow
The compound in which cation is isoelectronic with anion is:
A)
\[NaCl\]
done
clear
B)
\[CsF\]
done
clear
C)
\[Nal\]
done
clear
D)
\[{{K}_{2}}S\]
done
clear
View Answer play_arrow
The uncertainty in momentum of an electron is \[1\times {{10}^{-5}}kg\,m/s\] The uncertainty in its position will be \[(h=6.62\times {{10}^{-34}}kg{{m}^{2}}/s)\]:
A)
\[1.05\times {{10}^{-28}}m\]
done
clear
B)
\[1.05\times {{10}^{-26}}m\]
done
clear
C)
\[5.27\times {{10}^{-30}}m\]
done
clear
D)
\[5.25\times {{10}^{-28}}m\]
done
clear
View Answer play_arrow
Which of the properties remain unchanged on descending a group in the periodic table?
A)
Atomic size
done
clear
B)
Density
done
clear
C)
Valence electrons
done
clear
D)
Metallic character
done
clear
View Answer play_arrow
According to Fajan's rule, the extent of polarisation in an ionic compound is favoured by:
A)
Large cation and small anion
done
clear
B)
Large cation and anion
done
clear
C)
Small cation and small anion
done
clear
D)
Small cation and large anion
done
clear
View Answer play_arrow
Two ice cubes are pressed over each other until they unite to form one black. Which one of the following forcer dominates for holding them together?
A)
Dipole - dipole interaction
done
clear
B)
Vander waal's forces
done
clear
C)
Hydrogen bond
done
clear
D)
Covalent attraction
done
clear
View Answer play_arrow
The compressibility of a gas is less then unity at STP. Therefore:
A)
\[{{V}_{m}}>224\,litres\]
done
clear
B)
\[{{V}_{m}}<224\,litres\]
done
clear
C)
\[{{V}_{m}}=224\,litres\]
done
clear
D)
\[{{V}_{m}}=448\,litres\]
done
clear
View Answer play_arrow
Equal volumes of monoatomic and diatomic gases at same initial temperature and pressure are mixed The ratio of specified heats of the mixture \[({{C}_{p}}/{{C}_{r}})\] will be:
A)
1
done
clear
B)
2
done
clear
C)
1.67
done
clear
D)
1.5
done
clear
View Answer play_arrow
If equilibrium constants of reaction \[{{N}_{2}}{{O}_{2}}\,2NO\,\] is \[{{K}_{1}}\], and \[\frac{1}{2}{{N}_{2}}+\frac{1}{2}{{O}^{2}}NO\] is \[{{K}_{2}}\]then:
A)
\[{{K}_{1}}={{K}_{2}}\]
done
clear
B)
\[{{K}_{1}}=2{{K}_{2}}\]
done
clear
C)
\[{{K}_{2}}=\sqrt{{{K}_{1}}}\]
done
clear
D)
\[{{K}_{1}}=\frac{1}{2}{{K}_{2}}\]
done
clear
View Answer play_arrow
When an acid or alkali is mixed with buffer solution, then pH of buffer solution:
A)
Increases
done
clear
B)
Decreases
done
clear
C)
Changes slightly
done
clear
D)
Remains same
done
clear
View Answer play_arrow
In the following reaction, \[C{{r}_{2}}O_{7}^{2-}+14{{H}^{+}}+6{{l}^{-}}\xrightarrow{{}}\,2C{{r}^{3}}+3{{H}_{2}}O+3{{l}_{2}}\] Which element is reduced?
A)
Cr
done
clear
B)
H
done
clear
C)
0
done
clear
D)
I
done
clear
View Answer play_arrow
Ortho and para hydrogen differ in:
A)
Proton spin
done
clear
B)
Electron spin
done
clear
C)
Nuclear change
done
clear
D)
Nuclear reaction
done
clear
View Answer play_arrow
When burning magnesium ribbon is introduced into a jar of oxygen, it produces:
A)
\[Mg\]
done
clear
B)
\[MgO\]
done
clear
C)
\[Mg{{O}_{2}}\]
done
clear
D)
\[M{{g}_{2}}{{O}_{2}}\]
done
clear
View Answer play_arrow
In diborane, the two H-B-H angles are nearly?
