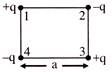
The work done required to put the four charges together at the comers of a square of side a, as shown in the figure is-
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{{{q}^{2}}}{a}\]
done
clear
B)
\[-\frac{2.6{{q}^{2}}}{4\pi {{\varepsilon }_{0}}}\,\,\frac{{{q}^{2}}}{a}\]
done
clear
C)
\[+\frac{2.6}{4\pi {{\varepsilon }_{0}}}\,\,\frac{{{q}^{2}}}{a}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
The bob of a pendulum carries an electric charge of \[39.2\,\,\times \,\,{{10}^{-}}^{10}\] coulomb in a horizontal electric field of \[20\text{ }\times \text{ }{{10}^{3}}\,V/m\] and it is at rest. The angle made by the pendulum with the vertical will be, if the mass of pendulum is \[8\,\,\times \,\,{{10}^{-6}}\text{ }kg\] and \[g\,\,=\,\,9.8\text{ }m/{{s}^{2}}\]-
A)
\[30{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[120{}^\circ \]
done
clear
View Answer play_arrow
Two identical capacitors, have the same capacitance C. One of them is charged to potential \[{{V}_{1}}\] and the other to\[{{V}_{2}}\]. The negative ends of the capacitors are connected together. When the positive ends are also connected, the decrease in energy of the combined system is-
A)
\[\frac{1}{4}\,\,C\,(V_{1}^{2}-V_{2}^{2})\]
done
clear
B)
\[\frac{1}{4}\,\,C\,(V_{1}^{2}+V_{2}^{2})\]
done
clear
C)
\[\frac{1}{4}\,\,C\,\,{{({{V}_{1}}-{{V}_{2}})}^{2}}\]
done
clear
D)
\[\frac{1}{4}\,\,C\,\,{{({{V}_{1}}+{{V}_{2}})}^{2}}\]
done
clear
View Answer play_arrow
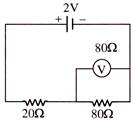
In the given figure, the emf of the cell is 2V and internal resistance is negligible. The resistance of the voltmeter is 80 ohm. The reading of voltmeter will be-
A)
2.00 volt
done
clear
B)
1.33 volt
done
clear
C)
1.60 volt
done
clear
D)
0.80 volt
done
clear
View Answer play_arrow
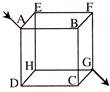
Twelve wires each of resistance \[6\,\Omega \] are connected to form a cube as shown in the adjoining figure. The current enters at a corner A and leaves at the diagonally opposite comer G. The equivalent resistance across the comers A and G is-
A)
\[12\,\Omega \]
done
clear
B)
\[6\,\Omega \]
done
clear
C)
\[3\,\Omega \]
done
clear
D)
\[5\,\Omega \]
done
clear
View Answer play_arrow
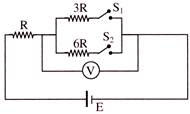
In the circuit shown in the figure, reading of voltmeter is \[{{V}_{1}}\] when only \[{{S}_{1}}\] is closed, reading of voltmeter is \[{{V}_{2}}\] when only \[{{S}_{2}}\] is closed and reading of voltmeter is \[{{V}_{3}}\] when both \[{{S}_{1}}\] and \[{{S}_{2}}\] are closed. Then-
A)
\[{{V}_{3}}>{{V}_{2}}>{{V}_{1}}\]
done
clear
B)
\[{{V}_{2}}>{{V}_{1}}>{{V}_{1}}\]
done
clear
C)
\[{{V}_{3}}>{{V}_{1}}>{{V}_{2}}\]
done
clear
D)
\[{{V}_{1}}>{{V}_{2}}>{{V}_{3}}\]
done
clear
View Answer play_arrow
The magnetic field existing in a region is given by\[\overrightarrow{B}={{B}_{0}}\left[ 1+\frac{x}{\ell } \right]\widehat{k}\]. A square loop of edge\[\ell \]. and carrying current I is placed with its edges parallel to the x-y axis. The magnitude of the net magnetic force experienced by the loop is-
A)
\[2{{B}_{0}}I\ell \]
done
clear
B)
\[{{B}_{0}}{{I}_{0}}\ell \]
done
clear
C)
\[{{B}_{0}}I\ell \]
done
clear
D)
\[BI\ell \]
done
clear
View Answer play_arrow
A long wire carries a steady current. It is bent into a circle of one turn and the magnetic field at the centre of the coil is B. It is then bent into a circular loop of n turns. The magnetic field at the centre of the coil will be-
A)
\[nB\]
done
clear
B)
\[{{n}^{2}}B\]
done
clear
C)
\[2nB\]
done
clear
D)
\[BI\ell \]
done
clear
View Answer play_arrow
A magnet is suspended in such a way that it oscillates in the horizontal plane. It makes 20 oscillations per minute at a place where dip angle is \[30{}^\circ \] and 15 oscillations per minute at a place where dip angle is \[60{}^\circ \]. The ratio of the total earth?s magnetic field at the two places is-
A)
\[3\sqrt{3}\,\,:\,\,8\]
done
clear
B)
\[16\,\,:\,\,9\sqrt{3}\]
done
clear
C)
\[4\,\,:\,\,9\]
done
clear
D)
\[2\sqrt{3}\,\,:\,\,9\]
done
clear
View Answer play_arrow
A circular coil of radius a and having N turns is placed at centre of a long solenoid, coaxially. The solenoid has radius \[b\,(b>>a)\] and number of turns per unit length is n. Their coefficient of mutual inductance will be-
A)
\[{{\mu }_{0}}n{{N}^{2}}\pi {{a}^{2}}\]
done
clear
B)
\[{{\mu }_{0}}nN\pi {{a}^{2}}\]
done
clear
C)
\[{{\mu }_{0}}{{n}^{2}}N\pi {{a}^{2}}\]
done
clear
D)
\[{{\mu }_{0}}{{n}^{2}}{{N}^{2}}\pi {{a}^{2}}\]
done
clear
View Answer play_arrow
Select the correct alternative (s): L, C and R denote inductance, capacitance and resistance respectively. The combinations which have the dimensions of frequency are-
[a] \[1/RC\] [b] \[R/L\] [c] \[R/L\] [d] \[C/L\]
A)
a, b, c
done
clear
B)
a, b, d
done
clear
C)
a, c, d
done
clear
D)
b, c, d
done
clear
View Answer play_arrow
The relation between electric field E and magnetic field induction B in an electromagnetic wave-
A)
\[E=\sqrt{\frac{{{\mu }_{0}}}{{{\varepsilon }_{0}}}}\,B\]
done
clear
B)
\[E\,\,=\,\,cB\]
done
clear
C)
\[E\,\,=\,\,\frac{B}{c}\]
done
clear
D)
\[E\,\,=\,\,\frac{B}{{{c}^{2}}}\]
done
clear
View Answer play_arrow
The horizontal range and maximum height attained by a projectile are R and H respectively. If a constant horizontal acceleration \[a=g/4\] is imparted to the projectile due to wind, then its horizontal range and maximum height will be-
A)
\[(R+H),\,\frac{H}{2}\]
done
clear
B)
\[\left( R+\frac{H}{2} \right),\,\,2H\]
done
clear
C)
\[\left( R+2H \right),\,\,H\]
done
clear
D)
\[\left( R+H \right),\,\,H\]
done
clear
View Answer play_arrow
The velocity of a particle is\[v={{v}^{0}}+gt+f{{t}^{2}}\]. If its position is \[x=0\text{ }at\text{ }t=0\], then its displacement after unit time \[\left( t=1 \right)\] is-
A)
\[{{v}_{0}}+2g+3f\]
done
clear
B)
\[{{v}_{0}}+g/2+f/3\]
done
clear
C)
\[{{v}_{0}}+g+f\]
done
clear
D)
\[{{v}_{0}}+g/2+f\]
done
clear
View Answer play_arrow
An object originally at the point (2, 5, 1) cm is given a displacement\[8\widehat{i}-2\widehat{j}+\widehat{k}\,\,cm\]. The co- ordinates of the new position are-
A)
(10, 3, 2) cm
done
clear
B)
(8,-2, 1) cm
done
clear
C)
(0, 0, 0) cm
done
clear
D)
data not correct
done
clear
View Answer play_arrow
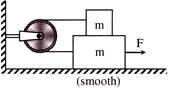
For the arrangement shown in figure the coefficient of friction between the two blocks is\[\mu \]. If both the block are identical, then the acceleration of each block is-
A)
\[\frac{F}{2m}-2\mu g\]
done
clear
B)
\[\frac{F}{2m}\]
done
clear
C)
\[\frac{F}{2m}-\mu g\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A block of mass m rests on a rough horizontal surface as shown in the figure. Coefficient of friction between the block and the surface is p. A force \[F=mg\] acting at angle \[\theta \] with the vertical side of the block pulls it. In which of the following cases can the block be pulled along the surface?
