A person throws balls into the air one after the other at an interval of one second. The next ball is thrown when the velocity of the ball thrown earlier is zero. To what height the ball rise? Take \[g=10\,m{{s}^{-2}}\]
A)
5 m
done
clear
B)
10 m
done
clear
C)
20 m
done
clear
D)
40 m
done
clear
View Answer play_arrow
Three point charges 1C, 2C and 3C are placed at the corners of an equilateral triangle of side 1m. Calculate the work required to move these charges to the corner of a smaller equilateral triangle of side \[0.5\text{ }m\]:-
A)
\[99\times {{10}^{9}}J\]
done
clear
B)
\[9.9\times {{10}^{5}}J\]
done
clear
C)
\[50\text{ }\times {{10}^{9}}\text{ }J\]
done
clear
D)
\[25\times {{10}^{9}}J\]
done
clear
View Answer play_arrow
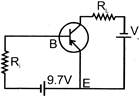
In circuit shown the base current is \[30\mu A\] and voltage drop accross terminal B & E is \[0.7\text{ }V\] then value of \[{{R}_{i}}\] is :-
A)
\[3\,k\Omega \]
done
clear
B)
\[30\,k\Omega \]
done
clear
C)
\[300\,k\Omega \]
done
clear
D)
\[3\times {{10}^{4}}\,k\Omega \]
done
clear
View Answer play_arrow
If the temperature of cold junction is lowered, then the neutral temperature :-
A)
Increases
done
clear
B)
Decreases
done
clear
C)
Approaches inversion temperature
done
clear
D)
Remains same
done
clear
View Answer play_arrow
A thin circular ring of mass M and radius R is rotating about its axis with a constant angular speed co. Two blocks each of mass m are attached gently to opposite ends of a diameter of the ring. The angular speed of the ring will be:-
A)
\[\frac{2M\omega }{M+2m}\]
done
clear
B)
\[\frac{(M-2m)\omega }{M+2m}\]
done
clear
C)
\[\frac{M\omega }{M+2m}\]
done
clear
D)
\[\frac{(M+2m)\omega }{M}\]
done
clear
View Answer play_arrow
Which of the following remains constant for a projectile fired from the earth :-
A)
Kinetic energy
done
clear
B)
Momentum
done
clear
C)
Horizontal component of velocity
done
clear
D)
Vertical component of velocity
done
clear
View Answer play_arrow
A proton moving with a constant velocity passes through a zero gravity region without change in velocity. If E and B represent electric and magnetic fields respectively, the region of zero gravity may not have:-
A)
\[E=0,\,B=0\]
done
clear
B)
\[E=0,\,B\ne 0\]
done
clear
C)
\[E\ne 0,\,B=0\]
done
clear
D)
\[E\ne 0,\,B\ne 0\]
done
clear
View Answer play_arrow
In Boolean algebra, which of the following is not equal to zero :-
A)
\[A.\overline{A}\]
done
clear
B)
\[A.0\]
done
clear
C)
\[\overline{A+A}\]
done
clear
D)
\[\overline{\overline{A}.0}\]
done
clear
View Answer play_arrow
A wheel is rotating at 900 r.p.m. about its axis. When power is cut off, it comes to rest in 1 minute. The angular retardation in \[rad/{{s}^{2}}\]is :-
A)
\[\frac{\pi }{2}\]
done
clear
B)
\[\frac{\pi }{4}\]
done
clear
C)
\[\frac{\pi }{6}\]
done
clear
D)
\[\frac{\pi }{8}\]
done
clear
View Answer play_arrow
A wheel is subjected to uniform angular acceleration about its axis. Initially its angular velocity is zero. In the first two seconds it rotates through angle \[{{\theta }_{1}}\]. In the next two second it rotates through angle \[{{\theta }_{2}}\]. What is the ratio \[\frac{{{\theta }_{2}}}{{{\theta }_{1}}}\] ?
A)
1
done
clear
B)
1
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
A wire of length 'L' is shaped into a circle and then bent about its one of the diameter in such a way that the two semi circles becomes perpendicular to each other. If i current flows through the loop then magnetic moment of system is :-
A)
\[\frac{\sqrt{2}i{{L}^{2}}}{8\pi }\]
done
clear
B)
\[\sqrt{3}\frac{i{{L}^{2}}}{4\pi }\]
done
clear
C)
\[\frac{i{{L}^{2}}}{4\pi }\]
done
clear
D)
\[\frac{i{{L}^{2}}}{2\pi }\]
done
clear
View Answer play_arrow
In the Bohr model of a hydrogen atom, the centripetal force is furnished by the coulomb attraction between the proton and the electron. If \[{{a}_{0}}\] is the radius of the ground state orbit, m is the mass, e is the charge on the electron and \[{{\varepsilon }_{0}}\] is the vacuum permittivity, the speed of the electron is.
A)
\[0\]
done
clear
B)
\[\frac{e}{\sqrt{{{\varepsilon }_{0}}{{a}_{0}}m}}\]
done
clear
C)
\[\frac{e}{\sqrt{4\pi {{\varepsilon }_{0}}{{a}_{0}}m}}\]
done
clear
D)
\[\frac{\sqrt{4\pi {{\varepsilon }_{0}}{{a}_{0}}m}}{e}\]
done
clear
View Answer play_arrow
Dimension of \[\frac{1}{{{\varepsilon }_{0}}{{\mu }_{0}}}\] is :-
A)
\[\frac{{{L}^{2}}}{{{T}^{2}}}\]
done
clear
B)
\[\frac{{{T}^{2}}}{{{L}^{2}}}\]
done
clear
C)
\[\frac{T}{L}\]
done
clear
D)
\[\frac{L}{T}\]
done
clear
View Answer play_arrow
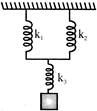
The resultant spring constant of the system of springs shown in fig. is :-
A)
\[\frac{{{k}_{1}}+{{k}_{2}}}{{{k}_{1}}+{{k}_{2}}+{{k}_{3}}}\]
done
clear
B)
\[\frac{(4{{k}_{1}}+2{{k}_{2}}){{k}_{3}}}{{{k}_{1}}+{{k}_{2}}+{{k}_{3}}}\]
done
clear
C)
\[\frac{{{k}_{1}}+{{k}_{2}}}{2({{k}_{1}}+{{k}_{2}}+{{k}_{3}})}\]
done
clear
D)
\[\frac{({{k}_{1}}+{{k}_{2}})({{k}_{3}})}{{{k}_{1}}+{{k}_{2}}+{{k}_{3}}}\]
done
clear
View Answer play_arrow
Two satellites of mass M and 16 M are put in the same orbit around earth. If \[{{T}_{1}}\] and \[{{T}_{2}}\]be their time periods, then \[\frac{{{T}_{1}}}{{{T}_{2}}}\]= ?
