The wavelength of \[{{K}_{d}}\] line from element of atomic number 51 is\[\lambda \]. From another element the wavelength of \[{{K}_{d}}\] line is\[4\lambda \]. What is the atomic number of the second element?
A)
25
done
clear
B)
26
done
clear
C)
100
done
clear
D)
99
done
clear
View Answer play_arrow
Suppose T be mean life of a radioactive sample 75% of the active nuclei present in the sample initially will decay in time:
A)
\[2T\]
done
clear
B)
\[1/2\,(\log \,2)\,T\]
done
clear
C)
\[4T\]
done
clear
D)
\[2\,(\log \,2)\,T\]
done
clear
View Answer play_arrow
The number of turn in primary and secondary turns of a transformer are 1000 and 3000 respectively. If 80V AC is supplied to the primary coil of the transformer, the potential difference per turn of the secondary coil would be:
A)
\[200\,V\]
done
clear
B)
\[2400\,V\]
done
clear
C)
\[0.24\,V\]
done
clear
D)
\[0.08\,V\]
done
clear
View Answer play_arrow
Which of the following does not fit into a group?
A)
Electron
done
clear
B)
Gravitation
done
clear
C)
Proton
done
clear
D)
Meson
done
clear
View Answer play_arrow
Water is cooled to form ice, if mass energy equivalence is taken into account this should cause the mass of water to:
A)
Increase
done
clear
B)
Decrease
done
clear
C)
Remain unchanged
done
clear
D)
First increase then decrease
done
clear
View Answer play_arrow
An electromagnetic pulse of energy \[3\times {{10}^{-2}}\] Joule when incident on a perfectly reflecting mirror produces an impulse of:
A)
\[3\times {{10}^{-10}}kg\,\,m{{s}^{-1}}\]
done
clear
B)
\[{{10}^{-10}}kg\,\,m{{s}^{-1}}\]
done
clear
C)
\[8\times {{10}^{-10}}kg\,\,m{{s}^{-1}}\]
done
clear
D)
\[2\times {{10}^{-10}}kg\,\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
Light passes through a glass plate of thickness d and refractive index \[\mu \]. For small angle of incidence \[\theta ,\] the lateral displacement will be:
A)
\[d(\theta )\]
done
clear
B)
\[d\,(\theta )\,\,(\mu -1)\]
done
clear
C)
\[d\,(\theta )\,\mu /\mu -1\]
done
clear
D)
\[d\,(\theta )\,\frac{(\mu -1)}{\mu }\]
done
clear
View Answer play_arrow
The intensity of different order maxima is nearly the same in :
A)
Young's double slit experiment
done
clear
B)
Single slit diffraction pattern
done
clear
C)
Double slit diffraction pattern
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
Which of the following function represent a simple harmonic oscillation?
A)
\[\sin \omega t-\cos \omega t\]
done
clear
B)
\[{{\sin }^{2}}\omega t\]
done
clear
C)
\[\sin \omega t+\sin 2\,\omega t\]
done
clear
D)
\[\sin \omega t-\sin 2\,\omega t\]
done
clear
View Answer play_arrow
In case of linearly polarized light the magnitude of the electric field vector :
A)
Does not change with time
done
clear
B)
Varies periodically with time
done
clear
C)
Increase and decrease linearly with time.
done
clear
D)
Is parallel to the direction of propagation.
done
clear
View Answer play_arrow
An electric bulb is rated \[220V,\text{ }60W,\] the electrical resistance is nearly:
A)
\[0.807\,\Omega \]
done
clear
B)
\[16\,\Omega \]
done
clear
C)
\[908\,\Omega \]
done
clear
D)
\[807\,\Omega \]
done
clear
View Answer play_arrow
The magnetic materials having negative magnetic susceptibility are!
A)
Non-magnetic
done
clear
B)
Para magnetic
done
clear
C)
Dia magnetic
done
clear
D)
Ferromagnetic
done
clear
View Answer play_arrow
A magnet of magnetic moment M is rotated through \[{{360}^{o}}\] in a magnetic field H, the work done is:
A)
\[MH\]
done
clear
B)
\[2MH\]
done
clear
C)
\[27\pi MH\]
done
clear
D)
zero
done
clear
View Answer play_arrow
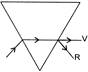
Which of the following diagram shows correct the dispersion of white light by a prism?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Two nucleons are at a separation of 1 fermi. The net force between them is \[{{f}_{1}}\] if both are neutrons, \[{{f}_{2}}\] if both are protons, and fg if one is a proton and the other is a neutron, then
A)
\[{{f}_{1}}>{{f}_{2}}>{{f}_{3}}\]
done
clear
B)
\[{{f}_{2}}>{{f}_{1}}>{{f}_{3}}\]
done
clear
C)
\[{{f}_{1}}={{f}_{3}}>{{f}_{2}}\]
done
clear
D)
\[{{f}_{1}}={{f}_{2}}>{{f}_{3}}\]
done
clear
View Answer play_arrow
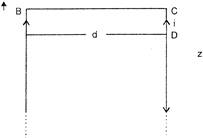
AB and CD are along straight conductor distance d apart carrying a current i. The magnetic field on BC due to the current in AB and CD.
A)
is zero at all points
done
clear
B)
to zero only at its mide point
done
clear
C)
has different magnitudes at different points
done
clear
D)
is maximum at its midpoint
done
clear
View Answer play_arrow
How does the internal energy change when the ice and wax melt at their normal melting points?
A)
Increase for ice, decrease for wax
done
clear
B)
Increase for wax, decrease for ice
done
clear
C)
Decrease for both ice and wax
done
clear
D)
Increase for both ice and wax.
done
clear
View Answer play_arrow
It there were no atmosphere, the earth would have been:
A)
Slightly hot
done
clear
B)
Less cold
done
clear
C)
More hot
done
clear
D)
Very cold
done
clear
View Answer play_arrow
To produce' a minimum reflaction of wavelength near the middle of visible spectrum (555nm) how should a coating of \[mg{{f}_{2}}(\mu =1.8)\] be vacuum coated on a glass surface?
A)
\[{{10}^{-7}}m\]
done
clear
B)
\[{{10}^{-6}}m\]
done
clear
C)
\[{{10}^{-9}}m\]
done
clear
D)
\[{{10}^{-12}}m\]
done
clear
View Answer play_arrow
To luminous flux from a 100 watt electric lamp is 6830 lumen. What is the efficiency of the lamp?
A)
1%
done
clear
B)
5%
done
clear
C)
10%
done
clear
D)
50%
done
clear
View Answer play_arrow
If the length of second pendulum is decreased by 2% how many seconds it will lose per day?