A)
\[\tan \,\theta \,\,\ge \,\,\mu \,\]
done
clear
B)
\[\cot \,\theta \,\,\ge \,\,\mu \,\]
done
clear
C)
\[\tan \,\,\frac{\theta }{2}\,\,\ge \,\,\mu \,\]
done
clear
D)
\[cot\,\,\frac{\theta }{2}\,\,\ge \,\,\mu \,\]
done
clear
View Answer play_arrow
A bullet when fired at a target has its velocity decreased to \[50\,%\] after penetrating 30 cm into it. Then the additional thickness it will penetrate before coming to rest is
A)
10 cm
done
clear
B)
30 cm
done
clear
C)
40 cm
done
clear
D)
60 cm
done
clear
View Answer play_arrow
A bomb of mass 9 kg explodes into the fragments of masses 3 kg and 6 kg. The velocity of mass 3 kg is\[16\text{ }m{{s}^{-}}^{1}\]. The energy of explosion is equal to-
A)
384 J
done
clear
B)
576 J
done
clear
C)
192 J
done
clear
D)
1152 J
done
clear
View Answer play_arrow
50 gm of ice is heated to increase its temperature by\[10{}^\circ C\]. If the same quantity of heat is given to 10 gm of water, the rise in its temperature is-
A)
\[25{}^\circ C\]
done
clear
B)
\[20{}^\circ C\]
done
clear
C)
\[17{}^\circ C~\]
done
clear
D)
\[18{}^\circ C\]
done
clear
View Answer play_arrow
Half a mole of helium at \[27{}^\circ C\] and at a pressure of 2 atmosphere is mixed with 1.5 mole of \[{{N}_{2}}\] at \[77{}^\circ C\] and at a pressure at 5 atmosphere so that the volume of the mixture is equal to the sum of their initial volumes. If the temperature of the mixture is \[69{}^\circ C\], its pressure in atmosphere is-
A)
3.5
done
clear
B)
3.8
done
clear
C)
3.95
done
clear
D)
4.25
done
clear
View Answer play_arrow
At \[27{}^\circ C\] two moles of an ideal monatomic gas occupy a volume V. The gas expands adiabatically to a volume 2V. Final temperature of the gas-
A)
200 K
done
clear
B)
250 K
done
clear
C)
173 K
done
clear
D)
189 K
done
clear
View Answer play_arrow
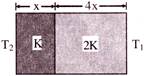
The temperatures of the two outer surfaces of a composite slab, consisting of two materials having coefficients of thermal conductivity K and 2K and thickness x and 4x respectively are\[{{T}_{2}}\,\,and\,\,{{T}_{1}}({{T}_{2}}>{{T}_{1}})\]. The rate of heat transfer through the slab, in a steady state is \[\left( \frac{A({{T}_{2}}-{{T}_{1}})K}{x} \right)f,\,\,\,\]with \[f\] equal to-
A)
\[1\]
done
clear
B)
\[1/2\]
done
clear
C)
\[2/3\]
done
clear
D)
\[1/3\]
done
clear
View Answer play_arrow
Two rods of lengths \[{{\ell }_{1}}\,\,and\,\,{{\ell }_{2}}\] are made of materials whose coefficient of linear expansions are\[{{\alpha }_{1}}\,\,and\,\,{{\alpha }_{2}}\]. If the difference between two lengths is independent of temperature-
A)
\[\frac{{{\ell }_{1}}}{{{\ell }_{2}}}\,\,=\,\,\frac{{{\alpha }_{1}}}{{{\alpha }_{2}}}\]
done
clear
B)
\[\frac{{{\ell }_{1}}}{{{\ell }_{2}}}\,\,=\,\,\frac{{{\alpha }_{2}}}{{{\alpha }_{1}}}\]
done
clear
C)
\[\ell _{2}^{2}{{\alpha }_{1}}\,\,=\,\,\ell _{1}^{2}{{\alpha }_{2}}\]
done
clear
D)
\[\frac{\alpha _{1}^{2}}{{{\ell }_{1}}}=\frac{\alpha _{2}^{2}}{{{\ell }_{2}}}\]
done
clear
View Answer play_arrow
A wave travels in a medium according to the equation of displacement given by \[y\,\,=\,\,0.03\text{ }sin\,\pi \left( 2t-0.0\text{1}x \right)\] where y and x are in metres and t in seconds. The wavelength of the wave is-
A)
200 m
done
clear
B)
100 m
done
clear
C)
20 m
done
clear
D)
10 m
done
clear
View Answer play_arrow
The velocity of the sound in a stationary medium is 'a'. A sound source is moving with a velocity 'b' toward a stationary observer. If the frequency of sound sources is n, then the fractional change in the frequency as heard by the observer is-
A)
\[\frac{a}{a-b}\]
done
clear
B)
\[\frac{b}{a-b}\]
done
clear
C)
\[\frac{a}{b}\,n\]
done
clear
D)
\[\frac{a+b}{a}\,\]
done
clear
View Answer play_arrow
In semiconductor the concentrations of electrons and holes are \[8\,\,\times \,\,{{10}^{18}}/{{m}^{3}}\] and \[5\,\,\times \,\,{{10}^{18}}/{{m}^{3}}\] respectively. If the mobilities of electrons and holes are \[2.3\,{{m}^{2}}/{{V}^{-2}}\] and 0.01 \[{{m}^{2}}/V-s\] respectively, then semiconductor is:
A)
n-type and its resistivity is \[0.34\,\Omega -m\]
done
clear
B)
p-type and its resistivity is \[0.034\,\Omega -m\]
done
clear
C)
n-type and its resistivity is \[0.034\,\Omega -m\]
done
clear
D)
p-type and its resistivity is \[3.4\,\,\Omega -m\]
done
clear
View Answer play_arrow
The volume of brick is 2.197 litres. The submerged brick is balanced by a 2.54 kg mass on the beam scale. The weight of the brick is-
A)
46 N
done
clear
B)
50 N
done
clear
C)
56 N
done
clear
D)
72 N
done
clear
View Answer play_arrow
The stress versus strain graphs for wires of two materials A and B are as shown in the figure. If \[{{y}_{A}}\,\,and\,\,{{y}_{B}}_{}\] are the Young's modulus of the materials, then-
A)
\[{{y}_{B}}=2{{y}_{A}}\]
done
clear
B)
\[{{y}_{A}}={{y}_{B}}\]
done
clear
C)
\[{{y}_{B}}=3{{y}_{A}}\]
done
clear
D)
\[{{y}_{A}}=3{{y}_{B}}\]
done
clear
View Answer play_arrow
The time period of a particle in simple harmonic motion is T. After a time (T/6) sec it passes from some position, its-
[a] velocity will be one-half its maximum velocity [b] displacement will be one-half its amplitude [c] acceleration will be nearly \[86\,%\] of its maximum acceleration [d] \[KE\,\,=\,\,PE\]
A)
a, b
done
clear
B)
b, c
done
clear
C)
a, c
done
clear
D)
c, d
done
clear
View Answer play_arrow
The weight of a body decrease by \[51%\] of the weight on the earth?s surface at a height of-
A)
\[\frac{10}{7}R\]
done
clear
B)
\[\frac{7}{10}R\]
done
clear
C)
\[\frac{3}{7}\,R\]
done
clear
D)
\[\frac{2}{7}\,R\]
done
clear
View Answer play_arrow
The moment of inertia of sphere is \[20\text{ }kg-{{m}^{2}}\] about the diameter. The moment of inertia about any tangent will be-
A)
\[70\text{ }kg-{{m}^{2}}\]
done
clear
B)
\[35\text{ }kg-{{m}^{2}}\]
done
clear
C)
\[50\text{ }kg-{{m}^{2}}\]
done
clear
D)
\[20\text{ }kg-{{m}^{2}}\]
done
clear
View Answer play_arrow
A particle is moving along a circular path of radius 3 meter in such a way that the distance travelled measured along the circumference is given by \[S\,\,=\,\,\frac{{{t}^{2}}}{2}\,\,+\,\,\frac{{{t}^{3}}}{3}\]. The acceleration of particle when \[t\text{ }=\text{ }2\] sec is-
A)
\[1.3\,m/{{s}^{2}}\]
done
clear
B)
\[13\,m/{{s}^{2}}\]
done
clear
C)
\[3\,m/{{s}^{2}}\]
done
clear
D)
\[10\,m/{{s}^{2}}\]
done
clear
View Answer play_arrow
When light of intensity \[1\text{ }W/{{m}^{2}}\] and wavelength \[5\,\,\times \,\,{{10}^{-}}^{7}\,m\] is incident on a surface. It is completely absorbed by the surface. If 100 photons emit one electron and area of the surface is \[1\text{ }c{{m}^{2}}\], then the photoelectric current will be-
A)
\[2m\,A\]
done
clear
B)
\[0.4\,\mu A\]
done
clear
C)
\[4.0\text{ }mA\]
done
clear
D)
\[4\,\mu A\]
done
clear
View Answer play_arrow
The radius of second orbit of an electron in hydrogen atom is\[2.116\text{ }\overset{{}^\circ }{\mathop{A}}\,\]. The de Broglie wavelength associated with this electron in this orbit would be-
A)
\[6.64\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
B)
\[1.058\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
C)
\[2.116\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
D)
\[13.28\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
View Answer play_arrow
Two coherent sources of intensity ratio \[1\text{ }:\text{ }4\] produce an interference pattern. The fringe visibility will be-
A)
1
done
clear
B)
0.8
done
clear
C)
0.4
done
clear
D)
0.6
done
clear
View Answer play_arrow
The magnify power of the objective of a compound microscope is 7 if the magnifying power of the microscope is 35, then the magnifying power is eyepiece will be-
A)
245
done
clear
B)
5
done
clear
C)
28
done
clear
D)
42
done
clear
View Answer play_arrow
A beam of light strikes a piece of glass at an angle of incidence of \[60{}^\circ \] and the reflected beam is completely plane polarised. The refractive index of the glass is-
A)
\[1.5\]
done
clear
B)
\[\sqrt{3}\]
done
clear
C)
\[\sqrt{2}\]
done
clear
D)
\[(3/2)\]
done
clear
View Answer play_arrow
When a light ray enters a refracting medium, it is found that the magnitude of the angle of refraction is equal to half the angle of reflection. If n is the refractive index of the medium, then the angle of incidence is-
A)
\[2\text{ si}{{\text{n}}^{-}}^{1}\left( n/2 \right)~\]
done
clear
B)
\[2\text{ }co{{s}^{-}}^{1}\left( n/2 \right)\]
done
clear
C)
\[co{{s}^{-}}^{1}\left( n/2 \right)\]
done
clear
D)
\[si{{n}^{-}}^{1}\left( n/2 \right)\]
done
clear
View Answer play_arrow
A prism having an apex angle \[4{}^\circ \] and refractive index 1.5 is located in front of a vertical plane mirror as shown in figure. Through what total angle is the ray deviated after reflection from the mirror?
