Two cylinders A and B fitted with pistons contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is free to move while that of B is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in A is 30 K, then the rise in temperature of the gas in B is:
A)
30 K
done
clear
B)
18 K
done
clear
C)
50 K
done
clear
D)
42 K
done
clear
View Answer play_arrow
Eight equal drops are falling through air each with a steady velocity of 5 cm/s. If the drops coalesce, the terminal velocity of the new drop will be
A)
10 cm/s
done
clear
B)
15 cm/s
done
clear
C)
20 cm/s
done
clear
D)
40 cm/s
done
clear
View Answer play_arrow
A flat plate of area \[10\text{ }c{{m}^{2}}\] is separated from a large plate by a layer of glycerine 1 mm thick. If the coefficient of viscosity of gylcerine is 20 poise, the force required to keep the plate moving with a velocity of 1 cm/sec is:
A)
80 dynes
done
clear
B)
200 dynes
done
clear
C)
800 dynes
done
clear
D)
2000 dynes
done
clear
View Answer play_arrow
The reading of a spring balance when a block is suspended from it in air is 60 Newton. This reading is changed to 40 Newton when the block is submerged in water. The specific gravity of the block must therefore be:
A)
3
done
clear
B)
2
done
clear
C)
6
done
clear
D)
\[\frac{3}{2}\]
done
clear
View Answer play_arrow
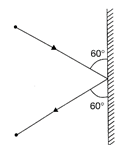
A 3 kg ball strikes a heavy rigid wall with a speed of 10 m/s at an angle of \[60{}^\circ \]. It gets reflected with the same speed and angle as shown in figure. If the ball is in contact with the wall for 0.2 sec, what is the average force exerted on the ball by the wall?
A)
0
done
clear
B)
150 N
done
clear
C)
\[150\sqrt{3}\,N\]
done
clear
D)
300 N
done
clear
View Answer play_arrow
A block rests on a rough inclined plane making an angle of \[30{}^\circ \] with the horizontal. The coefficient of static friction between the block and the plane is 0.8. If the frictional force on the block is ION, the mass of the block is (take\[g=10\text{ }m/{{s}^{2}}\])
A)
2.0 kg
done
clear
B)
4.0 kg
done
clear
C)
1.6kg
done
clear
D)
2.5kg
done
clear
View Answer play_arrow
particles of masses 2 kg each are placed such that the first lies on -ve \[x-\]axis, 2nd one lies on -ve y-axis and the third one lies on +ve z-axis at distances of 2 m, 3 m, and 1 m respectively from the origin. Then the square of the distance of the center of mass of the system from the origin is:
A)
\[1.55\,{{m}^{2}}\]
done
clear
B)
\[\sqrt{{}}(1.55){{m}^{2}}\]
done
clear
C)
\[1.55\,m\]
done
clear
D)
\[1.25\,{{m}^{2}}\]
done
clear
View Answer play_arrow
A uniform rod AB of mass m and length 2a is falling freely without rotation under gravity with AB horizontal. Suddenly the end A is fixed when the speed of the rod is u. The angular speed with which the rod begins to rotate is:
A)
\[\frac{u}{2a}\]
done
clear
B)
\[\frac{4u}{3a}\]
done
clear
C)
\[\frac{u}{3a}\]
done
clear
D)
\[\frac{3u}{4a}\]
done
clear
View Answer play_arrow
A potential difference of 1000 V is applied across an X - ray tube. The ratio of the de-Broglie wavelength of the incident electrons to the shortest wavelength of X - rays produced is (Take\[\frac{e}{m}=1.8\times {{10}^{14}}C/kg\]) for an electron.
A)
\[\frac{1}{20}\]
done
clear
B)
\[\frac{1}{100}\]
done
clear
C)
1
done
clear
D)
\[\frac{1}{{{10}^{4}}}\]
done
clear
View Answer play_arrow
The probability of survival of a radioactive nucleus for one mean life is
A)
\[\frac{1}{e}\]
done
clear
B)
\[1-\frac{1}{e}\]
done
clear
C)
\[\frac{\ln \,2}{3}\]
done
clear
D)
\[1-\frac{\ln \,2}{e}\]
done
clear
View Answer play_arrow
If \[\vec{E}\]and \[\vec{B}\]are the electric and magnetic field vectors of electromagnetic waves then the direction of propagation of electromagnetic wave is along the direction of
A)
\[\vec{E}\]
done
clear
B)
\[\vec{B}\]
done
clear
C)
\[\vec{E}\times \vec{B}\]
done
clear
D)
None of these.
