The dimensions of \[{{({{\mu }_{0}}{{\varepsilon }_{0}})}^{-1/2}}\]are
A)
\[[{{L}^{-1}}T]\]
done
clear
B)
\[[L{{T}^{-1}}]\]
done
clear
C)
\[[{{L}^{-1/2}}{{T}^{1/2}}]\]
done
clear
D)
\[[{{L}^{1/2}}{{T}^{-1/2}}]\]
done
clear
View Answer play_arrow
A particle starts its motion from rest under the action of a constant force. If the distance covered in first 10 s is \[{{s}_{1}}\]and that covered in the first 20 s is \[{{s}_{2}},\]then
A)
\[{{s}_{2}}=2{{s}_{1}}\]
done
clear
B)
\[{{s}_{2}}=3{{s}_{1}}\]
done
clear
C)
\[{{s}_{2}}=4{{s}_{1}}\]
done
clear
D)
\[{{s}_{2}}={{s}_{1}}\]
done
clear
View Answer play_arrow
A particle covers half of its total distance with speed \[{{v}_{1}}\]and the rest half distance with speed \[{{v}_{2}}.\]Its average speed during the complete journey is
A)
\[\frac{{{v}_{1}}{{v}_{2}}}{{{v}_{1}}+{{v}_{2}}}\]
done
clear
B)
\[\frac{2{{v}_{1}}{{v}_{2}}}{{{v}_{1}}+{{v}_{2}}}\]
done
clear
C)
\[\frac{2v_{1}^{2}v_{2}^{2}}{v_{1}^{2}+v_{2}^{2}}\]
done
clear
D)
\[\frac{{{v}_{1}}+{{v}_{2}}}{2}\]
done
clear
View Answer play_arrow
\[\vec{A}\]and\[\vec{B}\]are two vectors and\[\theta \]is the angle between them, if\[\left| \vec{A}\times \vec{B} \right|=\sqrt{3}(\vec{A}.\vec{B})\] the value of \[\theta \]is:
A)
\[\text{60 }\!\!{}^\circ\!\!\text{ }\]
done
clear
B)
\[\text{45 }\!\!{}^\circ\!\!\text{ }\]
done
clear
C)
\[30{}^\circ \]
done
clear
D)
\[\text{90 }\!\!{}^\circ\!\!\text{ }\]
done
clear
View Answer play_arrow
The horizontal range and the maximum height of a projectile are equal. The angle of projection of the projectile is
A)
\[\theta ={{45}^{o}}\]
done
clear
B)
\[\theta ={{\tan }^{-1}}4\left( \frac{1}{4} \right)\]
done
clear
C)
\[\theta ={{\tan }^{-1}}(4)\]
done
clear
D)
\[\theta ={{\tan }^{-1}}(2)\]
done
clear
View Answer play_arrow
If vectors \[\vec{A}=\cos \omega \,\hat{i}+\sin \omega t\,\hat{j}\]and \[\vec{B}=\cos \frac{\omega t}{2}\hat{i}+\sin \frac{\omega t}{2}\hat{j}\] are functions of time, then the value of t at which they are orthogonal to each other is:
A)
\[t=0\]
done
clear
B)
\[t=\frac{\pi }{4\omega }\]
done
clear
C)
\[t=\frac{\pi }{2\omega }\]
done
clear
D)
\[t=\frac{\pi }{\omega }\]
done
clear
View Answer play_arrow
Sand is being dropped on a conveyor belt at the rate of M kg/s. The force necessary to keep the belt moving with a constant velocity of v m/s will be
A)
Mv newton
done
clear
B)
2 Mv newton
done
clear
C)
\[\frac{Mv}{2}\]newton
done
clear
D)
zero
done
clear
View Answer play_arrow
A conveyor belt is moving at a constant speed of 2 m/s. A box is gently dropped on it. The co- efficient of friction between them is\[\mu =0.5.\]The distance that the box will move relative to belt before coming to rest on it taking \[g=10\,m{{s}^{-2}}\]is:
A)
1.2 m
done
clear
B)
0.6 m
done
clear
C)
zero
done
clear
D)
0.4 m
done
clear
View Answer play_arrow
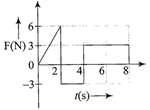
The force F acting on a particle of mass m is indicated by the force-time graph shown below. The change in momentum of the particle over the time interval from zero to 8 s is
A)
24 Ns
done
clear
B)
20 Ns
done
clear
C)
12Ns
done
clear
D)
6Ns
done
clear
View Answer play_arrow
A ball of mass 2 kg and another of mass 4 kg are dropped together from a 60 feet tall building. After a fall of 30 feet each towards earth, their respective kinetic energies will be in the ratio of
A)
\[\sqrt{2}:1\]
done
clear
B)
2 : 1
done
clear
C)
1 : 2
done
clear
D)
\[1:\sqrt{2}\]
done
clear
View Answer play_arrow
Water falls from a height of 60 m at the rate of 15 kg/s to operate a turbine. The losses due to frictional forces are 10% of energy. How much power is generated by the turbine? \[(g=10\,m/{{s}^{2}})\]
A)
8.1 kW
done
clear
B)
10.2 kW
done
clear
C)
12.3 kW
done
clear
D)
7.0 kW
done
clear
View Answer play_arrow
The potential energy of a system increases if work is done
A)
by the system against a conservative force
done
clear
B)
by the system against a nonconservative force
done
clear
C)
upon the system by a conservative force
done
clear
D)
upon the system by a nonconservative force
done
clear
View Answer play_arrow
The upper half of an inclined plane of inclination \[\theta \]is perfectly smooth while lower half is rough. A block starting from rest at the top of the plane will again come to rest at the bottom, if the coefficient of friction between the block and lower half of the plane is given by
A)
\[\mu =\frac{1}{\tan \theta }\]
done
clear
B)
\[\mu =\frac{2}{\tan \theta }\]
done
clear
C)
\[\mu =2\tan \theta \]
done
clear
D)
\[\mu =\tan \theta \]
done
clear
View Answer play_arrow
A ball is thrown vertically downwards from a height of 20 m with an initial velocity\[v_{0}^{2}.\]It collides with the ground, loses 50 percent of its energy in collision and rebounds to the same height. The initial velocity \[{{v}_{0}}\]is: (Take\[g=10\,m{{s}^{-2}}\])
A)
\[~10\,m{{s}^{-1}}\]
done
clear
B)
\[~14\,m{{s}^{-1}}\]
done
clear
C)
\[~20\,m{{s}^{-1}}\]
done
clear
D)
\[~28\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
The moment of inertia of a uniform circular disc of radius R and mass M about an axis touching the disc at its diameter and normal to the disc is
A)
\[M{{R}^{2}}\]
done
clear
B)
\[\frac{2}{5}M{{R}^{2}}\]
done
clear
C)
\[\frac{3}{2}M{{R}^{2}}\]
done
clear
D)
\[\frac{1}{2}M{{R}^{2}}\]
done
clear
View Answer play_arrow
If\[\vec{F}\]is the force acting on a particle having position vector \[\vec{r}\]and \[\vec{\tau }\]be the torque of this force about the origin, then
