A bar magnet of magnetic moment \[\overrightarrow{M}\]is placed in the magnetic field B. The torque acting on the magnet is:
A)
\[\overrightarrow{M}\times \overrightarrow{B}\]
done
clear
B)
\[\overrightarrow{M}-\overrightarrow{B}\]
done
clear
C)
\[{\scriptstyle{}^{1}/{}_{2}}\overrightarrow{M}\times \overrightarrow{B}\]
done
clear
D)
\[\overrightarrow{M}+\overrightarrow{B}\]
done
clear
View Answer play_arrow
A capacitor of capacitance \[6\mu f\]is charged up to 100 volt. The energy stored in the capacitor will be:
A)
\[0.6\]Joule
done
clear
B)
\[0.06\]Joule
done
clear
C)
\[0.03\]Joule
done
clear
D)
\[0.3\]Joule
done
clear
View Answer play_arrow
The radius of orbit of a planet in two times that of the earth. The time period of planet is:
A)
\[8.2\,T\]
done
clear
B)
\[2.8\,T\]
done
clear
C)
\[5.6\,T\]
done
clear
D)
\[8.4\,T\]
done
clear
View Answer play_arrow
A body falls from a height \[h=200m\]. The ratio of distance travelled in each \[2\text{ }sec\] during \[t=0\] to \[t=6\] second of the journey is
A)
\[1:4:9\]
done
clear
B)
\[5:3:1\]
done
clear
C)
\[1:3:5\]
done
clear
D)
\[1:2:3\]
done
clear
View Answer play_arrow
To make the frequency double of a spring oscillator we have to:
A)
reduce the mass to one fourth
done
clear
B)
quadruple the mass
done
clear
C)
double the mass
done
clear
D)
half the mass
done
clear
View Answer play_arrow
A particles executing SHM has amplitude \[0.01m\]and frequency\[60\,Hz\]. The maximum acceleration of particle will be:
A)
\[64\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
B)
\[80\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
C)
\[124\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
D)
\[144\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
View Answer play_arrow
When a wave travels in a medium the particles displacement in given by the equation \[y=0.03\,\sin \pi \] \[(2t-0.01x),\] where x and y are in metre and t in sec. The wavelength of the wave will be:
A)
\[200\,m\]
done
clear
B)
\[400\,m\]
done
clear
C)
\[40\,m\]
done
clear
D)
\[10\,m\]
done
clear
View Answer play_arrow
If \[300\,ml\] of a gas at \[{{27}^{o}}C\] is cooled to \[{{7}^{o}}C\] at constant pressure then its final volume is:
A)
\[740\,ml\]
done
clear
B)
\[250\,ml\]
done
clear
C)
\[280\,ml\]
done
clear
D)
\[135\,ml\]
done
clear
View Answer play_arrow
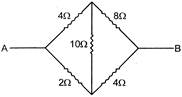
Find the equivalent resistant between the point A and B:
A)
\[2\,\Omega \]
done
clear
B)
\[4\,\Omega \]
done
clear
C)
\[32\,\Omega \]
done
clear
D)
\[16\,\Omega \]
done
clear
View Answer play_arrow
When sound wave travel from air to water which one of the following remains constant?
A)
Time period
done
clear
B)
Frequency
done
clear
C)
Velocity
done
clear
D)
Wavelength
done
clear
View Answer play_arrow
The minimum wavelength of x-rays emitted by x-ray tube is\[0.4125\overset{o}{\mathop{A}}\,\]. The acceleration voltage is:
A)
30 KV
done
clear
B)
50 KV
done
clear
C)
80 KV
done
clear
D)
60 KV
done
clear
View Answer play_arrow
A monoatomie gas supplied the heat Q very slowly keeping the pressure constant. The work done by the gas will:
A)
\[\frac{2}{3}Q\]
done
clear
B)
\[\frac{3}{5}Q\]
done
clear
C)
\[\frac{2}{5}Q\]
done
clear
D)
\[\frac{1}{5}Q\]
done
clear
View Answer play_arrow
The refractive index of the material of the prism and liquid are \[1.56\] and \[1.32\] respectively. What will be the value of \[\theta \] for the following refraction:
A)
\[\sin \theta \ge \frac{13}{11}\]
done
clear
B)
\[\sin \theta \ge \frac{11}{13}\]
done
clear
C)
\[\sin \theta \ge \frac{\sqrt{3}}{2}\]
done
clear
D)
\[\sin \theta \ge \frac{1}{\sqrt{2}}\]
done
clear
View Answer play_arrow
The temperature of the black body increase from T to 2T. The factor by which the rate of emission will increase is:
A)
4
done
clear
B)
2
done
clear
C)
16
done
clear
D)
8
done
clear
View Answer play_arrow
A police jeep is chasing with velocity of 45 km/h a thief in another jeep moving with velocity 153 km/h. Police fires a bullet with muzzle velocity of 180m/s. The velocity with which it will strike the jeep of the thief is:
A)
150m/5
done
clear
B)
27m/s
done
clear
C)
450m/s
done
clear
D)
250m/s
done
clear
View Answer play_arrow
In a sinusoidal wave, the time required maximum displacement to zero displacement is\[0.17\text{ }sec\]. The frequency of the wave is:
A)
\[1.47Hz\]
done
clear
B)
\[2.94Hz\]
done
clear
C)
\[0.73Hz\]
done
clear
D)
\[9.36Hz\]
done
clear
View Answer play_arrow
An LC circuit is in the state of resonance. If \[c=.1\mu f\]and \[L=0.25\,H\]. neglecting ohmic resistance of circuit. What is the frequency of oscillation?
A)
1007Hz
done
clear
B)
100Hz
done
clear
C)
1109Hz
done
clear
D)
600Hz
done
clear
View Answer play_arrow
A person who can see thing most clearly at a distance of 10cm, required spectacles to enable to see clearly thing at a distance of 30cm. What should be the focal length of the spectacles?
A)
15cm (concave)
done
clear
B)
15cm (convex)
done
clear
C)
20cm
done
clear
D)
40cm
done
clear
View Answer play_arrow
The dimensional formula for Young's modulus is:
A)
\[[M{{L}^{-1}}{{T}^{-2}}]\]
done
clear
B)
\[[{{M}^{o}}L{{T}^{-2}}]\]
done
clear
C)
\[[ML{{T}^{-2}}]\]
done
clear
D)
\[[M{{L}^{2}}{{T}^{-2}}]\]
done
clear
View Answer play_arrow
When temperature of an ideal gas is increased from \[{{27}^{o}}C\] to\[{{227}^{o}}C\]. Its rms speed is changed from 400 metre/sec to \[{{V}_{2}}\]. Then \[{{V}_{2}}\] will be:
A)
516 metre/sec
done
clear
B)
450 metre/sec
done
clear
C)
410 metre/sec
done
clear
D)
746 metre/sec
done
clear
View Answer play_arrow
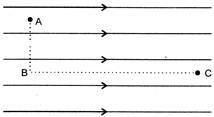
Figure shown three points A,B and C in a region of uniform electric field \[\overrightarrow{E}\]. The line AB is perpendicular and BC is parallel to the field line. Then which of the following hold goods.