done
clear
View Answer play_arrow
In a common emitter amplifier, input resistance is \[3\Omega \] and load resistance is \[24\Omega .\]What is the voltage gain? (Take\[\alpha =0.6\])
A)
8.4
done
clear
B)
4.8
done
clear
C)
2.4
done
clear
D)
1.2
done
clear
View Answer play_arrow
The displacement of a particle varies according to the relation \[x=4(\cos \pi t+\sin \pi t)\]The amplitude of the particle is:
A)
8
done
clear
B)
-4
done
clear
C)
4
done
clear
D)
\[4\sqrt{2}\]
done
clear
View Answer play_arrow
A body takes time t to reach the bottom of an inclined plane of angle \[\theta \] with the horizontal. If the plane is made rough, time taken now is 2t. The coefficient of friction of the rough surface is
A)
\[\frac{3}{4}\tan \theta \]
done
clear
B)
\[\frac{1}{2}\tan \theta \]
done
clear
C)
\[\frac{2}{3}\tan \theta \]
done
clear
D)
\[\frac{1}{4}\tan \theta \]
done
clear
View Answer play_arrow
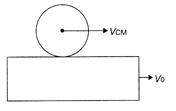
In the given figure, the sphere rolls without slipping on the plank which is moving with constant velocity \[{{v}_{0}}.\]The radius and angular velocity of the sphere is r and \[\omega \]respectively. The velocity of center of mass of the sphere is:
A)
\[{{v}_{0}}+r\omega \]
done
clear
B)
\[{{v}_{0}}-r\omega \]
done
clear
C)
\[r\omega \]
done
clear
D)
\[{{v}_{0}}\]1
done
clear
View Answer play_arrow
Two particles of masses\[{{m}_{1}}\]and\[{{m}_{2}}\]in projectile motion have velocities\[{{\vec{u}}_{1}}\]and \[{{\vec{u}}_{2}}\]respectively at time\[t=0.\]They collide at time\[{{t}_{0}}.\]Their velocities become\[{{\vec{v}}_{1}}\]and \[{{\vec{v}}_{2}}\]at time \[2{{t}_{0}}\]while still moving in air. The value of\[\left| ({{m}_{1}}{{{\vec{v}}}_{1}}+{{m}_{2}}{{{\vec{v}}}_{2}})-({{m}_{1}}{{{\vec{v}}}_{1}}+{{m}_{2}}{{{\vec{v}}}_{2}}) \right|\]
A)
0
done
clear
B)
\[({{m}_{1}}+{{m}_{2}})g{{t}_{0}}\]
done
clear
C)
\[2({{m}_{1}}+{{m}_{2}})g{{t}_{0}}\]
done
clear
D)
\[\frac{1}{2}({{m}_{1}}+{{m}_{2}})g{{t}_{0}}\]
done
clear
View Answer play_arrow
A gas in a container is compressed suddenly. Its temperature would
A)
increase
done
clear
B)
decrease
done
clear
C)
stay constant
done
clear
D)
charge depending upon surrounding temp.
done
clear
View Answer play_arrow
20 Joule of work is done on a gas when it is compressed suddenly. If the charge is adiabatic, the internal energy of the gas would increase by
A)
20 J
done
clear
B)
40 J
done
clear
C)
100 J
done
clear
D)
zero
done
clear
View Answer play_arrow
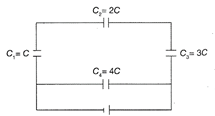
A network of four capacitors of capacity equal to \[{{C}_{1}}={{C}_{1}}=2C,\,{{C}_{3}}=3C\]and\[{{C}_{4}}=4C\]are connected to in a battery as shown in the figure. The ratio of the charges on \[{{C}_{2}}\]and \[{{C}_{4}}\]is:
A)
\[\frac{22}{3}\]
done
clear
B)
\[\frac{3}{22}\]
done
clear
C)
\[\frac{7}{4}\]
done
clear
D)
\[\frac{4}{7}\]
done
clear
View Answer play_arrow
A hollow sphere of radius 1R is charged to volt, V and another smaller sphere of radius, R is charged to V/2 volt. Then the smaller sphere is placed inside the bigger sphere without changing the net charge on each sphere. The potential difference between the two spheres would be:
A)
\[3\frac{V}{2}\]
done
clear
B)
\[\frac{V}{4}\]
done
clear
C)
\[\frac{V}{2}\]
done
clear
D)
V
done
clear
View Answer play_arrow
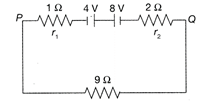
Two batteries of emf 4 V and 8 V with internal resistance\[1\Omega \] and \[2\Omega \]are connected in a circuit with a resistance of \[9\Omega \] as shown in figure. The current and potential difference between the points P and Q are:
A)
\[\frac{1}{3}A\]and\[3V\]
done
clear
B)
\[\frac{1}{6}A\] and\[4V\]
done
clear
C)
\[\frac{1}{9}A\] and\[9V\]