A)
\[60{}^\circ ,120{}^\circ \]
done
clear
B)
\[95{}^\circ ,120{}^\circ \]
done
clear
C)
\[95{}^\circ ,150{}^\circ \]
done
clear
D)
\[120{}^\circ ,180{}^\circ \]
done
clear
View Answer play_arrow
In kjeldahl's mehod, \[CuS{{O}_{4}}\] acts as a/an:
A)
Oxidising agent
done
clear
B)
Reducing agent
done
clear
C)
Hydrolysing agent
done
clear
D)
Catalytic agent
done
clear
View Answer play_arrow
Which of the following species is paramagnetic in nature:
A)
Free radical
done
clear
B)
Carboniumtion
done
clear
C)
Carbanion
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which of the following will have least hindered rotation about carbon-carbon bond?
A)
Ethane
done
clear
B)
Ethylene
done
clear
C)
Acetyline
done
clear
D)
Hexachloraethane
done
clear
View Answer play_arrow
Which of the following has a bond formed by overlap of \[sp-s{{p}^{3}}\] hybrid orbitals?
A)
\[C{{H}_{3}}-C=C-H\]
done
clear
B)
\[C{{H}_{3}}-CH=CH-C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
done
clear
D)
\[HC=CH\]
done
clear
View Answer play_arrow
The product formed when acetylene is passed through red hot iron tube at 873K is:
A)
Benzene
done
clear
B)
Cyclohexane
done
clear
C)
Neoprene
done
clear
D)
Ethane
done
clear
View Answer play_arrow
Benzene vapour when passed over catalyst at 773K gives:
A)
Glyoxal
done
clear
B)
Oxalic acid
done
clear
C)
Maleic anhydride
done
clear
D)
Fumaric acid
done
clear
View Answer play_arrow
The gas released in Bhopal tragedy was:
A)
\[Phosgine\]
done
clear
B)
\[MIC\]
done
clear
C)
\[COC{{l}_{2}}\]
done
clear
D)
\[All\text{ }of\text{ }these\]
done
clear
View Answer play_arrow
If 'Z' is the number of atoms in the unit cell that represents the closest packing sequence--- ABC ABC ----, the number of tetrahedral voids in the unit cell is equal to:
A)
\[2Z\]
done
clear
B)
\[Z\]
done
clear
C)
\[Z/2~\]
done
clear
D)
\[Z/4\]
done
clear
View Answer play_arrow
The osmatic pressure in atmospheres of 10% solution of cane sugar at \[69{}^\circ C\]is:
A)
724
done
clear
B)
8.24
done
clear
C)
8.21
done
clear
D)
7.21
done
clear
View Answer play_arrow
If the \[E{}^\circ \] cell or a given reaction has a negative value then which of the following gives the correct relationships for the value of \[\Delta G{}^\circ \]and\[{{K}_{eq}}\]?
A)
\[\Delta G{}^\circ >0;\,\,{{K}_{eq}}<1\]
done
clear
B)
\[\Delta G{}^\circ >0;\,\,{{K}_{eq}}>1\]
done
clear
C)
\[\Delta G{}^\circ <0;\,\,{{K}_{eq}}>1\]
done
clear
D)
\[\Delta G{}^\circ <0;\,\,{{K}_{eq}}<1\]
done
clear
View Answer play_arrow
A reaction is 50% completed in 2 hours and 70% completed in 4 hours. The order of reaction is:
A)
0
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
If gold number of A, B, C and D are 0.005, 0.05,0.5, and 5 respectively, then which of the following will have the highest protective power:
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Thomas slag is:
A)
\[CaSi{{O}_{3}}\]
done
clear
B)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\]
done
clear
C)
\[MnSi{{O}_{3}}\]
done
clear
D)
\[CaC{{O}_{3}}\]
done
clear
View Answer play_arrow
Which of the following hydrides has the lowest boiling point:
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}S~\]
done
clear
C)
\[{{H}_{2}}Se\]
done
clear
D)
\[{{H}_{2}}Te\]
done
clear
View Answer play_arrow
The least stable oxide at room temperature is
A)
\[A{{g}_{2}}O\]
done
clear
B)
\[CuO\]
done
clear
C)
\[S{{b}_{2}}{{O}_{3}}\]
done
clear
D)
\[ZnO\]
done
clear
View Answer play_arrow
The EAN of nickel of \[Ni{{(CO)}_{4}}\] is:
A)
38
done
clear
B)
36
done
clear
C)
28
done
clear
D)
54
done
clear
View Answer play_arrow
The most stable ion is:
A)
\[{{[Fe{{({{C}_{2}}{{O}_{4}})}_{3}}]}^{2-}}\]
done
clear
B)
\[{{[Fe{{(Cl)}_{6}}]}^{3-}}\]
done
clear
C)
\[{{[Fe{{(CN)}_{6}}]}^{3-}}\]
done
clear
D)
\[{{[Fe{{({{H}_{2}}O)}_{6}}]}^{3+}}\]
done
clear
View Answer play_arrow
Best method of preparing alkyl chloride is:
A)
\[ROH+PC{{l}_{3}}\xrightarrow{{}}\]
done
clear
B)
\[ROH+PC{{l}_{5}}\xrightarrow{{}}\]
done
clear
C)
\[ROH+SOC{{l}_{2}}\xrightarrow{{}}\]
done
clear
D)
\[ROH+SOC{{l}_{2}}\xrightarrow{anhy.\,Zncl}\]
done
clear
View Answer play_arrow
Phenol can be distinguished from aliphatic alcohol with:
A)
Tollen's reagent
done
clear
B)
Schiff?s base
done
clear
C)
\[FeC{{l}_{3}}\]
done
clear
D)
\[HCl\]
done
clear
View Answer play_arrow
The order of hydrolysis for the following is:
I. \[RCOCl~\] II. \[RCOOR\] III. \[RCON{{H}_{2}}\] IV \[{{(RCO)}_{2}}O\]
A)
IV > III > II > I
done
clear
B)
I > II > III > IV
done
clear
C)
I > III > II > IV
done
clear
D)
I > IV > II > III
done
clear
View Answer play_arrow
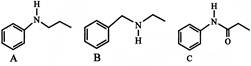
Which one of the following compound is most basic?
A)
A
done
clear
B)
C
done
clear
C)
B
done
clear
D)
All are equally basic
done
clear
View Answer play_arrow
Sodium dodecyl benzene sulphonate is used as a:
A)
Pesticide
done
clear
B)
Soap
done
clear
C)
Fertizer
done
clear
D)
Detergent
done
clear
View Answer play_arrow
Dacron is an example of:
A)
Polyamide
done
clear
B)
Palypropylene
done
clear
C)
Polyurethane
done
clear
D)
Polyester
done
clear
View Answer play_arrow
Which of the following is hypnotic drug?
A)
Luminal
done
clear
B)
Salal
done
clear
C)
Piperazine
done
clear
D)
Novalgin
done
clear
View Answer play_arrow
Which of the following is not an alkaloid?
A)
Reserpine
done
clear
B)
Morphine
done
clear
C)
Cluninine
done
clear
D)
Phenylbutazone
done
clear
View Answer play_arrow
Nitrate is confirmed by ring test. The brown colour of the ring is due to the formation of:
A)
Ferrous nitrite
done
clear
B)
\[FeS{{O}_{4}}NO\]
done
clear
C)
\[FeS{{O}_{4}}N{{O}_{2}}\]
done
clear
D)
Ferrous nitrate
done
clear
View Answer play_arrow
Among the following complexes the one which shows zero crystal field stabilization energy (CFSE) is:
A)
\[{{[Mn{{({{H}_{2}}O)}_{6}}]}^{3+}}\]
done
clear
B)
\[{{[Fe{{({{H}_{2}}O)}_{6}}]}^{3+}}\]
done
clear
C)
\[{{[Co{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
done
clear
D)
\[{{[Co{{({{H}_{2}}O)}_{6}}]}^{3+}}\]
done
clear
View Answer play_arrow
The transition elements have a general electronic configuration:
A)
\[n{{s}^{2}}n{{p}^{6}}n{{d}^{1-10}}\]
done
clear
B)
\[(n-1){{d}^{1-10}},n{{s}^{0-2}},n{{p}^{0-6}}\]
done
clear
C)
\[(n-1){{d}^{1-10}},n{{s}^{1-2}}\]
done
clear
D)
\[nd\,n{{s}^{-2}}\]
done
clear