A)
\[\vec{r}.\vec{\tau }\ne 0\]and\[\vec{F}.\vec{\tau }=0\]
done
clear
B)
\[\vec{r}.\vec{\tau }>0\]and\[\vec{F}.\vec{\tau }<0\]
done
clear
C)
\[\vec{r}.\vec{\tau }=0\]and\[\vec{F}.\vec{\tau }=0\]
done
clear
D)
\[\vec{r}.\vec{\tau }=0\]and\[\vec{F}.\vec{\tau }\ne 0\]
done
clear
View Answer play_arrow
The moment of inertia of a thin uniform rod of mass M and length L about an axis passing through its mid-point and perpendicular to its length is\[{{I}_{0}}.\]Its moment of inertia about an axis passing through one of its ends and perpendicular to its length is
A)
\[{{I}_{0}}+M{{L}^{2}}/4\]
done
clear
B)
\[{{I}_{0}}+2M{{L}^{2}}\]
done
clear
C)
\[{{I}_{0}}+M{{L}^{2}}\]
done
clear
D)
\[{{I}_{0}}+M{{L}^{2}}/2\]
done
clear
View Answer play_arrow
A solid cylinder of mass 3 kg is rolling on a horizontal surface with velocity \[4\,m{{s}^{-1}}.\] It collides with a horizontal spring of force constant \[200\,N{{m}^{-1}}.\]The maximum compression produced in the spring will be
A)
0.2 m
done
clear
B)
0.5 m
done
clear
C)
0.6 m
done
clear
D)
0.7 m
done
clear
View Answer play_arrow
A force \[\vec{F}=\alpha \hat{i}+3\hat{j}+6\hat{k}\]is acting at a point\[\vec{r}=2\hat{i}-6\hat{j}-12\hat{k}.\] The value of a for which angular momentum about origin is conserved is
A)
1
done
clear
B)
-1
done
clear
C)
2
done
clear
D)
zero
done
clear
View Answer play_arrow
The period of a satellite in a circular orbit of radius R is T, the period of another satellite in circular orbit of radius 4R is
A)
4T
done
clear
B)
T/4
done
clear
C)
8T
done
clear
D)
T/8
done
clear
View Answer play_arrow
Imagine a new planet having the same density as that of earth but it is 3 times bigger than the earth in size. If the acceleration due to gravity on the surface of earth is g and that on the surface of the new planet is g', then
A)
\[g'=3g\]
done
clear
B)
\[g'=\frac{g}{9}\]
done
clear
C)
\[g'=9g\]
done
clear
D)
\[g'=27g\]
done
clear
View Answer play_arrow
A particle of mass m is thrown upwards from the surface of the earth, with a velocity u. The mass and the radius of the earth are, respectively, M and R. G is gravitational constant and g is acceleration due to gravity on the surface of the earth. The minimum value of u so that the particle does not return back to earth is
A)
\[\sqrt{\frac{2GM}{R}}\]
done
clear
B)
\[\sqrt{\frac{2GM}{{{R}^{2}}}}\]
done
clear
C)
\[\sqrt{2g{{R}^{2}}}\]
done
clear
D)
\[\sqrt{\frac{4GM}{{{R}^{2}}}}\]
done
clear
View Answer play_arrow
Infinite number of bodies, each of mass 2 kg, are situated on x-axis at distance 1 m, 2 m, 4 m, 8 m, ..... respectively, from the origin. The resulting gravitational potential due to this system at the origin will be
A)
\[-G\]
done
clear
B)
\[-\frac{8}{3}G\]
done
clear
C)
\[-\frac{4}{3}G\]
done
clear
D)
\[-4G\]
done
clear
View Answer play_arrow
A remote-sensing satellite of earth revolves in a circular orbit at a height of \[0.25\times {{10}^{6}}\,m\]above the surface of earth. If earth's radius is \[6.38\times {{10}^{6}}\,m\]and \[g=9.8\,m{{s}^{-2}},\]then the orbital speed of the satellite is
A)
\[6.67\,km\,{{s}^{-1}}\]
done
clear
B)
\[7.76\,km\,{{s}^{-1}}\]
done
clear
C)
\[8.56\,km\,{{s}^{-1}}\]
done
clear
D)
\[9.13\,km\,{{s}^{-1}}\]
done
clear
View Answer play_arrow
Triple point of water is
A)
\[{{273.16}^{o}}F\]
done
clear
B)
\[273.16\,K\]
done
clear
C)
\[273.16{{\,}^{o}}C\]
done
clear
D)
\[273.16\,R\]
done
clear
View Answer play_arrow
If \[{{\lambda }_{m}}\]denotes the wavelength at which the radia-tive emission from a black body at a temperature is maximum, then
A)
\[{{\lambda }_{m}}\propto {{T}^{4}}\]
done
clear
B)
\[{{\lambda }_{m}}\]is independent
done
clear
C)
\[{{\lambda }_{m}}\propto T\]
done
clear
D)
\[{{\lambda }_{m}}\propto {{T}^{-1}}\]
done
clear
View Answer play_arrow
An electric kettle takes 4 A current at 220 V. How much time will it take to boil 1 kg of water from temperature\[\text{20 }\!\!{}^\circ\!\!\text{ C}\]? The temperature of boiling water is\[\text{100 }\!\!{}^\circ\!\!\text{ C}\].
A)
6.3 min
done
clear
B)
8.4 min
done
clear
C)
12.6 min
done
clear
D)
4.2 min
done
clear
View Answer play_arrow
The molar specific heats of an ideal gas at constant pressure and volume are denoted by\[{{C}_{p}}\] and \[{{C}_{\upsilon }}\]respectively. If\[\gamma =\frac{{{C}_{p}}}{{{C}_{\upsilon }}}\]and R is the universal gas constant, then \[{{C}_{\upsilon }}\]is equal to:
A)
\[\frac{1+\gamma }{1-\gamma }\]
done
clear
B)
\[\frac{R}{(\gamma -1)}\]
done
clear
C)
\[\frac{(\gamma -1)}{R}\]
done
clear
D)
\[\gamma R\]
done
clear
View Answer play_arrow
Which of the following processes is reversible?
A)
Transfer of heat by radiation
done
clear
B)
Electrical heating of a nichrome wire
done
clear
C)
Transfer of heat by conduction
done
clear
D)
Isothermal compression
done
clear
View Answer play_arrow
In thermodynamic processes which of the following statements is not true?
A)
In an adiabatic process the system is insulated from the surroundings
done
clear
B)
In an isochoric process pressure remains constant
done
clear
C)
In an isothermal process the temperature remains constant
done
clear
D)
In an adiabatic process\[p{{V}^{\gamma }}=\text{constant}\]
done
clear
View Answer play_arrow
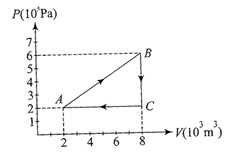
A gas is taken through the cycled \[A\to B\to C\to A\] , as shown. What is the net work done by the gas?