A)
\[{{V}_{A}}={{V}_{B}}={{V}_{C}}\]
done
clear
B)
\[{{V}_{A}}={{V}_{B}}>{{V}_{C}}\]
done
clear
C)
\[{{V}_{A}}={{V}_{C}}={{V}_{B}}\]
done
clear
D)
\[{{V}_{A}}>{{V}_{B}}>{{V}_{C}}\]
done
clear
View Answer play_arrow
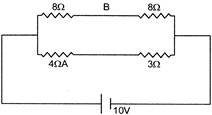
The potential difference between point A and B is:
A)
\[20/7V\]
done
clear
B)
\[40/7V\]
done
clear
C)
\[10/7V\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A short linear object of length b lies along the axis of a concave mirror of focal length f at a distance u from the pole of the mirror, what is the size of image?
A)
\[\left( \frac{f}{u-f} \right)b\]
done
clear
B)
\[{{\left( \frac{f}{u-f} \right)}^{2}}b\]
done
clear
C)
\[{{\left( \frac{f}{u-f} \right)}^{2}}{{b}^{2}}\]
done
clear
D)
\[\left( \frac{f}{u-f} \right){{b}^{2}}\]
done
clear
View Answer play_arrow
A closed organ pipe and an open organ pipe are funned to the same fundamental frequency. What is the ratio of their length?
A)
\[1:2\]
done
clear
B)
\[2:1\]
done
clear
C)
\[3:4\]
done
clear
D)
\[4:3\]
done
clear
View Answer play_arrow
Regarding a semiconductor which one of the following is wrong?
A)
There are no free electron at room temperature.
done
clear
B)
There are no free electron at OK
done
clear
C)
The number of free electron increases with rise of temperature.
done
clear
D)
The charge carriers are electron and holes.
done
clear
View Answer play_arrow
A steel scale measures the length of a copper wires as \[80.0cm,\] when both are at \[{{20}^{o}}C,\] the calibration temperature for the scale. What would the scale read for the length of the wire when both are at\[{{40}^{o}}C\]? Given a for steel \[=11\times {{10}^{-6}}\] per \[^{o}C\] and a for \[Cu=17\times {{10}^{-6}}\]per \[^{o}C\]:
A)
\[90.0096\,cm\]
done
clear
B)
\[80.0272\,cm\]
done
clear
C)
\[1\,cm\]
done
clear
D)
\[25.2\,cm\]
done
clear
View Answer play_arrow
A tank is filled with water up to height H. When a hole is made at a distance h below the level of water, what will be the horizontal range of water jet?
A)
\[2\sqrt{h(H-h)}\]
done
clear
B)
\[4\sqrt{h(H+h)}\]
done
clear
C)
\[4\sqrt{h(H-h)}\]
done
clear
D)
\[2\sqrt{h(H+h)}\]
done
clear
View Answer play_arrow
A raft of wood of mass 120kg floats in water. The weight that can be put on the raft to make Pt just sink, should be (draft\[=600kg/{{m}^{3}}\]):
A)
80 kg
done
clear
B)
50 kg
done
clear
C)
60 kg
done
clear
D)
30 kg
done
clear
View Answer play_arrow
Nuclear fusion is common to the part:
A)
thermonuclear reactor, uranium based nuclear reactor
done
clear
B)
energy production in sun, uranium based nuclear reactor
done
clear
C)
energy production in sun, hydrogen bomb
done
clear
D)
disintegration of heavy nuclear hydrogen bomb
done
clear
View Answer play_arrow
Which of the following statement is true for- n-type semi-conductor?
A)
The donor level lies, closely above. The bottom of the conduction band.
done
clear
B)
The donor level lies closely above the top of the valence band
done
clear
C)
The donor level lies at the half way mark of the forbidden energy gap.
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The length breadth and thickness of a block are given by \[l=12\,cm,\] \[b=cm\] and \[t=2.45cm\].
A)
\[1\times {{10}^{2}}c{{m}^{3}}\]
done
clear
B)
\[2\times {{10}^{2}}c{{m}^{3}}\]
done
clear
C)
\[1.764\times {{10}^{2}}c{{m}^{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Five particles of mass 2kg are attached to the rim of a circular disc of radius \[0.1m\] and negligible mass. Moment of inertia of the system about the axis passing through the centre of the disc and perpendicular to its plane is:
A)
\[3\,kg\,{{m}^{2}}\]
done
clear
B)
\[0.1\,kg\,{{m}^{2}}\]
done
clear
C)
\[4\,kg\,{{m}^{2}}\]
done
clear
D)
\[0.2\text{ }kg\text{ }{{m}^{2}}\]
done
clear
View Answer play_arrow
The radius of the convex surface of plano-convex lens is 20cm and the refractive index of the material of the lens is \[1.5\]. The focal length is:
A)
\[30\,cm\]
done
clear
B)
\[50\,cm\]
done
clear
C)
\[20\,cm\]
done
clear
D)
\[40\,cm\]
done
clear
View Answer play_arrow
An ice-cube of density \[900kg/{{m}^{3}}\] is floating in water of density \[1000g/{{m}^{3}}\]. The percentage of volume of ice-cube out site the water is:
A)
20%
done
clear
B)
35%
done
clear
C)
10%
done
clear
D)
25%
done
clear
View Answer play_arrow
A sphere of diameter \[0.2m\]and mass 2kg is rolling on an inclined plane with velocity \[V=0.5m/s\]. The kinetic energy of the sphere is:
A)
\[0.1\,J\]
done
clear
B)
\[0.3\,J\]
done
clear
C)
\[0.5\,J\]
done
clear
D)
\[0.42\,J\]
done
clear
View Answer play_arrow
An electron moves at right angle to a magnetic - field of \[1.5\times {{10}^{2}}\] tesia with a speed of \[6\times {{10}^{7}}m/s\]. If the specific charge of the electron is \[1.7\times {{10}^{11}}\] coulamb/kg. The radius of the circular path will be:
A)
\[2.9\,cm\]
done
clear
B)
\[3.9\,cm\]
done
clear
C)
\[2.35\,cm\]
done
clear
D)
\[2\,cm\]
done
clear
View Answer play_arrow
If work function of a metal is \[4.2e\,V,\] the cut of wavelength is:
A)
\[8000\overset{o}{\mathop{A}}\,\]
done
clear
B)
\[7000\overset{o}{\mathop{A}}\,\]
done
clear
C)
\[1472\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[2950\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
A particle executing the motion \[x=a\,\,\cos \,\,(\omega t-\theta )\]. The maximum velocity of the particle is:
A)
\[a\,\,\theta \,\,\cos \theta \]
done
clear
B)
\[a\,\omega \]
done
clear
C)
\[a\,\omega \,\sin \theta \]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A particle is executing two different simple harmonic motion mutually perpendicular different amplitudes and having phase difference of \[\frac{\pi }{2}\]. The path of the particle will be:
A)
circular
done
clear
B)
straight line
done
clear
C)
parabolic
done
clear
D)
elliptical
done
clear
View Answer play_arrow
Equation of motion in the same direction are given by: \[{{y}_{1}}=2a\,\sin (\omega t-kx),\,{{y}_{2}}=2a\,\sin (\omega t-kx-\theta )\] The amplitude of the medium particle will be:
A)
\[2a\,\cos \theta \]
done
clear
B)
\[\sqrt{2}a\,\cos \theta \]
done
clear
C)
\[4a\,\cos \frac{\theta }{2}\]
done
clear
D)
\[\sqrt{2}a\,\cos \frac{\theta }{2}\]
done
clear
View Answer play_arrow
A prism of refractive index \[\sqrt{2}\] has a refracting angle of\[{{60}^{o}}\]. At what angle a ray must incident on it so that when suffers a minimum deviation?