A)
4
done
clear
B)
1
done
clear
C)
\[0.5\]
done
clear
D)
\[0.25\]
done
clear
View Answer play_arrow
A magnetising field of \[1600\text{ }A/m\] produces a magnetic flux of \[2.4\times {{10}^{-5}}wb\] in an iron bar of cross sectional area\[0.2\text{ }c{{m}^{2}}\]. The susceptibility of iron bar is :-
A)
298
done
clear
B)
1192
done
clear
C)
596
done
clear
D)
1788
done
clear
View Answer play_arrow
If a proton and antiproton come close to each other and annihilate, how much energy will be released :-
A)
\[1.5\times {{10}^{-10}}J\]
done
clear
B)
\[3\times {{10}^{-10}}J\]
done
clear
C)
\[4.5\times {{10}^{-10}}J\]
done
clear
D)
\[6\times {{10}^{-10}}J\]
done
clear
View Answer play_arrow
A solid cylinder of mass M, radius R is attached to a horizontal mass less spring of spring constant k, so that it can roll without slipping along a horizontal surface. If the cylinder is slightly displaced and released, the time period of the SHM it executes is :-
A)
\[2\pi \sqrt{\frac{M}{k}}\]
done
clear
B)
\[2\pi \sqrt{\frac{2M}{k}}\]
done
clear
C)
\[2\pi \sqrt{\frac{M}{2k}}\]
done
clear
D)
\[2\pi \sqrt{\frac{3M}{2k}}\]
done
clear
View Answer play_arrow
The time period (T) of the artificial satellite of earth depends on the density (d) of the earth (assumed constant) as :-
A)
\[T\propto d\]
done
clear
B)
\[T\propto \sqrt{d}\]
done
clear
C)
\[T\propto \frac{1}{\sqrt{d}}\]
done
clear
D)
\[T\propto \frac{1}{d}\]
done
clear
View Answer play_arrow
A transfarmer has 100 turns in the primary coil and carries 8A current. If input power is one kilowatts, then number of turns in the secondary coil to have 500 V output will be :-
A)
100
done
clear
B)
200
done
clear
C)
400
done
clear
D)
300
done
clear
View Answer play_arrow
If T is the half life of a radioactive material, then the fraction that would remain after a time T/2 is.
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{3}{4}\]
done
clear
C)
\[\frac{1}{\sqrt{2}}\]
done
clear
D)
\[\frac{\sqrt{2}-1}{\sqrt{2}}\]
done
clear
View Answer play_arrow
A running man has half the KE to that of a boy of half the mass that man has. The man speeds up by \[1\text{ }m/s\] and then both have same KE. The original speeds of man and boy in m/s are :-
A)
\[(\sqrt{2}+1),\,(\sqrt{2}-1)\]
done
clear
B)
\[(\sqrt{2}+1),\,2(\sqrt{2}+1)\]
done
clear
C)
\[\sqrt{2},\sqrt{2}\]
done
clear
D)
\[(\sqrt{2}+1),2\,(\sqrt{2}-1)\]
done
clear
View Answer play_arrow
Chromatic aberration in a lens is caused by :-
A)
Reflection
done
clear
B)
Inteference
done
clear
C)
Diffraction
done
clear
D)
Dispersion
done
clear
View Answer play_arrow
The electric field in a region is given by \[\vec{E}=a\hat{i}+b\hat{j}\] . Here a and b are constant. The net flux passing through a square area of side \[\ell \] parallel to y-z plane will be :-
A)
\[a{{\ell }^{2}}\]
done
clear
B)
\[b{{\ell }^{2}}\]
done
clear
C)
\[\sqrt{{{a}^{2}}+{{b}^{2}}}{{\ell }^{2}}\]
done
clear
D)
\[\left( {{a}^{2}}+{{b}^{2}} \right){{\ell }^{2}}\]
done
clear
View Answer play_arrow
A thin bar magnet oscillates in the horizontal plane with a time period of\[2\text{ }sec\]. If bar magnet is broken into 4 equal parts perpendicular to its length than the time period of each part will be :-
A)
\[0.5\text{ }sec\]
done
clear
B)
\[1.0\text{ }sec\]
done
clear
C)
\[1.5\text{ }sec\]
done
clear
D)
\[2.0\text{ }sec\]
done
clear
View Answer play_arrow
The counting rate observed from a radioactive source at \[t=0\] second was 1600 counts per second and at \[t=8\] seconds it was 100 counts per second. The counting rate observed, as counts per second at \[t=4\] second will be :-
A)
400
done
clear
B)
300
done
clear
C)
200
done
clear
D)
150
done
clear
View Answer play_arrow
One of the refracting surfaces of a prism of angle \[{{30}^{o}}\] is silvered. A ray of light incident at an angle of \[{{60}^{o}}\]retraces its path. The refractive index of the material of prism is :-
A)
\[\sqrt{3}\]
done
clear
B)
\[\frac{3}{2}\]
done
clear
C)
\[2\]
done
clear
D)
\[\sqrt{3}\]
done
clear
View Answer play_arrow
Charge Q on a capacitor varies with voltage V as shown in the figure, where Q is taken along the X- axis and V along the Y-axis. The area of triangle will be represents.
A)
Capacitance
done
clear
B)
Capactive reactance
done
clear
C)
Electric field between the plates
done
clear
D)
Energy stored in the capacitor
done
clear
View Answer play_arrow
A coil of 100 turns and area of cross section \[1\,c{{m}^{2}}\] is lying in uniform transverse magnetic field, when coil is dragged out from the field then a charge of \[10\mu cb\]induced in it. If resistance of coil is \[10\,\,\Omega \].then external magnetic field will be:-
A)
\[{{10}^{-1}}T\]
done
clear
B)
\[{{10}^{-2}}T\]
done
clear
C)
\[{{10}^{-3}}T\]
done
clear
D)
\[{{10}^{-4}}T\]
done
clear
View Answer play_arrow
The spectrum of radiation \[1.0\times {{10}^{14}}Hz\] is in the infrared region. The energy of one photon of his in joules will be :-
A)
\[6.62\times {{10}^{-48}}\]
done
clear
B)
\[6.62\times {{10}^{-20}}\]
done
clear
C)
\[\frac{6.62}{3}\times {{10}^{-28}}\]
done
clear
D)
\[3\times 6.62\times {{10}^{-28}}\]
done
clear
View Answer play_arrow
If \[{{V}_{H}},{{V}_{N}}\] and \[{{V}_{O}}\] denotes the root mean square velocity of molecules of Hydrogen, Nitrogen and Oxygen respectively at a given temperature then.
A)
\[{{V}_{H}}>{{V}_{N}}>{{V}_{O}}\]
done
clear
B)
\[{{V}_{H}}={{V}_{N}}={{V}_{O}}\]
done
clear
C)
\[{{V}_{H}}<{{V}_{N}}<{{V}_{O}}\]
done
clear
D)
\[{{V}_{N}}>{{V}_{O}}>{{V}_{H}}\]
done
clear
View Answer play_arrow
Focal length of a glass lens in air is 2 cm. Its focal length when immersed in water would be :-
A)
4 cm
done
clear
B)
6 cm
done
clear
C)
8 cm
done
clear
D)
12 cm
done
clear
View Answer play_arrow
In the circuit shown in figure, \[{{V}_{A}}-{{V}_{B}}=19V.\] What is the potential difference across the \[3\mu F\] capacitor.