done
clear
D)
\[\frac{1}{2}A\] and\[12V\]
done
clear
View Answer play_arrow
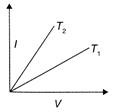
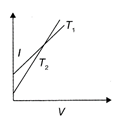
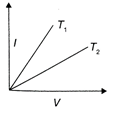
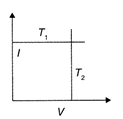
The current-voltage graphs for a given metallic wire at two different temperatures \[{{T}_{1}}\] and \[{{T}_{2}}({{T}_{2}}>{{T}_{1}})\] would be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
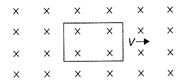
An electron beam is moving between two parallel plates having electric field \[1.125\times {{10}^{-6}}\,V/m.\] A magnetic field \[3\times {{10}^{-10}}T\] is also applied, so that beam of electrons does not deflect. The velocity of the electron is:
A)
4225 m/s
done
clear
B)
3750 m/s
done
clear
C)
2750 m/s
done
clear
D)
3200 m/s
done
clear
View Answer play_arrow
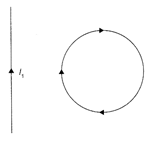
In the given figure, the loop is fixed but straight wire can move. The straight wire will:
A)
remain stationary
done
clear
B)
move towards the loop
done
clear
C)
move away from the loop
done
clear
D)
rotates about the axis
done
clear
View Answer play_arrow
A conducting square loop of side\[\ell \]and resistance R moves in its plane with a uniform velocity v perpendicular to one of its sides. A uniform and constant magnetic field B exists along the perpendicular to the plane of the loop as shown in figure. The current induced in the loop is:
A)
\[\frac{B\ell v}{R}\]clockwise
done
clear
B)
\[\frac{B\ell v}{R}\]anticlockwise
done
clear
C)
\[\frac{2B\ell v}{r}\]anticlockwise
done
clear
D)
0
done
clear
View Answer play_arrow
In a series circuit C = 2 fiF, L = 1 mH and R = 10Q, when the current in the circuit is maximum, at that time the ratio of the energies stored in the capacitor and the inductor will be
A)
1 : 1
done
clear
B)
1 : 2
done
clear
C)
2 : 1
done
clear
D)
1 : 5
done
clear
View Answer play_arrow
A person moves 30 m north and then 20 m towards east and finally \[30\sqrt{2}\,m\]in south west direction. The displacement of the person from the origin will be
A)
10 m along north
done
clear
B)
10 m along south
done
clear
C)
10 m along west
done
clear
D)
0.
done
clear
View Answer play_arrow
A body moves along a circular track of radius R. It starts from a point on the circular track, moves along the circular track and completes half a revolution. Find the ratio of its average speed to the magnitude of its average velocity
A)
\[\frac{2}{\pi }\]
done
clear
B)
\[\frac{3\pi }{2}\]
done
clear
C)
\[\frac{\pi }{2}\]
done
clear
D)
\[2\pi \]
done
clear
View Answer play_arrow
If \[\left| \vec{A}-\vec{B} \right|=\left| {\vec{A}} \right|=\left| {\vec{B}} \right|,\]the angle between A and B is
A)
\[60{}^\circ \]
done
clear
B)
\[0{}^\circ \]
done
clear
C)
\[120{}^\circ \]
done
clear
D)
\[90{}^\circ \]
done
clear
View Answer play_arrow
The angle between \[\vec{A}+\vec{B}\]and \[\vec{A}\times \vec{B}\] is
A)
0
done
clear
B)
\[\frac{\pi }{2}\]
done
clear
C)
\[\pi \]
done
clear
D)
\[\frac{\pi }{4}\]
done
clear
View Answer play_arrow
A body of mass 0.5 kg is projected under gravity with a speed of 98 m/s at an angle of \[30{}^\circ \] with the horizontal. The magnitude of its change in momentum is
A)
24.5 N-s
done
clear
B)
49.0 N-s
done
clear
C)
98.0 N-s
done
clear
D)
50.0 N-s
done
clear
View Answer play_arrow
A body is projected vertically upwards at time t = 0 and it is seen at a height Hat instants \[{{t}_{1}}\]and\[{{t}_{2}}\]seconds during its flight. The maximum height is
A)
\[\frac{g{{({{t}_{2}}-{{t}_{1}})}^{2}}}{8}\]
done
clear
B)
\[\frac{g{{({{t}_{2}}+{{t}_{1}})}^{2}}}{4}\]
done
clear
C)
\[\frac{g{{({{t}_{2}}+{{t}_{1}})}^{2}}}{8}\]