A)
\[1927\text{ }sec.\]
done
clear
B)
\[2727\text{ }sec.\]
done
clear
C)
\[2427\text{ }sec.\]
done
clear
D)
\[864\text{ }sec.\]
done
clear
View Answer play_arrow
For a simple pendulum graph between 1 and T will be a:
A)
Hyperbola
done
clear
B)
Parabola
done
clear
C)
Straight line
done
clear
D)
Ellipse
done
clear
View Answer play_arrow
The source X of unknown frequency produce 8 beats with a source of 250 Hz and 12 beats with a source of 270 Hz. The frequency of source X will be:
A)
258 Hz
done
clear
B)
242 Hz
done
clear
C)
262 Hz
done
clear
D)
382 Hz
done
clear
View Answer play_arrow
Ultrasonic waves are produced by :
A)
Piezoelectric effect
done
clear
B)
Peltier effect
done
clear
C)
Doppler's effect
done
clear
D)
Coulamb's effect
done
clear
View Answer play_arrow
Sound wave on air cannot be polarized becuase:
A)
Their speed in small
done
clear
B)
They require medium
done
clear
C)
These are longitudinal
done
clear
D)
Their speed is temperature dependent.
done
clear
View Answer play_arrow
Two spheres of radii \[{{R}_{1}}\] and \[{{R}_{2}}\] respectively are charged and joined by a wire. The ratio of electric field of sphere will be:
A)
\[\frac{2R_{2}^{2}}{R_{1}^{2}}\]
done
clear
B)
\[\frac{R_{1}^{2}}{R_{2}^{2}}\]
done
clear
C)
\[\frac{{{R}_{2}}}{{{R}_{1}}}\]
done
clear
D)
\[\frac{{{R}_{1}}}{{{R}_{2}}}\]
done
clear
View Answer play_arrow
The dielectric constant of metal is:
A)
1
done
clear
B)
zero
done
clear
C)
Infinite
done
clear
D)
None of these
done
clear
View Answer play_arrow
Three different capacitors are connected in series, then :
A)
They will have equal charges
done
clear
B)
They will have same potential
done
clear
C)
Both A and B
done
clear
D)
None of these
done
clear
View Answer play_arrow
Electric field at the centroid of triangle carrying q charge at each corner will be:
A)
\[\sqrt{2kq}\]
done
clear
B)
\[\frac{1}{2}.\frac{\sqrt{kq}}{{{r}^{2}}}\]
done
clear
C)
\[\frac{4kq}{{{r}^{2}}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
An electrical cable of copper has just one wire of radius 9mm. Its resistance is\[5\Omega \]. The single copper wire of the cable is replaced by different well insulated copper wires each of radius 3mm. The total resistance of the cable will now be equal to:
A)
\[7.5\Omega \]
done
clear
B)
\[45\,\Omega \]
done
clear
C)
\[90\,\Omega \]
done
clear
D)
\[270\,\Omega \]
done
clear
View Answer play_arrow
A body rolls down an inclined plane. If its kinetic energy of rotational motion is 40% of kinetic energy of translational motion, then the body is a:
A)
Ring
done
clear
B)
Cylinder
done
clear
C)
Spherical shell
done
clear
D)
Solid sphere
done
clear
View Answer play_arrow
A raw egg and a hard boiled egg are made to spin on a table with the same angular speed about the same axis. The ratio of the time taken by the two to step will be:
A)
\[=1\]
done
clear
B)
\[<1\]
done
clear
C)
\[>1\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If R is the radius of the earth and g is the acceleration due to gravity on the earth surface the mean density of earth will be:
A)
\[\frac{4\pi G}{3g\,R}\]
done
clear
B)
\[\frac{3\pi G}{4g\,R}\]
done
clear
C)
\[\frac{3g}{4\pi RG}\]
done
clear
D)
\[\frac{\pi RG}{2g}\]
done
clear
View Answer play_arrow
A spherical liquid drop of radius R is divided into eight equal droplets. If surface tension is T, then work done is process is:
A)
\[2\pi \,{{R}^{2}}T\]
done
clear
B)
\[8\pi \,{{R}^{2}}T\]
done
clear
C)
\[4\pi \,{{R}^{2}}T\]
done
clear
D)
\[2\pi \,R{{T}^{2}}\]
done
clear
View Answer play_arrow
The viscous force on a small sphere of radius R. moving in a fluid is directly proportional to:
A)
\[{{R}^{2}}\]
done
clear
B)
R
done
clear
C)
\[\frac{2}{R}\]
done
clear
D)
\[\frac{1}{{{R}^{2}}}\]
done
clear
View Answer play_arrow
In streamline flow to liquid, the total energy of liquid is constant at:
A)
All points
done
clear
B)
Inner points
done
clear
C)
Outer points
done
clear
D)
None of these
done
clear
View Answer play_arrow
The mass of water rises on capillary tube of radius R is M. The mass of water that rises on tube of radius 2R is:
A)
M
done
clear
B)
M/2
done
clear
C)
2M
done
clear
D)
4M
done
clear
View Answer play_arrow
If temperature of a black body is doubled the wave length at which spherical radiancy has its maximum will be:
A)
Doubled
done
clear
B)
Halved
done
clear
C)
Quandrupled
done
clear
D)
Tripled
done
clear
View Answer play_arrow
Let there be four articles having colour blue, red, black and white. When they are heated together and allowed to cool, which article will cool at the earliest?
A)
Blue
done
clear
B)
Red
done
clear
C)
Black
done
clear
D)
White
done
clear
View Answer play_arrow
The SI unit of thermal conductivity is:
A)
\[J{{S}^{-1}}M{{K}^{-1}}\]
done
clear
B)
\[J{{S}^{-1}}{{K}^{-1}}\]
done
clear
C)
\[JS{{M}^{-1}}K\]
done
clear
D)
\[J{{S}^{-1}}{{M}^{-1}}{{K}^{-1}}\]
done
clear
View Answer play_arrow
A particle of mass 4m which is at rest explodes into three fragments. Two of the fragments each of mass m are found to move with a speed V each is mutually perpendicular direction. The energy released in the process of explosion will be:
A)
\[1/4m{{V}^{2}}\]
done
clear
B)
\[3/2m{{V}^{2}}\]
done
clear
C)
\[1/2m{{V}^{2}}\]
done
clear
D)
\[3/4m{{V}^{2}}\]
done
clear
View Answer play_arrow
A mixture of \[{{n}_{1}}\]moles of monoatomic gas and \[{{n}_{2}}\]moles of diatomic gas has \[\frac{{{C}_{P}}}{{{C}_{V}}}=1.5=\gamma ,\] then.