A)
2000 J
done
clear
B)
1000 J
done
clear
C)
Zero
done
clear
D)
-2000 J
done
clear
View Answer play_arrow
One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure
The change in internal energy of the gas during the transition is
A)
20 kJ
done
clear
B)
-20 kJ
done
clear
C)
20 J
done
clear
D)
-12 kJ
done
clear
View Answer play_arrow
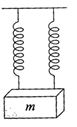
A mass m is suspended separately by two different springs of spring constant \[{{K}_{1}}\]and\[{{K}_{2}}\]gives the time period\[{{t}_{1}}\]and\[{{t}_{2}}\]respectively. If the same mass w is connected by both springs as shown in the figure, then time period t is given by the relation
A)
\[t={{t}_{1}}+{{t}_{2}}\]
done
clear
B)
\[t=\frac{{{t}_{1}}{{t}_{2}}}{{{t}_{1}}+{{t}_{2}}}\]
done
clear
C)
\[{{t}^{2}}=t_{1}^{2}+t_{2}^{2}\]
done
clear
D)
\[{{t}^{-2}}=t_{1}^{-2}+t_{2}^{-2}\]
done
clear
View Answer play_arrow
The resultant of two rectangular simple harmonic motions of the same frequency and unequal amplitudes but differing in phase by nil is
A)
Simple harmonic
done
clear
B)
Circular
done
clear
C)
Elliptical
done
clear
D)
Parabolic
done
clear
View Answer play_arrow
A simple pendulum performs simple harmonic motion about \[x=0\]with an amplitude a and time period T. The speed of the pendulum at \[x=a/2\]will be
A)
\[\frac{\pi a\sqrt{3}}{2T}\]
done
clear
B)
\[\frac{\pi a}{T}\]
done
clear
C)
\[\frac{3{{\pi }^{2}}a}{T}\]
done
clear
D)
\[\frac{\pi a\sqrt{3}}{T}\]
done
clear
View Answer play_arrow
When two displacements represented by \[{{y}_{1}}=a\sin (\omega t)\]and\[{{y}_{2}}=b\cos (\omega t)\]are superimposed the motion is
A)
Not a simple harmonic
done
clear
B)
Simple harmonic with amplitude \[\frac{a}{b}\]
done
clear
C)
Simple harmonic with amplitude\[\sqrt{{{a}^{2}}+{{b}^{2}}}\]
done
clear
D)
Simple harmonic with amplitude \[\frac{(a+b)}{2}\]
done
clear
View Answer play_arrow
A car is moving towards a high cliff. The car driver sounds a horn of frequency\[f\]The reflected sound heard by the driver has a frequency\[2f.\] If. If r be the velocity of sound, then the velocity of the car, in the same velocity units, will be:
A)
\[\frac{v}{\sqrt{2}}\]
done
clear
B)
\[\frac{V}{3}\]
done
clear
C)
\[\frac{V}{4}\]
done
clear
D)
\[\frac{V}{2}\]
done
clear
View Answer play_arrow
Each of the two strings of length 51.6 cm and 49.1 cm are tensioned separately by 20 N force. Mass per unit length of both the strings is the same and equal to \[1\,g{{m}^{-1}}.\]When both the strings vibrate simultaneously the number of beats is
A)
5
done
clear
B)
7
done
clear
C)
8
done
clear
D)
3
done
clear
View Answer play_arrow
Two sources of sound placed close to each other are emitting progressive waves given by \[{{y}_{1}}=4\,\sin 600\,\pi t\]and \[{{y}_{2}}=5\,\sin 608\,\pi t.\]An observer located near these two sources of sound will hear:
A)
4 beats per second with intensity ratio 81:1 between waxing and waning
done
clear
B)
4 beats per second with intensity ratio 25 : 16 between waxing and waning
done
clear
C)
8 beats per second with intensity ratio 25 : 16 between waxing and waning
done
clear
D)
8 beats per second with intensity ratio 81: 1 between waxing and waning
done
clear
View Answer play_arrow
The fundamental frequency of a closed organ pipe of length 20 cm is equal to the second overtone of an organ pipe open at both the ends. The length of organ pipe open at both the ends is
A)
80 cm
done
clear
B)
100 cm
done
clear
C)
120 cm
done
clear
D)
140 cm
done
clear
View Answer play_arrow
A charge Q is enclosed by a Gaussian spherical surface of radius R. If the radius is doubled, then the outward electric flux will
A)
be reduced to half
done
clear
B)
remain the same
done
clear
C)
be doubled
done
clear
D)
increase four times
done
clear
View Answer play_arrow
The electric potential at a point in free space due to a charge Q coulomb is \[Q\times {{10}^{11}}\]volts. The electric field at that point is
A)
\[4\pi {{\varepsilon }_{0}}Q\times {{10}^{22}}\,V/m\]
done
clear
B)
\[12\pi {{\varepsilon }_{0}}Q\times {{10}^{20}}\,V/m\]
done
clear
C)
\[4\pi {{\varepsilon }_{0}}Q\times {{10}^{20}}\,V/m\]
done
clear
D)
\[12\pi {{\varepsilon }_{0}}Q\times {{10}^{22}}\,V/m\]
done
clear
View Answer play_arrow
The potential energy of a particle in a force field is:\[U=\frac{A}{{{r}^{2}}}-\frac{B}{r},\] where A and B are positive constants and r is the distance of particle from the centre of the field. For stable equilibrium the distance of the particle is
A)
B/A
done
clear
B)
B/2A
done
clear
C)
2A/B
done
clear
D)
A/B
done
clear
View Answer play_arrow
If potential (in volts) in a region is expressed as \[V(x,y,z)=6xy-y+2yz,\]the electric field (in N/C) at point (1,1,0) is
A)
\[-\left( 6\hat{i}+9\hat{j}+\hat{k} \right)\]
done
clear
B)
\[-\left( 3\hat{i}+5\hat{j}+3\hat{k} \right)\]
done
clear
C)
\[-\left( 6\hat{i}+5\hat{j}+2\hat{k} \right)\]
done
clear
D)
\[-\left( 2\hat{i}+3\hat{j}+\hat{k} \right)\]
done
clear
View Answer play_arrow
To convert a 800 mV range milli voltmeter of resistance \[40\,\Omega \]into a galvanometer of 100 mA range, the resistance to be connected as shunt is
A)
\[10\,\Omega \]
done
clear
B)
\[20\,\Omega \]
done
clear
C)
\[30\,\Omega \]
done
clear
D)
\[40\,\Omega \]
done
clear
View Answer play_arrow
How many moles of lead (II) chloride will be formed from a reaction between 6.5 g of PbO and 3.2 g of HCl?
A)
0.333
done
clear
B)
0.011
done
clear
C)
0.029
done
clear
D)
0.044
done
clear
View Answer play_arrow
\[6.02\times {{10}^{20}}\]molecules of urea is present in 100 mL of its solution. The concentration of solution is
A)
0.01 M
done
clear
B)
0.001 M
done
clear
C)
0.1M
done
clear
D)
0.02 M
done
clear
View Answer play_arrow
If Avogadro number \[{{N}_{A}},\]is changed from \[6.022\times {{10}^{23}}\,mo{{l}^{-1}}\]to \[6.022\,\times {{10}^{23}}\,mo{{l}^{-1}},\] this would change:
A)
the ratio of chemical species to each other in a balanced equation
done
clear
B)
the ratio of elements to each other in a compound
done
clear
C)
the definition of mass in units of grams
done
clear
D)
the mass of one mole of grams
done
clear
View Answer play_arrow
A 0.66 kg ball is moving with a speed of 100 m/s. The associated wavelength will be
A)
\[6.6\times {{10}^{-32}}\,m\]
done
clear
B)
\[6.6\times {{10}^{-34}}\,m\]
done
clear
C)
\[1.0\times {{10}^{-35}}\,m\]
done
clear
D)
\[1.0\times {{10}^{-32}}\,m\]
done
clear
View Answer play_arrow
Calculate the energy in joule corresponding to light of wavelength 45 nm: (Planck's constant \[h=6.63\times {{10}^{-34}}\,Js;\]speed of light\[c=3\times {{10}^{8}}\,m{{s}^{-1}}\])
A)
\[6.67\times {{10}^{15}}\]
done
clear
B)
\[6.67\times {{10}^{11}}\]
done
clear
C)
\[4.42\times {{10}^{-15}}\]
done
clear
D)
\[4.42\times {{10}^{-18}}\]
done
clear
View Answer play_arrow
In which of the following bond angle is maximum?
A)
\[N{{H}_{3}}\]
done
clear
B)
\[NH_{4}^{+}\]
done
clear
C)
\[PC{{l}_{5}}\]
done
clear
D)
\[SC{{l}_{2}}\]
done
clear
View Answer play_arrow
In an octahedral structure, the pair of d orbitals involved in \[{{d}^{2}}s{{p}^{3}}\]hybridization is
A)
\[{{d}_{{{x}^{2-}}{{y}^{2}}}},{{d}_{{{z}^{2}}}}\]
done
clear
B)
\[{{d}_{xz}},{{d}_{{{x}^{2}}-{{y}^{2}}}}\]
done
clear
C)
\[{{d}_{{{z}^{2}}}},{{d}_{xz}}\]
done
clear
D)
\[{{d}_{xy}},{{d}_{yz}}\]
done
clear
View Answer play_arrow
Which is expected to show paramagnetism?
A)
\[Cl{{O}_{2}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[Si{{O}_{2}}\]
done
clear
View Answer play_arrow
The number of unpaired electrons in a para- magnetic diatomic molecule of an element with atomic number 16 is
A)
4
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
The correct order of decreasing second ionisation enthalpy of Ti (22), V (23), Cr (24) and Mn (25)is
A)
V > Mn > Cr > Ti
done
clear
B)
Mn > Cr > Ti > V
done
clear
C)
Ti > V > Cr > Mn
done
clear
D)
Cr > Mn > V > Ti
done
clear
View Answer play_arrow
In which one of the following species, the central atom has the type of hybridization which is not the same as that present in other three?