View Answer play_arrow
Standard electrode potential of three metals x, y and z are -1, 2V, +0.5V and -3.0V respectively. The reducing power of these metals will be:
A)
y > z > x
done
clear
B)
y > x > z
done
clear
C)
z > x > y
done
clear
D)
x > y > z
done
clear
View Answer play_arrow
Tautomerism is exhibited by:
A)
\[{{R}_{3}}CN{{O}_{2}}\]
done
clear
B)
\[RC{{H}_{2}}N{{O}_{2}}\]
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}CHO\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}}NH\]
done
clear
View Answer play_arrow
The correct order of increasing thermal stability of \[{{K}_{2}}C{{O}_{3}},MgC{{O}_{3}},CaC{{O}_{3}}\] and\[BeC{{O}_{3}}\]is:
A)
\[BeC{{O}_{3}}<MgC{{O}_{3}}<CaC{{O}_{3}}<{{K}_{2}}S{{O}_{3}}\]
done
clear
B)
\[MgC{{O}_{3}}<BeC{{O}_{3}}<CaC{{O}_{3}}<{{K}_{2}}S{{O}_{3}}\]
done
clear
C)
\[{{K}_{2}}S{{O}_{3}}<MgC{{O}_{3}}<CaC{{O}_{3}}<BeC{{O}_{3}}\]
done
clear
D)
\[BeC{{O}_{3}}<MgC{{O}_{3}}<{{K}_{2}}S{{O}_{3}}<CaC{{O}_{3}}\]
done
clear
View Answer play_arrow
Two words comprising the binomial nomenclature are:
A)
Family & Genus
done
clear
B)
Order & Family
done
clear
C)
Genus & Species
done
clear
D)
Species & Variety
done
clear
View Answer play_arrow
Ex-situ conservation and inbreeding between animals whose aim is?
A)
Zoo's
done
clear
B)
Museum
done
clear
C)
Botanical garden
done
clear
D)
Zoological park
done
clear
View Answer play_arrow
Where the British museum of Royal Botanical garden is located?
A)
Kew
done
clear
B)
Pairs
done
clear
C)
Kolkata
done
clear
D)
Dehradon
done
clear
View Answer play_arrow
Who promote the eco-tourism?
A)
Herbarium
done
clear
B)
Botanical park
done
clear
C)
Zoological park
done
clear
D)
Museum
done
clear
View Answer play_arrow
Viroids were discovered by
A)
Diener
done
clear
B)
Pasteur
done
clear
C)
Lyengar
done
clear
D)
Woese
done
clear
View Answer play_arrow
Plant of this group possess naked seed:
A)
Angiosperms
done
clear
B)
Gymnosperms
done
clear
C)
Pteridophytes
done
clear
D)
Bryophytes
done
clear
View Answer play_arrow
Which sentence is true for Bryophytes?
A)
They are autotropic
done
clear
B)
Fertilization takes place in the presence of water
done
clear
C)
Vascular tissues are absent
done
clear
D)
All of the three
done
clear
View Answer play_arrow
The body bears ____external rods of ciliated comb plates in Pleurobrachia:
A)
Eight
done
clear
B)
Sixteen
done
clear
C)
Ten
done
clear
D)
Four
done
clear
View Answer play_arrow
Which is correct w.r.t. earth worm:
A)
Segments
done
clear
B)
Nephridia
done
clear
C)
Parapodia
done
clear
D)
All of given
done
clear
View Answer play_arrow
Which type of thickening is found in protoxylem:
A)
Reticulated
done
clear
B)
Pitted
done
clear
C)
Spiral
done
clear
D)
Sclariform
done
clear
View Answer play_arrow
Safaranin stains by:
A)
Thick walled cells
done
clear
B)
Saberized cells
done
clear
C)
Lignified cells
done
clear
D)
Living cells
done
clear
View Answer play_arrow
In which tissue is called a living mechanical tissue:
A)
Parenchyma
done
clear
B)
Chlorenchyma
done
clear
C)
Arrenchyma
done
clear
D)
Collenchyma
done
clear
View Answer play_arrow
Which cells regulate the function of sieve tube?