done
clear
D)
\[\frac{g{{({{t}_{2}}-{{t}_{1}})}^{2}}}{4}\]
done
clear
View Answer play_arrow
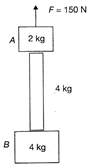
Consider the arrangement shown in figure where two blocks are connected by a uniform rope of mass 4 kg. The arrangement is lifted up vertically by a constant vertical force of magnitude 150 N. The ratio of tension in the rope at A to that at B would be
A)
2
done
clear
B)
4
done
clear
C)
\[\frac{3}{4}\]
done
clear
D)
\[\frac{5}{4}\]
done
clear
View Answer play_arrow
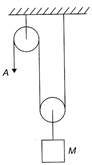
A load of mass M = 1000 kg is supported in a vertical plane by a string and pulleys. If the free end A of the string is pulled vertically downward with an acceleration of \[4\text{ }cm/{{s}^{2}},\] the tension in the string is
A)
1000 N
done
clear
B)
3000 N
done
clear
C)
4000 N
done
clear
D)
5000 N
done
clear
View Answer play_arrow
A uniform chain of mass m hangs from a light pulley, with unequal lengths of the chain hanging from the two sides of the pulley. The force exerted by the moving chain on the pulley is
A)
mg
done
clear
B)
<mg
done
clear
C)
>mg
done
clear
D)
variable
done
clear
View Answer play_arrow
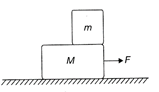
The mass m is placed on a body of mass M. There is no friction anywhere. The force F is applied on M and it moves with acceleration a. Then the force on the top body is:
A)
F
done
clear
B)
ma
done
clear
C)
F - ma
done
clear
D)
None of these
done
clear
View Answer play_arrow
A lift is moving down with acceleration a. A man in the lift drops a ball inside the lift. The acceleration of the ball as observed by the man in the lift and a man standing stationary on the ground respectively:
A)
\[g,g\]
done
clear
B)
\[g-a,\,g-a\]
done
clear
C)
\[g-a,\,g\]
done
clear
D)
\[a,g\]
done
clear
View Answer play_arrow
A tube of length L is filled completely with an incompressible liquid of mass M and closed at both ends. The tube is then rotated in a horizontal plane about one of its ends with a uniform angular velocity. The force exerted by the liquid at the other end is:
A)
\[\frac{ML{{\omega }^{2}}}{2}\]
done
clear
B)
\[ML{{\omega }^{2}}\]
done
clear
C)
\[\frac{ML{{\omega }^{2}}}{4}\]
done
clear
D)
\[\frac{M{{L}^{2}}{{\omega }^{2}}}{2}\]
done
clear
View Answer play_arrow
When forces \[{{F}_{1}},{{F}_{2}}\]and \[{{F}_{3}},\]are acting on a particle of mass m such that\[{{F}_{2}}\]and\[{{F}_{3}}\]are mutually perpendicular, then the particle remains stationary. If the force \[{{F}_{1}}\]is now removed then the acceleration of the particle is:
A)
\[\frac{{{F}_{1}}}{m}\]
done
clear
B)
\[\frac{{{F}_{2}}{{F}_{3}}}{m{{F}_{1}}}\]
done
clear
C)
\[\frac{({{F}_{2}}-{{F}_{3}})}{m}\]
done
clear
D)
\[\frac{{{F}_{3}}}{m}\]
done
clear
View Answer play_arrow
An object is placed at a distance of 10 cm from a co-axial combination of two lenses A and B in contact. The combination forms a real image three times the size of the object. If lens B is concave with a focal length of 30 cm, what is the nature and focal length of lens A?
A)
Convex, 12 cm
done
clear
B)
Concave, 12 cm
done
clear
C)
Convex, 6 cm
done
clear
D)
Convex, 18 cm
done
clear
View Answer play_arrow
A plano-convex lens acts like a concave mirror of 28 cm focal length when its plane surface is silvered and like a concave mirror of 10 cm focal length when its curved surface is silvered. What is the refractive index of the material of the lens?
A)
1.50
done
clear
B)
1.55
done
clear
C)
1.60
done
clear
D)
1.65
done
clear
View Answer play_arrow
In the expression \[A={{A}_{0}}{{e}^{-a/kT}},\]is Boltzmann constant and T is the absolute temperature. The dimensions of a are the same as those of.