A)
\[176{}^\circ \]
done
clear
B)
\[4{}^\circ \]
done
clear
C)
\[178{}^\circ \]
done
clear
D)
\[2{}^\circ \]
done
clear
View Answer play_arrow
A light ray is incident on a plane mirror \[{{M}_{1}}\] at an angle of incidence\[45{}^\circ \]. At what angle from mirror \[{{M}_{1}}\] a second plane mirror \[{{M}_{2}}\] be placed in order that the finally reflected ray from mirror \[{{M}_{2}}\] goes parallel to the mirror\[{{M}_{1}}\]?
A)
\[22.5{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[67.5{}^\circ \]
done
clear
View Answer play_arrow
The rest mass of an electron as well as that of positron is 0.51 MeV. When an electron and positron are annihilate, they produce 2 gamma- photons of wavelength(s)-
A)
\[0.012\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
B)
\[0.024\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
C)
\[0.012\text{ }\overset{{}^\circ }{\mathop{A}}\,~\text{ }to\text{ }\infty \]
done
clear
D)
\[0.024\text{ }\overset{{}^\circ }{\mathop{A}}\,~\text{ }to\text{ }\infty \]
done
clear
View Answer play_arrow
hi X-ray tube when the accelerating voltage V is halved, the difference between the wavelength of \[K{{ }_{a}}\] line and minimum wavelength of continuous X-ray spectrum-
A)
remain constant
done
clear
B)
increases
done
clear
C)
becomes half
done
clear
D)
decreases
done
clear
View Answer play_arrow
A radioactive substance X decays into another radioactive substance Y. Initially only X was present. \[{{\lambda }_{x}}\text{ }and\text{ }{{\lambda }_{y}}\] are the disintegration constant of X and Y. \[{{N}_{x}}\text{ }and\text{ }{{N}_{y}}\] are the number of nuclei of X and Y at any time t. Number of nuclei \[{{N}_{y}}\] will be maximum when-
A)
\[\frac{{{N}_{y}}}{{{N}_{x}}-{{N}_{y}}}\,\,=\,\,\frac{{{\lambda }_{y}}}{{{\lambda }_{x}}-{{\lambda }_{y}}}\]
done
clear
B)
\[\frac{{{N}_{y}}}{{{N}_{x}}-{{N}_{y}}}\,\,=\,\,\frac{{{\lambda }_{y}}}{{{\lambda }_{x}}-{{\lambda }_{y}}}\]
done
clear
C)
\[{{\lambda }_{y}}{{N}_{y}}\,=\,\,{{\lambda }_{x}}{{N}_{x}}\]
done
clear
D)
\[{{\lambda }_{y}}{{N}_{x}}\,=\,\,{{\lambda }_{x}}{{N}_{y}}\]
done
clear
View Answer play_arrow
A beam of cathode rays accelerated across potential difference V is subjected to crossed Electric (E) and Magnetic fields [B]. The fields are adjusted such that the beam is not deflected. The specific charge of the cathode rays is given by-
A)
\[\frac{{{E}^{2}}}{2V{{B}^{2}}}\]
done
clear
B)
\[\frac{{{B}^{2}}}{2V{{B}^{2}}}\]
done
clear
C)
\[\frac{2V{{B}^{2}}}{{{E}^{2}}}\]
done
clear
D)
\[\frac{2V{{E}^{2}}}{{{B}^{2}}}\]
done
clear
View Answer play_arrow
Which set is expected to show the smallest difference in first ionisation energy.
A)
He, Ne, Ar
done
clear
B)
B, N, O
done
clear
C)
\[Mg,\text{ }M{{g}^{+}},\text{ }M{{g}^{+2}}\]
done
clear
D)
Fe, Co, Ni
done
clear
View Answer play_arrow
In which of the following set do we have \[s{{p}^{3}}d\] hybridization-
A)
\[Xe{{F}^{2}},\text{ }IB{{r}_{3}},\text{ }Xe{{O}_{3}}\]
done
clear
B)
\[IB{{r}_{3}},SF_{5}^{\oplus },SF_{5}^{\Theta }\]
done
clear
C)
\[Xe{{F}_{2}},\text{ }IB{{r}_{3}},SF_{5}^{\oplus }\]
done
clear
D)
\[SF_{5}^{\oplus }\text{ }\And \text{ }SF_{5}^{\Theta }\]
done
clear
View Answer play_arrow
Which of the following reactions are not possible?
(I) \[{{(C{{H}_{3}})}_{2}}O+B{{F}_{3}}\to \,\,{{(C{{H}_{3}})}_{2}}O\,\,\to \,\,B{{F}_{3}}\] (II) \[{{(Si{{H}_{3}})}_{2}}O+B{{F}_{3}}\to {{(Si{{H}_{3}})}_{2}}O\,\,\to \,\,B{{F}_{3}}\] (III) \[{{H}_{3}}N+B{{F}_{3}}\to \,\,{{H}_{3}}N\to B{{F}_{3}}\] (IV) \[{{(C{{H}_{3}})}_{2}}O+BF_{4}^{\Theta }\,\,\to \,\,{{(C{{H}_{3}})}_{2}}O\to BF_{4}^{\Theta }\]
Correct is-
A)
I, II
done
clear
B)
II, III
done
clear
C)
II, IV
done
clear
D)
Only IV
done
clear
View Answer play_arrow
Which of the following molecule is planar?
A)
\[\left[ I{{\left( CN \right)}_{2}} \right]{{~}^{\Theta }}\]
done
clear
B)
\[PC{{l}_{3}}{{F}_{2}}\]
done
clear
C)
\[PC{{l}_{5}}\]
done
clear
D)
\[S{{F}_{4}}\]
done
clear
View Answer play_arrow
In the complex \[{{\left[ Pt\left( {{O}_{2}} \right){{\left( en \right)}_{2}}\left( Br \right) \right]}^{+2}}\], co- ordination number & oxidation number of platinum are-
A)
4, 3
done
clear
B)
4, 5
done
clear
C)
4, 6
done
clear
D)
6, 4
done
clear
View Answer play_arrow
The pair in which both species have same magnetic moment (spin only value) is-
A)
\[{{\left[ Cr{{({{H}_{2}}O)}_{6}} \right]}^{+2}},\,\,{{\left[ CoC{{l}_{4}} \right]}^{-2}}\]
done
clear
B)
\[{{\left[ Cr{{({{H}_{2}}O)}_{6}} \right]}^{+2}},\,\,{{\left[ Fe{{({{H}_{2}}O)}_{6}} \right]}^{+2}}\]
done
clear
C)
\[{{\left[ Cr{{({{H}_{2}}O)}_{6}} \right]}^{+2}},\,\,{{\left[ Fe{{({{H}_{2}}O)}_{6}} \right]}^{+2}}\]
done
clear
D)
\[{{\left[ CoC{{l}_{4}} \right]}^{-2}},\,\,{{\left[ Fe{{({{H}_{2}}O)}_{6}} \right]}^{+2}}\]
done
clear
View Answer play_arrow
Primary, secondary and tertiary amines can be distinguished by:
A)
Schiff' s test
done
clear
B)
Fehling' s test
done
clear
C)
Hinsberg test
done
clear
D)
Tollens' test
done
clear
View Answer play_arrow
Calcium imide on hydrolysis give gas (P) which on oxidation by bleaching powder give gas (Q). Gas (Q) on reaction with magnesium gives compound(R) which on hydrolysis again give gas (P). Identify (P), (Q), (R).