A)
\[{{45}^{o}}\]
done
clear
B)
\[{{60}^{o}}\]
done
clear
C)
\[{{90}^{o}}\]
done
clear
D)
\[{{180}^{o}}\]
done
clear
View Answer play_arrow
A cone filled with water is revolved in a vertical circle of radius 4m and the water does not fall down. What must be the maximum period of revolution?
A)
\[2sec\]
done
clear
B)
\[4sec\]
done
clear
C)
\[1sec\]
done
clear
D)
\[6sec\]
done
clear
View Answer play_arrow
A transparent cube of \[15cm\] edge contains a small air bubble. It apparent depth when viewed through one face is \[6cm\] and when viewed through the opposite face is\[4cm\]. Then the refractive index of the material of the cube will be:
A)
\[2.0\]
done
clear
B)
\[2.5\]
done
clear
C)
\[1.6\]
done
clear
D)
\[1.5\]
done
clear
View Answer play_arrow
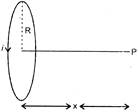
A coil having N turns carries a current as shown in figure. The magnetic field intensity at point P is:
A)
\[\frac{{{\mu }_{0}}Ni{{r}^{2}}}{2{{({{R}^{2}}+{{x}^{2}})}^{3/2}}}\]
done
clear
B)
\[\frac{{{\mu }_{0}}Ni}{2R}\]
done
clear
C)
\[\frac{{{\mu }_{0}}Ni{{R}^{2}}}{{{(R+x)}^{2}}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A capacitor in connected to a cell of emf \[\varepsilon \] having some internal resistance r. The potential difference across the:
A)
Cell is \[<\varepsilon \]
done
clear
B)
Cell is \[\varepsilon \]
done
clear
C)
Capacitor is \[>\varepsilon \]
done
clear
D)
Capacitor is \[<\varepsilon \]
done
clear
View Answer play_arrow
The electrons identified by quantum numbers
I. \[n=4,\,l=1\] II. \[n=4,\,l=0\] III. \[n=3,\,l=2\] IV. \[n=2,\,l=1\]
can be placed in order of increasing energy from the lowest to highest as:
A)
IV < II < III < I
done
clear
B)
II < IV < I < III
done
clear
C)
I < III < II < IV
done
clear
D)
III < I < IV < II
done
clear
View Answer play_arrow
The first emission line in the electronic spectrum of hydrogen in the Balmer series appears at \[c{{m}^{-1}}\]
A)
\[\frac{9R}{400}c{{m}^{-1}}\]
done
clear
B)
\[\frac{7R}{144}c{{m}^{-1}}\]
done
clear
C)
\[\frac{3R}{4}c{{m}^{-1}}\]
done
clear
D)
\[\frac{5R}{36}c{{m}^{-1}}\]
done
clear
View Answer play_arrow
\[KMn{{O}_{4}}\] reacts with oxalic acid according to the equation \[2MnO_{4}^{-}+5{{C}_{2}}O_{4}^{2-}+16{{H}^{+}}\to 2M{{n}^{2+}}+10C{{O}_{2}}+8{{H}_{2}}O\]Here, \[20\text{ }ml\] of \[0.1\,M\,\,KMn{{O}_{4}}\] is equivalent to:
A)
\[20ml\] of \[0.5\,M\] \[{{H}_{2}}{{C}_{2}}{{O}_{4}}\]
done
clear
B)
\[50\,ml\] of \[1\,M\,\,{{H}_{2}}{{C}_{2}}{{O}_{4}}\]
done
clear
C)
\[50\,ml\] of \[0.01\,M\,\,{{H}_{2}}{{C}_{2}}{{O}_{4}}\]
done
clear
D)
\[20\,ml\] of \[0.1\,M\,\,{{H}_{2}}{{C}_{2}}{{O}_{4}}\]
done
clear
View Answer play_arrow
An aqueous solution of \[6.3\text{ }g\] oxalic acid dehydrate is made up to \[250\text{ }ml.\]The volume of \[0.1\text{ }N\] sodium hydroxide required to completely neutralise 10 ml of this solution is:
A)
\[40\text{ }ml\]
done
clear
B)
\[20\text{ }ml\]
done
clear
C)
\[10\,\,ml\]
done
clear
D)
\[4\,\,ml\]
done
clear
View Answer play_arrow
Which of the following salts is most soluble?
A)
\[B{{i}_{2}}{{S}_{3}}({{K}_{SP}}=1\times {{10}^{-17}})\]
done
clear
B)
\[MnS({{K}_{SP}}=7\times {{10}^{-16}})\]
done
clear
C)
\[CuS({{K}_{SP}}=8\times {{10}^{-37}})\]
done
clear
D)
\[A{{g}_{2}}S({{K}_{SP}}=6\times {{10}^{-51}})\]
done
clear
View Answer play_arrow
In the reaction \[2A+B\to {{A}_{2}}B,\] if the concentration of A is doubled and that of B is halved, then the rate of reaction will:
A)
increase by 4 times
done
clear
B)
decrease by 2 times
done
clear
C)
increase by 2 times
done
clear
D)
remains the same
done
clear
View Answer play_arrow
The Van't Hoff factor for \[0.1M\] \[Ba{{(N{{O}_{3}})}_{2}}\] solution is\[2.74\]. The degree of dissociation is:
A)
\[91.3%\]
done
clear
B)
\[87%\]
done
clear
C)
\[100%\]
done
clear
D)
\[74%\]
done
clear
View Answer play_arrow
The occurrence of the reaction is impossible if:
A)
\[\Delta H\] is \[+ve;\] \[\Delta S\] is also \[+ve\]
done
clear
B)
\[\Delta H\] is \[-ve;\] \[\Delta S\] is also \[-ve\]
done
clear
C)
\[\Delta H\] is \[-ve;\] \[\Delta S\] is \[+ve\]
done
clear
D)
\[\Delta H\] is \[+ve;\] \[\Delta S\] is \[-ve\]
done
clear
View Answer play_arrow
\[50\text{ }ml\] of hydrogen diffuses through small hole from a vessel in 20 min time. Time taken for \[40\text{ }ml\] of oxygen to diffuse out under similar conditions will be
A)
12 min
done
clear
B)
32 min
done
clear
C)
8 min
done
clear
D)
64 min
done
clear
View Answer play_arrow
In which of the following, Tyndall effect is not observed?
A)
Smoke
done
clear
B)
Emulsions
done
clear
C)
Sugar solution
done
clear
D)
Gold sol
done
clear
View Answer play_arrow
\[{{N}_{2}}\] and \[{{O}_{2}}\] are converted to \[N_{2}^{+}\] and \[O_{2}^{+}\] respectively. Which of the following is not correct?