A)
7 V
done
clear
B)
8 V
done
clear
C)
23 V
done
clear
D)
4 V
done
clear
View Answer play_arrow
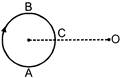
A small source of sound moves on a circle as shown in figure and an observer is sitting at O. Let \[{{n}_{1}},{{n}_{2}},{{n}_{3}}\] be the frequencies heard when the source is A, B and C respectively :-
A)
\[{{n}_{1}}>{{n}_{2}}>{{n}_{3}}\]
done
clear
B)
\[{{n}_{1}}={{n}_{2}}>{{n}_{3}}\]
done
clear
C)
\[{{n}_{2}}>{{n}_{3}}>{{n}_{1}}\]
done
clear
D)
\[{{n}_{1}}>{{n}_{3}}>{{n}_{2}}\]
done
clear
View Answer play_arrow
For photoelectric effect the stopping potential depends on :-
A)
The intensity of light
done
clear
B)
The nature of surface of the metal
done
clear
C)
Both the intensity of light and nature of surface of metal
done
clear
D)
Both the frequency of incident light and nature of surface of metal
done
clear
View Answer play_arrow
A gaseous mixture consists of 16 g of helium and 16 g of oxygen. The ratio of \[{{C}_{P}}/{{C}_{V}}\] of the mixture is :-
A)
\[1.4\]
done
clear
B)
\[1.54\]
done
clear
C)
\[1.59\]
done
clear
D)
\[1.62\]
done
clear
View Answer play_arrow
Given that \[\vec{P}+\vec{Q}+\vec{R}=\vec{0}\]. Two out of the three vectors are equal in magnitude. The magnitude of the third vector is \[\sqrt{2}\] times that of the either out of the other two. Which of the following can be the angles between these vectors :-
A)
\[{{45}^{o}},{{45}^{o}},{{90}^{o}}\]
done
clear
B)
\[{{45}^{o}},{{90}^{o}},{{135}^{o}}\]
done
clear
C)
\[{{90}^{o}},{{135}^{o}},{{135}^{o}}\]
done
clear
D)
\[{{30}^{o}},{{60}^{o}},{{90}^{o}}\]
done
clear
View Answer play_arrow
A potentiometer wire of length 100 cm has a resistance of \[10\Omega \]. It is connected in series with a resistance and a cell of emf 2V and of negligible internal resistance. A source of emf \[10mV\] is balanced against a length of 40 cm of the potentiometer wire. What is the value of external resistance :-
A)
\[810\,\Omega \]
done
clear
B)
\[790\,\Omega \]
done
clear
C)
\[1020\,\Omega \]
done
clear
D)
\[480\,\Omega \]
done
clear
View Answer play_arrow
The fundamental frequency of a closed organ pipe is equal to the first overtone frequency of an open organ pipe. If the length of the open pipe is 60 cm. What is the length of the closed pipe :-
A)
\[15\text{ }cm\]
done
clear
B)
\[30\text{ }cm\]
done
clear
C)
\[60\text{ }cm\]
done
clear
D)
\[7.5\text{ }cm\]
done
clear
View Answer play_arrow
The \[{{K}_{\alpha }}\] line form molybdenum (atomic number = 42) has a wavelength of\[0.7078\overset{o}{\mathop{A}}\,\]. The wavelength of \[{{K}_{\alpha }}\]line of zinc (atomic number = 30) will be :-
A)
\[1\overset{o}{\mathop{A}}\,\]
done
clear
B)
\[1.4148\overset{o}{\mathop{A}}\,\]
done
clear
C)
\[0.3541\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[0.5\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
Two rods of equal length and area of cross-section are kept parallel and lagged between temperatures \[{{20}^{o}}C\] and \[{{80}^{o}}C\]. The ratio of the effective thermal conductivity to that of the first rod is (the ratio of \[\frac{{{K}_{1}}}{{{K}_{2}}}=\frac{3}{4}\]):-
A)
\[7:4\]
done
clear
B)
\[7:6\]
done
clear
C)
\[4:7\]
done
clear
D)
\[7:8\]
done
clear
View Answer play_arrow
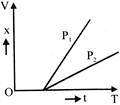
Fig shows two displacement time graphs of particles \[{{P}_{1}}\] and \[{{P}_{2}}\]. Their relative velocity :-
A)
Is zero
done
clear
B)
Is non zero but constant
done
clear
C)
Continuously decreases
done
clear
D)
Continuously increases
done
clear
View Answer play_arrow
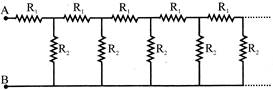
An infinite sequence of resistance is shown in the figure. The resultant resistance between A and B will be, when \[{{R}_{1}}=1\Omega \] and \[{{R}_{2}}=2\Omega \] :-
A)
Infinity
done
clear
B)
\[1\Omega \]
done
clear
C)
\[2\Omega \]
done
clear
D)
\[1.5\Omega \]
done
clear
View Answer play_arrow
When the voltage drop across a P-N Junction diode is increased from \[0.65\]to \[0.70,\]the change in the diode current is\[5\text{ }mA\]. The dynamic resistance of diode is :-
A)
\[10\Omega \]
done
clear
B)
\[20\Omega \]
done
clear
C)
\[50\Omega \]
done
clear
D)
\[80\Omega \]
done
clear
View Answer play_arrow
Fifty electric bulbs are connected in series across a 220 V supply and the illumination produced is \[{{I}_{1}}\]. Five bulbs are fused and remaining forty five bulbs are connected in series, the illumination produced is \[{{I}_{2}}\]. Which of the following is true :-
A)
\[{{I}_{1}}>{{I}_{2}}\]
done
clear
B)
\[{{I}_{1}}<{{I}_{2}}\]
done
clear
C)
\[{{I}_{1}}={{I}_{2}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
\[{{H}_{2}}S\] gas when passed through a solution of cations containing \[HCl\] precipitates the cations of second group of qualitative analysis but not those belonging to the fourth group. It is because -
A)
Presence of \[HCl\] decreases the sulphide ion concentration.
done
clear
B)
Sulphides of group IV cations are unstable in \[HCl\]
done
clear
C)
Solubility product of group II sulphides is more than that of group IV sulphides
done
clear
D)
Presence of \[HCl\] increases the sulphide ion concentration
done
clear
View Answer play_arrow
The rapid change of pH near the stoichiometric point of an acid-base titration is the basis of indicator detection. pH of the solution is related to ratio of the concentrations of the conjugate acid (HIn) and base \[(I{{n}^{-}})\]forms of the indicator by the expression :-
A)
\[\log \,\frac{[HIn]}{[I{{n}^{-}}]}=p{{K}_{In}}+pH\]
done
clear
B)
\[\log \,\frac{[HIn]}{[I{{n}^{-}}]}=pH-p{{K}_{In}}\]
done
clear
C)
\[\log \,\frac{[I{{n}^{--}}]}{[HIn]}=pH-p{{K}_{In}}\]
done
clear
D)
\[\log \,\frac{[I{{n}^{--}}]}{[HIn]}=p{{K}_{In}}-pH\]
done
clear
View Answer play_arrow
In Haber process 30 litres of dihydrogen and 30 litres of dinitrogen were taken for reaction which yielded only 50% of the expected product. What will be the composition of gaseous mixture under the above condition in the end :-
A)
20 litres ammonia, 20 litres nitrogen 20 litres hydrogen
done
clear
B)
10 litres ammonia, 25 litres nitrogen 15 litres hydrogen
done
clear
C)
20 litres ammonia, 10 litres nitrogen 30 litres hydrogen
done
clear
D)
20 litres ammonia, 25 litres nitrogen 15 litres hydrogen
done
clear
View Answer play_arrow
The number of moles of \[KMn{{O}_{4}}\] reduced by one mole of \[KI\] in alkaline medium is :-
A)
One
done
clear
B)
Two
done
clear
C)
Five
done
clear
D)
One fifth
done
clear
View Answer play_arrow
The maximum number of molecules is present in :-
A)
\[5L\] of \[{{N}_{2}}\] gas at STP
done
clear
B)
\[0.5\text{ }g\] of \[{{H}_{2}}\] gas
done
clear
C)
\[10\text{ }g\] of 0, gas
done
clear
D)
15 L of \[{{H}_{2}}\] gas at STP
done
clear
View Answer play_arrow