A)
energy
done
clear
B)
time
done
clear
C)
acceleration
done
clear
D)
velocity
done
clear
View Answer play_arrow
If \[{{\ell }_{1}}\]and \[{{\ell }_{2}}\]are the length of an column for the first and second Resonance when a turning fork of frequency n is sounded on a resonance tube, then the distance of the anti-node from the top end of the resonance tube is
A)
\[2({{\ell }_{1}}-{{\ell }_{1}})\]
done
clear
B)
\[\frac{1}{2}(2{{\ell }_{1}}-{{\ell }_{2}})\]
done
clear
C)
\[\frac{{{\ell }_{2}}-3{{\ell }_{1}}}{2}\]
done
clear
D)
\[\frac{{{\ell }_{1}}-{{\ell }_{2}}}{2}\]
done
clear
View Answer play_arrow
The radius of second orbit of an electron in hydrogen atom is 2. 116 A. The de-Broglie wavelength associated with this electron in this orbit would be:
A)
\[6.64\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[~1.058\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[2.116\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[~13,28\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
A spherical hole is made in a solid sphere of radius R. The mass of the sphere before hollowing was M. The gravitational field at the center of the hole due to the remaining mass is:
A)
0
done
clear
B)
\[\frac{GM}{8{{R}^{2}}}\]
done
clear
C)
\[\frac{GM}{2{{R}^{2}}}\]
done
clear
D)
\[\frac{GM}{{{R}^{2}}}\]
done
clear
View Answer play_arrow
According to Heisenberg's uncertainty principle, the product of uncertainties in position and velocities for an electron of mass \[9.1\times {{10}^{-31}}\,kg\]is
A)
\[3.8\times {{10}^{-5}}{{m}^{2}}{{s}^{-1}}\]
done
clear
B)
\[6.8\times {{10}^{-3}}{{m}^{2}}{{s}^{-1}}\]
done
clear
C)
\[5.8\times {{10}^{-5}}{{m}^{2}}{{s}^{-1}}\]
done
clear
D)
\[1.8\times {{10}^{-4}}{{m}^{2}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
The state of hybridization of B in\[BC{{l}_{3}}\]is
A)
\[sp\]
done
clear
B)
\[~s{{p}^{2}}\]
done
clear
C)
\[~s{{p}^{3}}\]
done
clear
D)
\[~s{{p}^{2}}{{d}^{2}}\]
done
clear
View Answer play_arrow
If 20 g of a solute was dissolved in 500 ml of water and osmotic pressure of the solution was found to be 600 mm of Hg at \[15{}^\circ C\] , then molecular weight of the solute is
A)
900
done
clear
B)
1200
done
clear
C)
1100
done
clear
D)
1600
done
clear
View Answer play_arrow
The significant figures in 3400 are
A)
2
done
clear
B)
0
done
clear
C)
4
done
clear
D)
8
done
clear
View Answer play_arrow
For a Car not engine, the source is at 500 K and the sink is at 300 K. What is the efficiency of this engine?
A)
1.2
done
clear
B)
0.4
done
clear
C)
0.9
done
clear
D)
0.3
done
clear
View Answer play_arrow
Work done during isothermal expansion of one mole of an ideal gas form 10 atm to 1 atm at 300 K is
A)
2.938.6 Joules
done
clear
B)
1.138.8 Joules
done
clear
C)
5.744.1 Joules
done
clear
D)
7342.2 Joules
done
clear
View Answer play_arrow
The number of the sodium atoms in 2 moles of sodium Ferro cyanide is
A)
\[~10\times {{10}^{23}}\]
done
clear
B)
\[~35\times {{10}^{23}}\]
done
clear
C)
\[~28\times {{10}^{23}}\]
done
clear
D)
\[~48\times {{10}^{23}}\]
done
clear
View Answer play_arrow
According to Bohr's principle, the relation between principle quantum number (n) and radius of orbit is
A)
\[r\propto n\]
done
clear
B)
\[r\propto {{n}^{2}}\]
done
clear
C)
\[r\propto 1/n\]
done
clear
D)
\[r\propto 1/{{n}^{2}}\]
done
clear
View Answer play_arrow
For an exothermic reaction, the value of
A)
\[\Delta E \] is positive
done
clear
B)
\[\Delta E\] is negative
done
clear
C)
\[\Delta H\] is negative
done
clear
D)
\[\Delta H\] is equal to zero
done
clear
View Answer play_arrow
Wolframite ore is separated from tinstone ore by the process of
A)
Roasting
done
clear
B)
Electromagnetic separation
done
clear
C)
Calcinations
done
clear
D)
Smelting
done
clear
View Answer play_arrow
If equilibrium constant of reaction, \[{{N}_{2}}+{{O}_{2}}2NO\]is \[{{K}_{1}},\] And \[\frac{1}{2}{{N}_{2}}+\frac{1}{2}{{O}_{2}}NO\]is\[{{K}_{2}},\] then
A)
\[{{K}_{1}}=2{{K}_{2}}\]
done
clear
B)