A)
In \[N_{2}^{+},\] the \[N-N\] bond weakens
done
clear
B)
In \[O_{2}^{+},\] \[O-O\] bond order increases
done
clear
C)
In \[O_{2}^{+},\] paramagnetism decreases
done
clear
D)
\[N_{2}^{+},\] becomes diamagnetic
done
clear
View Answer play_arrow
The decreasing order of basic character of the amines and ammonia is
A)
\[N{{H}_{3}}>C{{H}_{3}}N{{H}_{2}}>{{C}_{2}}{{H}_{5}}N{{H}_{2}}>{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}N{{H}_{2}}>C{{H}_{3}}N{{H}_{2}}>N{{H}_{3}}>{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}>{{C}_{2}}{{H}_{5}}N{{H}_{2}}>C{{H}_{3}}N{{H}_{2}}>N{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}N{{H}_{2}}>{{C}_{2}}{{H}_{5}}N{{H}_{2}}>{{C}_{6}}{{H}_{6}}N{{H}_{2}}>N{{H}_{3}}\]
done
clear
View Answer play_arrow
Which of the following is not an amino acid?
A)
Glycine
done
clear
B)
Alanine
done
clear
C)
Histidine
done
clear
D)
Benzidine
done
clear
View Answer play_arrow
The boiling point of water decreases at high altitudes because
A)
the atmospheric pressure is low
done
clear
B)
the temperature is low
done
clear
C)
the atmospheric pressure is high
done
clear
D)
the temperature is high
done
clear
View Answer play_arrow
Which of the following statement is not correct?
A)
Phenol is more acidic than acetic acid
done
clear
B)
Ethanol is less acidic than phenol
done
clear
C)
Ethanol has lower boiling point than ethane
done
clear
D)
Ethane is a non-linear molecule
done
clear
View Answer play_arrow
A solid has a structure in which W atoms are located at the corners of a cubic lattice, O atoms at the centre of edges and Na atom at centre of the cube. The formula for the compound is
A)
\[NaW{{O}_{2}}\]
done
clear
B)
\[NaW{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}W{{O}_{3}}\]
done
clear
D)
\[NaW{{O}_{4}}\]
done
clear
View Answer play_arrow
The bond angles of \[N{{H}_{3}},NH_{4}^{+}\] and \[NH_{2}^{-}\] are in the order
A)
\[NH_{2}^{-}>N{{H}_{3}}>NH_{4}^{+}\]
done
clear
B)
\[NH_{4}^{+}>N{{H}_{3}}>NH_{2}^{-}\]
done
clear
C)
\[N{{H}_{3}}>NH_{2}^{-}>NH_{4}^{+}\]
done
clear
D)
\[N{{H}_{3}}>NH_{4}^{+}>NH_{2}^{-}\]
done
clear
View Answer play_arrow
The freezing point of a dilute solution of acetamide in glacial acetic acid is 298 K. This is the value when crystals of
A)
Acetamide first appears
done
clear
B)
Acetic acid first appears
done
clear
C)
Both appear together
done
clear
D)
Ice first appears
done
clear
View Answer play_arrow
A radiactive isotope having a half life of \[2.3\]days was received after \[9.2\]days. It was found that there were 300 mg of the isotope in the container. The initial amount of the isotope was:
A)
1200 mg
done
clear
B)
3600mg
done
clear
C)
4800 mg
done
clear
D)
2400mg
done
clear
View Answer play_arrow
In a \[0.2\]molal aqueous solution of a weaks acid HX, the degree of ionization is\[0.3\]. Taking k. for water as \[1.85,\]the freezing point of the solution will be nearest to
A)
\[-{{4.80}^{o}}C\]
done
clear
B)
\[-{{0.360}^{o}}C\]
done
clear
C)
\[-{{0.260}^{o}}C\]
done
clear
D)
\[-{{2.56}^{o}}C\]
done
clear
View Answer play_arrow
Which one of the following is a compound most likely to have a dipole moment?
A)
\[C{{S}_{2}}\]
done
clear
B)
\[CC{{l}_{2}}{{F}_{2}}\]
done
clear
C)
\[S{{O}_{3}}\]
done
clear
D)
\[SnC{{l}_{2}}\]
done
clear
View Answer play_arrow
\[C{{r}_{2}}O_{7}^{2-}+{{I}^{-}}\xrightarrow{{}}{{I}_{2}}+C{{r}^{3+}}\] \[E_{cell}^{o}=0.79V,\] \[E_{C{{r}_{2}}O_{7}^{2-}}^{o}=1.33V\] \[E_{{{I}_{2}}}^{o}=?\]
A)
\[0.54\,V\]
done
clear
B)
\[-0.54\,V\]
done
clear
C)
\[0.18\,V\]
done
clear
D)
\[-0.18\,V\]
done
clear
View Answer play_arrow
The time required to coat a metal surface of \[800\text{ }c{{m}^{2}}\] with \[5\times {{10}^{-3}}cm\] thick layer of silver (density\[=10.5\text{ }g\text{ }c{{m}^{-3}}\]) with the passage of 3A current through a silver nitrate solution is
A)
\[115\text{ }sec\]
done
clear
B)
\[125\text{ }sec\]
done
clear
C)
\[135\text{ }sec\]
done
clear
D)
\[145\text{ }sec\]
done
clear
View Answer play_arrow
Which of the following compound is optically active?
A)
1-butanol
done
clear
B)
Isopropyi alcohol
done
clear
C)
Acetaldehyde
done
clear
D)
2-butanol
done
clear
View Answer play_arrow
Relation between hydrolysis constant and dissociation constant are given. Which is the correct formula for \[MgC{{l}_{2}}\]?
A)
\[{{K}_{h}}=\frac{{{K}_{w}}}{{{K}_{a}}}\]
done
clear
B)
\[{{K}_{h}}=\frac{{{K}_{w}}}{{{K}_{b}}}\]
done
clear
C)
\[{{K}_{h}}=\frac{{{K}_{w}}}{{{K}_{a}}\times {{K}_{b}}}\]
done
clear
D)
\[{{K}_{w}}=\frac{{{K}_{h}}}{{{K}_{b}}}\]
done
clear
View Answer play_arrow
When \[{{F}_{2}}\] reacts with hot and concentrated alkali then following will be obtained:
I. \[O{{F}_{2}}\] II. \[{{O}_{2}}\] III. \[{{H}_{2}}O\] IV. NaF
A)
I, III and IV
done
clear
B)
II and III
done
clear
C)
II, III and IV
done
clear
D)
all of the above
done
clear
View Answer play_arrow
The process used in conversion of triolein to tristearin is:
A)
hydrolysis
done
clear
B)
hydration
done
clear
C)
hydrogenation
done
clear
D)
dehydrogenation
done
clear
View Answer play_arrow
The product obtained on fusion of \[BaS{{O}_{4}}\] and \[N{{a}_{2}}C{{O}_{3}}\] is
A)
\[BaC{{O}_{3}}\]
done
clear
B)
\[BaO\]
done
clear
C)
\[Ba{{(OH)}_{2}}\]
done
clear
D)
\[BaHS{{O}_{4}}\]
done
clear
View Answer play_arrow
Which of the following statement is wrong?