A)
\[{{n}_{1}}={{n}_{2}}\]
done
clear
B)
\[2{{n}_{1}}={{n}_{2}}\]
done
clear
C)
\[3{{n}_{1}}=2{{n}_{2}}\]
done
clear
D)
\[2{{n}_{1}}=3{{n}_{2}}\]
done
clear
View Answer play_arrow
The function \[F(t)=\sin \omega t+\cos \omega t\] corresponds a:
A)
Simple harmonic function
done
clear
B)
Periodic function
done
clear
C)
Cylindrical functions
done
clear
D)
None of the above.
done
clear
View Answer play_arrow
The work function of a metal is \[1.5\text{ }eV,\] light of wavelength \[6600\overset{o}{\mathop{A}}\,\]is made incident on it. The maximum \[k.\varepsilon .\] of emitted photo-electron is:
A)
\[0.6\times {{10}^{-19}}J\]
done
clear
B)
\[2.6\times {{10}^{-19}}J\]
done
clear
C)
\[1.6\times {{10}^{-19}}J\]
done
clear
D)
\[1.6\,eV\]
done
clear
View Answer play_arrow
The time dependence of a physical quantity p is given by \[p={{p}_{0}}\exp \,(-\alpha {{t}^{2}})\]where a constant and t is time. The constant \[\alpha \]:
A)
has no dimension
done
clear
B)
has dimension of \[{{T}^{-2}}\]
done
clear
C)
has dimension of \[{{T}^{2}}\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
A smuggler could not carry gold by depositing iron on gold surface since
A)
gold is denser
done
clear
B)
iron rusts
done
clear
C)
gold has higher reduction potential than iron
done
clear
D)
gold has lower reduction potential than iron.
done
clear
View Answer play_arrow
The highest electrical conductivity of the following aqueous solutions is of:
A)
\[0.1\text{ }M\] difluoroacetic acid
done
clear
B)
\[0.1\text{ }M\] fluoroacetic acid
done
clear
C)
\[0.1\text{ }M\] chloroaceticacid
done
clear
D)
\[0.1\text{ }M\] acetic acid
done
clear
View Answer play_arrow
The safest and the most common alternative of sugar is
A)
glucose
done
clear
B)
aspartame
done
clear
C)
saccharin
done
clear
D)
cyclodextrin
done
clear
View Answer play_arrow
Plexiglass is commercial name of
A)
glyptal
done
clear
B)
polyacrylonitrile
done
clear
C)
polymethyl methacrylate
done
clear
D)
polyethylacrylate
done
clear
View Answer play_arrow
A bad smelling substance formed by the action of alcoholic caustic potash on chloroform and aniline is
A)
nitrobenzene
done
clear
B)
phenyl isocyanide
done
clear
C)
phenyl cyanide
done
clear
D)
phenyl isocynate
done
clear
View Answer play_arrow
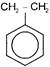
In the following reaction
The organic product X is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The increasing order of the rate of HCN addition to compound A-D is
A. HCHO B. \[C{{H}_{3}}COC{{H}_{3}}\] C. \[PhCOC{{H}_{3}}\] D. \[PhCOPh\]
A)
\[A<B<C<D\]
done
clear
B)
\[D<B<C<A\]
done
clear
C)
\[D<C<B<A\]
done
clear
D)
\[C<D<B<A\]
done
clear
View Answer play_arrow
The enthalpies of formation of \[{{C}_{2}}{{H}_{2(g)}}\] and \[{{C}_{6}}{{H}_{6(g)}}\]at 298 K are 230 and \[85\text{ }kJ\text{ }mo{{l}^{-1}}\]respectively. The enthalpy change \[\Delta H\] for the polymerisation of acetylene at 298 K is
A)
\[+205\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
B)
\[-205\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[+605\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
D)
\[-605\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
The correct order of bond angles in \[{{H}_{2}}S,\,N{{H}_{3}},B{{F}_{3}}\] and \[Si{{H}_{4}}\] is
A)
\[{{H}_{2}}S>Si{{H}_{4}}<N{{H}_{3}}<B{{F}_{3}}\]
done
clear
B)
\[N{{H}_{3}}<{{H}_{2}}S<Si{{H}_{4}}<B{{F}_{3}}\]
done
clear
C)
\[{{H}_{2}}S<N{{H}_{3}}<Si{{H}_{4}}<B{{F}_{3}}\]
done
clear
D)
\[{{H}_{2}}S<N{{H}_{3}}<B{{F}_{3}}<Si{{H}_{4}}\]
done
clear
View Answer play_arrow
When during electrolysis of a solution of \[AgN{{O}_{3}},\]9650 coulombs of charge pass through the electroplating both, the mass of silver deposited on the cathode will be
A)
\[1.08\text{ }g\]
done
clear
B)
\[10.8\text{ }g\]
done
clear
C)
\[21.6\text{ }g\]
done
clear
D)
\[108\,g\]
done
clear
View Answer play_arrow
At what temperature will the RMS velocity of \[S{{O}_{2}}\]be the same as that of \[{{O}_{2}}\] at 303 K?
A)
606 K
done
clear
B)
273 K
done
clear
C)
403 K
done
clear
D)
303 K
done
clear
View Answer play_arrow
During dehydration of alcohols to alkenes by heating with concentrated \[{{H}_{2}}S{{O}_{4}}\] the initiation step is
A)
profanation of alcohol molecule
done
clear
B)
formation of carbocation
done
clear
C)
elimination of water
done
clear
D)
formation of an ester
done
clear
View Answer play_arrow
The correct order of magnetic moment among is
A)
\[{{[MnC{{l}_{4}}]}^{2-}}>{{[CoC{{l}_{4}}]}^{2-}}>{{[Fe{{(CN)}_{6}}]}^{4-}}\]
done
clear
B)
\[{{[MnC{{l}_{4}}]}^{2-}}>{{[Fe{{(CN)}_{6}}]}^{4-}}>{{[CoC{{l}_{4}}]}^{2-}}\]
done
clear
C)
\[{{[Fe{{(CN)}_{6}}]}^{4-}}>{{[MnC{{l}_{4}}]}^{2-}}>{{[CoC{{l}_{4}}]}^{2-}}\]
done
clear
D)
\[{{[Fe{{(CN)}_{6}}]}^{4-}}>{{[CoC{{l}_{4}}]}^{2-}}>{{[MnC{{l}_{4}}]}^{2-}}\]
done
clear
View Answer play_arrow
For the reaction \[2N{{O}_{2(g)}}2N{{O}_{(g)}}+{{O}_{2(g)}}\] (\[{{K}_{C}}=1.8\times {{10}^{-6}}\] at \[{{184}^{o}}C\] and \[R=0.0831\,kJ.mol.K\]) When \[{{K}_{P}}\] and \[{{K}_{C}}\] are compared at \[{{184}^{o}}C,\] it is found that
A)
\[{{K}_{P}}\] is greater than \[{{K}_{C}}\]
done
clear
B)
\[{{K}_{P}}\] is less than \[{{K}_{C}}\]
done
clear
C)
\[{{K}_{P}}={{K}_{C}}\]
done
clear
D)
Whether \[{{K}_{P}}\] is greater than, less than or equal to \[{{K}_{C}}\] depends upon the total gas pressure.
done
clear
View Answer play_arrow
The only cations present in a slightly acidic solution and \[F{{e}^{3+}},Z{{n}^{2+}}\] and \[C{{u}^{2+}}\]. The reagent that when added in excess to this solution would identify and separate \[F{{e}^{3+}}\] in one step is:
A)
\[2\text{ }M\text{ }HCl\]
done
clear
B)
\[\text{6 }M\text{ N}{{\text{H}}_{3}}\]
done
clear
C)
\[\text{6 }M\text{ NaOH}\]
done
clear
D)
\[{{\text{H}}_{2}}\text{S}\] gas
done
clear
View Answer play_arrow
In atomic reactor cadmium rods are used to control the chain reaction. This is because cadmium
A)
absorbs some neutrons
done
clear
B)
slows down the neutrons
done
clear
C)
speeds up neutrons
done
clear
D)
emits neutrons
done
clear
View Answer play_arrow
Which of the following solutions shows positive deviation?