A)
\[{{N}_{2}},\text{ }N{{H}_{3}},\,\,MgNH\]
done
clear
B)
\[N{{H}_{3}},\,\,{{N}_{2}},\text{ }M{{g}_{3}}{{N}_{2}}\]
done
clear
C)
\[N{{H}_{3}},\text{ }N{{O}_{2}},\,\,Mg{{\left( N{{O}_{2}} \right)}_{2}}\]
done
clear
D)
\[{{N}_{2}},\text{ }{{N}_{2}}{{O}_{5}},\text{ }Mg{{\left( N{{O}_{3}} \right)}_{2}}\]
done
clear
View Answer play_arrow
Which one of the following processes will produce hard water-
A)
Saturation of water with \[CaC{{O}_{3}}\]
done
clear
B)
Addition of \[N{{a}_{2}}S{{O}_{4}}\] to water
done
clear
C)
Saturation of water with \[MgC{{O}_{3}}\]
done
clear
D)
Saturation of water with \[CaS{{O}_{4}}\]
done
clear
View Answer play_arrow
\[PbC{{l}_{4}}\] exist but \[Pb{{I}_{4}}\] does not exist because of-
A)
Large size of \[{{I}^{\Theta }}\]
done
clear
B)
Strong oxidising character of \[P{{b}^{+4}}\]
done
clear
C)
Strong reducing character of \[P{{b}^{+4}}\]
done
clear
D)
Low electronegativity of \[{{I}^{\Theta }}\]
done
clear
View Answer play_arrow
Which of the following does not have S-S-linkage?
A)
\[{{S}_{2}}{{O}_{8}}{{^{-}}^{2}}\]
done
clear
B)
\[{{S}_{2}}{{O}_{6}}{{^{-}}^{2}}\]
done
clear
C)
\[{{S}_{2}}{{O}_{5}}{{^{-}}^{2}}\]
done
clear
D)
\[{{S}_{2}}{{O}_{3}}{{^{-}}^{2}}\]
done
clear
View Answer play_arrow
The compound of Vanadium has magnetic moment of 1.73 B.M. The Vanadium chloride has the formula-
A)
\[VC{{l}_{2}}\]
done
clear
B)
\[VC{{l}_{3}}\]
done
clear
C)
\[VC{{l}_{4}}\]
done
clear
D)
\[VC{{l}_{5}}\]
done
clear
View Answer play_arrow
Both geometrical and optical isomerism are shown by-
A)
\[{{\left[ Co{{(en)}_{2}}C{{l}_{2}} \right]}^{+}}\]
done
clear
B)
\[{{\left[ Co{{(N{{H}_{3}})}_{5}}Cl \right]}^{+2}}\]
done
clear
C)
\[{{\left[ Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}} \right]}^{+}}\]
done
clear
D)
\[{{[Cr{{(ox)}_{3}}]}^{-3}}\]
done
clear
View Answer play_arrow
In which reaction Hs02 act as a reducing agent.
A)
\[2HI+{{H}_{2}}{{O}_{2}}\to \,\,{{I}_{2}}\,\,+\,\,2{{H}_{2}}O\]
done
clear
B)
\[FeS{{O}_{4}}+{{H}_{2}}S{{O}_{4}}+{{H}_{2}}{{O}_{2}}\to \,\,\,F{{e}_{2}}{{\left( S{{O}_{4}} \right)}_{3}}+2{{H}_{2}}O\]
done
clear
C)
\[MnO+{{H}_{2}}{{O}_{2}}\,\,\to \,\,Mn{{O}_{2}}+{{H}_{2}}O\]
done
clear
D)
\[{{\text{I}}_{\text{2}}}+2NaOH\,\,+\,\,{{H}_{2}}{{O}_{2}}\to 2NaI+{{H}_{2}}O+{{O}_{2}}\]
done
clear
View Answer play_arrow
A human body required the \[0.01\,\,\mu \] activity of radioactive substance after 24 hours. Half life of radioactive is 6 hours. Than injection of max. activity of radioactive substance that can be injected:
A)
0.08
done
clear
B)
0.04
done
clear
C)
0.16
done
clear
D)
0.32
done
clear
View Answer play_arrow
The shortest wavelength of H-atom in Lyman series is x, then longest wavelength in Balmer series of \[H{{e}^{+}}\] is-
A)
\[\frac{9x}{5}\]
done
clear
B)
\[\frac{36x}{5}\]
done
clear
C)
\[\frac{x}{4}\]
done
clear
D)
\[\frac{5x}{9}\]
done
clear
View Answer play_arrow
Heavy water is used as
A)
washing water
done
clear
B)
detergent
done
clear
C)
drinking water
done
clear
D)
moderator
done
clear
View Answer play_arrow
Insulin contains \[3.4\,%\] sulphur. What will be the minimum molecular weight of insulin?
A)
94.176
done
clear
B)
1884
done
clear
C)
941.176
done
clear
D)
976
done
clear
View Answer play_arrow
A solution containing \[10g\text{ }per\text{ }d{{m}^{3}}\] of urea \[\left( molecular\text{ }mass\text{ }=\text{ }60g\text{ }mo{{l}^{-1}} \right)\] is isotonic with a \[5%\] solution of a nonvolatile solute. The molecular mass of this nonvolatile solute is-
A)
\[250g\text{ }mo{{l}^{-}}^{1}\]
done
clear
B)
\[300g\text{ }mo{{l}^{-}}^{1}\]
done
clear
C)
\[350g\text{ }mo{{l}^{-}}^{1}\]
done
clear
D)
\[200g\text{ }mo{{l}^{-}}^{1}\]
done
clear
View Answer play_arrow
Propene on reaction with mCPBA produces-
A)
1, 3-Epoxy propane
done
clear
B)
1, 2-Epoxy propane
done
clear
C)
2-propanone
done
clear
D)
2-propanol
done
clear
View Answer play_arrow
Identify the product (Y) in the following reaction sequence:
A)
cyclobutane
done
clear
B)
cyclopentane
done
clear
C)
pentane
done
clear
D)
cyclopentanone
done
clear
View Answer play_arrow
\[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\,\,\to \,\,{{H}_{2}}O\,(\ell )\] \[B.E\left( H-H \right)={{x}_{1}},\,\,B.E\left( O=O \right)={{x}_{2}};\,\,B.E\left( O-H \right)={{x}_{3}}\]Latent heat of vaporisation of water liquid into water vapour \[\text{=}\,\,{{x}_{4}}\text{ }then\text{ }\Delta {{H}_{f}}\] (that of formation of water) is-
A)
\[{{x}_{1}}+\frac{{{x}_{2}}}{2}-{{x}_{3}}+{{x}_{4}}\]
done
clear
B)
\[2{{x}_{3}}-{{x}_{1}}-\frac{{{x}_{2}}}{2}-{{x}_{4}}\]
done
clear
C)
\[{{x}_{1}}+\frac{{{x}_{2}}}{2}-2{{x}_{3}}-{{x}_{4}}\]
done
clear
D)
\[{{x}_{1}}+\frac{{{x}_{2}}}{2}-2{{x}_{3}}+{{x}_{4}}\]
done
clear
View Answer play_arrow
4.5 g of aluminium (at. mass 27 amu) is deposited at cathode from \[A{{l}^{3+}}\] Solution by a certain quantity of electric charge. The volume of hydrogen produced at STP from \[{{H}^{+}}\] ions in solution by the same quantity of electric charge will be-
A)
44.8 L
done
clear
B)
11.2 L
done
clear
C)
22.4 L
done
clear
D)
5.6 L
done
clear
View Answer play_arrow
If the bond dissociation energies of XY, Xz and Yz (all diatomic molecules) are in the ratio \[1\text{ }:\text{ }1\text{ }:\text{ }0.5\] and \[{{\Delta }_{f}}H\] of XY is\[-200\text{ }kJ\text{ }mo{{l}^{-1}}\]. The mol dissociation energy of Xs will be-
A)
\[800\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
B)
\[200\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[300\,\,kJ\,\,mo{{l}^{-1}}\]
done
clear
D)
\[400\,\,kJ\,\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
In which of the following compounds, the oxidation number of iodine is fractional?