A)
Salicylic acid is a monobasic acid.
done
clear
B)
Methyl salicylate is an ester
done
clear
C)
Salicylic acid gives violet colour with neutral ferric chloride as well as brisk effervescence with sodium bicarbonate
done
clear
D)
Methyl salicylate does not occur in mineral oils.
done
clear
View Answer play_arrow
An organic compound 'A' having molecular formula \[{{C}_{2}}{{H}_{3}}N\] on reduction gave another compound 'B', upon treatment with nitrous acid 'B', gave ethyl alcohol. On warming with chlroform and alcoholic \[KOH,\]it formed an offensive smelling compounds 'C' The compound 'C'. is
A)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N\overset{\to }{\mathop{=}}\,C\]
done
clear
C)
\[C{{H}_{3}}C\equiv N\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
IUPC name of the compound Day
A)
5-methyl-4-isopropyl-6,6' diethyloctane
done
clear
B)
3,3-dimethyl, 3-ethyl-5-isopropyl octane
done
clear
C)
3,3-diethyl-4-methyl-5(l, 1-dimethyl) octane
done
clear
D)
3,3-diethyl-4-methyl-5(r-methylethyl) octane
done
clear
View Answer play_arrow
Apatite is an ore of:
A)
fluorine
done
clear
B)
chlorine
done
clear
C)
bromine
done
clear
D)
iodine
done
clear
View Answer play_arrow
Which of the following forms vortex ring?
A)
\[{{P}_{2}}{{O}_{5}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[P{{H}_{3}}\]
done
clear
D)
\[{{P}_{4}}{{O}_{10}}\]
done
clear
View Answer play_arrow
Tincture of iodine is
A)
Solution of iodine in \[CC{{l}_{4}}\]
done
clear
B)
Solution of iodine in ethanol
done
clear
C)
\[KI{{O}_{3}}\]
done
clear
D)
\[CH{{I}_{3}}\]
done
clear
View Answer play_arrow
Chloramine-T is used as
A)
antipyretic
done
clear
B)
analgesic
done
clear
C)
antiseptic
done
clear
D)
disinfectant
done
clear
View Answer play_arrow
The decomposition temperature is maximum for
A)
\[MgC{{O}_{3}}\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[SrC{{O}_{3}}\]
done
clear
D)
\[BaC{{O}_{3}}\]
done
clear
View Answer play_arrow
In the dichromate dianion
A)
\[4\text{ }Cr-O\]bonds are equivalent
done
clear
B)
\[\text{6 }Cr-O\] bonds are equivalent
done
clear
C)
all \[Cr-O\] bonds are equivalent
done
clear
D)
all \[Cr-O\] bonds are non-equivalent
done
clear
View Answer play_arrow
Among the following metal carbonyls the \[C-O\] bonds order is lowest in
A)
\[{{[Mn{{(CO)}_{6}}]}^{+}}\]
done
clear
B)
\[[Fe{{(CO)}_{5}}]\]
done
clear
C)
\[[Cr{{(CO)}_{6}}]\]
done
clear
D)
\[{{[V{{(CO)}_{6}}]}^{-}}\]
done
clear
View Answer play_arrow
Which kind of isomerism is shown by \[CO{{(N{{H}_{3}})}_{4}}\] \[B{{r}_{2}}Cl\]?
A)
geometrical and ionisation
done
clear
B)
optical and ionisation
done
clear
C)
geometrical and optical
done
clear
D)
geometrical
done
clear
View Answer play_arrow
A metal nitrate reacts with \[KI\]to give a black precipitate which on additon of excess o \[KI\]convert into orage colour solution. The cation of metal nitrate is
A)
\[H{{g}^{2+}}\]
done
clear
B)
\[B{{i}^{3+}}\]
done
clear
C)
\[S{{n}^{2+}}\]
done
clear
D)
\[P{{b}^{2+}}\]
done
clear
View Answer play_arrow
A gas 'X' is passed through water to form a saturated solution. The aqueous solution on treatment with silver nitrate gives a white precipitate. The saturated aqueous solution also dissolves magnesium ribbon with evolution of a colourless gas Y. Identify 'X' and Y.
A)
\[X=C{{O}_{2}}\text{,}Y=C{{l}_{2}}\]
done
clear
B)
\[X=C{{l}_{2}}\text{,}Y=C{{O}_{2}}\]
done
clear
C)
\[X=C{{l}_{2}},Y={{H}_{2}}\]
done
clear
D)
\[X={{H}_{2}},Y=C{{l}_{2}}\]
done
clear
View Answer play_arrow
Identify the correct order of boiling points of the following compounds \[\underset{I}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH}}\,\] \[\underset{II}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COH}}\,\] \[\underset{III}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COOH}}\,\]
A)
\[I>II>III\]
done
clear
B)
\[III>I>II\]
done
clear
C)
\[I>III>II\]
done
clear
D)
\[III>II>I\]
done
clear
View Answer play_arrow
A solution of (+)-2-chloro-2-phenylethane in toluene racemises slowly in the presence of small amount of \[SbC{{l}_{2}}\] due to the formation of
A)
carbanion
done
clear
B)
carbene
done
clear
C)
free radical
done
clear
D)
carbocation
done
clear
View Answer play_arrow
Which of the following pairs give positive Tollen's test?
A)
glucose, sucrose
done
clear
B)
glucose, fructose
done
clear
C)
fructose, sucrose
done
clear
D)
hexanal, acetophenone
done
clear
View Answer play_arrow
Which of the following is not oxidised by\[{{O}_{3}}\]?
A)
\[KI\]
done
clear
B)
\[FeS{{O}_{4}}\]
done
clear
C)
\[KMn{{O}_{4}}\]
done
clear
D)
\[{{K}_{2}}Mn{{O}_{4}}\]
done
clear
View Answer play_arrow
Biological concept of species is based upon reproductive isolation which is not true for one of the following pair of taxon
A)
Brassica and pisum species
done
clear
B)
Solanum and Nicotiana species
done
clear
C)
Raphanobrassica and Triticale
done
clear
D)
homo sapians and panther
done
clear
View Answer play_arrow
According to Woese, all the six Kingdoms were naturally clustered into three domains on the basis of ____ and it comes under the branch ___.
A)
23s rRNA, Omega taxonomy
done
clear
B)
16s rRNA, Chemo taxonomy
done
clear
C)
Cell wall composition, Cytotaxonomy
done
clear
D)
DNA, Karyotaxonomy
done
clear
View Answer play_arrow
In case of snake bites, the injection given to the patients contain antitoxins against snake venoms. This type of immunization is called
A)
Artificially acquired passive immunity
done
clear
B)
Naturally acquired passive immunity
done
clear
C)
Artificially acquired active immunity
done
clear
D)
Naturally acquired active immunity
done
clear
View Answer play_arrow
Which of the following is a direct method of gene transfer?
A)
Agrobacterium carrying recombinant disarmed Ti plasmid vector
done
clear
B)
Cosmid
done
clear
C)
Electroporation
done
clear
D)
BAG
done
clear
View Answer play_arrow
Self-feeder bacteria using non-sulphur aliphatic organic compounds as \[{{e}^{-}}\]and \[{{H}^{+}}\] donors for \[C{{O}_{2}}\]reduction are
A)
Photoheterotrophs
done
clear
B)
Photolithotrophs
done
clear
C)
Chemolithotrophs
done
clear
D)
Chemo-organoheterotrophs
done
clear
View Answer play_arrow
Photosynthetic protestans with siliceous cell wall show
A)
Haplontic life cycle
done
clear
B)
Auxospore stage
done
clear
C)
Heterokont flagellation
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
Find the correct match
A)
Black bread mould - Coenocytic, trisporic acid and dikaryophase
done
clear
B)
Pink bread mould - Ascospore, perithecium and exogenous asexual spore
done
clear
C)
Rust fungi - Basidiomycetes, dolipore septa and short-lived primary mycelium
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which one of the following is common type of motile asexual spore in chlamydomonas?