On the basis of the information available from the reaction : \[\frac{4}{3}Al+{{O}_{2}}\to \frac{2}{3}A{{l}_{2}}{{O}_{3}},\Delta G=-827KJ\,mo{{l}^{-1}}\] of \[{{O}_{2}}\], the minimum e.m.f. required to carry out electrolysis of \[A{{l}_{2}}{{O}_{3}}\] is \[(F=96500\,Cmo{{l}^{-1}})\]
A)
\[2.14\text{ }V\]
done
clear
B)
\[4.28\text{ }V\]
done
clear
C)
\[6.42\text{ }V\]
done
clear
D)
\[8.56\text{ }V\]
done
clear
View Answer play_arrow
The e.m.f. of a Daniell cell at \[298\text{ }K\] is \[{{E}_{1}}\]. \[Zn/ZnS{{O}_{4}}\,\,(0.01M||CuS{{O}_{4}}\,(1.0M)/Cu\] When the concentration of \[ZnS{{O}_{4}}\] is \[1.0\text{ }M\] and that of \[CuS{{O}_{4}}\] is \[0.01\text{ }M,\] the e.m.f. is changed to \[{{E}_{2}}\]. What is the relationship between \[{{E}_{1}}\] and \[{{E}_{2}}\] :-
A)
\[{{E}_{1}}>{{E}_{2}}\]
done
clear
B)
\[{{E}_{1}}<{{E}_{2}}\]
done
clear
C)
\[{{E}_{1}}={{E}_{2}}\]
done
clear
D)
\[{{E}_{2}}=0\ne {{E}_{1}}\]
done
clear
View Answer play_arrow
The percentage of C, H and N in an organic compound are 40%, 13.3% and 46.7% respectively then empirical formula is :-
A)
\[{{C}_{3}}{{H}_{13}}{{N}_{3}}\]
done
clear
B)
\[C{{H}_{2}}N\]
done
clear
C)
\[C{{H}_{4}}N\]
done
clear
D)
\[C{{H}_{6}}N\]
done
clear
View Answer play_arrow
Oxidation numbers of A, B and C are \[+2,+5\] and \[-2\] respectively possible formula of compound is :-
A)
\[{{A}_{2}}{{(B{{C}_{2}})}_{2}}\]
done
clear
B)
\[{{A}_{3}}{{(B{{C}_{4}})}_{2}}\]
done
clear
C)
\[{{A}_{2}}{{(B{{C}_{3}})}_{2}}\]
done
clear
D)
\[{{A}_{3}}{{({{B}_{2}}C)}_{2}}\]
done
clear
View Answer play_arrow
Mole fraction of solute is \[0.2\]in solution then lowering in V.P. \[\Delta P=10\]. If lowering in V.P. \[\Delta P=20\] then mole fraction of solvent will be in solution:-
A)
\[0.2\]
done
clear
B)
\[0.4\]
done
clear
C)
\[0.6\]
done
clear
D)
\[0.8\]
done
clear
View Answer play_arrow
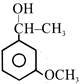
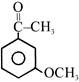
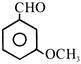
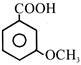
Consider the following compounds:
(i) \[{{C}_{6}}{{H}_{5}}COCl\] (ii) (iii) (iv)
The correct decreasing order of their reactivity towards hydrolysis is:
A)
(ii) > (iv) > (i) > (iii)
done
clear
B)
(ii) > (iv) > (iii) > (i)
done
clear
C)
(i) > (ii) > (iii) > (iv)
done
clear
D)
(iv) > (ii) > (i) > (iii)
done
clear
View Answer play_arrow
Which one of the following on treatment with 50% aqueous sodium hydroxide yields the corresponding alcohol and acid :
A)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}CHO\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHO\]
done
clear
View Answer play_arrow
During the process of digestion, the proteins present in food materials are hydrolysed to amino acids. The two enzymes involved in the process \[\text{Proteins}\xrightarrow{Enzyme\,\,\,(A)}Polypeptides\]\[\xrightarrow{Enzyme\,\,\,(B)}\text{Amino}\,\text{acids,}\]are respectively :-
A)
Amylase and Maltase
done
clear
B)
Diastase and Lipase
done
clear
C)
Pepsin and Trypsin
done
clear
D)
Invertase and Zymase
done
clear
View Answer play_arrow
Nucleophilic addition reaction will be most favoured in :-
A)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\]
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C=O\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
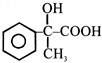
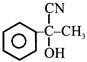
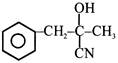
In a set of reactions acetic acid yielded a product D :- \[C{{H}_{3}}COOH\xrightarrow{SOC{{l}_{2}}}A\xrightarrow[Anhy.\,AlC{{l}_{3}}]{Benzene}B\xrightarrow{HCN}\]\[C\xrightarrow{{{H}_{2}}O}D\] The structure of D would be -
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
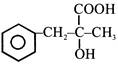
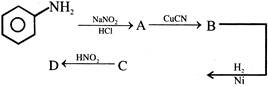
Aniline in a set of reactions yielded a product D
The structure of the product D would be -
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}OH\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}NHOH\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}NHC{{H}_{2}}C{{H}_{3}}\]
done
clear
View Answer play_arrow
Which one of the following is a chain growth polymer :-
A)
Nucleic acid
done
clear
B)
Polystyrene
done
clear
C)
Protein
done
clear
D)
Starch
done
clear
View Answer play_arrow
IUPAC name of the compound given below is:
A)
4-Ethyl-3-methyloctane
done
clear
B)
3-Methyl-4-ethyloctane
done
clear
C)
2,3-Diethylheptane
done
clear
D)
5-Ethyl-6-methyloctane
done
clear
View Answer play_arrow
Which one of the following is a free-radical substitution reaction :-
A)
done
clear
B)
done
clear
C)
done
clear
D)
\[C{{H}_{3}}CHO+HCN\to C{{H}_{3}}CH(OH)CN\]
done
clear
View Answer play_arrow
The final product C, obtained in this reaction, would be :
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Product 'P' in the above reaction is :-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
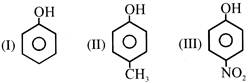
The correct acidic order of following is :-
A)
\[I>II>III\]
done
clear
B)
\[III>I>II\]
done
clear
C)
\[II>III>I\]
done
clear
D)
\[I>III>II\]
done
clear
View Answer play_arrow
Intermediates formed during reaction of \[R\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,N{{H}_{2}}\] with \[B{{r}_{2}}\] and \[KOH\] are :-
A)
RCONHBr and RNCO
done
clear
B)
RNHCOBr and RNCO
done
clear
C)
RNH - Br and RCONHBr
done
clear
D)
\[RCONB{{r}_{2}}\]
done
clear
View Answer play_arrow
An organic compound \[A({{C}_{4}}{{H}_{9}}Cl)\] on reaction with Na/diethyl ether gives a hydrocarbon which on monochlorination gives only one chloro derivative then, A is :-
A)
t-butyl chloride
done
clear
B)
sec. butyl chloride
done
clear
C)
Iso butyl chloride
done
clear
D)
n-butyl chloride
done
clear
View Answer play_arrow
Polarization in acrolein as:-
A)
\[\overset{+\delta }{\mathop{C{{H}_{2}}}}\,=CH-\overset{-\delta }{\mathop{CHO}}\,\]
done
clear
B)
\[\overset{-\delta }{\mathop{C{{H}_{2}}}}\,=CH-\overset{+\delta }{\mathop{CHO}}\,\]
done
clear
C)
\[\overset{-\delta }{\mathop{C{{H}_{2}}}}\,=CH-\overset{+\delta }{\mathop{CHO}}\,\]
done
clear
D)
\[\overset{+\delta }{\mathop{C{{H}_{2}}}}\,=CH-\overset{-\delta }{\mathop{CHO}}\,\]
done
clear
View Answer play_arrow
In Friedal craft reaction Toluene can be prepared by :-
A)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{3}}Cl\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}Cl+C{{H}_{4}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{2}}C{{l}_{2}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{3}}COCl\]
done
clear
View Answer play_arrow
The correct sequence of increasing covalent character is represented by :-
A)
\[NaCl<KCl<RbCl\]
done
clear
B)
\[AlC{{l}_{3}}<MgC{{l}_{2}}<NaCl\]
done
clear
C)
\[KCl<NaCl<MgC{{l}_{2}}\]
done
clear
D)
\[LiCl<NaCl<BaC{{l}_{2}}\]
done
clear