A)
\[I{{F}_{7}}\]
done
clear
B)
\[I_{3}^{-}\]
done
clear
C)
\[I{{F}_{5}}\]
done
clear
D)
\[I{{F}_{3}}\]
done
clear
View Answer play_arrow
Among the metals Cr, Fe, Mn, Ti and Mg, the one that cannot by obtained by reduction of metal oxide by aluminium is-
A)
Cr
done
clear
B)
Fe
done
clear
C)
Mn
done
clear
D)
Mg
done
clear
View Answer play_arrow
A sudden large jump between the values of second and third ionization energy of an element would be associated with the electronic configuration-
A)
\[1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{6}}\,3{{s}^{2}}\,3{{p}^{4}}\]
done
clear
B)
\[1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{6}}\,3{{s}^{2}}\,3{{p}^{2}}\]
done
clear
C)
\[1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{6}}\,3{{s}^{2}}\,3{{p}^{2}}\]
done
clear
D)
\[1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{6}}\,3{{s}^{2}}\]
done
clear
View Answer play_arrow
The correct formula of salt formed by the neutralization of hypophosphorus acid with \[NaOH\] is-
A)
\[N{{a}_{3}}P{{O}_{3}}\]
done
clear
B)
\[N{{a}_{3}}P{{O}_{2}}\]
done
clear
C)
\[Na{{H}_{2}}P{{O}_{2}}\]
done
clear
D)
\[N{{a}_{2}}HP{{O}_{2}}\]
done
clear
View Answer play_arrow
In the electrolysis of a \[CuS{{O}_{4}}\] solution, how many grams of \[Cu\] are plated out on the cathode in the time that is takes to liberate 5.6 litre of \[{{O}_{2}}\](g), measured at STP, at the anode?
A)
31.75
done
clear
B)
14.2
done
clear
C)
4.32
done
clear
D)
None of these
done
clear
View Answer play_arrow
Among the following acids, which has the lowest \[p{{K}_{a}}\] value?
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[HCOOH\]
done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHCOOH\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
\[{{(C{{H}_{3}})}_{2}}C=CHC{{H}_{3}}\,\xrightarrow[Heat]{X}\,\,Acetone+AcOH\] In the above reaction X is-
A)
\[KMn{{O}_{4}}/{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[{{O}_{3}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}COOH\]
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following will not react with NaOH?
A)
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{3}}CON{{H}_{2}}\]
done
clear
D)
\[CH{{\left( CN \right)}_{3}}\]
done
clear
View Answer play_arrow
The IUPAC name of the following compound is
A)
2-ethyl-3, 4-dimethylpentanal
done
clear
B)
2, 3-dimethyl-4-aldo hexane
done
clear
C)
3-aldo-4, 5-dimethyl hexane
done
clear
D)
1, 3, 4, 5-tetraethyl butanal
done
clear
View Answer play_arrow
Reagent [A] can be-
A)
\[{{H}_{2}}O/{{H}^{+}}\]
done
clear
B)
\[B{{H}_{3}}.THF/{{H}_{2}}{{O}_{2}}-O{{H}^{-}}\]
done
clear
C)
\[Hg{{\left( OCOC{{H}_{3}} \right)}_{2}}.THF/NaB{{H}_{4}}.NaOH\]
done
clear
D)
all the above are true
done
clear
View Answer play_arrow
Lassaigne?s test for the detection of nitrogen fails in-
A)
\[{{H}_{2}}N-CO-NHN{{H}_{2}}.HCl\]
done
clear
B)
\[N{{H}_{2}}-N{{H}_{2}}.HCl\]
done
clear
C)
\[N{{H}_{2}}-CO-N{{H}_{2}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}-NH-N{{H}_{2}}.HCl\]
done
clear
View Answer play_arrow
Which set of terms correctly identifies the carbohydrate shown?
1. Pentose 2. Hexose 3. Aldose 4. Ketose 5. Pyranose 6. Furanose
A)
1, 3 and 6
done
clear
B)
1, 3 and 5
done
clear
C)
2, 3 and 5
done
clear
D)
2, 3 and 5
done
clear
View Answer play_arrow
Which of the following statements best describes the most stable conformation of trans-1, 3-dimethylcyclohexane?
A)
Both methyl groups are axial
done
clear
B)
Both methyl groups are equatorial
done
clear
C)
One methyl group is axial, the other equatorial
done
clear
D)
The molecule is severely strained and cannot exist
done
clear
View Answer play_arrow
The major products from the following sequence of reactions are \[{{(C{{H}_{3}})}_{2}}CHC{{H}_{2}}N{{(C{{H}_{2}}C{{H}_{3}})}_{2}}\xrightarrow{C{{H}_{3}}I}\xrightarrow[{{H}_{2}}O]{A{{g}_{2}}O}\xrightarrow{heat}\]?
A)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHC{{H}_{2}}N{{H}_{2}}+{{H}_{2}}C=C{{H}_{2}}\]
done
clear
B)
\[{{\left( C{{H}_{3}} \right)}_{2}}NC{{H}_{2}}C{{H}_{3}}+{{H}_{2}}C=C{{\left( C{{H}_{3}} \right)}_{2}}\]
done
clear
C)
done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{3}}\overset{+}{\mathop{N}}\,C{{H}_{2}}C{{H}_{3}}{{I}^{-}}+{{H}_{2}}C=C{{H}_{2}}\]
done
clear
View Answer play_arrow
A primary amine on treatment with carbon disulphide and \[HgC{{l}_{2}}\] produce
A)
alkyl cyanide
done
clear
B)
alkyl isothiocyanate
done
clear
C)
alkyl cyanate
done
clear
D)
carbylamine
done
clear
View Answer play_arrow
Major product A is-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[HC\equiv CH~~\xrightarrow[H{{g}^{+2}}]{{{H}_{2}}O}\left( A \right)~\xrightarrow[NaOH]{dilute}dilute\left( B \right)\] The compound [B] is-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following reagents will not react with above compound?
A)
Na metal
done
clear
B)
\[AgN{{O}_{3}}+N{{H}_{4}}OH\]
done
clear
C)
\[C{{u}_{2}}C{{l}_{2}}+N{{H}_{4}}OH\]
done
clear
D)
\[NaHC{{O}_{3}}\]
done
clear
View Answer play_arrow
If \[20\text{ }c{{m}^{3}}\] gas at 1 atm. is expanded to \[50\text{ }c{{m}^{3}}\] at constant T, then what is the final pressure?
A)
\[20\times \frac{1}{50}\]
done
clear
B)
\[50\times \frac{1}{20}\]
done
clear
C)
\[1\times \frac{1}{20}\times 50\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which is most basic?
A)
\[C{{H}_{3}}C{{H}_{2}}-N{{H}_{2}}\]
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Two gases P and Q both decolourise aqueous bromine but only one of them gives white ppt with Tollen?s reagent. P and Q are likely to be
A)
\[{{H}_{2}}C=C{{H}_{2}}\text{ }and\text{ }C{{H}_{3}}-C\equiv C-C{{H}_{3}}\]
done
clear
B)
\[HC\equiv CH\,\,and\,\,C{{H}_{3}}-C{{H}_{2}}-C\equiv CH\]
done
clear
C)
\[HC\equiv CH\,\,and\,\,C{{H}_{3}}-C\equiv CH\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-C\equiv CH\,\,and\,\,C{{H}_{3}}-C\equiv C-C{{H}_{3}}\]
done
clear
View Answer play_arrow
Which of the following is an autoimmune disease?