A)
Water + nitric acid
done
clear
B)
Acetone + aniline
done
clear
C)
Acetone + benzene
done
clear
D)
Acetone + chloroform
done
clear
View Answer play_arrow
Which of the following has no unpaired electrons but is coloured?
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
B)
\[{{K}_{2}}Mn{{O}_{4}}\]
done
clear
C)
\[CuS{{O}_{4}}.5{{H}_{2}}O\]
done
clear
D)
\[MnC{{l}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is iron pyrites
A)
\[F{{e}_{2}}{{O}_{3}}.3{{H}_{2}}O\]
done
clear
B)
\[FeS\]
done
clear
C)
\[Fe{{S}_{2}}\]
done
clear
D)
\[F{{e}_{2}}{{S}_{3}}\]
done
clear
View Answer play_arrow
Gem dihalides are hydrolysed with either acid or basic catalysts to give
A)
aldehydes or ketones
done
clear
B)
halohydrins
done
clear
C)
carboxylic acids
done
clear
D)
gem diols
done
clear
View Answer play_arrow
3-pentanol upon reaction with \[PB{{r}_{3}}\]gives 2 and 3-bromopentane. Such rearrangement can be avoided by:
A)
using excess of \[PB{{r}_{3}}\]
done
clear
B)
choosing low temperature form reaction
done
clear
C)
converting alcohol into sulphuric or sulphonic add
done
clear
D)
converting alcohol into carboxylate esters.
done
clear
View Answer play_arrow
For the reversible system \[{{X}_{(g)}}{{Y}_{(g)}}+{{Z}_{(g)}}\] a quantity of X was heated at constant pressure P at a certain temperature. The equilibrium partial pressure of X was found to be \[\frac{P}{7}\]. What is the vlaue of \[{{K}_{P}}\] at given temperature?
A)
\[\frac{6P}{7}\]
done
clear
B)
\[\frac{9P}{7}\]
done
clear
C)
\[\frac{36P}{7}\]
done
clear
D)
\[6P\]
done
clear
View Answer play_arrow
The enthalpy of neutralization of \[NaOH\] with acetic acid is \[55\text{ }kJ\] and with \[HCl\] is\[57.1\text{ }kJ\]. This happens because
A)
some heat is required for complete ionisation of acetic acid
done
clear
B)
acetic acid is an organic acid
done
clear
C)
acetic acid is less soluble in water
done
clear
D)
acetic acid is a weak acid and required lesser alkali
done
clear
View Answer play_arrow
Choose the incorrect statement from the following
A)
The order and the molecularity of an elementary reaction are equal
done
clear
B)
The rate constant of a reaction varies with temperature
done
clear
C)
The order of a reaction is a theoretical concept
done
clear
D)
The hydrolysis of an ester in basic medium is second order reaction
done
clear
View Answer play_arrow
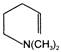
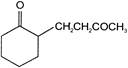
The IUPAC name of
is
A)
4'(2-oxocyclohexyl) butan-2-one
done
clear
B)
3 -(2-oxocyclohexyl) butan-3-one
done
clear
C)
4-(2-oxobutyl) cyclohexan-1-one
done
clear
D)
2-(3-oxobutyl) cyclohexan-1-one
done
clear
View Answer play_arrow
Sugar containing an impurity of common salt can be purified by crystallization from
A)
benzene
done
clear
B)
alcohol
done
clear
C)
petroleum ether
done
clear
D)
water
done
clear
View Answer play_arrow
A solid Y melts slightly above 273 K and is a poor conductor of heat and electricity. To which of the following categories does it belong?
A)
ionic solid
done
clear
B)
covalent solid
done
clear
C)
metallic
done
clear
D)
molecular
done
clear
View Answer play_arrow
In two H-atoms A and B the electrons move around the nucleus in circular orbits of radius r and 4r respectively. The ratio of the times taken by them to complete one revolution is
A)
\[1:4\]
done
clear
B)
\[1:2\]
done
clear
C)
\[1:8\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
Which of the following does not contain any co-ordinate bond?
A)
\[{{H}_{3}}{{O}^{+}}\]
done
clear
B)
\[BF_{4}^{-}\]
done
clear
C)
\[HF_{2}^{-}\]
done
clear
D)
\[NH_{4}^{+}\]
done
clear
View Answer play_arrow
A solution containing 6g urea per litre is isotonic with a solution containing
A)
\[5.85\,g\] \[NaCl\] per litre
done
clear
B)
\[34.2\text{ }g\] sucrose per litre
done
clear
C)
\[36.0\text{ }g\] glucose per litre
done
clear
D)
\[6.0\text{ }g\] sucrose per litre
done
clear
View Answer play_arrow
How many ml of \[1M\] \[{{H}_{2}}S{{O}_{4}}\] is required to neutralise \[10\text{ }ml\] of \[1\text{ }M\text{ }NaOH\] solution?
A)
\[2.5\]
done
clear
B)
\[5.0\]
done
clear
C)
\[10.0\]
done
clear
D)
\[20.0\]
done
clear
View Answer play_arrow
The half cell reactions of the cells used in hearing aids are \[Zn\xrightarrow{{}}Z{{n}^{2+}}+2{{e}^{-}}\] \[({{E}^{o}}=+0.76\,V)\] \[A{{g}_{2}}O+{{H}_{2}}O+2{{e}^{-}}\xrightarrow{{}}2Ag+2O{{H}^{-}}\] \[({{E}^{o}}=0.34\,V)\] \[{{E}^{o}}\] of the cell will be
A)
\[1.1\,V\]
done
clear
B)
\[0.42\,V\]
done
clear
C)
\[0.84\,V\]
done
clear
D)
\[3.2\,V\]
done
clear
View Answer play_arrow
Crude common salt is hygroscopic because of the presence of impurities of
A)
\[CaS{{O}_{4}}\] and \[MgS{{O}_{4}}\]
done
clear
B)
\[CaC{{l}_{2}}\] and \[MgC{{l}_{2}}\]
done
clear
C)
\[CaB{{r}_{2}}\] and \[MgB{{r}_{2}}\]
done
clear
D)
\[Ca{{(HC{{O}_{3}})}_{2}}\] and \[Mg{{(HC{{O}_{3}})}_{2}}\]
done
clear
View Answer play_arrow
Which of the following statements is wrong?
A)
Beryllium salts are extensively hydrolysed
done
clear
B)
\[C{{a}^{2+}}\] and \[M{{g}^{2+}}\] ions form complexes with EDTA
done
clear
C)
Anhydrone can be used to dry organic materials
done
clear
D)
Amongst the sulphates of alkaline earth metals \[BeS{{O}_{4}}\] is thermally least stable
done
clear
View Answer play_arrow
'Ammonal? used in bombs is a mixture of
A)
\[A{{l}_{2}}{{O}_{3}}+C\]
done
clear
B)
\[Al+A{{l}_{2}}{{O}_{3}}+{{B}_{2}}{{O}_{3}}\]
done
clear
C)
\[Al+KN{{O}_{3}}\]
done
clear
D)
\[Al+Al{{(N{{O}_{3}})}_{3}}\]
done
clear
View Answer play_arrow
Among the trihalides of nitrogen which one of most basic in nature?