View Answer play_arrow
Which of the following species has trigonal planar geometry :-
A)
\[Cl{{F}_{3}}\]
done
clear
B)
\[B{{O}_{3}}^{3-}\]
done
clear
C)
\[NC{{l}_{3}}\]
done
clear
D)
\[Xe{{O}_{3}}\]
done
clear
View Answer play_arrow
\[S{{O}_{2}}\] is dipolar, whereas \[C{{O}_{2}}\] is not, it is because
A)
EN of S > C
done
clear
B)
\[S-O\] bond is more polar than \[C-O\]bond
done
clear
C)
\[S{{O}_{2}}\] is angular and \[C{{O}_{2}}\] is linear
done
clear
D)
\[S{{O}_{2}}\] is symmetrical and \[C{{O}_{2}}\] is unsymmetrical
done
clear
View Answer play_arrow
Which of the following is not an organometallic compound :-
A)
\[{{C}_{2}}{{H}_{5}}MgBr\]
done
clear
B)
\[[{{R}_{3}}Al]TiC{{l}_{4}}\]
done
clear
C)
\[B\,{{(OC{{H}_{3}})}_{2}}C{{H}_{3}}\]
done
clear
D)
\[{{K}_{4}}\,[Fe\,\,{{(CN)}_{6}}]\]
done
clear
View Answer play_arrow
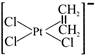
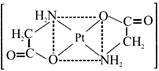
Which of the following is used to be an anticancer species :-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In \[Cl{{O}_{4}}^{-}\] formal charge and bond order on every oxygen is :-
A)
\[0.25\]and \[1.75\]
done
clear
B)
\[0.5\]and\[~0.25\]
done
clear
C)
\[0.75\] and \[1.25\]
done
clear
D)
\[0.66\]and \[1.33\]
done
clear
View Answer play_arrow
An atom has electronic configuration \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{d}^{5}}4{{s}^{1}},\] you will place it is :-
A)
first group and 4th period
done
clear
B)
s-block and 1st group
done
clear
C)
6th group and 4th period
done
clear
D)
11th group and d-block
done
clear
View Answer play_arrow
In which of the following pair species are is o-electronic :-
A)
\[{{S}^{2-}},{{O}^{2-}}\]
done
clear
B)
\[CN,NO\]
done
clear
C)
\[S{{O}_{4}}^{2-},P{{O}_{4}}^{3-}\]
done
clear
D)
\[{{F}_{2}},{{H}_{2}}O\]
done
clear
View Answer play_arrow
The most convenient method to protect the bottom of ship made of iron is :-
A)
Coating it with Red lead oxide
done
clear
B)
White tin plating
done
clear
C)
Connecting it with Mg block
done
clear
D)
Connecting it with Pb block
done
clear
View Answer play_arrow
Nitrogen form \[{{N}_{2}},\]but phosphorous form \[{{P}_{2}},\] it's at a time convert in \[{{P}_{4}},\] reason is :-
A)
Triple bond present between phosphorous atom
done
clear
B)
\[{{p}_{\pi }}-{{p}_{\pi }}\] bonding is weak
done
clear
C)
\[{{p}_{\pi }}-{{p}_{\pi }}\] bonding is strong
done
clear
D)
Multiple bond form easilly
done
clear
View Answer play_arrow
According to hardy Schultze law the order of coagulation power of cations will be:-
A)
\[N{{a}^{+}}>B{{a}^{+2}}>A{{l}^{+3}}\]
done
clear
B)
\[A{{l}^{+3}}>B{{a}^{+2}}>N{{a}^{+}}\]
done
clear
C)
\[B{{a}^{+2}}>A{{l}^{+3}}>N{{a}^{+}}\]
done
clear
D)
\[A{{l}^{+3}}>N{{a}^{+}}>B{{a}^{+2}}\]
done
clear
View Answer play_arrow
Which of the following example behave as a lewis acid \[B{{F}_{3}},SnC{{l}_{2}},SnC{{l}_{4}}\]:-
A)
Stenus chloride, stenic chloride
done
clear
B)
\[B{{F}_{3}},\] stenus chloride
done
clear
C)
Only \[B{{F}_{3}},\]
done
clear
D)
\[B{{F}_{3}},\] stenus chloride, stenic chloride
done
clear
View Answer play_arrow
Which one of the following statements about the zeolites is false :-
A)
They have open structure which enables them to take up small molecules
done
clear
B)
Zeolites are alumino silicates having three dimensional network
done
clear
C)
Some of the \[SiO_{4}^{4-}\] units are replaced by \[AlO_{4}^{5-}\]and \[AlO_{6}^{9-}\] ions in zeolites
done
clear
D)
They are used as cation exchangers.
done
clear
View Answer play_arrow
Which one of the following orders is not in according with the property state a against it?
A)
\[{{F}_{2}}>C{{l}_{2}}>B{{r}_{2}}>{{I}_{2}};\] Oxidising power
done
clear
B)
\[HI>HBr>HCl>HF;\] Acidic property in water
done
clear
C)
\[{{F}_{2}}>C{{l}_{2}}>B{{r}_{2}}>{{I}_{2}};\] Electronegativity
done
clear
D)
\[{{F}_{2}}>C{{l}_{2}}>B{{r}_{2}}>{{I}_{2}};\] Bond dissociation energy
done
clear
View Answer play_arrow
Consider the following reactions:
(a) \[{{H}^{+}}_{(aq)}+O{{H}^{-}}_{(aq)}={{H}_{2}}{{O}_{(l)}},\,\,\,\Delta H=-{{X}_{1}}kJ\,mo{{l}^{-1}}\] (b) \[{{H}_{2(g)}}+\frac{1}{2}{{O}_{2(g)}}={{H}_{2}}{{O}_{(l)}},\,\,\,\Delta H=-{{X}_{2}}\,kJ\,mo{{l}^{-1}}\] (c) \[C{{O}_{2(g)}}+{{H}_{2(g)}}=C{{O}_{(g)}}+{{H}_{2}}{{O}_{(l)}}-{{X}_{3}}kJ\,mo{{l}^{-1}}\] (d) \[{{C}_{2}}{{H}_{2(g)}}+\frac{5}{2}{{O}_{2(g)}}=2C{{O}_{2(g)}}+{{H}_{2}}{{O}_{(l)}}+{{X}_{4}}kJmo{{l}^{-1}}\]
Enthalpy of formation of \[{{H}_{2}}{{O}_{(l)}}\] is:
A)
\[+{{X}_{1}}\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[-{{X}_{2}}\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[+{{X}_{3}}\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-{{X}_{4}}\,kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
The orientation of an atomic orbital is governed by
A)
Azimuthal quantum number
done
clear
B)
Spin quantum number
done
clear
C)
Magnetic quantum number
done
clear
D)
Principal quantum number
done
clear
View Answer play_arrow
For a first order reaction \[A\to B\] the reaction rate at reactant concentration of \[0.01M\] is found to be \[2.0\times {{10}^{-5}}mol\,\,{{L}^{-1}}{{s}^{-1}}.\] The half life period of the reaction is :-
A)
300 s
done
clear
B)
30 s
done
clear
C)
220 s
done
clear
D)
347 s
done
clear
View Answer play_arrow
Considering entropy (S) as a thermodynamic parameter, the criterion for the spontaneity of any process is :-
A)
\[\Delta {{S}_{system}}-\Delta {{S}_{surroundings}}>0\]
done
clear
B)
\[\Delta {{S}_{system}}>0\] only
done
clear
C)
\[\Delta {{S}_{surroundings}}>0\] only
done
clear
D)
\[\Delta {{S}_{system}}+\Delta {{S}_{surroundings}}>0\]
done
clear
View Answer play_arrow
The molar heat capacity of water at constant pressure, C, is\[75\text{ }J{{K}^{-1}}\text{ }mo{{l}^{-1}}\]. When \[1.0\text{ }KJ\] of heat is supplied to 100 g of water which is free to expand, the increase in temperature of water is :-
A)
\[1.2\text{ }K\]
done
clear
B)
\[2.4\text{ }K\]
done
clear
C)
\[4.8\text{ }K\]
done
clear
D)
\[6.6\text{ }K\]
done
clear
View Answer play_arrow
All of the following statements are correct about plasmid except that :-
A)
They are extra chromosomal genetic elements
done
clear
B)
They can be transmitted to another bacterium during conjugation
done
clear
C)
They do not impart any essential properties to the bacteria
done
clear
D)
They are transmitted to daughter cells by binary fission
done
clear
View Answer play_arrow
The largest carpuscles of human blood are :-
A)
Neutrophils
done
clear
B)
Lymphocytes
done
clear
C)
Monocytes
done
clear
D)
Eosinophils
done
clear
View Answer play_arrow
How many different type of gametes are produced by A/a, B/B, C/c, D/d produce :-
A)
2
done
clear
B)
4
done
clear
C)
8
done
clear
D)
16
done
clear
View Answer play_arrow
Which is called smokers disease :-
A)
Gastritis
done