A)
Cancer
done
clear
B)
Asthma
done
clear
C)
Erythroblastosis foetalis
done
clear
D)
Rhematoid heart disease
done
clear
View Answer play_arrow
The gradient of temperature over surface of earth is-
A. Increase progressively from equator towards pole B. Decrease progressively from pole towards equator C. Increase progressively from pole towards equator D. Decrease progressively from plains to mountain top
How many of the above are correct
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Alcohol is produced by the activities of
A)
Leuconostoc
done
clear
B)
Saccharomyces cerevisiae
done
clear
C)
Torulopsis
done
clear
D)
Clostridium botulinum
done
clear
View Answer play_arrow
How many ATP are produced during fermentation-
A)
4
done
clear
B)
6
done
clear
C)
2
done
clear
D)
Zero
done
clear
View Answer play_arrow
A leaf primordium grows into adult leaf lamina by means of
A)
At first by apical meristem and later largely by marginal meristems
done
clear
B)
Apical meristem
done
clear
C)
Lateral meristem
done
clear
D)
Marfinal meristems
done
clear
View Answer play_arrow
The term 'niche' of a species refers to-
A)
Specific and habitual function
done
clear
B)
Specific place where an organism lives
done
clear
C)
Competitive power of an organism
done
clear
D)
Specific function of organism
done
clear
View Answer play_arrow
In our body, antibodies are formed against pathogens in-
A)
Lymphnodes by Lympho cytes
done
clear
B)
Liver by RBCs
done
clear
C)
Blood by platelets
done
clear
D)
Brain by macrophages
done
clear
View Answer play_arrow
The subunits of chromatids are:
A)
Chromonemata
done
clear
B)
Chromomeres
done
clear
C)
Telomeres
done
clear
D)
Secondary constrictions
done
clear
View Answer play_arrow
When the persons die in old age after completing its life span, the survivalship curve is-
A)
J-shaped
done
clear
B)
Concave
done
clear
C)
S-shaped
done
clear
D)
Convex
done
clear
View Answer play_arrow
In \[{{C}_{4}}\] plants the bundle sheath cells-
A)
Have thin walls to facilitate the gaseous exchange
done
clear
B)
Have large intercellular spaces
done
clear
C)
Are rich in PEP carboxylase
done
clear
D)
Have a high density of chloroplasts
done
clear
View Answer play_arrow
Match the column I with Column II
Column-I Column-II I Many people have nutrient deficiencies as they cannot buy fruits & vegetables [A] Single cell proteins II Crops with higher vitamins, proteins & fats are breed [B] Micropropagation III Growing microbes as the alternative source of protein [C] Somoclones IV Capacity to generate plant from single cell or explants [D] Hidden hunger V Production of thousand plants through tissue culture [E] Biofortification VI Genetically identical plants [F] Totipotency
A)
I-D, II-E, III-F, IV-A.V-B.VI-C
done
clear
B)
I-D, II-E, III-F, IV-A, V-B, VI-C
done
clear
C)
I-D, II-E, III-A, IV-F, V-B, VI-C
done
clear
D)
I-F, II-E, III-A, IV-D, V-B, VI-C
done
clear
View Answer play_arrow
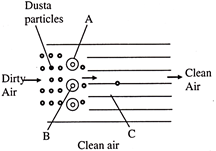
The below diagram shows electrostatic precipitator identify A, B and C
A)
A-Discharge corona, B-Negatively charged wire, C-Collection plate
done
clear
B)
A-Discharge corona, B-Positively charged wire, C-collection plate grounded
done
clear
C)
A-Discharge corona, B-Negatively charged, wire, C-Air filters
done
clear
D)
A-Uncharge corona, B-Positively charged wire, C-Dust removar:
done
clear
View Answer play_arrow
Cell-mediated immune responses are-
A)
Enhanced by depletion of complement
done
clear
B)
Enhanced by depletion of T-cells
done
clear
C)
Suppressed by antihistamine
done
clear
D)
Suppressed by cortisone
done
clear
View Answer play_arrow
INDC (Intended Nationally Determined Contribution) of India in Cop-21 is-
A)
\[60-65\text{ }%\] decrease in carbon emission in GDP from level of 2005
done
clear
B)
\[33-35\text{ }%\] decrease in carbon emission in GDP from level of 2005
done
clear
C)
\[26-28\text{ }%\] decrease in carbon emission in GDP from level of 2005
done
clear
D)
\[6-12\text{ }%\] decrease in carbon emission from level of 1990
done
clear
View Answer play_arrow
Secondary growth is absent in-
A)
Hydrophytes
done
clear
B)
Mesophytes
done
clear
C)
Halophytes
done
clear
D)
Xerophytes
done
clear
View Answer play_arrow
In an ecosystem-
A)
Cycling of energy and nutrients is a coupled process
done
clear
B)
Cycling of energy is an independent process
done
clear
C)
Movement of energy is unidirectional
done
clear
D)
Macro and micronutrients cycle at the same pace
done
clear
View Answer play_arrow
Which of the following mineral cannot be reutilized by plants-
A)
Ca
done
clear
B)
S
done
clear
C)
Mg
done
clear
D)
P
done
clear
View Answer play_arrow
In which stage of cell cycle each chromosome have two DNA:
A)
\[{{G}_{1}}\]
done
clear
B)
\[{{G}_{2}}\]
done
clear
C)
Anaphase-II
done
clear
D)
None of these
done
clear
View Answer play_arrow
Basal bodies are most closely associated with which one of the following cell components
A)
Nucleus
done
clear
B)
Cilia
done
clear
C)
Golgi apparatus
done
clear
D)
Mitochondria
done
clear
View Answer play_arrow
Sialic acid is present in:
A)
Chromatids
done
clear
B)
Golgi bodies
done
clear
C)
Cell wall
done
clear
D)
Cell membrane
done
clear
View Answer play_arrow
Desmosomes are concerned with-
A)
cell adherence
done
clear
B)
cytolysis
done
clear
C)
cellular excretion
done
clear
D)
cell division
done
clear
View Answer play_arrow
The study of chromosomes at meiotic diplotene shows that
A)
The intimately paired chromosomes repel each other and begin to separate
done
clear
B)
The pairing of homologous chromosomes which had initiated in the earlier stage is completed
done
clear
C)
The homologous chromosomes remain united by chiasmata
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Few events regarding meiosis-I is given below
A The chromosome gradually visible under light microscope during zygotene B. Bivalent are formed by a pair of synapsed homologous chromosome during pachytene C. Recombinase enzyme is involved in nonhomologous chromosome recombination during pachytene D. Chromosome pairing occurs during laptotene E. Chiasma is formed during diplotene
How many of these statement are true-
A)
Three
done
clear
B)
Four
done
clear
C)
Two
done
clear
D)
One
done
clear
View Answer play_arrow
When volume of body increases due to increase in the volume of cells, not in the number of cells, then it is called 'auxetic growth'. Such growth is found in-
A)
Lizards
done
clear
B)
Men
done
clear
C)
Frogs
done
clear
D)
Nematodes
done
clear
View Answer play_arrow
In which of the following stage of cell division, DNA content and chromosome number is equal to haploid-
A)
Anaphase
done
clear
B)
Telophase
done
clear
C)
Telophase-I
done
clear
D)
Telophase-II
done
clear
View Answer play_arrow
Kidneys of mammals are present on either side of vertebral column at the level of-
A)
10th thoracic to 3rd lumbar vertebrae
done
clear
B)
12th thoracic to 5th lumbar vertebrae
done
clear
C)
12th thoracic to 3rd lumbar vertebrae
done
clear
D)
10th thoracic to 5th lumbar vertebrae
done
clear
View Answer play_arrow
A. ICBN is used for naming of plant and bacteria. B. Taxonomic key are tools that help in classification of organism only. C. Animal species are more than plant species D. All living organism are linked to one another by sharing common genetic material.
A)
A, B, C & D are correct
done
clear
B)
A, C & D are true
done
clear
C)
A & C are true
done
clear
D)
C & D are true
done
clear
View Answer play_arrow
Taxonomic key is based on the set of contrasting characters known as-
A)
Lead
done
clear
B)
Doublet
done
clear
C)
Couplet
done
clear
D)
Catalogues
done
clear
View Answer play_arrow
Floral formula of ray floret in compositae
A)
\[Br\,\overset{{}^\circ }{\mathop{\,A}}\,\,\,K\,p\,\,{{C}_{(5)}}\,{{A}_{0\,}}{{G}_{(2)}}\]
done
clear
B)
\[Br%\,\,\,K\,p\,\,{{C}_{(4)}}\,{{A}_{0\,}}{{G}_{(2)}}\]
done
clear
C)
\[Br%\,\,\,Kp\,{{C}_{(5)}}\,{{A}_{0\,}}{{G}_{(2)}}\]
done
clear
D)
\[Br\overset{{}^\circ }{\mathop{\,A}}\,K{{p}_{(4)}}\,{{A}_{0\,}}{{G}_{(2)}}\]
done
clear
View Answer play_arrow
One direct use of earthworms to farmers is-
A)
Allowing sunlight to pass in upper layers of soil
done
clear
B)
Enrichment of soil by nephridial excretion
done
clear
C)
Removal of dead leves
done
clear
D)
Killing of some harmful insects
done
clear
View Answer play_arrow
Fungi that help in decomposition of litter and mineral cycling basically belong to-
A)
Bracket fungi
done
clear
B)
Sac fungi
done
clear
C)
Imperfect fungi
done
clear
D)
Lichens
done
clear
View Answer play_arrow
Orange rots is due to member of-
A)
Phycomycetes
done
clear
B)
Ascomycetes
done
clear
C)
Basidiomycetes
done
clear
D)
Deuteromycetes
done
clear
View Answer play_arrow
Bulbil is a modification of-
A)
Underground stem
done
clear
B)
Bases of leaves
done
clear
C)
Buds
done
clear
D)
Radicle
done
clear
View Answer play_arrow
1. Isogamy, oogamy or anisogamy 2. Stored food complex polysaccharide 3. Bitlagellated or tetraflagellated zoospore 4. Vacoulated cytoplasm 5. Isokont flagella
Which of the above mentioned character are present in pheophyceae?
A)
1, 2, 3, 4
done
clear
B)
1, 2, 4
done
clear
C)
1, 2, 4, 5
done
clear
D)
1, 2
done
clear
View Answer play_arrow
Which of the following is commonly present in fresh water?
[a] Ulothrix [b] Spirogyra [c] Ectocarpus [d] Baterachospermem
A)
A & B
done
clear
B)
A, B & D
done
clear
C)
A, B, C & D
done
clear
D)
A, B & C
done
clear
View Answer play_arrow
A. True root stem or leaves are not present B. Female sex organ is known as archegonium C. Unicellular or multicellular rhizoid are present D. Union of gametes occur in free water
Which of the above mentioned character are common in thallophyta and bryophyte both?