A)
Guard cells
done
clear
B)
Bulliform cells
done
clear
C)
Companion cells
done
clear
D)
Passage cells
done
clear
View Answer play_arrow
In roots the lateral root originated from:
A)
Cortex
done
clear
B)
Pericycle
done
clear
C)
Endodermis
done
clear
D)
Epidermis
done
clear
View Answer play_arrow
Name the connective tissue present in larynx
A)
White fibrous cartilage
done
clear
B)
Hyaline cartilage
done
clear
C)
Areolar tissue
done
clear
D)
Yellow elastic cartilage
done
clear
View Answer play_arrow
Epiphytic roots are found in:
A)
Indian rubber
done
clear
B)
Orchid
done
clear
C)
Tinospora
done
clear
D)
Cuscuta
done
clear
View Answer play_arrow
Adventitious roots developed by:
A)
Develop from radical
done
clear
B)
Develop from flower
done
clear
C)
Develop from embryo
done
clear
D)
Develop from any part of plant body except radical
done
clear
View Answer play_arrow
Petiole is modified into tendril in:
A)
Passiflora
done
clear
B)
Gloriosa
done
clear
C)
Pisum
done
clear
D)
Clematis
done
clear
View Answer play_arrow
Root that grow from any part of the plant body other than the radical are called?
A)
Tap root
done
clear
B)
Adventitious root
done
clear
C)
Modified roots
done
clear
D)
Aerial roots
done
clear
View Answer play_arrow
Example of Geocarpic fruit:
A)
Potato
done
clear
B)
Pea nut
done
clear
C)
Onion
done
clear
D)
Garlic
done
clear
View Answer play_arrow
Which of the following pairs is not correct?
A)
Corymb - Candytuft
done
clear
B)
Capitulum - Sunflower
done
clear
C)
Catkin - Mulberry
done
clear
D)
Raceme - Wheat
done
clear
View Answer play_arrow
Individual components of perianth are called:
A)
Sepals
done
clear
B)
Petals
done
clear
C)
Tepals
done
clear
D)
Brackets
done
clear
View Answer play_arrow
Zygomorphic condition can be represented as:
A)
C
done
clear
B)
%
done
clear
C)
P
done
clear
D)
G
done
clear
View Answer play_arrow
Which is the path of blood circulation in cockroach:
A)
Heart - artery - organs - heart
done
clear
B)
Heart - Sinuses - heart
done
clear
C)
Sinuses - artery - organs - vein - heart
done
clear
D)
Heart - artery - heart
done
clear
View Answer play_arrow
Which structure possess flagellin protein?
A)
Muscles fiber
done
clear
B)
Flagellum
done
clear
C)
Pilli
done
clear
D)
All of these
done
clear
View Answer play_arrow
What is the function of SER?
A)
Synthesis of steroid hormone
done
clear
B)
Synthesis of protein
done
clear
C)
Synthesis of enzyme
done
clear
D)
All of the above
done
clear
View Answer play_arrow
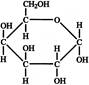
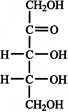
Which of the following structure shows the molecules?
A)
Glucose
done
clear
B)
Fructose
done
clear
C)
Galactose
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following structure show the types of sugar?
A)
Aldo triose sugar
done
clear
B)
Aldo pentose sugar
done
clear
C)
Keto triose sugar
done
clear
D)
Keto pentose sugar
done
clear
View Answer play_arrow
Full form of RUBISCO is:
A)
Ribulose Bisulphate carboxylase Oxygenase
done
clear
B)
Ribuose Biphosphate Carboxylase Oxygenase
done
clear
C)
Ribuose Bisulphate Carboxylase Oxygenase
done
clear
D)
Ribulose Bisulphate Carboxylase Oxygenase
done
clear
View Answer play_arrow
At the end of which stage does cell enter mitosis?