A)
\[N{{F}_{3}}\]
done
clear
B)
\[NC{{l}_{3}}\]
done
clear
C)
\[N{{I}_{3}}\]
done
clear
D)
\[NB{{r}_{3}}\]
done
clear
View Answer play_arrow
During etching of glass one of the final products formed is
A)
\[Si{{F}_{4}}\]
done
clear
B)
\[N{{a}_{2}}Si{{F}_{6}}\]
done
clear
C)
\[{{H}_{2}}Si{{O}_{3}}\]
done
clear
D)
\[Si{{(OH)}_{4}}\]
done
clear
View Answer play_arrow
\[PC{{l}_{3}}\] is stored in a well stoppered bottle because
A)
It is decomposed by light
done
clear
B)
It is highly volatile
done
clear
C)
It reacts with air to form \[POC{{l}_{3}}\]
done
clear
D)
It reacts with moisture of air
done
clear
View Answer play_arrow
IUPAC nomenclature of the given organic compound will be \[{{(C{{H}_{3}})}_{2}}C(C{{H}_{2}}C{{H}_{3}})C{{H}_{2}}CH(Cl)C{{H}_{3}}\]
A)
5-chloro-3, 3-dimethylhexane
done
clear
B)
4-chloro-2-ethyl-2-methlpentane
done
clear
C)
2-chloro-4-ethyl-4-methylpentane
done
clear
D)
2-chloro-4, 4-dimethylhexane
done
clear
View Answer play_arrow
Which of the following does not show geometrical isomerism?
A)
1, 2-dichloro-l-pentene
done
clear
B)
1, 3-dichloro-2-pentene
done
clear
C)
1, 1-dichloro-l-pentene
done
clear
D)
1,4-dichloro-2-pentene
done
clear
View Answer play_arrow
The most stable carbanion among the following is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following compound gives similar products obeying markovnikov's rule and peroxide effect?
A)
\[C{{H}_{3}}-CH=C{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}CH=CHC{{H}_{3}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}-CH=CH-C{{H}_{3}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Hydration of3-phenyl but-1-ene with dil \[{{H}_{2}}S{{O}_{4}}\]mainly gives
A)
3-phenylbutan-l-ol
done
clear
B)
3-phenyl butan-2-ol
done
clear
C)
2-phenylb'utan-l-ol
done
clear
D)
2-phenyl butan-2-ol
done
clear
View Answer play_arrow
The order of increasing boiling points of
I. \[C{{H}_{3}}COCl\] II. \[{{(C{{H}_{3}}CO)}_{2}}O\] III. \[C{{H}_{3}}CON{{H}_{2}}\] IV \[C{{H}_{3}}COOH\]
A)
\[I>IV>II>III\]
done
clear
B)
\[II<I<III<IV\]
done
clear
C)
\[IV>I>II>III\]
done
clear
D)
\[I<IV<II<III\]
done
clear
View Answer play_arrow
The compound formed when malonic ester is heated with urea is
A)
Cinnamic acid
done
clear
B)
Buyric acid
done
clear
C)
Barbituric acid
done
clear
D)
Crotonic acid
done
clear
View Answer play_arrow
Mark the incorrect one (with respect to economic importance)
A)
Gelidium - Agar; Chondrus - Carrageenin
done
clear
B)
Alaria - Sarvmen; Fucus - Alginate
done
clear
C)
Rhodymenia - laver; Porphyra - Funori
done
clear
D)
Laminaria - Kombu; Macrocystic - Potash
done
clear
View Answer play_arrow
Which one of the following features are common in potato and lily family?
A. Pentamerous flower B. Actinomorphic C. Adhesion of stamens D. Bicarpellary E. Axile placentation F. Superior placentation G. Superior ovary
A)
B, D and F
done
clear
B)
A, B and E
done
clear
C)
B, C and D
done
clear
D)
B, C, E and F
done
clear
View Answer play_arrow
Chloroplast as well as mitochondria
A)
Called as semiautonomous because all enzymes of these organelles are coded by nuclear DNA
done
clear
B)
Contain cytochromes and ETS for ATP synthesis
done
clear
C)
Have SS - DNA, RNA and ribosomes
done
clear
D)
Show photophosphorylation and binary fission
done
clear
View Answer play_arrow
How many electrons (X), ATP (Y) and NADPH (Z) molecules are required for the reduction of \[2C{{O}_{2}}\]in a \[{{C}_{3}}\] plant?
A)
\[X=24,\text{ }Y=18,\text{ }Z=12\]
done
clear
B)
\[X=24,\text{ }Y=30,\text{ }Z=12\]
done
clear
C)
\[X=8,Y=6,Z=4\]
done
clear
D)
\[X=4,\text{ }Y=6,\text{ }Z=8\]
done
clear
View Answer play_arrow
Grafting is a technique of connecting two parts one scion and other stock. The stock is different from scion in
A)
Absence of buds
done
clear
B)
Presence of cambium
done
clear
C)
Presence of leaves
done
clear
D)
Absence of eustelic condition
done
clear
View Answer play_arrow
A diploid organism is heterozygous for 5 loci, if first three loci showing complete linkage and last two independent assortment then how many types of gametes can be produced?
A)
32
done
clear
B)
16
done
clear
C)
8
done
clear
D)
2
done
clear
View Answer play_arrow
Maximum number of ATP molecules are obtained after the
A)
Incomplete oxidation of two Pyruvic acid
done
clear
B)
Complete oxidation of one PGAL
done
clear
C)
Complete oxidation of one 1, 3 - di PGA
done
clear
D)
Incomplete oxidation of two glucose
done
clear
View Answer play_arrow
Biodetergents are obtained by all, except
A)
Aspergillus
done
clear
B)
Monascus
done
clear
C)
Mortierella
done
clear
D)
Bacillus
done
clear
View Answer play_arrow
As the exhaust passes through the catalytic converter
A)
\[HCs\] are converted into \[CO\] and \[{{H}_{2}}O\]
done
clear
B)
Nitric Oxides are changed to \[{{N}_{2}}\] and \[CO\]
done
clear
C)
\[CO\] and HCs are removed by oxidation
done
clear
D)
\[NO\] and \[N{{O}_{2}}\] are converted to \[C{{O}_{2}}\] and HCs
done
clear
View Answer play_arrow
Why migratory birds accumulate good amount of lipids before their migration?
A)
Lipid acts as insulator
done
clear
B)
Lipid is hydrophobic in nature
done
clear
C)
It provide high energy
done
clear
D)
It protects internal organs from mechanical stress
done
clear
View Answer play_arrow
The modified sebaceous glands present in prepuce and glands penis in human are
A)
Meibomian glands
done
clear
B)
Glands of Tyson
done
clear
C)
Harderian glands
done
clear
D)
Natrial glands
done
clear
View Answer play_arrow
Which of the following secretion contains the enzyme nucleotidase and nucleosidase?