A)
\[S{{F}_{4}}\]
done
clear
B)
\[I_{3}^{-}\]
done
clear
C)
\[SbCl_{5}^{2-}\]
done
clear
D)
\[PC{{l}_{5}}\]
done
clear
View Answer play_arrow
Some of the properties of the two species, \[NO_{3}^{-}\]and\[{{H}_{3}}{{O}^{+}}\]are described below. Which one of them is correct?
A)
Dissimilar in hybridization for the central atom with different structure
done
clear
B)
Isostructural with the same hybridization for the central atom
done
clear
C)
Isostructural with different hybridization for the central atom
done
clear
D)
Similar in hybridization for the central atom with different structure
done
clear
View Answer play_arrow
Which of the following species contains three bond pairs and one lone pan- around the central atom?
A)
\[NH_{2}^{-}\]
done
clear
B)
\[PC{{l}_{3}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[B{{F}_{3}}\]
done
clear
View Answer play_arrow
Which of the following is paramagnetic?
A)
\[O_{2}^{-}\]
done
clear
B)
\[C{{N}^{-}}\]
done
clear
C)
\[N{{O}^{+}}\]
done
clear
D)
CO
done
clear
View Answer play_arrow
Which of the following species contains equal number of\[\sigma -\]and\[\pi -\]bonds?
A)
\[{{(CN)}_{2}}\]
done
clear
B)
\[C{{H}_{2}}{{(CN)}_{2}}\]
done
clear
C)
\[HCO_{3}^{-}\]
done
clear
D)
\[Xe{{O}_{4}}\]
done
clear
View Answer play_arrow
The pressure exerted by 6.0 g of methane gas ma \[0.03\,{{m}^{3}}\]vessel at \[129{}^\circ C\] is: (Atomic masses of C \[=12.01,\,H=1.01\]and\[R=8.314\,J{{K}^{-1}}mo{{l}^{-1}}\]
A)
215216 Pa
done
clear
B)
13409 Pa
done
clear
C)
41648 Pa
done
clear
D)
31684 Pa
done
clear
View Answer play_arrow
50 mL of each gas A and of gas B takes 150 and 200 seconds respectively for effusing through a pin hole under the similar condition. It molecular mass of gas B is 36, then the molecular mass of gas A will be
A)
96
done
clear
B)
128
done
clear
C)
32
done
clear
D)
64
done
clear
View Answer play_arrow
The unit of entropy is
A)
\[J{{K}^{-1}}\,mo{{l}^{-1}}\]
done
clear
B)
\[k{{J}^{-1}}mo{{l}^{-1}}\]
done
clear
C)
\[kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[{{J}^{-1}}{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
For the reaction, \[{{C}_{3}}{{H}_{8}}(g)+5{{O}_{2}}\xrightarrow{{}}3C{{O}_{2}}(g)+4{{H}_{2}}O(l)\] at constant temperature, \[\Delta H -\Delta U\] is
A)
\[+RT\]
done
clear
B)
\[-3RT\]
done
clear
C)
\[+3RT\]
done
clear
D)
\[-RT\]
done
clear
View Answer play_arrow
Identify the correct statement for change of Gibbs energy for a system\[(\Delta {{G}_{system}})\] at constant temperature and pressure:
A)
if \[\Delta {{G}_{system}}=0,\] the system is still moving in a particular direction
done
clear
B)
if\[\Delta {{G}_{system}}=-ve,\]not spontaneous
done
clear
C)
if \[\Delta {{G}_{system}}=+ve,\]the Process is spontaneous
done
clear
D)
if \[\Delta {{G}_{system}}=0,\]the system has attained equilibrium
done
clear
View Answer play_arrow
For the gas phase reaction, \[PC{{l}_{5}}(g)\rightleftharpoons PC{{l}_{3}}(g)+C{{l}_{2}}(g)\] Which of the following conditions are correct?
A)
\[\Delta H>0\]and \[\Delta S>0\]
done
clear
B)
\[\Delta H<0\]and\[\Delta S<0\]
done
clear
C)
\[\Delta H>0\]and\[\Delta S<0\]
done
clear
D)
\[\Delta H=0\]and\[\Delta S<0\]
done
clear
View Answer play_arrow
For vaporization of water at 1 atmospheric pressure, the values of\[\Delta H\]and\[\Delta S\]are \[40.63\text{ }kJ\text{ }mo{{l}^{-1}}\]\[\text{ }and\text{ }108.8\text{ }JK-1\text{ }mol-1,\] respectively The temperature when Gibbs energy change \[(\Delta G)\] for this transformation will be zero is
A)
273.4 K
done
clear
B)
393.4 K
done
clear
C)
373.4 K
done
clear
D)
293.4 K
done
clear
View Answer play_arrow
Which of the following statements is correct tor the spontaneous adsorption of a gas?
A)
\[\Delta S\]is negative and therefore\[\Delta H\]should be highly positive.
done
clear
B)
\[\Delta S\] is negative and therefore,\[\Delta H\]should be highly negative.
done
clear
C)
\[\Delta S\]is positive and therefore\[\Delta H\]should be negative.
done
clear
D)
\[\Delta S\]is positive and therefore \[\Delta H\]should also be highly positive.
done
clear
View Answer play_arrow
The dissociation constants for acetic acid and HCN at \[25{}^\circ C\] are\[1.5\times {{10}^{-5}}\]and \[4.5\times {{10}^{-10}},\] respectively. The equilibrium constant for the equilibrium. \[C{{N}^{-}}+C{{H}_{3}}COOH\rightleftharpoons HCN+C{{H}_{3}}CO{{O}^{-}}\]would be
A)
\[3.0\times {{10}^{5}}\]
done
clear
B)
\[3.0\times {{10}^{-5}}\]
done
clear
C)
\[3.0\times {{10}^{-4}}\]
done
clear
D)
\[3.0\times {{10}^{4}}\]
done
clear
View Answer play_arrow
Which of the following statements is correct for a reversible process in a state of equilibrium?
A)
\[\Delta {{G}^{o}}=-2.30\,RT\,\log \,K\]
done
clear
B)
\[\Delta {{G}^{o}}=-2.30\,RT\,\log \,K\]
done
clear
C)
\[\Delta G=-2.30\,RT\,\log \,K\]
done
clear
D)
\[\Delta G=2.30\,RT\,\log \,K\]
done
clear
View Answer play_arrow
Solution of \[0.1\,N\,N{{H}_{4}}OH\]and \[0.1\,N\,N{{H}_{4}}Cl\]has pH 9.25, then find out \[p{{K}_{b}}\]of \[N{{H}_{4}}OH.\]
A)
9.25
done
clear
B)
4.75
done
clear
C)
3.75
done
clear
D)
8.25
done
clear
View Answer play_arrow
Equimotar solutions of the following were prepared in water separately. Which one of the solutions will record the highest pH?
A)
\[BC{{l}_{2}}\]
done
clear
B)
\[MgC{{l}_{2}}\]
done
clear
C)
\[CaC{{l}_{2}}\]
done
clear
D)
\[SrC{{l}_{2}}\]
done
clear
View Answer play_arrow
Which of the following describes correct sequence for decreasing Lewis acid nature?
A)
\[BC{{l}_{3}}>B{{E}_{3}}>BB{{r}_{3}}\]
done
clear
B)
\[BB{{r}_{3}}>BC{{l}_{3}}>B{{F}_{3}}\]
done
clear
C)
\[BB{{r}_{3}}>B{{F}_{3}}>BC{{l}_{3}}\]
done
clear
D)
\[B{{F}_{3}}>BC{{l}_{3}}>BB{{r}_{3}}\]
done
clear
View Answer play_arrow
In which of the following arrangements the given sequence is not strict according to the property indicated against it?