clear
B)
Emphysema
done
clear
C)
Asthma
done
clear
D)
Liver cirrhosis
done
clear
View Answer play_arrow
Which is strongest endopeptidase :-
A)
Trypsin
done
clear
B)
Pepsin
done
clear
C)
Rennin
done
clear
D)
Chymotrypsin
done
clear
View Answer play_arrow
5-ATGCCATACCAG-3 \[\leftarrow \] Antisense strand 3-TACGGTATGGTC-5 according to this DNA, which of the following mRNA is formed on transcription :-
A)
\[5-AUGCCAUACCAG-3\]
done
clear
B)
\[5-UACGGUAUGGUC-3\]
done
clear
C)
\[5-CUGGUAUGGCAU-3\]
done
clear
D)
\[5-GACCAUACCGUA-3\]
done
clear
View Answer play_arrow
Iliac artery carries blood to the :-
A)
Lungs
done
clear
B)
Ileum
done
clear
C)
Hind limbs
done
clear
D)
Brain
done
clear
View Answer play_arrow
In human embryology the cranial end is marked by appearance of :-
A)
Primitive streak
done
clear
B)
Prochordal plate
done
clear
C)
Notochord
done
clear
D)
Neural tube
done
clear
View Answer play_arrow
Which groups depend on external source of free water for fertilization :-
A)
Algae, mosses and ferns
done
clear
B)
Algae, mosses and Gymnosperms
done
clear
C)
Mosses, ferns and Gymnosperms
done
clear
D)
Only algae and mosses
done
clear
View Answer play_arrow
Facultative saprophyte is one, which is :-
A)
Always parasitic
done
clear
B)
Always saprophytic
done
clear
C)
Normally a saprophyte but accquires a parasitic mode
done
clear
D)
Normally a parasite but can be a saprophyte
done
clear
View Answer play_arrow
Which of the statements is not true for frog :-
A)
Neck and tail is absent in adult frog
done
clear
B)
Nictitating membrane is also present besides lower and upper eye lids
done
clear
C)
Exo-skeleton is present
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Dosase componsation is related to :-
A)
Watson and crick
done
clear
B)
Barr bodies
done
clear
C)
Y-body
done
clear
D)
Muller - 1932
done
clear
View Answer play_arrow
Which is most common enzymetic deficiency disorder in human being :-
A)
Thalassemia
done
clear
B)
G-6-PD deficiency syndrome
done
clear
C)
P.K.U.
done
clear
D)
Tay-sac disease
done
clear
View Answer play_arrow
Which vitamin prevent blindness in children
A)
vit. - A
done
clear
B)
vit. - C
done
clear
C)
vit. - D
done
clear
D)
vit. - B
done
clear
View Answer play_arrow
C-value of homosapiens is 2.8 picogram. One picogram is simillar to:-
A)
\[6.1\times {{10}^{11}}\]dalton
done
clear
B)
\[956\times {{10}^{6}}\]base pair
done
clear
C)
Both (a) and (b)
done
clear
D)
None
done
clear
View Answer play_arrow
The only human system that is derived from all the three germ layers is :-
A)
Digestive system
done
clear
B)
Excretory system
done
clear
C)
Respiratory system
done
clear
D)
Nervous sytem
done
clear
View Answer play_arrow
In case of drowning, death will occur early if the person is drowned m :-
A)
Sea water (salt water)
done
clear
B)
River water (fresh water)
done
clear
C)
Death will occur in equal time
done
clear
D)
It depends on the flow of water
done
clear
View Answer play_arrow
When moss spores germinate, they form :-
A)
Leafy gametophyte
done
clear
B)
Capsule
done
clear
C)
Protonema
done
clear
D)
Prothallus
done
clear
View Answer play_arrow
For the basis of classification of kingdom plantae the floral characters of higher plants are taken as a basis because :-
A)
Flowers can be easily preserved
done
clear
B)
show great variety in colour and structure
done
clear
C)
Reproductive parts are more conservative
done
clear
D)
can by study easily
done
clear
View Answer play_arrow
A man who carries an x-linked allele will pass it on to :-
A)
All his daughters
done
clear
B)
Half of his daughter
done
clear
C)
All his sons
done
clear
D)
All his children
done
clear
View Answer play_arrow
Which is not related to ELISA :-
A)
Simple and sensitive test
done
clear
B)
Quantative test
done
clear
C)
Qualitative test
done
clear
D)
None of above
done
clear
View Answer play_arrow
A person have bacterial throat infection with febrile and rhinorrhea which drug will be effective in it :-
A)
Antibiotics
done
clear
B)
Antihistaminic
done
clear
C)
Antipyretics
done
clear
D)
All the above
done
clear
View Answer play_arrow
How many pair of salivery gland is present in cat :-
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
tRNA attaches amino acid at its :-
A)
3' end
done
clear
B)
5' end
done
clear
C)
Anticodon loop
done
clear
D)
DHU loop
done
clear
View Answer play_arrow
Which one of the following muscles are responsible for moving and shaking the skin :-
A)
Arrector pilli
done
clear
B)
Collagen fibres
done
clear
C)
Paniculus camosus
done
clear
D)
Sphincter muscles
done
clear
View Answer play_arrow
The dissociation of oxyhaemoglobin and release of oxygen to the cells at tissue level is due :-
A)
\[\uparrow pC{{O}_{2}}\]
done
clear
B)
\[\downarrow pC{{O}_{2}}\]
done
clear
C)
\[\uparrow p{{O}_{2}}\]
done
clear
D)
\[\downarrow p{{O}_{2}}\]
done
clear
View Answer play_arrow
Which statement is true :-
A)
Spores and gametes are invariably diploids
done
clear
B)
Spores and gametes are invariably haploids
done
clear
C)
Only gametes are invariably haploids
done
clear
D)
Only spores are invariably diploids
done
clear
View Answer play_arrow
Character common in bryophyta and pteridophytes is :-
A)
Both are vascular
done
clear
B)
Sporophytic generation is dominant in both
done
clear
C)
Both have spiral and Muldflagellated Antherozoides
done
clear
D)
Both have jacketed and multicellular sexorgan
done
clear
View Answer play_arrow
In neurospora every mutation expressed as :-
A)
Recessive character
done
clear
B)
Dominant character
done
clear
C)
Complementry character
done
clear
D)
Duplicate gene
done
clear
View Answer play_arrow
Which enzyme is used in ELISA test :-
A)
\[\beta \]-galactosidase
done
clear
B)
Peroxidase
done
clear
C)
Both of above
done
clear
D)
None of above
done
clear
View Answer play_arrow
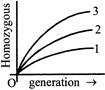
The accompanying diagram shows percentage of homozygous in generation under different digree of inbreeding through brother and sister, first cousin and second cousins match the lines numbered 1,2 and 3 with the above mentioned breeding pairs :-
A)
1, 2, 3
done
clear
B)
2, 1, 3
done
clear
C)
3, 1, 2
done
clear
D)
3, 2, 1
done
clear
View Answer play_arrow
Which type of teeth present in human :-
A)
Heterodont
done
clear
B)
Monophydont
done
clear
C)
Diphyodont
done
clear
D)
All the above
done
clear
View Answer play_arrow
In DNA like double stranded part of tRNA ratio of A-U/G-C may be :-
A)
\[1.0\]
done
clear
B)
\[1.2\]
done
clear
C)
\[0.8\]
done
clear
D)
\[2.3\]
done
clear
View Answer play_arrow
Ovulation occurs :-
A)
Alteranately from two ovaries
done
clear
B)
Simultaneously from both the ovaries
done
clear
C)
From one ovary alone throughout the life
done
clear
D)
According to the season from two ovaries