A)
Salivary secretion
done
clear
B)
Gastric juice
done
clear
C)
Pancreatic juice
done
clear
D)
Intestinal juice
done
clear
View Answer play_arrow
Musculi pectinati are present in
A)
Atria
done
clear
B)
Ventricles
done
clear
C)
VenaCava
done
clear
D)
Capillaries
done
clear
View Answer play_arrow
Cells of the human body exhibit all the following types of movement except, mark it
A)
Amoeboid movement
done
clear
B)
Muscular movement
done
clear
C)
Ciliary movement
done
clear
D)
Wriggling movement
done
clear
View Answer play_arrow
The medulla oblongata contains centres which control
A)
Respiration
done
clear
B)
Cardiovascular reflex
done
clear
C)
Vomiting
done
clear
D)
All of these
done
clear
View Answer play_arrow
An adult male manufactures over \[{{10}^{-12}}\] to \[{{10}^{-13}}\]sperms each day. After spermiation, they are stored in
A)
Epididymis
done
clear
B)
Rete testis
done
clear
C)
AmpuIIa ofvas deferens
done
clear
D)
Vasa efferentia
done
clear
View Answer play_arrow
Progymnosperms were evolved in
A)
Ordovician period
done
clear
B)
Silurian period
done
clear
C)
Devonian period
done
clear
D)
Carboniferous period
done
clear
View Answer play_arrow
Which of the following restriction endonuclease can be related to forming blunt cut/or leave flush ends?
A)
E. Coli
done
clear
B)
Barn HI
done
clear
C)
Sma I
done
clear
D)
Sal I
done
clear
View Answer play_arrow
A : Coenogametes of Rhizopus stolonifer are morphologically similar and multinucleate. B : These are made by coenocytic and septate hyphae. Select correct options.
A)
Both A, B
done
clear
B)
Only A
done
clear
C)
Only B
done
clear
D)
None
done
clear
View Answer play_arrow
A : Cork cambium is always secondary in origin B : It is made by hypodermis always.
A)
Both A, B
done
clear
B)
Only A
done
clear
C)
Only B
done
clear
D)
None
done
clear
View Answer play_arrow
A : Opuntia and Agave plants are drought avoiding xerophytes. B : These plants show chylocauly
A)
Both A, B
done
clear
B)
Only A
done
clear
C)
Only B
done
clear
D)
None
done
clear
View Answer play_arrow
When Mosquito Day is celebrated?
A)
August 22
done
clear
B)
August 25
done
clear
C)
August 27
done
clear
D)
August 20
done
clear
View Answer play_arrow
Noise is measured in units called
A)
Hertz
done
clear
B)
Decibels
done
clear
C)
Amperes
done
clear
D)
Horse power
done
clear
View Answer play_arrow
Where is Tandoba National Park?
A)
Madhya Pradesh
done
clear
B)
Orissa
done
clear
C)
Karnataka
done
clear
D)
Maharashtra
done
clear
View Answer play_arrow
Where India's first Biosphere Reserve was first established?
A)
Namdapha
done
clear
B)
Nanda Devi
done
clear
C)
Nilgiri
done
clear
D)
Kanha
done
clear
View Answer play_arrow
Surrogate mother is
A)
Artificially inseminated female
done
clear
B)
Future carrying several embryos
done
clear
C)
Future mother with embryo implanted from another source
done
clear
D)
A mother without location
done
clear
View Answer play_arrow
Which of the fibres is not a plat product?
A)
Hemp
done
clear
B)
Cotton
done
clear
C)
Flax
done
clear
D)
Silk
done
clear
View Answer play_arrow
Population explosion has occurred in the last
A)
500 years
done
clear
B)
300 years
done
clear
C)
100 years
done
clear
D)
50 years
done
clear
View Answer play_arrow
The Tropic of cancer does not pass through
A)
Gujarat
done
clear
B)
Orissa
done
clear
C)
Tripura
done
clear
D)
West Bengal
done
clear
View Answer play_arrow
Which of the following parts of the poppy plants is used for the extraction of opium?
A)
Capsules
done
clear
B)
Flowers
done
clear
C)
Leaves
done
clear
D)
Roots
done
clear
View Answer play_arrow
In a museum large animals are kept
A)
Only in the form of Skeletons
done
clear
B)
By keeping the body stuffed
done
clear
C)
In large containers filled with preservative solution
done
clear
D)
In the form of clay modles
done
clear
View Answer play_arrow
Chromatium is
A)
Photolithotrophic
done
clear
B)
Chemolithotrophic
done
clear
C)
Photoorganotrophic
done
clear
D)
Chemoorganotrophic
done
clear
View Answer play_arrow
Non flagellated male and female gametes are formed by
A)
Ulothrix and marchantia
done
clear
B)
Cycas and Pinus
done
clear
C)
Selaginella and Batrachospermum
done
clear
D)
Spirogyra, Batrachospermum
done
clear
View Answer play_arrow
Find the correct set
A)
Tomato - Epipetalous stamens - Axile placentation - Berry fruit
done
clear
B)
Sweet Pea - Diadelphous stamens - Basal placentation - legume fruit
done
clear
C)
Shepherd's purse - Epipetalous stamens - parietal placentation - silicula fruit
done
clear
D)
Bamboo - polyandrous stamens - Basal placentation - lomentum fruit
done
clear
View Answer play_arrow
A part of the cortex is formed by tunica when
A)
Tunica shows both anticlinal and periclinal divisions
done
clear
B)
Corpus is single layered
done
clear
C)
Corpus shows only aticlinal division
done
clear
D)
Tunica is multi layered
done
clear
View Answer play_arrow
Match the column
Column - I Column - II A. Cell Membrane 1. CAC B. Mitochondria 2. Glycogen metabolism C. SER 3. Immune response D. Peroxisomes 4. Glyoxylate synthesis
A)
A-3 B-1 C-4 D-2
done
clear
B)
A-3 B-4 C-2 D-1
done
clear
C)
A-2 B-3 C-4 D-1
done
clear
D)
A-3 B-1 C-2 D-4
done
clear
View Answer play_arrow
Read the statements given below
A. Cell cycle is regulated by cyclin dependent protein kinases B. MPF is formed by cyclin \[M+CdC2\]kinase
A)
Both A and B are correct
done
clear
B)
Only A is correct
done
clear
C)
Only B is correct
done
clear
D)
Both A and B are incorrect
done
clear
View Answer play_arrow
What is incorrect for movement of water from root hair cell to xylem?
A)
Both apoplastic and symplastic
done
clear
B)
Only apoplastic from endodermis to pericycle
done
clear
C)
Mainly due to transpiration
done
clear
D)
Apoplastic in cortical area
done
clear
View Answer play_arrow
Find the incorrectly matched pair for mineral and its functions
A)
Phosphorus - Structural component of nucleoproteins and \[NAD{{P}^{+}}\]
done
clear
B)
Magnesium - Structural component of catalase and peroxidase
done
clear
C)
Copper - Structural component of cytochrome oxidase
done
clear
D)
Manganese - Photolysis of water
done
clear
View Answer play_arrow
What is incorrect for PSI?