A)
HF < HCl < HBr < HI: increasing acidic strength
done
clear
B)
\[{{H}_{2}}O<{{H}_{2}}S<{{H}_{2}}Se<{{H}_{2}}Te:\] increasing \[p{{K}_{a}}\] values
done
clear
C)
\[N{{H}_{3}}<\text{ }P{{H}_{3}}<\text{ }As{{H}_{3}}<\text{ }Sb{{H}_{3}}:\]increasing acidic character
done
clear
D)
\[C{{O}_{2}}<Si{{O}_{2}}<Sn{{O}_{2}}<Pb{{O}_{2}}:\]increasing oxidizing power.
done
clear
View Answer play_arrow
Which one of the following pairs of solution is not an acidic buffer?
A)
\[{{H}_{2}}C{{O}_{3}}\]and\[~N{{a}_{2}}C{{O}_{3}}\]
done
clear
B)
\[{{H}_{2}}P{{O}_{4}}\]and\[N{{a}_{3}}P{{O}_{4}}\]
done
clear
C)
\[HCl{{O}_{4}}\]and\[NaCl{{O}_{4}}\]
done
clear
D)
\[C{{H}_{3}}COOH\]and\[C{{H}_{3}}COONa\]
done
clear
View Answer play_arrow
Which of the following oxidation state is the most common among the lanthanoides?
A)
4
done
clear
B)
2
done
clear
C)
5
done
clear
D)
3
done
clear
View Answer play_arrow
The reaction of aqueous\[KMn{{O}_{4}}\]with\[{{H}_{2}}{{O}_{2}}\] acidic conditions gives
A)
\[M{{n}^{4+}}\]and\[{{O}_{2}}\]
done
clear
B)
\[M{{n}^{2+}}\]and\[{{O}_{2}}\]
done
clear
C)
\[M{{n}^{2+}}\]and\[{{O}_{3}}\]
done
clear
D)
\[M{{n}^{4+}}\]and\[Mn{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following groups of ions makes the water hard?
A)
Sodium and bicarbonate
done
clear
B)
Magnesium and chloride
done
clear
C)
Potassium and sulphate
done
clear
D)
Ammonium and chloride
done
clear
View Answer play_arrow
Some statements about heavy water are given below:
(i) Heavy water is used as a moderator in nuclear reactors (ii) Heavy water is more associated than ordinary water. (iii) Heavy water is more effective solvent than ordinary water
Which of the above statements are correct?
A)
(i) and (ii)
done
clear
B)
(i), (ii) and (iii)
done
clear
C)
(ii) and (iii)
done
clear
D)
(i) and (iii)
done
clear
View Answer play_arrow
Which of the following compounds has the lowest melting point?
A)
\[Ca{{F}_{2}}\]
done
clear
B)
\[CaC{{l}_{2}}\]
done
clear
C)
\[~CaB{{r}_{2}},\]
done
clear
D)
\[Ca{{I}_{2}}\]
done
clear
View Answer play_arrow
In the hydrocarbon, \[\underset{6}{\mathop{C}}\,{{H}_{3}}-\underset{5}{\mathop{C}}\,H=\underset{4}{\mathop{C}}\,H-\underset{3}{\mathop{C}}\,{{H}_{2}}-\underset{2}{\mathop{C}}\,\equiv \underset{1}{\mathop{C}}\,H\] the state of hybridization of carbons 1, 3 and 5 are in the following sequence
A)
\[sp,s{{p}^{3}},s{{p}^{2}}\]
done
clear
B)
\[sp,s{{p}^{2}},s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}},s{{p}^{2}},sp\]
done
clear
D)
\[s{{p}^{2}},sp,s{{p}^{3}}\]
done
clear
View Answer play_arrow
The structure of isobutyl group in an organic compound is
A)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-\]
done
clear
C)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\]
done
clear
D)
done
clear
View Answer play_arrow
The number of structure isomers possible from the molecular formula\[{{C}_{3}}{{H}_{9}}N\]is:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
The product of the following reaction are \[C{{H}_{3}}C\equiv C\,.C{{H}_{2}}C{{H}_{3}}=\xrightarrow[(ii)Hydrolysis]{(i){{O}_{3}}}?\]
A)
\[C{{H}_{3}}COOH+C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}COOH+C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}CHO+C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
D)
\[C{{H}_{3}}COOH+C{{O}_{2}}\]
done
clear
View Answer play_arrow
In the following reaction, \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Br\] \[\xrightarrow[2.{{H}_{3}}{{O}^{+}}]{1\,Mg.\,Ether}X,\]the product ?X? is
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}C{{H}_{2}}{{C}_{6}}{{H}_{5}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}OC{{H}_{2}}{{C}_{6}}{{H}_{5}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}OH\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\]
done
clear
View Answer play_arrow
If NaCl is dropped with \[{{10}^{-4}}\] mole % of \[SrC{{l}_{2}},\]the concentration of cation vacancies will be
A)
\[6.02\times {{10}^{6}}mo{{l}^{-1}}\]
done
clear
B)
\[6.02\times {{10}^{17}}mo{{l}^{-1}}\]
done
clear
C)
\[6.02\times {{10}^{14}}mo{{l}^{-1}}\]
done
clear
D)
\[6.02\times {{10}^{15}}mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
The number of carbon atoms per unit cell of diamond unit cell is
A)
8
done
clear
B)
6
done
clear
C)
1
done
clear
D)
4
done
clear
View Answer play_arrow
The freezing point depression constant for water is \[-1.86{{\,}^{o}}C\,{{m}^{-1}}.\]If \[5.00\,g\,N{{a}_{2}}S{{O}_{4}}\]is dissolved in \[45.0\,g\,{{H}_{2}}O,\]the freezing point is changed by \[-3.82{{\,}^{o}}C,\] Calculate the van't Hoff factor for \[N{{a}_{2}}S{{O}_{4}}.\]
A)
0.381
done
clear
B)
2.63
done
clear
C)
2.05
done
clear
D)
3.11
done
clear
View Answer play_arrow
A hypothetical electrochemical cell is shown below\[\overset{O-}{\mathop{A}}\,|{{A}^{+}}(xM)||{{B}^{+}}(yM)|\overset{O-}{\mathop{B}}\,\]The emf measured is + 0.20 V. The cell reaction is
A)
The cell reaction cannot be predicted
done
clear
B)
\[A+{{B}^{+}}\xrightarrow{{}}{{A}^{+}}+B\]
done
clear
C)
\[{{A}^{+}}+B\xrightarrow{{}}A+{{B}^{+}}\]
done
clear
D)
\[{{A}^{+}}+{{e}^{-}}\xrightarrow{{}}A;{{B}^{+}}+{{e}^{-}}\xrightarrow{{}}B\]
done
clear
View Answer play_arrow
\[A{{l}_{2}}{{O}_{3}}\]is reduced by electrolysis at low potentials and high currents. If \[4.0\times {{10}^{4}}\]amperes of cur- rent is passed through molten \[A{{l}_{2}}{{O}_{3}}\]for 6 hours, what mass of aluminium is produced? (Assume 100% current efficiency. At mass of Al = 27g \[mo{{l}^{-1}}\])
A)
\[1.3\times {{10}^{4}}g\]
done
clear
B)
\[1.3\times {{10}^{4}}g\]
done
clear
C)
\[8.05\times {{10}^{4}}g\]
done
clear
D)
\[2.4\times {{10}^{5}}\,g\]
done
clear
View Answer play_arrow
Select the correct statement from the following:
A)
Mutations are random and directional
done
clear
B)
Darwinian variations are small and directionless
done
clear
C)
Fitness is the end result of the ability to adapt and gets selected by nature
done
clear
D)
All mammals except whales and camels have seven cervical vertebrae
done
clear
View Answer play_arrow
The hemoglobin human foetus:
A)
has only 2 protein subunits instead of 4
done
clear
B)
has a higher affinity for oxygen than that of an adult
done
clear
C)
has a lower affinity for oxygen than that of an adult
done
clear
D)
its affinity for oxygen is the same as that of an adult
done
clear
View Answer play_arrow
Which one of the following is considered important in the development of seed habit?