A)
A & D
done
clear
B)
A, B & D
done
clear
C)
Only A
done
clear
D)
Only D
done
clear
View Answer play_arrow
The female hormone that causes deposition of fat in the breast and hips and the growth of pubic hairs during puberty is-
A)
Estrogen
done
clear
B)
Progesterone
done
clear
C)
LH
done
clear
D)
FSH
done
clear
View Answer play_arrow
Pew diagram are given below. How many of the following belongs to monocot plant only
A)
Two
done
clear
B)
Three
done
clear
C)
One
done
clear
D)
Zero
done
clear
View Answer play_arrow
Which of the following is true regarding collenchyma
A)
It form major bulk of ground tissue in dicot stem
done
clear
B)
It is a mechanical and physiological tissue
done
clear
C)
It provide mechanical support to leaf petiole and leaf base
done
clear
D)
All of the above
done
clear
View Answer play_arrow
In a flower, if the megaspore mother cell forms megaspores without undergoing meiosis and if one of the megaspores develops into an embryo sac, its nuclei would be-
A)
Haploid
done
clear
B)
Diploid
done
clear
C)
A few haploid and a few diploid
done
clear
D)
With varying ploidy
done
clear
View Answer play_arrow
Which of the following is not a stem modification?
A)
Phyllode
done
clear
B)
Cladode
done
clear
C)
Phylloclade
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Floral character which is described by floral formula but not by floral diagram
A)
Placentation
done
clear
B)
Position of ovary
done
clear
C)
[a] & [b] both
done
clear
D)
Astivation
done
clear
View Answer play_arrow
Match the given below organelles with their function
I II a. Krebs cycle i. Stalked particles of mitochondria b. Photorespiration ii. Cytoplasm c. Oxidative iii. Peroxisomes d. Glycolysis iv. Inner surface of membrane of mitochondria
A)
a-ii b-iii c-iv d-i
done
clear
B)
a-i b-ii c-iii d-iv
done
clear
C)
a-iv b-iii c-i d-ii
done
clear
D)
a-iii b-ii c-iv d-i
done
clear
View Answer play_arrow
Few character regarding china rose is given below.
Which and how many are observed in china rose?
A)
Three A, C & E
done
clear
B)
Four A, B, C & D
done
clear
C)
Three A, C & D
done
clear
D)
Two A & C
done
clear
View Answer play_arrow
Whom among the following are known for their work on plant breeding in India?
A)
M. S. Swaminathan and B. P. Pal
done
clear
B)
M. S. Swaminathan and B. P. Pal and N. E. Borlaug
done
clear
C)
G. A. Gani, B. S. Kadam and N. E. Borlaug
done
clear
D)
M. S. Swaminathan and N. E. Borlaug
done
clear
View Answer play_arrow
Match the following
Column-I Column-II A SNPs (i) 3164.7 million B Genes of chromosomes N.1 (ii) 1.4 million C Total No. of Human genes (iii) 30000 D Total nucleotides of human genome (iv) 2968
A)
A-ii B-iii C-iv D-i
done
clear
B)
A-ii B-iv C-iii D-i
done
clear
C)
A-ii B-iv C-i D-iii
done
clear
D)
A-iv B-ii C-iii D-i
done
clear
View Answer play_arrow
Which of the following will be included under innate immunity?
A)
Mucus membrane
done
clear
B)
Interferon
done
clear
C)
Natural killer cells
done
clear
D)
All of these
done
clear
View Answer play_arrow
Match the list-
List-I List-II A. Diameter of the helix 1. \[34\overset{{}^\circ }{\mathop{A}}\,\] B. Distance between two adjacent 2. \[20\overset{{}^\circ }{\mathop{A}}\,\] C. Distance between two complete turns 3. \[3\overset{{}^\circ }{\mathop{A}}\,\] D. Length of the hydrogens 4. \[4.4\overset{{}^\circ }{\mathop{A}}\,\]
A)
A=1, B=3, C=2, D=4
done
clear
B)
A=2, B=4, C=1, D=3
done
clear
C)
A=4, B=2, C=3, D=1
done
clear
D)
A=1, B=4, C=2, D=3
done
clear
View Answer play_arrow
Factors which determine polarity of eggs are-
A)
Direct electric current
done
clear
B)
Presence of neighbouring eggs
done
clear
C)
Position of sperm entry
done
clear
D)
Unequal distribution of substances in egg
done
clear
View Answer play_arrow
In DNA finger printing-
A)
Restriction enzyme digests or generate unique fragments
done
clear
B)
The polymerase chain reaction amplifies fewer DNA
done
clear
C)
The variability of repeated sequences between two restriction sites is evaluated
done
clear
D)
All of these
done
clear
View Answer play_arrow
For a T-cell to recognize an antigen it must interact with-
A)
Complement
done
clear
B)
B cell
done
clear
C)
A macrophage
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Match the column-I with column-II and identify the correct match-
Column-I Column-II A. Cytidine P. Nucleoside B. Lecithin Q. Nucleotide C. Alanine R. Phospholipid D. Uracil S. Amino acid E. Trypsin T. Nitrogen base U. Enzyme
A)
A = P, B = R, C = S, D = T, E = U
done
clear
B)
A = Q, B = R, C = S, D = T, E = U
done
clear
C)
A = P, B = R, C = S, D = Q, E = U
done
clear
D)
A = Q, B = R, C = S, D = P, E = U
done
clear
View Answer play_arrow
Interferons are-
A)
Antiviral proteins
done
clear
B)
Antibacterial proteins
done
clear
C)
Anticancer proteins
done
clear
D)
None of above
done
clear
View Answer play_arrow
If two persons with heterozygous A and B blood group marries, then possibility of genotypes in their progeny are-
A)
\[Only\,\,{{I}^{0}}{{I}^{0}}\]
done
clear
B)
\[Only\,\,{{I}^{A}}{{I}^{B}}\]
done
clear
C)
\[{{I}^{A}}{{I}^{0}}\,\,and\,\,\,{{I}^{B}}{{I}^{0}}\]
done
clear
D)
All the above
done
clear
View Answer play_arrow
The possible blood groups of children bom to parents having A x AB groups are-
A)
O, A, B
done
clear
B)
O, A, B, AB
done
clear
C)
O, A
done
clear
D)
A, B, AB
done
clear
View Answer play_arrow
If \[{{6}^{n}}\] is the number of genotypes where 'n' is the number of different chromosome pairs with each carrying one heterozygous alleles. What would be the different genotypes produced by garden pea plant?
A)
\[{{6}^{10}}\]
done
clear
B)
\[{{6}^{12}}\]
done
clear
C)
\[{{6}^{14}}\]
done
clear
D)
\[{{6}^{7}}\]
done
clear
View Answer play_arrow
Each immunoglobin has two heavy chains & two light chains, the antigen binding site is present in-
A)
Variable region of heavy chain
done
clear
B)
Variable region of both heavy and light chain
done
clear
C)
Variable region of light chain
done
clear
D)
Constant region of both light and heavy chain
done
clear
View Answer play_arrow
Active immunity is obtained by-
A)
Natural resistance
done
clear
B)
Antibiotics
done
clear
C)
Weakened germs infection
done
clear
D)
None of these
done
clear
View Answer play_arrow
Genotype of male plant is \[{{S}_{2}}{{S}_{3}}\] while of female plant is\[{{S}_{1}}{{S}_{2}}\]. Which of the following pollen grain is comptible for the stigma of given female plant
A)
\[{{S}_{1}}\]
done
clear
B)
\[{{S}_{2}}\]
done
clear
C)
\[{{S}_{3}}\]
done
clear
D)
\[Both\,\,{{S}_{2}},\,\,{{S}_{3}}\]
done
clear
View Answer play_arrow
Which of the following immunity is conferred by transfer of immune products like antibodies from another individual into a non-immune individual?
A)
Adaptive immunity
done
clear
B)
Specific immunity
done
clear
C)
Active immunity
done
clear
D)
Passive immunity
done
clear
View Answer play_arrow
Which of the following statement is incorrect w.r.t. fishes?
A)
Cartilaginous fish like shark has placoid scales
done
clear
B)
Ammocoete larva of true fishes (after metamorphosis) shows anadromous migration
done
clear
C)
Lateral line system is well developed in fishes
done
clear
D)
In sea horse, male bears a brood pouch
done
clear
View Answer play_arrow
Tick mark the incorrect match
A)
Antennal gland - Crayfish
done
clear
B)
Metanephridia - Earthworm
done
clear
C)
Protonephridia - Planaria
done
clear
D)
Malpighian corpuscle - Cockroach
done
clear
View Answer play_arrow
Select the correct statement w.r.t. anatomical structure of cockroach?
A)
Ventral diaphragm in the haemocoel is non-perforated
done
clear
B)
Chitinous membrane is permeable to digestive enzyme and digested substance
done
clear
C)
Collateral gland forms outermost covering of ootheca
done
clear
D)
Crystalline cone of ommatidium is made up of 4 type of conical cells
done
clear
View Answer play_arrow
Which of the following will not make minerals more available to plants-
A)
Increasing the rainfall in a wet forested area
done
clear
B)
Raising the pH of a very acidic soil
done
clear
C)
Tillering a packed - down or water - logged soil
done
clear
D)
Introducing fungi that can form mycormiza into a soil that lacks them
done
clear
View Answer play_arrow
Which one of the following is correctly matched?