\[{{K}_{2}}=\sqrt{{{K}_{2}}}\]
done
clear
C)
\[{{K}_{1}}={{K}_{2}}\]
done
clear
D)
\[{{K}_{1}}=\frac{1}{2}{{K}_{2}}\]
done
clear
View Answer play_arrow
The number of lattice arrangements in different crystal systems are
A)
10
done
clear
B)
8
done
clear
C)
3
done
clear
D)
14
done
clear
View Answer play_arrow
If solubility product of \[HgS{{O}_{4}}\]is\[6.4\times {{10}^{-5}}\] then its solubility is
A)
\[8\times {{1}^{-3}}\,\text{mol/litre}\]
done
clear
B)
\[2.2\times {{10}^{-5}}\,\text{mol/litre}\]
done
clear
C)
\[4.2\times {{10}^{-4}}\,\text{mil/litre}\]
done
clear
D)
\[1.4\times {{10}^{-8}}\,\text{mol/litre}\]
done
clear
View Answer play_arrow
Disintegration constant for a radioactive substance is \[0.58\,h{{r}^{-1}}.\]Its half-life period is
A)
6.2 hr
done
clear
B)
2.3 hr
done
clear
C)
1.2 hr
done
clear
D)
4.6 hr
done
clear
View Answer play_arrow
The maximum number of hydrogen bonds formed by a water molecular in ice is
A)
0
done
clear
B)
5
done
clear
C)
7
done
clear
D)
3
done
clear
View Answer play_arrow
A cylinder of 5 litre capacity, filled with air at N. T. P is connected with another evacuated cylinder of 30 litre of capacity. The resultant air pressure in both the cylinder will be
A)
10.8 cm of Hg
done
clear
B)
18.9 cm of Hg
done
clear
C)
28.8 cm of Hg
done
clear
D)
39.9 cm of Hg
done
clear
View Answer play_arrow
The rate constant of a reaction depends upon
A)
Time of reaction
done
clear
B)
Extent of reaction
done
clear
C)
Temperature of the system
done
clear
D)
Concentration of the system
done
clear
View Answer play_arrow
The volume strength of\[1.5\,N\,{{H}_{2}}{{O}_{2}}\]solution is
A)
8.4 litres
done
clear
B)
2.2 litres
done
clear
C)
5.5 litres
done
clear
D)
3.9 litres
done
clear
View Answer play_arrow
The chemical formula of diammine silver (I) chloride is
A)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
B)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
C)
\[[Ag(N{{H}_{3}})]Cl\]
done
clear
D)
\[{{[Ag(N{{H}_{4}})]}_{2}}Cl\]
done
clear
View Answer play_arrow
Acetone reacts with iodine to form iodoform in the presence of
A)
\[KOH\]
done
clear
B)
\[NaOH\]
done
clear
C)
\[MgC{{O}_{3}}\]
done
clear
D)
\[~CaC{{O}_{3}}\]
done
clear
View Answer play_arrow
When a substance is dissolved in a solvent the vapour pressure of that solvent decreases. It brings
A)
A decrease in freezing point of the solution
done
clear
B)
An increase in boiling point of the solution
done
clear
C)
A decrease in boiling point of the solution.
done
clear
D)
An increase in freezing point of the solution
done
clear
View Answer play_arrow
When alkyl halide is heated with dry \[A{{g}_{2}}O,\]it produces
A)
Ketone
done
clear
B)
Ether
done
clear
C)
Ester
done
clear
D)
Alcohol
done
clear
View Answer play_arrow
Which of the following is used widely in the man facturing of lead storage battery?
A)
Bismuth
done
clear
B)
Arsenic
done
clear
C)
Lithium
done
clear
D)
Antimony
done
clear
View Answer play_arrow
Which of the following has the highest first ionization energy?
A)
B
done
clear
B)
Li
done
clear
C)
Be
done
clear
D)
C
done
clear
View Answer play_arrow
The IUPAC name of the compound \[C{{H}_{3}}-C{{H}_{2}}-\overset{OC{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}-OH\]
A)
2-methoxy-1 -butanol
done
clear
B)
2-methoxy-1-butanol
done
clear
C)
1, 2-methoxy-butanol
done
clear
D)
3 -methoxy-1 -butanol
done
clear
View Answer play_arrow
By annealing, steel
A)
Becomes soft
done
clear
B)
Becomes hard and brittle
done
clear
C)
Is covered with a thin film of\[F{{e}_{3}}{{O}_{4}}\]
done
clear
D)
Becomes liquid
done
clear
View Answer play_arrow
Which of the following acid is formed when \[Si{{F}_{4}}\]reacts with water?
A)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[{{H}_{2}}Si{{F}_{4}}\]
done
clear
C)
\[Si{{F}_{4}}\]
done
clear
D)
\[{{H}_{2}}Si{{F}_{6}}\]
done
clear
View Answer play_arrow
How many atoms of calcium will be deposited from a solution of \[CaC{{l}_{2}}\]by a current of 5 mA flowing for 60 seconds?