A)
Free-living gametophyte
done
clear
B)
Dependent sporophyte
done
clear
C)
Heterospory
done
clear
D)
Haplontic life cycle
done
clear
View Answer play_arrow
What is true for archaebacteria?
A)
All Halophiles
done
clear
B)
All photosynthetic
done
clear
C)
All fossils
done
clear
D)
Oldest living beings
done
clear
View Answer play_arrow
Which of the following secretes toxins during storage conditions of crop plants?
A)
Aspergillus
done
clear
B)
Pemcillium
done
clear
C)
Fusarium
done
clear
D)
Colletotrichum
done
clear
View Answer play_arrow
Some bacteria are able to grow in Streptomycin containing medium due to
A)
Natural selection
done
clear
B)
Induced mutation
done
clear
C)
Reproductive isolation
done
clear
D)
Genetic drift
done
clear
View Answer play_arrow
Which of the following is without exception in angiosperms?
A)
Presence of vessels
done
clear
B)
Double fertilisation
done
clear
C)
Secondary growth
done
clear
D)
Autotrophic nutrition
done
clear
View Answer play_arrow
Moss peat is used as a packing material for sending flowers and live plants to distant places because
A)
It is easily available
done
clear
B)
It is hygroscopic
done
clear
C)
It reduces transpiration
done
clear
D)
It serves as a disinfectant
done
clear
View Answer play_arrow
Which one of the following plants is monoecious?
A)
Papaya
done
clear
B)
Marchantia
done
clear
C)
Pinus
done
clear
D)
Cycas
done
clear
View Answer play_arrow
In which of the animal dimorphic nucleus is found:
A)
Amoeba proteus
done
clear
B)
Trypanosoma gambiense
done
clear
C)
Plasmodhim vivax
done
clear
D)
Paramecium caudatum
done
clear
View Answer play_arrow
From the following statements select the wrong one:
A)
Prawn has two pairs of antennae
done
clear
B)
Millipedes have two pairs of appendages in each segment of the body
done
clear
C)
Animals belonging to phylum Porifera are exclusively marine
done
clear
D)
Nematocysts are characteristic of the phylum cnidaria.
done
clear
View Answer play_arrow
Biradial symmetry and lack of cnidoblasts are the characteristics of:
A)
Starfish and sea anemone
done
clear
B)
Ctenoplana and Beroe
done
clear
C)
Aurelia and Paramecium
done
clear
D)
Hydra and starfish
done
clear
View Answer play_arrow
What is the eye of potato?
A)
Axillary bud
done
clear
B)
Accessory bud
done
clear
C)
Adventitious bud
done
clear
D)
Apical bud
done
clear
View Answer play_arrow
What type of placentation is seen in sweet pea?
A)
Basal
done
clear
B)
Axile
done
clear
C)
Free central
done
clear
D)
Marginal
done
clear
View Answer play_arrow
The floral formula
Is that of:
A)
Tobacco
done
clear
B)
Tulip
done
clear
C)
Soybean
done
clear
D)
Sunn hemp
done
clear
View Answer play_arrow
Vessels are found in:
A)
All angiosperms and some gymnosperm
done
clear
B)
Most of the angiosperm and few gymnosperms
done
clear
C)
All angiosperms, all gymnosperms and some pteridophyta
done
clear
D)
All pteridophyta
done
clear
View Answer play_arrow
For a critical study of secondary growth in plants, which one of the following pairs is suitable?
A)
Wheat and maiden hair fem
done
clear
B)
Sugarcane and sunflower
done
clear
C)
Teak and pine
done
clear
D)
Deodar and fem
done
clear
View Answer play_arrow
Palisade parenchyma is absent in the leaves of
A)
Gram
done
clear
B)
Sorghum
done
clear
C)
Mustard
done
clear
D)
Soybean
done
clear
View Answer play_arrow
Mast cells of connective tissue contain:
A)
Heparin and histamine
done
clear
B)
Heparin and calcitonin
done
clear
C)
Serotonin and melanin
done
clear
D)
Vasopressin and relaxin
done
clear
View Answer play_arrow
Mast cells secrete:
A)
Hippurin
done
clear
B)
Myoglobin
done
clear
C)
Histamine
done
clear
D)
Haemoglobin
done
clear
View Answer play_arrow
Which one of the following is correct pairing of a body part and like kind of muscle tissue that moves it?
A)
Iris - Involuntary smooth muscle
done
clear
B)
Heart wall - Involuntary unstriated muscle
done
clear
C)
Biceps of upper arm - smooth muscle fibres
done
clear
D)
Abdominal wall - smooth muscle
done
clear
View Answer play_arrow
In chloroplasts, chlorophyll is present in the:
A)
Inner membrane
done
clear
B)
Thylakoids
done
clear
C)
Stroma
done
clear
D)
Outer membrane
done
clear
View Answer play_arrow
The two subunits of ribosome remain united at a critical ion level of:
A)
Magnesium
done
clear
B)
Calcium
done
clear
C)
Copper
done
clear
D)
Manganese
done
clear
View Answer play_arrow
Middle lamella is composed mainly of:
A)
Phosphoglycerides
done
clear
B)
Hemicellulose
done
clear
C)
Muramic acid
done
clear
D)
Calcium pectate
done
clear
View Answer play_arrow
Which of the following is a reducing sugar?
A)
Galactose
done
clear
B)
Gluconic acid
done
clear
C)
\[\beta -\]methyl galactoside
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
Carbohydrates, the most abundant biomolecules on earth, are produced by:
A)
Some bacteria, algae and green plant cells
done
clear
B)
All bacteria, fungi and algae
done
clear
C)
Fungi, algae and green plants cells
done
clear
D)
Viruses, fungi and bacteria
done
clear
View Answer play_arrow
A competitive inhibitor of succinic dehydrogenase is:
A)
\[\alpha \]ketoglutarate
done
clear
B)
malate
done
clear
C)
malonate
done
clear
D)
oxaloacetate
done
clear
View Answer play_arrow
At what stage of the cell cycle are histone proteins synthesized in a eukaryotic cell:
A)
During telophase
done
clear
B)
During S-phase
done
clear
C)
During G-2 stage of prophase
done
clear
D)
During entire prophase
done
clear
View Answer play_arrow
During gamete formation, the enzyme recom- binase participates during:
A)
Prophase I
done
clear
B)
Prophase II
done
clear
C)
Metaphase I
done
clear
D)
Anaphase II
done
clear
View Answer play_arrow
A somatic cell that has just completed the S phase of its cell cycle, as compared to gamete of the same species, has:
A)
four times the number of chromosomes and twice the amount of DNA
done
clear
B)
twice the number of chromosomes and twice the amount of DNA
done
clear
C)
same number of chromosomes but twice the amount of DNA
done
clear
D)
twice the number of chromosomes and four times the amount of DNA
done
clear
View Answer play_arrow
Which one of the following mineral elements plays an important role in biological nitrogen fixation?
A)
Copper
done
clear
B)
Manganese
done
clear
C)
Zinc
done
clear
D)
Molybdenum
done
clear
View Answer play_arrow
A plant requires magnesium for:
A)
Cell wall development
done
clear
B)
Holding cells together
done
clear
C)
Protein synthesis
done
clear
D)
Chlorophyll synthesis
done
clear
View Answer play_arrow
Manganese is required in:
A)
Chlorophyll synthesis
done
clear
B)
Nucleic acid synthesis
done
clear
C)
Plant cell wall formation
done
clear
D)
Photolysis of water during photosynthesis
done
clear
View Answer play_arrow
Nitrifying bacteria:
A)
reduce nitrates to free nitrogen
done
clear
B)
oxidize ammonia to nitrates
done
clear
C)
convert free nitrogen to nitrogen to com- pounds
done
clear
D)
convert proteins into ammonia
done
clear
View Answer play_arrow
The first stable product of fixation of atmospheric nitrogen in leguminous plants is:
A)
\[NO_{2}^{-}\]
done
clear
B)
Ammonia
done
clear
C)
\[NO_{3}^{-}\]
done
clear
D)
Glutamate
done
clear
View Answer play_arrow
Root pressure develops due to;
A)
Increase in transpiration
done
clear
B)
Active absorption
done
clear
C)
Low osmotic potential in soil
done
clear
D)
Passive absorption
done
clear
View Answer play_arrow
Which one of the following is wrong in relation to photorespiration?