A)
Zoospore
done
clear
B)
Aplanospore
done
clear
C)
Hypnospore
done
clear
D)
Palmella stage
done
clear
View Answer play_arrow
Northern blot test is used for
A)
DNA analysis
done
clear
B)
RNA analysis
done
clear
C)
Analysis of proteins
done
clear
D)
Enzyme analysis
done
clear
View Answer play_arrow
The genetically engineered 'golden rice' rich in
A)
P - carotene only
done
clear
B)
P-carotene and iron
done
clear
C)
Iron only
done
clear
D)
Folic acid and iron
done
clear
View Answer play_arrow
Type of stele represented by walking fern is
A)
Dictyostele
done
clear
B)
Solenostele
done
clear
C)
Plectostele
done
clear
D)
Atactostele
done
clear
View Answer play_arrow
Monocot stem is anatomically different from dicot stem in
A)
Having homoxylous condition
done
clear
B)
Presence of hypodermis
done
clear
C)
Absence of pith
done
clear
D)
Absence of ground tissue
done
clear
View Answer play_arrow
Double membrane system is absent in
A)
Chlorella and chromatium
done
clear
B)
Chlorobium and Anabaena
done
clear
C)
Chondrus and frullaria
done
clear
D)
Ricciocarpous and Sphaerocarpous
done
clear
View Answer play_arrow
Tertiary lysosomes are
A)
Small vesicle - like with inactive enzymes
done
clear
B)
Heterophagosomes
done
clear
C)
Residual bodies
done
clear
D)
Autophagic vacuoles
done
clear
View Answer play_arrow
Cell cycle is regulated by MPF during transition of cell from
A)
\[{{G}_{1}}\to S\]
done
clear
B)
\[S\to {{G}_{2}}\]
done
clear
C)
\[{{G}_{2}}\to M\]
done
clear
D)
Prophase \[\to \]Metaphase
done
clear
View Answer play_arrow
OEC enzyme is associated with non-cyclic photo phosphorylation as donor of electrons to
A)
\[{{P}^{+}}_{680}\]
done
clear
B)
\[{{P}_{700}}\]
done
clear
C)
\[{{P}_{680}}\]
done
clear
D)
\[NADP{{H}^{+}}+H\]
done
clear
View Answer play_arrow
The common type of pollen tetrad in major group of flowering plants is
A)
Linear tetrad
done
clear
B)
Isobilateral tetrad
done
clear
C)
Formed after simultaneous cytokinesis
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
Ovule with microphyle end which lies close to hilum in
A)
Campylotropous
done
clear
B)
Antropous
done
clear
C)
Amphitropous
done
clear
D)
Hemianatropous
done
clear
View Answer play_arrow
Autogamy is prevented and allogamy is promoted injasminumby
A)
Prepotency
done
clear
B)
Dicliny
done
clear
C)
Heterostyly
done
clear
D)
Herkogamy
done
clear
View Answer play_arrow
Which one of the following dwarf rice variety was developed by conventional breeding techniques
A)
Atomita-2
done
clear
B)
Jagannath
done
clear
C)
IR-24
done
clear
D)
Reimei
done
clear
View Answer play_arrow
Improved variety of sugarcane with short duration in life-cycle developed by
A)
Somaclonal Variations
done
clear
B)
Embryo Culture
done
clear
C)
Haploid Culture
done
clear
D)
Mutation
done
clear
View Answer play_arrow
Select the gene of tliJ following disorders which can be expressed in hemizygous condition in organisms
A)
Myotonic dystrophy
done
clear
B)
Muscular dystrophy
done
clear
C)
Cystic fibrosis
done
clear
D)
TSD
done
clear
View Answer play_arrow
Which one of the following genetic combination will produce off springs with one phenotype and four genotypes?
A)
\[AaBbcc\times aabbcc~~\]
done
clear
B)
\[AABBCC\times AaBbCc\]
done
clear
C)
\[Aa\text{ }BbCC\times aaBbCc\]
done
clear
D)
\[AA\text{ }Bb\text{ }Cc\times aa\text{ }BB\text{ }CC\]
done
clear
View Answer play_arrow
RNA polymerase type enzyme is involved in autocatalytic behaviour of DNA in which one of the following steps?
A)
Removal of super coiling of DNA
done
clear
B)
Initiation process
done
clear
C)
Chain elongation and proofreading
done
clear
D)
Unwinding of helix.
done
clear
View Answer play_arrow
Select incorrect with respect to lac operon
A)
Inhibitory gene (i) is constitutive gene
done
clear
B)
RNA polymerase is normally in blocked state
done
clear
C)
Controls anabolic pathway
done
clear
D)
Shows glucose inhibition
done
clear
View Answer play_arrow
Annually, plants fix \[75\times {{10}^{12}}\text{ }kg\]of carbon through photo synthesis and produce about 170 billion tonnes of dry mater in the form of
A)
GPP
done
clear
B)
NPP
done
clear
C)
NPE
done
clear
D)
Community productivity
done
clear
View Answer play_arrow
Find the group of secondary pollutants
A)
\[{{O}_{3}},\] PAN and \[S{{O}_{2}}\]
done
clear
B)
\[{{O}_{3}}\]and PAN
done
clear
C)
\[CO\]and NO
done
clear
D)
SPM, Ketone and VOCs
done
clear
View Answer play_arrow
During the life cycle, Fasciola hepatica (liver fluke) infects its intermediate host and primary host at following larval stages, respectively
A)
Redia and Miracidium
done
clear
B)
Cercaria and Redia
done
clear
C)
Metacercaria and Cercaria
done
clear
D)
Miracidium and Metacercariacaria
done
clear
View Answer play_arrow
Which of the following incorrect match about the pathogen and disease caused?
A)
Try panosoma cruzi - Chagas disease
done
clear
B)
Leishmania donovani - Kala azar
done
clear
C)
Entamoeba histolytica - Amoebiasis
done
clear
D)
Aedes aegypti - Dengue fever
done
clear
View Answer play_arrow
Which of the following is not a matching pair of the kind of excretory organ and an animal?
A)
Organ of Bojanus - Pila
done
clear
B)
Protonephridia - Amphioxus
done
clear
C)
Green glands - Prawn
done
clear
D)
Flame cells - Ascaris
done
clear
View Answer play_arrow
Given below are four matching of an animal and its kind of respiratory organ
A. Scorpion book lungs B. Nereis parapodia C. Herdmania gills D. Pila pulmonary sac
The correct matching are
A)
A and B
done
clear
B)
A, B and C
done
clear
C)
A, B and D
done
clear
D)
A, B, C and D
done
clear
View Answer play_arrow
Which of the following is infective stage of Entamoeba histolytica?
A)
Magnaform
done
clear
B)
Minutaform
done
clear
C)
Mature quabrinucteated cyst
done
clear
D)
Trophozoite
done
clear
View Answer play_arrow
Which type of joint is present between the upper ends of the forearm bones?
A)
Ball and socket joint
done
clear
B)
Hinge joint
done
clear
C)
Pivot joint
done
clear
D)
Ellipsoid joint
done
clear
View Answer play_arrow
Which of the following hormones would be released in response to increase in blood flow to the atria of the heart?