A)
\[4.68\times {{10}^{18}}\]
done
clear
B)
\[~4.68\times {{10}^{9}}\]
done
clear
C)
\[~6.46\times {{10}^{14}}\]
done
clear
D)
\[~4.68\times {{10}^{23}}\]
done
clear
View Answer play_arrow
In the reaction \[A{{g}_{2}}O+{{H}_{2}}{{O}_{2}}\to 2Ag+{{H}_{2}}O+{{O}_{2}},\]the\[{{H}_{2}}{{O}_{2}}\]acts as
A)
Reducing agent
done
clear
B)
Bleaching agent
done
clear
C)
Oxidizing agent
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Iron pipes, lying in acidic soil are often attached the blocks of magnesium for their protection from rusting, because magnesium
A)
Forms a corrosion-resistant alloy with iron
done
clear
B)
Is readily converted into positive ion iron.
done
clear
C)
Prevents air from reaching the surface of iron
done
clear
D)
Is higher than iron.
done
clear
View Answer play_arrow
Which of the following statements is correct with respect to the property of elements with an increase in atomic number in the carbon family (group 14)?
A)
Metallic character decreases
done
clear
B)
Atomic size decreases
done
clear
C)
lionization energy increases
done
clear
D)
Stability of +2 oxidation state increases
done
clear
View Answer play_arrow
Phenol can be converted into salicylaldehyde by
A)
Perkin reaction
done
clear
B)
Cannizzaro reaction
done
clear
C)
Etard reaction
done
clear
D)
Reimer-Tiemann reaction
done
clear
View Answer play_arrow
The most efficient agent for the absorption of \[S{{O}_{3}}\]is
A)
\[50%\,{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[98%\,{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[80%\,{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[10%\,{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
Alkaline earth metals are denser than alkali metals, because metallic bonding in alkaline earth metal is
A)
Stronger
done
clear
B)
Not present
done
clear
C)
Weaker
done
clear
D)
Volatile
done
clear
View Answer play_arrow
Toluene is oxidized to benzoic acid by
A)
\[KMn{{O}_{4}}\]
done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
Both and
done
clear
View Answer play_arrow
Methane and ethane both can be prepared in one step by which of the following compound?
A)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
C)
\[C{{H}_{3}}Br\]
done
clear
D)
\[C{{H}_{3}}O\]
done
clear
View Answer play_arrow
Boron halides behave as Lewis base, because of their
A)
Covalent nature
done
clear
B)
Ionic nature
done
clear
C)
Acidic nature
done
clear
D)
Electron deficient nature
done
clear
View Answer play_arrow
Which of the following oil is obtained from benzene after fractional distillation of coal tar?
A)
Light oil
done
clear
B)
Anthracene oil
done
clear
C)
Heavy oil
done
clear
D)
Middle oil
done
clear
View Answer play_arrow
A biological catalyst is essentially
A)
An enzyme
done
clear
B)
An amino acid
done
clear
C)
A carbohydrate
done
clear
D)
A nitrogen compound
done
clear
View Answer play_arrow
Which of the following is the strongest base
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is the most stable compound?
A)
\[P{{h}_{3}}\overset{+}{\mathop{C}}\,\]
done
clear
B)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
C)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,H\]
done
clear
D)
\[Ph\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
View Answer play_arrow
Azo dyes are prepared from
A)
Phenol
done
clear
B)
Benzoic acid
done
clear
C)
Aniline
done
clear
D)
Benzaldehyde
done
clear
View Answer play_arrow
Haemoglobin is an example of
A)
Glycoprotein
done
clear
B)
Blood
done
clear
C)
Fibrous protein
done
clear
D)
Globular protein
done
clear
View Answer play_arrow
The complex combustion of \[C{{H}_{4}}\]gives
A)
\[CO+{{N}_{2}}O\]
done
clear
B)
\[CO+{{H}_{2}}\]
done
clear
C)
\[C{{O}_{2}}+{{H}_{2}}O\]
done
clear
D)
\[CO+{{N}_{2}}\]
done
clear
View Answer play_arrow
When glucose reacts with bromine water, the main product is
A)
Saccharin acid
done
clear
B)
Acetic acid
done
clear
C)
Glyceraldehydes
done
clear
D)
Gluconic acid.
done
clear
View Answer play_arrow
In the reaction \[C{{H}_{3}}CN+2H\xrightarrow[Either]{HCl}X\xrightarrow[{{H}_{2}}O]{Boiling}Y:\] The term Y is?
A)
Ethyl amine
done
clear
B)
Dimethyl amine
done
clear
C)
Acetaldehyde
done
clear
D)
Acetone
done
clear
View Answer play_arrow
In elastomer, intermolecular forces are
A)
Strong
done
clear
B)
Weak
done
clear
C)
Nil
done
clear
D)
Very strong
done
clear
View Answer play_arrow
Which of the following is industrially prepared by passing ethylene into hypochlorous acid?