A)
It occurs in chloroplasts
done
clear
B)
It occurs in daytime only
done
clear
C)
It is a characteristic of \[{{C}_{4}}\] plants
done
clear
D)
It is a characteristic of \[{{C}_{3}}\]plants
done
clear
View Answer play_arrow
In sugarcane plant \[^{14}C{{O}_{2}}\]is fixed in malic acid, in which the enzyme that fixes \[C{{O}_{2}}\]is:
A)
Ribulose biphosphate carboxylase
done
clear
B)
Phosphoenol pyruvic acid carboxylase
done
clear
C)
Ribulose phosphate kinase
done
clear
D)
Fructose phosphatase
done
clear
View Answer play_arrow
In photosystem I, the first electron acceptor is:
A)
Ferredoxin
done
clear
B)
Cytochrome
done
clear
C)
Plastocyanin
done
clear
D)
An iron sulphur protein
done
clear
View Answer play_arrow
The chemiosmotic coupling hypothesis of oxidative phosphorylation proposes that adenosine triphosphate (ATP) is formed because:
A)
A proton gradient forms across the inner membrane
done
clear
B)
There is a change in the permeability of the inner mitochondrial membrane toward adenosine diphosphate (ADP)
done
clear
C)
High energy bonds are formed in mitochondrial proteins
done
clear
D)
ADP is pumped out of the matrix into the intermembrane space
done
clear
View Answer play_arrow
In germinating seeds fatty acids are degraded exclusively in the:
A)
peroxisomes
done
clear
B)
mitochondria
done
clear
C)
proplastids
done
clear
D)
glyoxysomes
done
clear
View Answer play_arrow
The energy-releasing process in which the substrate is oxidized without an external electron acceptor is called:
A)
aerobic respiration
done
clear
B)
glycolysis
done
clear
C)
fermentation
done
clear
D)
photorespiration
done
clear
View Answer play_arrow
Anthesis is a phenomenon which refers to:
A)
Formation of pollen
done
clear
B)
Development of anther
done
clear
C)
Opening of flower bud
done
clear
D)
Reception of pollen by stigma
done
clear
View Answer play_arrow
Which one of the following acids is a derivative of carotenoids?
A)
Abscisic acid
done
clear
B)
Indole butyric acid
done
clear
C)
Indole-3-acetic acid
done
clear
D)
Gibberellic acid
done
clear
View Answer play_arrow
During seed germination its stored food is mobilized by:
A)
Ethylene
done
clear
B)
Cytokinin
done
clear
C)
ABA
done
clear
D)
Gibberellin
done
clear
View Answer play_arrow
The richest sources of vitamin B12 are:
A)
Chocolate and green gram
done
clear
B)
Rice and hen's egg
done
clear
C)
Carrot and chicken's breast
done
clear
D)
Goat's liver and spirulina
done
clear
View Answer play_arrow
When breastfeeding is replaced by less nutritive food low in proteins and calories, the infants be- low the age of one year are likely to suffer from:
A)
Pellagra
done
clear
B)
Marasmus
done
clear
C)
Rickets
done
clear
D)
Kwashiorkor
done
clear
View Answer play_arrow
If for some reason our goblet cells are nonfunctional, this will adversely affect:
A)
production of somatostatin
done
clear
B)
secretion of sebum from the sebaceous glands
done
clear
C)
maturation of sperms
done
clear
D)
smooth movement of food down the intestine
done
clear
View Answer play_arrow
Globulins contained in human blood plasma are primarily involved in;
A)
Clotting of blood
done
clear
B)
Defence mechanisms of body
done
clear
C)
Osmotic balance of body fluids
done
clear
D)
Oxygen transport in the blood
done
clear
View Answer play_arrow
Compared to blood our lymph has;
A)
More RBCs and less WBCs
done
clear
B)
No plasma
done
clear
C)
Plasma without proteins
done
clear
D)
More WBCs and no RBCs
done
clear
View Answer play_arrow
Which one of the following statements is correct regarding blood pressure?
A)
190/110 mm Hg may harm vital organs like brain and kidney
done
clear
B)
130/90 mm Hg is considered high and requires treatment
done
clear
C)
100/55 mm Hg is considered an ideal blood pressure
done
clear
D)
105/50 mm Hg makes one very active
done
clear
View Answer play_arrow
Person with blood group AB is considered as universal recipient because he has:
A)
both A and B antigens on RBC but no antibodies in the plasma
done
clear
B)
both A and B antibodies in the plasma
done
clear
C)
no antigen on RBC and no antibody in the plasma
done
clear
D)
both A and B antigens in the plasma but no antibodies
done
clear
View Answer play_arrow
Which one of the following is correct?
A)
Blood = Plasma + RBC + WBC + Platelets
done
clear
B)
Plasma = Blood - Lymphocytes
done
clear
C)
Serum = Blood + Fibrinogen
done
clear
D)
Lymph = Plasma + RBC + WBC
done
clear
View Answer play_arrow
Which one of the following animals has two separate circulatory pathways?
A)
Shark
done
clear
B)
Frog
done
clear
C)
Lizard
done
clear
D)
Whale
done
clear
View Answer play_arrow
The principal nitrogenous excretory compound in humans is synthesised:
A)
in kidneys but eliminated mostly through liver
done
clear
B)
in kidneys as well as eliminated by kidneys
done
clear
C)
in liver and also eliminated by the same through bile
done
clear
D)
in the liver, but eliminated mostly through kidneys
done
clear
View Answer play_arrow
A fall in glomerular filtration rate (GFR) activates:
A)
Adrenal cortex to release aldosterone
done
clear
B)
Adrenal medulla to release adrenaline
done
clear
C)
Posterior pituitary to release vasopressin
done
clear
D)
Juxtaglomerular cells to release rennin
done
clear
View Answer play_arrow
Which of the following causes an increase in sodium reabsorption in distal convoluted tubule?
A)
Increase in aldosterone levels
done
clear
B)
Decrease in antidiuretic hormone levels
done
clear
C)
Decrease in aldosterone levels
done
clear
D)
Decrease in antidiuretic hormone levels
done
clear
View Answer play_arrow
One of the examples of the action of the autonomous nervous system is:
A)
Knee-jerk response
done
clear
B)
Pupillary reflex
done
clear
C)
Peristalsis of the intestines
done
clear
D)
Swallowing of food
done
clear
View Answer play_arrow
Which one of the following pairs of structures distinguishes a nerve cell from other types of cell?
A)
Nucleus and mitochondria
done
clear
B)
Perikaryon and dendrites
done
clear
C)
Vacuoles and fibres
done
clear
D)
Flagellum and medullary sheath
done
clear
View Answer play_arrow
Bowman's glands are located in the
A)
Olfactory epithelium of our nose
done
clear
B)
Proximal end of uriniferous tubules
done
clear
C)
Anterior pituitary
done
clear
D)
Female reproductive system of cockroach be positive
done
clear
View Answer play_arrow
The nerve centres which control the body temperature and the urge for eating are contained in:
A)
Hypothalamus
done
clear
B)
Pons
done
clear
C)
Cerebellum
done
clear
D)
Thalamus
done
clear
View Answer play_arrow
Which part of the human ear plays no role in hearing as such but is otherwise very much required?