A)
Renin
done
clear
B)
Aldosterone
done
clear
C)
Angiotensin-II
done
clear
D)
Atrial Natriuretic factor
done
clear
View Answer play_arrow
Avian pheumonia is also called as
A)
Birds flu
done
clear
B)
New castle's disease
done
clear
C)
Pollorum
done
clear
D)
Coccidiosis
done
clear
View Answer play_arrow
The age sex pyramid of developed countries is
A)
Less steep, representing stable population
done
clear
B)
Steep pyramid, representing growing population
done
clear
C)
Steep pyramid, representing stable population
done
clear
D)
Less steep, representing a rapidly growing population
done
clear
View Answer play_arrow
In which of the following organs the immature lymphocytes differentiate into antigen - sensitive lymphocytes.
A)
Spleen and lymph nodes
done
clear
B)
Peyer's patches of small intestine and appendix
done
clear
C)
Bone marrow and thymus
done
clear
D)
Tonsils and MALT
done
clear
View Answer play_arrow
What would happen to cells lacking telomerase enzyme activity?
A)
They will keep dividing
done
clear
B)
They will develop into normal differentiated cells
done
clear
C)
They will develop into actively dividing cancerous cells
done
clear
D)
The cells will show senescence and eventul death
done
clear
View Answer play_arrow
Aminiotic eggs evolved as a means to
A)
Protect the embryos from predators
done
clear
B)
Protect the embryos from adsorption
done
clear
C)
Prevent the embryo from desiccation
done
clear
D)
Provide space for the storage of food
done
clear
View Answer play_arrow
In which of the following the toxin blocks the release of acetylcholine from presynaptic terminals, and produces paralysis of respiratory muscles and death?
A)
Botulism
done
clear
B)
Myasthenia gravis
done
clear
C)
Curare
done
clear
D)
Tetanus
done
clear
View Answer play_arrow
Lactational amenorrhoea is based on the fact that ovulation and menstruation do not occur during the period of intense lactation following parturition. This is due to high levels of
A)
Prolactin hormone
done
clear
B)
FSH and LH
done
clear
C)
Oxytocin
done
clear
D)
Progesterone
done
clear
View Answer play_arrow
Example of compound tubular gland is
A)
Salivary glands
done
clear
B)
Crypts of Lieberkuhn
done
clear
C)
Brunners glands
done
clear
D)
Pancrease
done
clear
View Answer play_arrow
Maximum amount of carbon dioxide is transported
A)
in dissolved state
done
clear
B)
as carbamino hemoglobin
done
clear
C)
as sodium bicarbonate
done
clear
D)
as potassium bicarbonate
done
clear
View Answer play_arrow
Which of the following epithelium lines the cornea?
A)
Stratified cuboidal epithelium
done
clear
B)
Stratified squamous non - keratinised epithelium
done
clear
C)
Transitional epithelium
done
clear
D)
Stratified squamous keratinized epithelium
done
clear
View Answer play_arrow
Blood cholesterol lowering agent which acts by competitively inhibiting the enzyme responsible for synthesis of cholesterol is produced by
A)
Trichoderma polysporum
done
clear
B)
Mortierella renispora
done
clear
C)
Clostridium butylicum
done
clear
D)
Monascus purpureus
done
clear
View Answer play_arrow
In crassinucellate ovules, the sporogenous cell becomes sub-hypodermal due to
A)
formation of connective tissue
done
clear
B)
divisions in integument epidermis
done
clear
C)
formation of partial cells
done
clear
D)
proliferation of endothelial cells
done
clear
View Answer play_arrow
Ratio of \[C{{O}_{2}}\] evolved is three times more in aerobic than anaerobic respiration in
A)
Warburg effect
done
clear
B)
Crabtree effect
done
clear
C)
Kutusky effect
done
clear
D)
Pasteur effect
done
clear
View Answer play_arrow
Mutation which result in formation of a short protein due to early stoppage of polypeptide chain is
A)
Missense mutation
done
clear
B)
Non-sense mutation
done
clear
C)
Same sense mutation
done
clear
D)
Frame shift mutation
done
clear
View Answer play_arrow
Red algae differ from blue green algae in showing the presence of
A)
phycobilins
done
clear
B)
non flagellate cells
done
clear
C)
single thylakoid per lamella
done
clear
D)
sulphated polysaccharides
done
clear
View Answer play_arrow
Of all the ecosystem services, which of the following accounts for more than 10% of total cost of various ecosystem services?
A)
Recreation and nutrient cycling
done
clear
B)
Climate regulation
done
clear
C)
Soil formation
done
clear
D)
Habitat for wildlife
done
clear
View Answer play_arrow
Fruiting body of ergot disease causing pathogen is
A)
Hysterothecium
done
clear
B)
Cleistothecium
done
clear
C)
Ascostroma
done
clear
D)
Perithecium
done
clear
View Answer play_arrow
Find the incorrect match with respect to disease resistant crops
A)
Pusa Shubhra - Chilly mosaic virus
done
clear
B)
Himgiri - Hill bunt
done
clear
C)
Pusa Snow Ball K-l- Black rot
done
clear
D)
Pusa Komal - Bacterial blight
done
clear
View Answer play_arrow
If a flaccid cell is placed in hypertonic solution its DPD will become
A)
= OP
done
clear
B)
> OP
done
clear
C)
< OP
done
clear
D)
< OP
done
clear
View Answer play_arrow
The element which is not used in catalytic converters for automobiles is
A)
Platinum
done
clear
B)
Palladium
done
clear
C)
Strontium
done
clear
D)
Rhodium
done
clear
View Answer play_arrow
Different forms of shapes of leaves occur at different places on the same plants in its development period in
A)
Sonchus, Eucalyptus
done
clear
B)
Jack fruit, ficus heterophylla
done
clear
C)
Ranunclus aquatilis, sagittaria
done
clear
D)
Mustard, Limnophila heterophylla
done
clear
View Answer play_arrow
Find the incorrect match
A)
Cortical vascular bundles - Nyctanthus
done
clear
B)
Living sclerenchyma fibres - Tamarix
done
clear
C)
Polystelic condition - Primula
done
clear
D)
Medullary bundles ? Casuarina
done
clear
View Answer play_arrow
Blue light induces maximum opening of stomata because it increases the activity of
A)
PEP case
done
clear
B)
RUBISCO
done
clear
C)
Phosphorylase
done
clear
D)
Phosphatase
done
clear
View Answer play_arrow
Codon UGA in mitocondria
A)
Acts as a stop codon
done
clear
B)
Codes for glutamine
done
clear
C)
Codes for tryptophan
done
clear
D)
Codes for arginine
done
clear
View Answer play_arrow
Match the following
Column-I Column-II A. Bryophyllm 1. Tubers B. Potamogeton 2. Rhizome C. Aspidium 3. Vivipary D. Selaginella 4. Turions
A)
A-1, B-4, C-3, D-2
done
clear
B)
A-2, B-4, C-1, D-3
done
clear
C)
A-3, B-4, C-2, D-1
done
clear
D)
A-4, B-1, C-2, D-3
done
clear
View Answer play_arrow
Which of the following is a photolitho -organotrophic diazotroph?