done
clear
View Answer play_arrow
During diastole auricles receive :-
A)
Oxygenated blood
done
clear
B)
Deoxygenated blood
done
clear
C)
Mixed blood
done
clear
D)
Venous blood
done
clear
View Answer play_arrow
Which of the following statement is correct:-
A)
Cycas is a living fossil
done
clear
B)
Gnetum is a connecting link between higher and lower Gymnosperm
done
clear
C)
In Aspergillus fruiting body is perithecium
done
clear
D)
In Funaria capsule represents gametophytic generation
done
clear
View Answer play_arrow
The Gymnosperms differ from the ferns in that
A)
They have heterosporous condition
done
clear
B)
They have vascular tissue
done
clear
C)
Sexual fusion does not occur in water
done
clear
D)
They have a larger gametophyte
done
clear
View Answer play_arrow
The genetic concept of segregation and recombination are most likely to be associated with :-
A)
Meiosis and gametes formation
done
clear
B)
Meiosis and mitosis
done
clear
C)
Meiosis and cleavage
done
clear
D)
Meiosis and fertilization
done
clear
View Answer play_arrow
Which is a immunopotentiating drug :-
A)
Cyclosporin
done
clear
B)
Interferon
done
clear
C)
Corticosteriod
done
clear
D)
Antibiotic
done
clear
View Answer play_arrow
Which of the following is a part of innate immunity :-
A)
NK-cell
done
clear
B)
Lymphocyte
done
clear
C)
Complementry protein
done
clear
D)
Both (a) and (c)
done
clear
View Answer play_arrow
How many milk canine is present in human :-
A)
4
done
clear
B)
20
done
clear
C)
2
done
clear
D)
S
done
clear
View Answer play_arrow
In a polypeptide chain seven amino acid are present. In the mRNA which form this polypeptide chain how many ester bonds are present :-
A)
47
done
clear
B)
41
done
clear
C)
30
done
clear
D)
32
done
clear
View Answer play_arrow
Obturator foramen in pelvic girdle of mammals is formed by :-
A)
Pubis and ischium
done
clear
B)
Pubis and ilium
done
clear
C)
Ilium and ischium
done
clear
D)
Ilium, ischium and pubis
done
clear
View Answer play_arrow
Which increase the fluidity of plasma membrane :-
A)
Having a larger number of membrane protein
done
clear
B)
Cholesterol present in the membrane
done
clear
C)
Number of double bonds
done
clear
D)
Number of single bonds
done
clear
View Answer play_arrow
Meiosis occurs in Lycopsida when :-
A)
Gametes are formed
done
clear
B)
Prothallus is formed
done
clear
C)
Sex organs are formed
done
clear
D)
Spores are formed
done
clear
View Answer play_arrow
Which process does not occur in yeast :-
A)
Ascospore formation
done
clear
B)
Budding
done
clear
C)
Plasmogamy
done
clear
D)
Oogamy
done
clear
View Answer play_arrow
In \[{{F}_{2}}\] generation the phenotype and genotype ratio are same in case of :-
A)
Mendelian inheritance
done
clear
B)
Coupling phenomenon
done
clear
C)
Partial dominance
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which laser is used for LASIK laser :-
A)
U.V. laser
done
clear
B)
\[C{{O}_{2}}\] laser
done
clear
C)
Argon-ion laser
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which is not true about the prokaryotic genes
A)
The are ready to transcribed any time
done
clear
B)
Their expression is regulated by promoter sequence
done
clear
C)
They have intron and exon
done
clear
D)
They are shorter than the eukaryotic gene
done
clear
View Answer play_arrow
Absorption of iron take place in which part of intestine :-
A)
Duodenom
done
clear
B)
Jejunum
done
clear
C)
Ilium
done
clear
D)
Stomach
done
clear
View Answer play_arrow
At the time of transcription, the sequence of \[{{N}_{2}}\] bases may be present at-terminator site :-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If \[P{{O}_{2}}\] of inspired air is \[104\text{ }mm\text{ }Hg\] then the \[P{{O}_{2}}\] of first \[150\text{ }ml\] of expired air during normal breathing would be about :-
A)
\[40\text{ }mm\text{ }Hg\]
done
clear
B)
\[46\text{ }mm\text{ }Hg\]
done
clear
C)
\[104\text{ }mm\text{ }Hg\]
done
clear
D)
\[150\text{ }mm\text{ }Hg\]
done
clear
View Answer play_arrow
Protein that stay within the cell are produced:-
A)
On free ribosome in the cytoplasm
done
clear
B)
In the nucleous
done
clear
C)
On ribosome attached to rough ER
done
clear
D)
On ribosome and pass through the golgi apparatus
done
clear
View Answer play_arrow
Gymnosperms are called naked seeded plants:-
A)
because their seeds are not surrounded by ovary wall
done
clear
B)
Integument surrounding their seeds is thin
done
clear
C)
Seeds are produced inside the fruits
done
clear
D)
They have heterosporous nature
done
clear
View Answer play_arrow
Best source to obtain areolar connective tissue for histology is :-
A)
Periosteum
done
clear
B)
Perimycium
done
clear
C)
Perichondrium
done
clear
D)
Epimycium
done
clear
View Answer play_arrow
A trait in a diploid via single gene with five alleles. How many different allele would be present in an individual :-
A)
2
done
clear
B)
5
done
clear
C)
15
done
clear
D)
10
done
clear
View Answer play_arrow
Which is most common problem of adolesence
A)
Neuroasthenia
done
clear
B)
Acne
done
clear
C)
Drug addiction
done
clear
D)
Phobia
done
clear
View Answer play_arrow
Function of IgE is :-
A)
Agglutination
done
clear
B)
Opsonisation
done
clear
C)
Hypersensitivity reaction
done
clear
D)
All the above
done
clear
View Answer play_arrow
Even after disruption of all the hydrogen bonds, which structural levels of a protein molecule still remain intact :-
A)
Tertiary
done
clear
B)
Primary
done
clear
C)
Secondary
done
clear
D)
Quaternary
done
clear
View Answer play_arrow
Father of lichenology is :-
A)
Morrison
done
clear
B)
Crombie
done
clear
C)
De bary
done
clear
D)
Erik acharius
done
clear
View Answer play_arrow
The amount of haemoglobin present in an average adult human blood is about :-
A)
20 gm
done
clear
B)
15 gm
done
clear
C)
75 gm
done
clear
D)
750 gm
done
clear
View Answer play_arrow
The ear detect sound by the movement of :-
A)
The basilar membrane
done
clear
B)
The tectorial membrane
done
clear
C)
The cupula
done
clear
D)
Otoconia
done
clear
View Answer play_arrow
Among the following which is not characteristic feature of bryophyta :-
A)
Motile sperms
done
clear
B)
Presence of Archegonium
done
clear
C)
Water essential for fertilization
done
clear
D)
Photosynthetically independent sporophyte
done
clear
View Answer play_arrow
In order to study the efficiency of stereocilia one can refer to :-
A)
Fallopian tube
done
clear
B)
Ependymal epithelium
done
clear
C)
Epididymes
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Human skin cell contain....chromosomes and sperm contian.....chromosome :-
A)
23, 46
done
clear
B)
46, 23
done
clear
C)
46, 46
done
clear
D)
23, 23
done
clear
View Answer play_arrow
Which test you will prescribe to find out the risk of heart attack :-
A)
E.E.G.