A)
\[{{G}_{1}}-phase\]
done
clear
B)
\[S-phase\]
done
clear
C)
\[M-phase\]
done
clear
D)
\[{{G}_{2}}-phase\]
done
clear
View Answer play_arrow
Membrane protein is responsible for transport of:
A)
Water molecules
done
clear
B)
Transpiration of \[{{H}_{2}}O\]
done
clear
C)
Active transport
done
clear
D)
Passive transport
done
clear
View Answer play_arrow
Which group is included in macronutrients:
A)
H, Mn, S
done
clear
B)
S, P, Ca, Mg
done
clear
C)
Mn, Cu, N
done
clear
D)
Na, Cl
done
clear
View Answer play_arrow
One photon of blue light contains ____Kcal and of red light ____ Kcal:
A)
70; 40
done
clear
B)
220; 70
done
clear
C)
10; 90
done
clear
D)
90; 10
done
clear
View Answer play_arrow
Chloroplast has maximum quantity of____ in stroma:
A)
Dehydrogenase
done
clear
B)
RuBP carboxylase
done
clear
C)
Pyruvic carboxylase
done
clear
D)
Hexokinase
done
clear
View Answer play_arrow
What is the first stage in respiration:
A)
Glycolysis
done
clear
B)
Krebs cycle
done
clear
C)
ETS
done
clear
D)
Glycogenesis
done
clear
View Answer play_arrow
Spiral development of tendrils?
A)
Thigmotropism
done
clear
B)
Thermotropism
done
clear
C)
Hydrotropism
done
clear
D)
Phototropism
done
clear
View Answer play_arrow
Vernalization is a/an:
A)
Growth graph related to light
done
clear
B)
Effect of photoperiod on the growth of plant, which results flowering
done
clear
C)
Rapid growth in low temperature
done
clear
D)
Daily photoperiod
done
clear
View Answer play_arrow
Our Buccal cavity open?
A)
Oesophagus
done
clear
B)
Salivary gland
done
clear
C)
Pharynx
done
clear
D)
Stomach
done
clear
View Answer play_arrow
Duodenum is a part of _____
A)
Stomach
done
clear
B)
Oesophagus
done
clear
C)
Digestive track
done
clear
D)
Small Intestine
done
clear
View Answer play_arrow
One RBC can transport about how many molecules of oxygen?
A)
One million
done
clear
B)
Ten million
done
clear
C)
One billion
done
clear
D)
Ten billion
done
clear
View Answer play_arrow
Which one is not viral infection:
A)
Vocational lungs disease
done
clear
B)
Bronchitis
done
clear
C)
Asthma
done
clear
D)
Emphysema
done
clear
View Answer play_arrow
Thrombokinase is produced in:
A)
RBC
done
clear
B)
WBC
done
clear
C)
Blood vessels
done
clear
D)
Blood platelets
done
clear
View Answer play_arrow
Lymph consists of:
A)
RBCs, WBCs and plasma
done
clear
B)
RBCs, proteins and platelets
done
clear
C)
All components of blood except RBCs & some protein
done
clear
D)
WBCs and Serum
done
clear
View Answer play_arrow
Transamination process takes place in:
A)
Liver
done
clear
B)
Heart
done
clear
C)
Kidney
done
clear
D)
All of the above
done
clear
View Answer play_arrow
According to solubility of water:
A)
\[N{{H}_{3}}>uricacid>urea\]
done
clear
B)
\[Uric\text{ }acid>urea>N{{H}_{3}}\]
done
clear
C)
\[Uric>acid>N{{H}_{3}}>ures\]
done
clear
D)
\[N{{H}_{3}}>urea>uric\,acid\]
done
clear
View Answer play_arrow
Which phase of conjugation is impossible in gamete?
A)
Post - fertilization phase
done
clear
B)
Fertilization phase
done
clear
C)
Pre - fertilization phase
done
clear
D)
Gamete phase
done
clear
View Answer play_arrow
Micropylar end lacks:
A)
Egg cell
done
clear
B)
Synergids
done
clear
C)
Egg - apparatus
done
clear
D)
Integument
done
clear
View Answer play_arrow
Transfer of a pollen grain to the stigma of the same plant is called:
A)
Autogamy
done
clear
B)
Geitonogamy
done
clear
C)
Allogamy
done
clear
D)
Homogamy
done
clear
View Answer play_arrow
Suspensor is produced from:
A)
Apical cell
done
clear
B)
Small upper basal cell
done
clear
C)
Large lower basal cell
done
clear
D)
Hypothesis
done
clear
View Answer play_arrow
Which gland secrete alkaline fluid?
A)
Seminal vesicle gland
done
clear
B)
Prostate gland
done
clear
C)
Bulbourethral gland
done
clear
D)
[b] and [c] both
done
clear
View Answer play_arrow
What is pH of semen?