A)
Vestibular apparatus
done
clear
B)
Ear ossicles
done
clear
C)
Eustachian tube
done
clear
D)
Organ of corti
done
clear
View Answer play_arrow
Stimulation of a muscle fiber by a motor neuron occurs at:
A)
the neuromuscular junction
done
clear
B)
the transverse tubules
done
clear
C)
the myofibril
done
clear
D)
the sarcoplasmic reticulum
done
clear
View Answer play_arrow
Sertoli cells are regulated by the pituitary hormone known as
A)
FSH
done
clear
B)
GH
done
clear
C)
Prolactin
done
clear
D)
LH
done
clear
View Answer play_arrow
Chemically hormones are:
A)
Proteins, steroids and biogenic amines
done
clear
B)
Proteins only
done
clear
C)
Steroids only
done
clear
D)
Biogenic amines only
done
clear
View Answer play_arrow
Which one of the following pairs of organs includes only the endocrine glands?
A)
Thymus and testes
done
clear
B)
Adrenal and ovary
done
clear
C)
Parathyroid and adrenal
done
clear
D)
Pancreas and parathyroid
done
clear
View Answer play_arrow
Which one of the following is surrounded by a callose wall?
A)
Pollen grain
done
clear
B)
Microspore mother cell
done
clear
C)
Male gamete
done
clear
D)
Egg
done
clear
View Answer play_arrow
Dry indehiscent single-seeded fruit formed from bicarpellary, syncarpous, inferior ovary is:
A)
berry
done
clear
B)
cremocarp
done
clear
C)
caryopsis
done
clear
D)
cypsela
done
clear
View Answer play_arrow
An example of a seed with endosperm, perisperm and caruncle is:
A)
Castor
done
clear
B)
Cotton
done
clear
C)
Coffee
done
clear
D)
Lily
done
clear
View Answer play_arrow
Apomictic embryos in citrus arise from:
A)
Synergids
done
clear
B)
Maternal sporophytic tissue in ovule
done
clear
C)
Antipodal cells
done
clear
D)
Diploid egg
done
clear
View Answer play_arrow
In which one of the following pollination 1s autogamous?
A)
Cleistogamy
done
clear
B)
Geitonogamy
done
clear
C)
Xenogamy
done
clear
D)
Chasmogamy
done
clear
View Answer play_arrow
An organic substance that can withstand environmental extremes and cannot be degraded by any enzyme is:
A)
Lignin
done
clear
B)
Cellulose
done
clear
C)
Cuticle
done
clear
D)
Sporopollenin
done
clear
View Answer play_arrow
Which one of the following statements about morula in humans is correct?
A)
It has almost equal quantity of cytoplasm as an uncleaved zygote but much more DNA
done
clear
B)
It has far less cytoplasm as well as less DNA than in an uncleaved zygote
done
clear
C)
It has more or less equal quantity of cytoplasm and DNA as in uncleaved zygote
done
clear
D)
It has more cytoplasm and more DNA than an uncleaved zygote
done
clear
View Answer play_arrow
The signals for parturition originate from:
A)
placenta only
done
clear
B)
placenta as well as fully developed foetus
done
clear
C)
oxytocin released from maternal pituitary
done
clear
D)
fully developed foetus only
done
clear
View Answer play_arrow
In vitro fertilization is a technique that involves transfer of which one of the following into the fallopian tube?
A)
Embryo only, up to 8 cell stage
done
clear
B)
Either zygote or early embryo up to 8 cell stage
done
clear
C)
Embryo of 32 cell stage
done
clear
D)
Zygote only
done
clear
View Answer play_arrow
The linkage map of X-chromosome of fruit fly has 66 units, with yellow body gene (y) at one end and bobbed hair gene at the other end. The recombination frequency between these two genes (y and b) should be:
A)
60%
done
clear
B)
>50%
done
clear
C)
<50%
done
clear
D)
100%
done
clear
View Answer play_arrow
Which one of the following traits of garden pea studied by Mendel was a recessive feature?
A)
Axial flower position
done
clear
B)
Green seed colour
done
clear
C)
Green pod colour
done
clear
D)
Round seed shape
done
clear
View Answer play_arrow
A normal woman, whose father was colour blind is married to a normal man. The sons would be:
A)
50% colour blind
done
clear
B)
All normal
done
clear
C)
All colour blind
done
clear
D)
75% colour blind
done
clear
View Answer play_arrow
Lack of independent assortment of two genes A and B in fruit fly Drosophila is due to:
A)
Recombination
done
clear
B)
Linkage
done
clear
C)
Crossing over
done
clear
D)
Repulsion
done
clear
View Answer play_arrow
A woman with normal vision, but whose father was colour bind, marries a colour blind man. Suppose that the fourth child of this couple wasa boy. This boy:
A)
Must have normal colour vision
done
clear
B)
May be colour blind or may be normal vision
done
clear
C)
Will be partially colour blind since he is heterozygous for the colour blind mutant allele
done
clear
D)
Must be colour blind
done
clear
View Answer play_arrow
In order to find out the different types of gametes produced by a pea plants having the genotype AaBb, it should be crossed to a plant with the genotype:
A)
AaBb
done
clear
B)
aabb
done
clear
C)
AABB
done
clear
D)
aaBB
done
clear
View Answer play_arrow
Degeneration of a genetic code is attributed to the:
A)
First member of a codon
done
clear
B)
Second member of a codon
done
clear
C)
Entire codon
done
clear
D)
Third member of a codon
done
clear
View Answer play_arrow
During translation initiation in prokaryotes, a GTP molecule is needed in:
A)
Formation of formyl-met-tRNA
done
clear
B)
Binding of 30S subunit of ribosome with mRNA
done
clear
C)
Association of 30 S-mRNA with formyl-met-tRNA
done
clear
D)
Association of 50 S subunit of ribosome with initiation complex
done
clear
View Answer play_arrow
In transgenics expression of transgene in target tissue is determined by:
A)
Transgene
done
clear
B)
Promoter
done
clear
C)
Reporter
done
clear
D)
Enhancer
done
clear
View Answer play_arrow
Which of the following is correct order of the evolutionary history of man?
A)
Peking man. Homo sapiens, Neanderthal man, Cro-Magnon man
done
clear
B)
Peking man. Neanderthal man, Homo sapiens Cro-Magnon man
done
clear
C)
Peking man, Heidelberg man, Neanderthal man, Cro-Magnon man
done
clear
D)
Peking man. Neanderthal man. Homo sapiens, Heidelberg man
done
clear
View Answer play_arrow
Two different species cannot live for long duration in the same niche or habitat. This law is:
A)
Alien's law
done
clear
B)
Cause's law
done
clear
C)
Competitive exclusion principal
done
clear
D)
Weiseman's theory
done
clear
View Answer play_arrow
Darwin in his 'Natural Selection Theory' did not believe in any role of which one of the following in organic evolution:
A)
Parasites and predators as natural enemies
done
clear
B)
Survival of the fittest
done
clear
C)
Struggle for existence
done
clear
D)
Discontinuous variations
done
clear
View Answer play_arrow
Carcinoma refers to:
A)
Malignant tumours of the connective tissue
done
clear
B)
Malignant tumours of the skin or mucous membrane
done
clear
C)
Malignant tumours of the colon
done
clear
D)
Benign tumours of the connective tissue
done
clear
View Answer play_arrow
Which one of the following depresses brain activity and produces feelings of calmness. relaxation and drowsiness?
A)
Morphine
done
clear
B)
Valium
done
clear
C)
Hashish
done
clear
D)
Amphetamines
done
clear
View Answer play_arrow
A person showing unpredictable moods, out- bursts of emotion, quarrelsome behavior and conflicts with others is suffering from
A)
Schizophrenia
done
clear
B)
Borderline Personality Disorder (BPD)
done
clear
C)
Mood disorders
done
clear
D)
Addictive disorders
done
clear
View Answer play_arrow
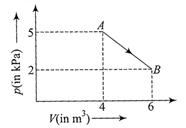 The change in internal energy of the gas during the transition is
The change in internal energy of the gas during the transition is