A)
Reaction centre is \[{{P}_{700}}\]
done
clear
B)
Found in non appressed part of grana lamellae
done
clear
C)
Not involved in photophosphorylation that releases
done
clear
D)
Lies on outer side of thylakoid membrane
done
clear
View Answer play_arrow
Select the correct statement for \[{{C}_{4}}\] plants
A)
Primary \[C{{O}_{2}}\] acceptor is PEP case
done
clear
B)
Operation of both \[{{C}_{2}}\] and \[{{C}_{4}}\] cycle
done
clear
C)
Bundle sheath cells have well developed granal chloroplast
done
clear
D)
Rubisco is primary \[C{{O}_{2}}\] acceptor
done
clear
View Answer play_arrow
Which of the following conversation will yield maximum number of ATP molecules?
A)
Pyruvic acid to \[\alpha \] - ketoglutaric acid
done
clear
B)
\[\alpha \] - ketoglutaric acid to fumaric acid
done
clear
C)
Succinyl CoA to oxaloacetic acid
done
clear
D)
All will yield same number of ATP molecules
done
clear
View Answer play_arrow
Which of the given enzyme is not concerned with the irreversible reaction of glycolytic pathway?
A)
Phosphofructo kinase
done
clear
B)
Hexakinase
done
clear
C)
Triose phosphate mutase
done
clear
D)
Pyruvate kinase
done
clear
View Answer play_arrow
Family has a group of related _____ with still less number of similarities as compared to ______ and ____.
A)
Genera, Genus, Species
done
clear
B)
Classes, Genera, Species
done
clear
C)
Species, Genus, Classes
done
clear
D)
Species, Genera, Genus
done
clear
View Answer play_arrow
HaIophiles differ from eubacteria in
A)
Having different cell wall and cell membrane structure
done
clear
B)
They survival in extreme conditions
done
clear
C)
Presence of introns in DNA
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
Mark the correct statement (with respect to mycoplasma)
A)
survive in presence of oxygen only
done
clear
B)
having cell wall like cyanobacteria
done
clear
C)
such organism is generally motile
done
clear
D)
can exhibit variety of shapes
done
clear
View Answer play_arrow
A pair of leaves arise at each node and lie opposite to each other in
A)
Mustard and sunflower
done
clear
B)
Calotropic and Guava
done
clear
C)
Alstonia and Chinarose
done
clear
D)
Sunflower and Syzgium
done
clear
View Answer play_arrow
In a group of brassicaceae, fabaceae, solanaceae, liliaceae, how many family have marginal and axile placentation respectively?
A)
2, 2
done
clear
B)
1, 2
done
clear
C)
1, 1
done
clear
D)
2, 1
done
clear
View Answer play_arrow
Algae with sulphated phycocolloids are
A)
Unicellular only
done
clear
B)
Multicellular filamentous only
done
clear
C)
Mostly fresh water forms
done
clear
D)
Oogamous only
done
clear
View Answer play_arrow
Which one of the following is not a characteristic of sapwood?
A)
Present in peripheral region of secondary xylem
done
clear
B)
Physiologically inactive
done
clear
C)
Helps in water conduction
done
clear
D)
Lighter in colour
done
clear
View Answer play_arrow
Choose the correct match
A)
Proteinoplast - Protein - Castor seed
done
clear
B)
Alevroplast - Protein - Maize seed
done
clear
C)
Elaioplast - Oil and fat - Castor seed
done
clear
D)
Amyloplast - Starch - Potato Tubers
done
clear
View Answer play_arrow
During the translocation, phloem transports
A)
Water and sucrose
done
clear
B)
Ammo acids
done
clear
C)
Hormones
done
clear
D)
All of these
done
clear
View Answer play_arrow
Transpiration cools leaf surfaces, sometimes ______ by evaporative cooling.
A)
2 to 3 degrees
done
clear
B)
10 to 15 degrees
done
clear
C)
30 to 40 degrees
done
clear
D)
5 to 8 degrees
done
clear
View Answer play_arrow
CAM plants differ from \[{{C}_{4}}\] plants in
A)
Absence of wreath anatomy
done
clear
B)
Spatial separation in both carboxylations
done
clear
C)
Having Rubisco activity during day
done
clear
D)
Use of assimilatory power for \[C{{O}_{2}}\] fixation
done
clear
View Answer play_arrow
Slow oxidation of NADH to \[NA{{D}^{+}}\]occurs in
A)
Aerobic respiration
done
clear
B)
Fermentation
done
clear
C)
Dicarboxylic acid cycle
done
clear
D)
PIMP - pathway
done
clear
View Answer play_arrow
Apomictic variable or recurrent embryo cannot be produced by
A)
Adventitive embryony
done
clear
B)
Diplospory
done
clear
C)
Apospory
done
clear
D)
Aplospory
done
clear
View Answer play_arrow
Number of chromosomes in a gamete of rice is
A)
24
done
clear
B)
7
done
clear
C)
12
done
clear
D)
14
done
clear
View Answer play_arrow
Euglena shows,_______ binary fission
A)
Simple
done
clear
B)
Longitudinal
done
clear
C)
Transverse
done
clear
D)
Oblique
done
clear
View Answer play_arrow
The genes for body colour and eye colour in Drosophila are located on
A)
Chromosome - I
done
clear
B)
Chromosome - X
done
clear
C)
Chromosome - Y
done
clear
D)
Chromosome - 4
done
clear
View Answer play_arrow
Choose odd one out with respect to enzymes involved in post transcriptional processing
A)
Guanyl transferase
done
clear
B)
Ribonuclease-P
done
clear
C)
SnRNPs
done
clear
D)
Translocase
done
clear
View Answer play_arrow
The intrinsic rate of natural increase in population is represented by
A)
\[\frac{dN}{dt}\]
done
clear
B)
\[\frac{K-N}{K}\]
done
clear
C)
\[r\]
done
clear
D)
\[\frac{\Delta Nn}{N\Delta t}\]
done
clear
View Answer play_arrow
Find the odd one with respect to disease resistance variety
A)
Karan Rai
done
clear
B)
Pusa Komal
done
clear
C)
Pusa Gaurav
done
clear
D)
Pusa Shubhra
done
clear
View Answer play_arrow
Citric acid and gluconic acid are obtained through the fermentation by
A)
Acetobacter aceti
done
clear
B)
Aspergillus riger
done
clear
C)
Penicillium chrysogenum
done
clear
D)
Lacto bacillus bulgaricus
done
clear
View Answer play_arrow
Which is incorrect statement with respect to GFC or predator food chain?
A)
Major conduct energy flow in aquatic system
done
clear
B)
Begins with producers
done
clear
C)
Based on energy got from sun
done
clear
D)
Size of organisms finally reduces at higher trophic level
done
clear
View Answer play_arrow
In Delhi between 1997 and 2005 a substantial fall has been found in
A)
\[C{{O}_{2}}\] and \[S{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\] and \[CO\]
done
clear
C)
\[S{{O}_{2}}\]and \[N{{O}_{2}}\]
done
clear
D)
\[CO\] and \[N{{O}_{2}}\]
done
clear
View Answer play_arrow
The aquatic food chain, increase in concentration of toxicant at successive tropic levels is called
A)
Eutrophication
done
clear
B)
Biomagnification
done
clear
C)
Biofortification
done
clear
D)
Bioprospecting
done
clear
View Answer play_arrow
Available biomass for the consumption to herbivores and decomposers is
A)
GPP
done
clear
B)
Standing state
done
clear
C)
NPP
done
clear
D)
TLE
done
clear
View Answer play_arrow
Which of the following technique does not serve the purpose of early diagnosis?