A)
7.3 to 7.7
done
clear
B)
7.2 to 7.6
done
clear
C)
7.4 to 7.8
done
clear
D)
7.5 to 7.9
done
clear
View Answer play_arrow
Which tissue layer covers the ovary?
A)
Columnar epithelium
done
clear
B)
Squamoces epithelium
done
clear
C)
Cuboidal epithelium
done
clear
D)
Ciliary epithelium
done
clear
View Answer play_arrow
Which method is used to detect the gender of the fetus?
A)
ART
done
clear
B)
IVF
done
clear
C)
AFT
done
clear
D)
GIFT
done
clear
View Answer play_arrow
In amino contests the fluid is taken from
A)
Foetal blood
done
clear
B)
Mother's blood
done
clear
C)
Body fluid of mother
done
clear
D)
Fluid surrounding foetus
done
clear
View Answer play_arrow
Text cross is:
A)
\[Tt\times Tt\]
done
clear
B)
\[Tt\times TT\]
done
clear
C)
\[TT\times TT\]
done
clear
D)
\[~Tt\times tt\]
done
clear
View Answer play_arrow
If distance between gene on chromosome in more, then gene shows:
A)
Less linkage
done
clear
B)
Strong linkage
done
clear
C)
Weak linkage
done
clear
D)
Incomplete linkage
done
clear
View Answer play_arrow
The couple has four daughters. The percentage probability of fifth child to be a Daughter is:
A)
10
done
clear
B)
5
done
clear
C)
75
done
clear
D)
100
done
clear
View Answer play_arrow
Dihybrid cross proves the law of:
A)
Segregation
done
clear
B)
Purity of gametes
done
clear
C)
Dominance
done
clear
D)
Independent assortment
done
clear
View Answer play_arrow
Multiplication of DNA is called:
A)
Translation
done
clear
B)
Replication
done
clear
C)
Transduction
done
clear
D)
Transcription
done
clear
View Answer play_arrow
Which of the following enzymes can detect and correct the wrong inserted base during DNA replication?
A)
DNA polymerase-I
done
clear
B)
DNA polymerase-II
done
clear
C)
Primase
done
clear
D)
Ligase
done
clear
View Answer play_arrow
Humans have approximately _____ times more than genes E. Coli:
A)
8
done
clear
B)
15
done
clear
C)
100
done
clear
D)
50
done
clear
View Answer play_arrow
The scientist who cut off the tails of mice of successive generations to prove Lamarck's theory was wrong:
A)
Weismann
done
clear
B)
Haeckel
done
clear
C)
Darwin
done
clear
D)
Wallace
done
clear
View Answer play_arrow
Which of the following are kingdoms?
A)
Monera
done
clear
B)
Protista
done
clear
C)
Animalae
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Gonyaulax is responsible for:
A)
Cancer
done
clear
B)
Red tides
done
clear
C)
Asthma
done
clear
D)
Black tides
done
clear
View Answer play_arrow
Which substance is twice amount of in maize hybride:
A)
Lysine
done
clear
B)
Leusine
done
clear
C)
Iso leusine
done
clear
D)
Glycine
done
clear
View Answer play_arrow
In which plant the seeds do not contain stored food?
A)
Atropa beladona
done
clear
B)
Canbis sativa
done
clear
C)
Orchids
done
clear
D)
Opium
done
clear
View Answer play_arrow
Which test is carried out if disease is occurred by Salmonella typhi?
A)
Widal test
done
clear
B)
Lisman test
done
clear
C)
Blood test
done
clear
D)
Barium test
done
clear
View Answer play_arrow
Mark odd one?
A)
Chickengunia
done
clear
B)
Elephantiasis
done
clear
C)
Diphtheria
done
clear
D)
Malaria
done
clear
View Answer play_arrow
What is reason for causing AIDS:
A)
Cereto immunity
done
clear
B)
Interferon
done
clear
C)
Destroying and Helper T-cells
done
clear
D)
Destroying of killer T-cells
done
clear
View Answer play_arrow
Which plant responsible for doing hallucination?
A)
Atropa baladona
done
clear
B)
Datura
done
clear
C)
Erythroxylumcoca
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which one is related with steroids:
A)
Citric acid
done
clear
B)
Batyric acid
done
clear
C)
Acetic acid
done
clear
D)
Palmitic acid
done
clear
View Answer play_arrow
Which bacteria is useful for purification in sewage treatment?