A)
Gloeotheca
done
clear
B)
Nostac
done
clear
C)
Anabaena
done
clear
D)
Rhodopseudomonas
done
clear
View Answer play_arrow
Fluorescence phenomenon is observed when electron returns from
A)
Triplet excited state to ground state
done
clear
B)
Excited second singlet state to ground state
done
clear
C)
Excited first singlet state to ground state
done
clear
D)
Excited second singlet state to triplet excited
done
clear
View Answer play_arrow
Antarctic fishes can survive below
due to the
A)
Internal oxidation of fat
done
clear
B)
Development of extra solutes in cytosol
done
clear
C)
Presence of antifreeze protein in cell organelles
done
clear
D)
Development of ice nucleating proteins in the extra cellular spaces
done
clear
View Answer play_arrow
Higher heterogenity in habitats in a region and greater dissimilarity between communities indicates a higher
A)
local diversity
done
clear
B)
a-diversity
done
clear
C)
P-diversity
done
clear
D)
y-diversity
done
clear
View Answer play_arrow
Eflux of
ions to the subsidiary cells is increased by
A)
Auxin
done
clear
B)
GA
done
clear
C)
ABA
done
clear
D)
CK
done
clear
View Answer play_arrow
Which of the following control the trans boundary movement of hazardous wastes and their dispersal?
A)
IPCC
done
clear
B)
Basel convention
done
clear
C)
Beijing protocol
done
clear
D)
CCC
done
clear
View Answer play_arrow
Equisetum shows ___ sporangium and ____ embryogeny.
A)
Leptosporangiate, Exoscopic
done
clear
B)
Eusporangiate, Endoscopic
done
clear
C)
Leptosporangiate, Endoscopic
done
clear
D)
Eusporangiate, Exoscopic
done
clear
View Answer play_arrow
On the inhibition of succinate dehydrogenase enzyme complex in aerobic respiration in cells of kidney, what will be the number of ATP molecules produced through ETS only by three molecule of 3- phosphoglyceraldehyde?
A)
27
done
clear
B)
37
done
clear
C)
36
done
clear
D)
45
done
clear
View Answer play_arrow
Find the correct match with respect to dispersal mechanism
A)
Ecballium - Tumble weed
done
clear
B)
Argemone - Rolling mechanism
done
clear
C)
Rudlia - Jaculator mechanism
done
clear
D)
Amaranthus - Fountain mechanism
done
clear
View Answer play_arrow
Ethylene does not play a major role in ripening of
A)
Mango
done
clear
B)
Peach
done
clear
C)
Plum
done
clear
D)
Strawberry
done
clear
View Answer play_arrow
The antibiotic which binds to C -terminus of growing peptide chain causes premature termination of polypeptide chain is
A)
Chloramphenicol
done
clear
B)
Rifampicin
done
clear
C)
Neomycin
done
clear
D)
Puromycin
done
clear
View Answer play_arrow
Which of the following is not a reason for resistance shown by endospores?
A)
Thick and impereable spore coat
done
clear
B)
Low water contant
done
clear
C)
Ca-DAPA complex
done
clear
D)
Low metabolic activity
done
clear
View Answer play_arrow
A population of 250 phytoplanktons increased to 400 after two hours. Calculate the percent growth rate in this population
A)
100%
done
clear
B)
30%
done
clear
C)
67%
done
clear
D)
75%
done
clear
View Answer play_arrow
Tyioses and Callose plugs are associated with
A)
Duramen and sieve tube respectively
done
clear
B)
Alburnum and endothecium respectively
done
clear
C)
Heart wood and sapwood respectively
done
clear
D)
Primary xylem and primary phloem respectively
done
clear
View Answer play_arrow
The structure of puccinia which behaves as a female reproductive unit is called
A)
Spermatia
done
clear
B)
Epibasidium
done
clear
C)
Flexuous hyphae
done
clear
D)
Urediopustules
done
clear
View Answer play_arrow
Crossing over frequency decreases with increase in
A)
Exposure to X-rays
done
clear
B)
Distance between genes
done
clear
C)
Distance from centromere
done
clear
D)
Interference
done
clear
View Answer play_arrow
Rootlet of flagellum which joints two adjacent basal bodies is called
A)
Rhizoplast
done
clear
B)
Paradesmos
done
clear
C)
Blepharoplast
done
clear
D)
Flimmer
done
clear
View Answer play_arrow
Vitamin C enriched crop produced by IARI, New Delhi is
A)
Carrot
done
clear
B)
Pumpkin
done
clear
C)
Bitter gaurd
done
clear
D)
Spinach
done
clear
View Answer play_arrow
Most variable phase of cell cycle having monads in dispersed state is
A)
\[{{G}_{1}}\] phase
done
clear
B)
\[{{G}_{2}}\] phase
done
clear
C)
\[{{G}_{0}}\] phase
done
clear
D)
S phase
done
clear
View Answer play_arrow
Coenomegaspore is involved in formation of ______ type of embryo sac
A)
Polygonum
done
clear
B)
Oenothera
done
clear
C)
Endymion
done
clear
D)
Plumbago
done
clear
View Answer play_arrow
Which of the following animal is a deuterostomic
A)
Sea star
done
clear
B)
Sea pen
done
clear
C)
Sea fan
done
clear
D)
Sea mouse
done
clear
View Answer play_arrow
In earthworm insects and snails are placed together, which of the following is applicable to all of them?
A)
Acoelomates
done
clear
B)
Deuterostomes
done
clear
C)
Protostomes
done
clear
D)
Enterocoelic
done
clear
View Answer play_arrow
The bat differs from bird in having
A)
wings
done
clear
B)
four chambered heart
done
clear
C)
feathers
done
clear
D)
sweat glands in skin
done
clear
View Answer play_arrow
In structure the Keratin of mammalian hairs is
A)
a - helical like silk fibrion
done
clear
B)
p - pleated like silk
done
clear
C)
a - helical unlike fibrion
done
clear
D)
p - pleated unlike silk
done
clear
View Answer play_arrow
In cockroach the blood filled coelom is _____.
A)
Absent
done
clear
B)
Haemocoel
done
clear
C)
Filled with parenchyma
done
clear
D)
Well developed
done
clear
View Answer play_arrow
Read the following characters
A. Mostly terrestrial B. Body is covered by dry and cornified skin; epidermal scales or scutes C. Heart is three chambered D. Fertilization is internal
All the given characters are applicable for
A)
Exocoetus
done
clear
B)
Ichthyophis
done
clear
C)
Calotes
done
clear
D)
Columba
done
clear
View Answer play_arrow
In process of moving fluids in urine, which organ is not involved
A)
Collecting duct
done
clear
B)
Renal pelvis
done
clear
C)
Distal convoluted tubles
done
clear
D)
Ureter
done
clear
View Answer play_arrow
The function between Schwann cells is
A)
Synapse
done
clear
B)
Dendrons
done
clear
C)
Nodes of Ranvier
done
clear
D)
Plasmalemma
done
clear
View Answer play_arrow
Poor nutrients in top soil related to
A)
Tropical rain forest
done
clear
B)
Tundra region
done
clear
C)
Coniferous forest
done
clear
D)
Grass land biome
done
clear
View Answer play_arrow
Which type of hepatitis virus has ds DNA?
A)
HAV
done
clear
B)
HBV
done
clear
C)
HCV
done
clear
D)
HDV
done
clear
View Answer play_arrow
The most active ingredient delta-9 THC is present in
A)
Psilocybin
done
clear
B)
Marijuana
done
clear
C)
Morphine
done
clear
D)
Mescaline
done
clear
View Answer play_arrow