A)
Enzyme Linked Immunosorbent Assay
done
clear
B)
Polymerase Chain Reaction
done
clear
C)
Recombinant DNA technology
done
clear
D)
Serum Analysis
done
clear
View Answer play_arrow
A useful drug that damages gastric mucosa if it is taken along with alcohol
A)
Antihistamines
done
clear
B)
Barbiturates
done
clear
C)
Aspirin
done
clear
D)
Morphine
done
clear
View Answer play_arrow
A person who has recovered from an attack of small pox or measles or mumps develops
A)
Naturally acquired passive immunity
done
clear
B)
Naturally acquired active immunity
done
clear
C)
Artificially acquired active immunity
done
clear
D)
Artificially acquired passive immunity
done
clear
View Answer play_arrow
Complete the following analogy Sponge : Archaeocytes : Hydra :
A)
Cnidoblasts
done
clear
B)
Nematocysts
done
clear
C)
Interstitial cells
done
clear
D)
Choanocytes
done
clear
View Answer play_arrow
The general body plan, body symmetry and coelom are similar in
A)
Porifera and Coelenterate
done
clear
B)
Ctenophora and platyhelminthes
done
clear
C)
Annelida and Arthropoda
done
clear
D)
Coelenterata and Echinodermata
done
clear
View Answer play_arrow
During course of evolution, complete digestive system appeared for the first time in
A)
Sponges
done
clear
B)
Mollusca
done
clear
C)
Platyhelminthes
done
clear
D)
Aschelminthes
done
clear
View Answer play_arrow
Choose the correct match
A)
Loligo - squid
done
clear
B)
Sepia-Cuttlefish
done
clear
C)
Ophiura - Sea urchin
done
clear
D)
Antedon - Sea lily
done
clear
View Answer play_arrow
If the systolic blood pressure is 130 mm of Hg and diastolic pressure is 70 mm of Hg. What will be the mean actual blood pressure?
A)
90 mm of Hg
done
clear
B)
100 mm of Hg
done
clear
C)
60 mm of Hg
done
clear
D)
50 mm of Hg
done
clear
View Answer play_arrow
Which of the following does not constitute appendicular skeleton?
A)
Coxal bone
done
clear
B)
Scapula
done
clear
C)
Humerus
done
clear
D)
Frontal
done
clear
View Answer play_arrow
In polymerase chain reaction, multiple copies of the gene of in trest is synthesized in vitro using two sets of primers and the enzyme DNA polymerase. This thermostable DNA polymerase isolated from
A)
Thermoplasma
done
clear
B)
Thermus proteus
done
clear
C)
Agrobacterium tumefaciens
done
clear
D)
Thermus aquaticus
done
clear
View Answer play_arrow
Animal husbandry is the agricultural practice of breeding and raising livestock. It is estimated that more than _______of the world livestock is in India and China
A)
20%
done
clear
B)
70%
done
clear
C)
50%
done
clear
D)
90%
done
clear
View Answer play_arrow
Which of the following are the two key concepts of Darwinian Theory of evolution?
A)
Mutation and adaptive ability
done
clear
B)
Fitness and genetic drift
done
clear
C)
Gene migration and natural selection
done
clear
D)
Branching descent and natural selection
done
clear
View Answer play_arrow
Complete the following analogy Filoriasis : Culex : Malaria :
A)
Male anopheles
done
clear
B)
Female anopheles
done
clear
C)
Male Culex
done
clear
D)
Female Aedex
done
clear
View Answer play_arrow
Which of the following drug does not depress brain activity, leads to increased energy and euphoria?
A)
Barbiturates
done
clear
B)
Benzodiazepines
done
clear
C)
Amphetamine
done
clear
D)
Morphine
done
clear
View Answer play_arrow
Choose the incorrect option with respect to cyclostomes
A)
They have an elogated body bearing 6-15 pairs of gills slits for respiration
done
clear
B)
They have a sucking and circular mouth with well-developed jaws
done
clear
C)
Their body is devoid of scales and paired fins
done
clear
D)
All living members of class cyclostomata are ectoparasite on fishes
done
clear
View Answer play_arrow
Choose odd one out
A)
Bungarus
done
clear
B)
Ancistrodon
done
clear
C)
Ophiophagus
done
clear
D)
Python
done
clear
View Answer play_arrow
Choose the correct match
A)
Dense regular connective tissue - Skin
done
clear
B)
Dense irregular connective tissue - Attach skeletal muscle to bones
done
clear
C)
Ligament - Attach one bone to another bone
done
clear
D)
Smooth muscles fibres - Heat
done
clear
View Answer play_arrow
Within the rise/increase in substrate concentration the velocity of enzymatic reaction
A)
Increases continuously
done
clear
B)
Decreases
done
clear
C)
Unaffected
done
clear
D)
Increases at first, then reaction ultimately reaches a maximum velocity which is not exceeded by any further rise in concentration of the substrate
done
clear
View Answer play_arrow
Choose the correct match with respect to vitamins and its deficiency disorder
A)
Vitamin Deficiency disorder Biotin Macrocytic anaemia
done
clear
B)
Vitamin Deficiency disorder Thiamine Severe bleeding
done
clear
C)
Vitamin Deficiency disorder Retinol Nyctalopia
done
clear
D)
Vitamin Deficiency disorder Calciferol Dermatitis
done
clear
View Answer play_arrow
Vertebrochondral ribs are
A)
1st to 7th ribs
done
clear
B)
8th, 9th and 10th ribs
done
clear
C)
11th and 12th ribs
done
clear
D)
7th, 8th and 9th ribs
done
clear
View Answer play_arrow
For which of the following hormones, there is no inhibitory control directly by hypothalamus?
A)
Prolactin
done
clear
B)
Somatrpin
done
clear
C)
Melanocyte stimulating hormone
done
clear
D)
Gonadotropin
done
clear
View Answer play_arrow
Partial pressure of \[C{{O}_{2}}\] in inspired and expired air are __ and _______ respectively
A)
159 and 116 mm of Hg
done
clear
B)
40 and 45 mm of Hg
done
clear
C)
\[0.3\]and 45 mm of Hg
done
clear
D)
\[0.3\]and 32 mm of Hg
done
clear
View Answer play_arrow
The walls of blood vassels and air sacs of lungs are involved in a functions like forming a diffusion boundary, made up of
A)
Smooth muscle fibres
done
clear
B)
A single thin layer of flattened cells with irregular boundaries
done
clear
C)
A single layer of tall and slender cells
done
clear
D)
Two or more layers of flattened cells with irregular boundaries
done
clear
View Answer play_arrow
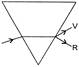
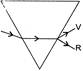
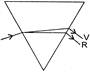
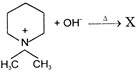 The organic product X is
The organic product X is 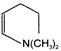
 is
is