A)
Hetrotrops
done
clear
B)
Symbiont
done
clear
C)
Free living
done
clear
D)
Parasist
done
clear
View Answer play_arrow
Ideal host for the amplification of DNA molecules:
A)
Viruses
done
clear
B)
Plants
done
clear
C)
Bacteria
done
clear
D)
Animals
done
clear
View Answer play_arrow
The first hormone artificially produced by culturing bacteria is:
A)
Insulin
done
clear
B)
Thyroxine
done
clear
C)
Testosterone
done
clear
D)
Adrenaline
done
clear
View Answer play_arrow
Exploitation of patent of biological resources of other nations is called as:
A)
Biosafety
done
clear
B)
Biopiracy
done
clear
C)
Biowar
done
clear
D)
Bioabuse
done
clear
View Answer play_arrow
Biofuel is made by utilizing which strain of bacteria?
A)
Bacillus amylolicfue faciens
done
clear
B)
Klebsiella planticola
done
clear
C)
E. Coli
done
clear
D)
Phanerochaete chrysosporium
done
clear
View Answer play_arrow
In birth rate and death rate is equal, then what will be the vital Index?
A)
= 1
done
clear
B)
=100
done
clear
C)
>100
done
clear
D)
<100
done
clear
View Answer play_arrow
Which path is following by plants as a part of adaptation in an ecosystem where living?
A)
\[{{C}_{3}}\]path
done
clear
B)
\[{{C}_{4}}\] plant
done
clear
C)
CAM path
done
clear
D)
TCA path
done
clear
View Answer play_arrow
Which of the following is trophic level of the orchid staying on mango tree?
A)
First
done
clear
B)
Tertiary third
done
clear
C)
Second
done
clear
D)
Fourth
done
clear
View Answer play_arrow
Which age group in pyramid indicates less reproductive potential?
A)
Bell shaped
done
clear
B)
Triangular
done
clear
C)
Inverted bell shaped
done
clear
D)
All of them have equal potential
done
clear
View Answer play_arrow
What is the original source of energy for the living organisms?
A)
Carbohydrate
done
clear
B)
Sunlight
done
clear
C)
ATP
done
clear
D)
Lipid
done
clear
View Answer play_arrow
In which of the following curd eating people are included?
A)
Producer
done
clear
B)
First
done
clear
C)
Tertory
done
clear
D)
Second
done
clear
View Answer play_arrow
Which scientist has classified species diversity?
A)
Thoeprestus
done
clear
B)
Lineus
done
clear
C)
Whittaker
done
clear
D)
Treshaw
done
clear
View Answer play_arrow
Which can be used for cryopreservation at \[196{}^\circ c\] temperature:
A)
Liquid \[{{N}_{2}}\]
done
clear
B)
Free \[{{N}_{2}}\]
done
clear
C)
Liquid \[C{{O}_{2}}\]
done
clear
D)
Solid \[{{N}_{3}}\]
done
clear
View Answer play_arrow
Biodiversity of which organisms is more in eastern ghat in comparison to western ghat?
A)
Reptilia
done
clear
B)
Amphibian
done
clear
C)
Aves
done
clear
D)
Mammals
done
clear
View Answer play_arrow
Which gas is responsible for increase in atmospheric temperature?
A)
\[{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
Sound waves of _____ decibels causes damages to eardrum or permanently impair hearing?
A)
150 db or more
done
clear
B)
120 db or more
done
clear
C)
More than 80 db
done
clear
D)
110 db
done
clear
View Answer play_arrow
Which chemical of petrol in activates the catalysts of filter for reducing emission of poisonous gass?
A)
Lead
done
clear
B)
Rhodium
done
clear
C)
Straunsium
done
clear
D)
Iron
done
clear
View Answer play_arrow
Chipko movement is associated with prevention of
A)
Air pollution
done
clear
B)
Cutting of trees
done
clear
C)
River pollution
done
clear
D)
Construction of dames
done
clear
View Answer play_arrow
Jhoom cultivation refers to
A)
Crop cultivation
done
clear
B)
Social forestry
done
clear
C)
Shifting cultivation
done
clear
D)
Large scale agriculture
done
clear
View Answer play_arrow