A)
Gout - Painful urination
done
clear
B)
Ketonuria - Presence of P-hydroxy butyric acid and acetone in urine
done
clear
C)
Nephroptosis - Dropping of the kidney, floating kidney
done
clear
D)
Oligouria - Presence of albumin in urine
done
clear
View Answer play_arrow
Osmosis is driven by following factor(s)
A)
density
done
clear
B)
pressure difference
done
clear
C)
solute concentration
done
clear
D)
both ?b and ?c
done
clear
View Answer play_arrow
Cochlea is composed of three parallel fluid-filled canals separated by Reissners membrane and basilar membrane. Which of the following canal is filled with endolymph?
A)
Median canal
done
clear
B)
Vestibular canal
done
clear
C)
Tympanic canal
done
clear
D)
Helicotrema
done
clear
View Answer play_arrow
If zona pellucida is digested during morula stage in fallopian tube then which of the following condition is likely to happen?
A)
It leads to ectopic pregnancy
done
clear
B)
Mobility is affected
done
clear
C)
Cleavage is affected
done
clear
D)
Corona radiata behaves like zona pellucida
done
clear
View Answer play_arrow
When an Amoeba comes near it's food, it stop moving and Pseudopodia are formed above, below and on the sides of food to form a food cup, which does not touch the food. Finally edges of food cup fase around the food to form a non-contractile food vacuole. The above method of ingestion is used by Amoeba to-
A)
Capture a immobile prey
done
clear
B)
Capture a live prey
done
clear
C)
Protection itself from enemy
done
clear
D)
Capture a prey
done
clear
View Answer play_arrow
Which of the following are examples of hormone releasing IUDs?
A)
CuT, Multiload 375
done
clear
B)
Progestasert, LNG-20
done
clear
C)
Lippes loop
done
clear
D)
Mala-D and Mala-N
done
clear
View Answer play_arrow
In Chlamydomonas the meiosis occurs in-
A)
Gamete
done
clear
B)
Zygote
done
clear
C)
Sporogonium
done
clear
D)
Zoospore
done
clear
View Answer play_arrow
Hb-F has a higher affinity for \[{{O}_{2}}\], because
A)
It binds BPG less strongly
done
clear
B)
Its oxygen haemoglobin dissociation curve appears on the left side
done
clear
C)
Hb-F can carry up to \[30%\] more \[{{O}_{2}}\] than maternal Hb-A
done
clear
D)
It having gamma polypeptide chain
done
clear
View Answer play_arrow
It causes cancer of vagina-
A)
Cadmium oxide
done
clear
B)
Diethylstibestrol
done
clear
C)
Afflatoxin
done
clear
D)
Vinylchloride
done
clear
View Answer play_arrow
The spaces in between the trabeculae of long bone, contain highly vascularised red bone marrow which takes part in forming erythrocytes and granular leucocytes. It is not present in which of the following?
A)
Proximal ends of long bones
done
clear
B)
Vertebrae
done
clear
C)
Skull bones
done
clear
D)
Clavicles
done
clear
View Answer play_arrow
Which of the following epithelium, serves to expel secretions, such as sweat, milk and saliva from the sweat gland, mammary gland and salivary glands respectively?
A)
Glandular epithelium
done
clear
B)
Myoepithelium
done
clear
C)
Neurosensory epithelium
done
clear
D)
Holocrine glands
done
clear
View Answer play_arrow
Which of the following property of plasma membrane of erythrocyte helps in hamburger phenomenon-
A)
Chloride shift
done
clear
B)
Haldane effect
done
clear
C)
Bohr effect
done
clear
D)
Donan Phenomenon
done
clear
View Answer play_arrow
The indusium in Pteridium is-
A)
True outside and false inside
done
clear
B)
Always true
done
clear
C)
False outside and true inside
done
clear
D)
Always false
done
clear
View Answer play_arrow
In the anal region, the mucous membrane is thrown into longitudinal folds called as
A)
Crypts of Lieberkuhn
done
clear
B)
Column of Bertini
done
clear
C)
Folds of kerkrings
done
clear
D)
Rectal columns of Morgagni
done
clear
View Answer play_arrow
Which statement is incorrect w.r.t. muscle structure and muscle contraction?
A)
Zwischenschiebe line and Dobie?s line are synonymous to each other
done
clear
B)
Temperature above \[42{}^\circ C\] produces heat rigor due to coagulation of proteins present a in the muscle
done
clear
C)
The resting muscle membrane is permeable to \[\,{{K}^{+}}\] but not to\[N{{a}^{+}}\].
done
clear
D)
In case of rigor mortis the muscle loses its excitability, shortens in length, gradually becomes soft and pH of muscle increases
done
clear
View Answer play_arrow
Metacarpophalangeal joints allow the movement in two directions, it is an example of which type of joint?
A)
Angular/Ellipsoid joint
done
clear
B)
Gliding joint
done
clear
C)
Pivotal joint
done
clear
D)
Ball and socket joint
done
clear
View Answer play_arrow
Which of the following is common to both avian and mammalian development?
A)
Primitive streak
done
clear
B)
Trophoblast
done
clear
C)
Holoblastic cleavage
done
clear
D)
Yolk plug
done
clear
View Answer play_arrow
Human heart is myogenic and it is supplied with sympathetic and vagus nerve branches that are acceleratory and inhibitory in nature respectively. Which one of the following statement is incorrect w.r.t. the heart beat?
A)
In foetal heart the heart rate is \[140-130\] beats per minute
done
clear
B)
Mental status such as depression and grief decrease heart rate
done
clear
C)
An excess of \[C{{a}^{++}}\] ion decreases heart beat rate
done
clear
D)
High levels of \[N{{a}^{+}}\text{ }and\text{ }{{K}^{+}}\] decreases the strength of contraction
done
clear
View Answer play_arrow
Which of the given function is related to parasympathetic nervous system?
a. Constriction of pupil b. Stimulates secretion of gastric juice c. Relaxes urinary bladder d. Inhibits perspiration
A)
a and d only
done
clear
B)
a, c, d
done
clear
C)
a, b, d
done
clear
D)
a, b, c, d
done
clear
View Answer play_arrow
During the accomodation reflex for near objects, which of the following does not happen?
A)
The third cranial nerve of parasympathetic nervous system causes contraction of the ciliary muscles
done
clear
B)
There is slackening of suspensory ligaments
done
clear
C)
The radius of curvature increases
done
clear
D)
The lens increases its curvature due to elasticity and its power is enhanced
done
clear
View Answer play_arrow
Which of the following unicellular organism has a macro-nucleus for trophic function and one or more micro-nuclei for reproduction?
A)
Euglena
done
clear
B)
Amoeba
done
clear
C)
Paramecium
done
clear
D)
Typanosoma
done
clear
View Answer play_arrow
A fight or flight response in the body is controlled by the
A)
Somatic nervous system
done
clear
B)
Release of acetylcholine from post-ganglion neuron
done
clear
C)
Parasympathetic division of the nervous system
done
clear
D)
A sympathetic division of the nervous system
done
clear
View Answer play_arrow
Find out the incorrect statement
A)
Darwin perceived an important connection between natural selection and the capacity of organisms to over reproduce
done
clear
B)
The unequal ability of individuals to survive and reproduce is natural selection
done
clear
C)
Evolution can be measured only as changes in variations in a population of same generation
done
clear
D)
Natural selection can lead to the adaptation of organisms to their environment
done
clear
View Answer play_arrow
The disadvantage of parthenogenesis is-
A)
Establishment of polyploid generation
done
clear
B)
Elimination of variety in population
done
clear
C)
Means of reproduction
done
clear
D)
Does not encourage the appearance of new and advantageous combinations of genes
done
clear
View Answer play_arrow
What is the DPD of a cell having \[OP\,\,=\,\,10\] and \[T.P.=4\]:
A)
10
done
clear
B)
4
done
clear
C)
14
done
clear
D)
6
done
clear
View Answer play_arrow
Following are the steps in MOET programmes for herd improvement in which a cow has been administered hormones with FSH like activity
A. Transferred to surrogate mother B. It is either mated with an elite bull or artificially inseminated C. Fertilised eggs at 32 cell stage are recovered non-surgically D. It produces 6-8 eggs instead of one egg which they normally yield per cycle
The correct sequence is
A)
B, D, C, A
done
clear
B)
D, B, C, A
done
clear
C)
A, D, C, B
done
clear
D)
A, B, C, D
done
clear
View Answer play_arrow
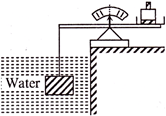 The volume of brick is 2.197 litres. The submerged brick is balanced by a 2.54 kg mass on the beam scale. The weight of the brick is-
The volume of brick is 2.197 litres. The submerged brick is balanced by a 2.54 kg mass on the beam scale. The weight of the brick is- 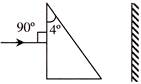
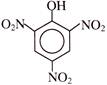
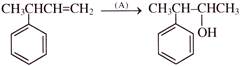 Reagent [A] can be-
Reagent [A] can be- 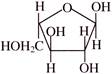
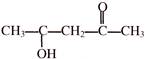
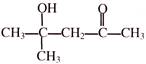
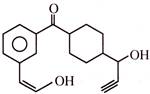 Which of the following reagents will not react with above compound?
Which of the following reagents will not react with above compound?