done
clear
B)
Lipid profile
done
clear
C)
E.S.R.
done
clear
D)
B.T. &C.T. test
done
clear
View Answer play_arrow
Which is a brain stimulant :-
A)
Smack
done
clear
B)
Opium
done
clear
C)
Hashish
done
clear
D)
Cocaine
done
clear
View Answer play_arrow
In a double stranded DNA sample 20 molecules of pentose sugar are present. In this sample what will be the number of purine nitrogen bases :-
A)
10
done
clear
B)
5
done
clear
C)
6
done
clear
D)
20
done
clear
View Answer play_arrow
'Reindeer moss' is common name of :-
A)
Cladonia
done
clear
B)
Cetraria islandica
done
clear
C)
Letharia valpina
done
clear
D)
Cladonia rangifera
done
clear
View Answer play_arrow
If a person takes deep and fast breaths for some time forcefully then after this act his desire to breath reduces for some time. This is due to:-
A)
Increased \[P{{O}_{2}}\] in blood
done
clear
B)
Decreased \[PC{{O}_{2}}\] in blood
done
clear
C)
Decreased \[P{{O}_{2}}\] in blood
done
clear
D)
Increased \[PC{{O}_{2}}\] in blood
done
clear
View Answer play_arrow
Which of the following statement is incorrect:-
A)
Vertebrate focus the eye by changing the shape of lens
done
clear
B)
Flow of sensory information in the retina is opposite to the path of light
done
clear
C)
The vertebrate eye adjust the amount of light entering the eye by contracting the ciliary muscle
done
clear
D)
Inner segment of rod and cones have more mitochondria
done
clear
View Answer play_arrow
Which of the two kingdoms have equal rank in the five kingdom classification :-
A)
Monera and Protista
done
clear
B)
Protista and Plantae
done
clear
C)
Protista and Animalia
done
clear
D)
Plantae and Animalia
done
clear
View Answer play_arrow
Mast cells are also known to occur as :?
A)
Monocyte
done
clear
B)
Eosinophils
done
clear
C)
Acidophil
done
clear
D)
Basophil
done
clear
View Answer play_arrow
The genetic event that result in Jacob syndrome is probably :-
A)
Non disjunction
done
clear
B)
Deletion
done
clear
C)
Duplication
done
clear
D)
Monoploidy
done
clear
View Answer play_arrow
Which gene is called 'guardian of genome :-
A)
\[{{P}^{53}}\]gene
done
clear
B)
Ti gene
done
clear
C)
\[{{P}^{BR\,\,322}}\]
done
clear
D)
Promoter gene
done
clear
View Answer play_arrow
'Window period' is related to which virus :-
A)
HIV-I
done
clear
B)
L.A.V.
done
clear
C)
H.T.L.V-III
done
clear
D)
All the above
done
clear
View Answer play_arrow
Biuret test is specific test for protein. It determine the presence of :-
A)
Peptide bond
done
clear
B)
a-amino acid
done
clear
C)
\[-COOH\] group
done
clear
D)
\[-N{{H}_{2}}\] group
done
clear
View Answer play_arrow
Which structure regulate the volume and composition of the body fluid of earthworm :-
A)
Typhlosole
done
clear
B)
Lateral heart
done
clear
C)
Nephridia
done
clear
D)
Dorsal vessel
done
clear
View Answer play_arrow
Dialation or contraction of which blood vessel can alter the amount blood flow to an organ :-
A)
Artery
done
clear
B)
Arterioles
done
clear
C)
Capillary
done
clear
D)
Veins
done
clear
View Answer play_arrow
Individual with type-I diabetes :-
A)
Produce enough insulin but lack functional receptor on their cells
done
clear
B)
Lack P cells in the islets of Langerhans
done
clear
C)
Lack a cells in the islets of langerhans
done
clear
D)
Can contral diabetes with diet and exercise
done
clear
View Answer play_arrow
Which of following statement is incorrect :-
A)
DNA in the nucleus is usually coiled into chromosome
done
clear
B)
The nucleoues is the sets for ribosomal RNA synthesis
done
clear
C)
RNA can pass through nucleur pore
done
clear
D)
Red blood cell cannot synthesize RNA
done
clear
View Answer play_arrow
Choose the wrong statement :-
A)
Chlorellin antibiotic is obtained from Chlorella
done
clear
B)
Chlorella causes Red rust disease in the leaves of tea plant
done
clear
C)
Chlorella is non motile algae
done
clear
D)
In space Chlorella is used as food and\[{{O}_{2}}\]source
done
clear
View Answer play_arrow
Compound tubulo - alveolar gland is :-
A)
Parotid gland
done
clear
B)
Sweat gland
done
clear
C)
Submandibular gland
done
clear
D)
None of the above
done
clear
View Answer play_arrow
\[1:1:1:1\]ratio of offspring from a dihybrid test cross indicates that :-
A)
Linked gene
done
clear
B)
Dominant organism was homozygous
done
clear
C)
Crossing over
done
clear
D)
Non linked gene
done
clear
View Answer play_arrow
Which drug is used in Narcolepsy :-
A)
Valium
done
clear
B)
Pethinine
done
clear
C)
Amphetamine
done
clear
D)
L.S.D.
done
clear
View Answer play_arrow
Which is true for pulmonery cancer :-
A)
Smoking is most common cause
done
clear
B)
Does not present in female
done
clear
C)
Most common sarcoma
done
clear
D)
All the above
done
clear
View Answer play_arrow
A bacterial mRNA with a length of 360 nucleotides code for a protein of how many amino acid :-
A)
360
done
clear
B)
1080
done
clear
C)
120
done
clear
D)
119
done
clear
View Answer play_arrow
Chitinous asymmetrical structures surrounding the male gonopore of cockroach :-
A)
Ovipositor
done
clear
B)
Ootheca
done
clear
C)
Gonopophysis
done
clear
D)
Gynatrium
done
clear
View Answer play_arrow
Increase in Aldosterone secretion will not cause
A)
Increase in plasma concentration of sodium
done
clear
B)
Rise in blood pressure
done
clear
C)
Fall in urinary excretion of salt
done
clear
D)
Concentration of urine
done
clear
View Answer play_arrow
Second messenger CAMP are activated in response to :-
A)
Steroid hormones
done
clear
B)
Thyroxine
done
clear
C)
Insulin
done
clear
D)
Peptide hormone
done
clear
View Answer play_arrow
 The structure of the product D would be -
The structure of the product D would be - 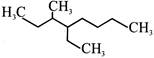
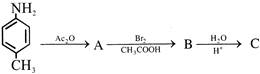
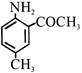
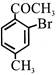
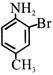
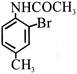
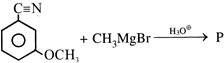 Product 'P' in the above reaction is :-
Product 'P' in the above reaction is :-