A)
Ethylene glycol
done
clear
B)
Ethane
done
clear
C)
Ethylene dinitrate
done
clear
D)
Ethylene oxide
done
clear
View Answer play_arrow
A primary amine is formed as an amide by the treatment of bromine and alkali. The primary amine has
A)
1 carbon atom less than amide.
done
clear
B)
1 hydrogen atom more than amide.
done
clear
C)
1 carbon atom more than amide.
done
clear
D)
1 hydrogen atom less than amide.
done
clear
View Answer play_arrow
A reagent used to test for unsaturation of alkene is
A)
Ammoniacal \[AgN{{O}_{3}}\]
done
clear
B)
Concentrated \[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
Ammoniacal\[C{{u}_{2}}C{{l}_{2}}\]
done
clear
D)
Solution of \[B{{r}_{2}}\]in \[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
Which is the incorrect statement about water hyacinth?
A)
Introduced in India for their lovely flowers and shape of leaves.
done
clear
B)
It is worlds the most problematic terrestrial weed.
done
clear
C)
It reproduces asexually through offsets.
done
clear
D)
Its botanical name is Eichhornia eras sip es.
done
clear
View Answer play_arrow
Select the odd one out
A)
Tuber
done
clear
B)
Rhizome
done
clear
C)
Bulbil
done
clear
D)
Zoospores
done
clear
View Answer play_arrow
Most wild mammals are
A)
Continuous breeder
done
clear
B)
Seasonal breeder
done
clear
C)
Continuous for half year, seasonal for next half year
done
clear
D)
None of these
done
clear
View Answer play_arrow
Internal fertilization is seen in
A)
Bryophytes
done
clear
B)
Pteridophytes
done
clear
C)
Gymnosperm and Angiosperm
done
clear
D)
All of these
done
clear
View Answer play_arrow
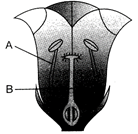
Identify A and B in the below figure?
A)
A: Anther, B: Sepals
done
clear
B)
A: Petals, B: Anther
done
clear
C)
A: Stamen, B: Carpel
done
clear
D)
A: Pistil, B: Stamen
done
clear
View Answer play_arrow
After fertilization in angiosperms, ovules develop into
A)
Pericarp
done
clear
B)
Fruit
done
clear
C)
Seed
done
clear
D)
Embryo
done
clear
View Answer play_arrow
Biologist consider flowers to be objects of
A)
Morphological marvels
done
clear
B)
Embryological marvels
done
clear
C)
Sites of sexual reproduction
done
clear
D)
All the above
done
clear
View Answer play_arrow
What is A, B, C and D the below figure?
A)
A: Thalamus, B: Style, C: Ovary, D: Stigma
done
clear
B)
A: Style, B: Ovary, C: Stigma, D: Thalamus
done
clear
C)
A: Stigma, B: Style, C: Ovary, D: Thalamus
done
clear
D)
A: Ovary, B: Stigma, C: Thalamus, D: Style
done
clear
View Answer play_arrow
Intine of pollen grain is
A)
Inner layer of pollen grain
done
clear
B)
A thin and continuous layer
done
clear
C)
Made up of cellulose and pectin
done
clear
D)
All are correct
done
clear
View Answer play_arrow
Match the following:
Column - I Column - II (A) Vegetative cell (1) Sporopollenin (B) Generative cell (2) Spindle-shaped cell (C) Exine (3) Large sized and has abundant food reserve (D) Intine (4) Cellulose and pectin
A)
A:1, B:2, C:3, D:4
done
clear
B)
A:4, B:3, C:2, D:1
done
clear
C)
A:3 B:2, C:1, D:4
done
clear
D)
A:2, B:1, C:4, D:3
done
clear
View Answer play_arrow
The landing platform for pollen grains is
A)
Stigma
done
clear
B)
Style
done
clear
C)
Ovary
done
clear
D)
None of the above
done
clear
View Answer play_arrow
How many embryo sacs are present in an ovule?
A)
One embryo sac
done
clear
B)
More than one embryo sac
done
clear
C)
One embryo sac (Generally)
done
clear
D)
Two embryo sacs
done
clear
View Answer play_arrow
Male germ cells are known as
A)
Sperms
done
clear
B)
Spermatogonia
done
clear
C)
Spermatid
done
clear
D)
Sertoli cells
done
clear
View Answer play_arrow
Which one of the following is not an accessory male duct in context of male reproductive system?
A)
Rete testis
done
clear
B)
Testes
done
clear
C)
Epididymis
done
clear
D)
Vas deferens
done
clear
View Answer play_arrow
The clitoris is a tiny ____ shaped structure which lies above the urethral opening.
A)
Flagellated
done
clear
B)
Finger like
done
clear
C)
Bean shaped
done
clear
D)
Pear
done
clear
View Answer play_arrow
The average time span of human gestation is
A)
8 months
done
clear
B)
9 months
done
clear
C)
10 months
done
clear
D)
1 year
done
clear
View Answer play_arrow
Each testis has how many testicular tubules?
A)
200
done
clear
B)
250
done
clear
C)
300
done
clear
D)
150
done
clear
View Answer play_arrow
Which of the following undergoes unequal division?
A)
Primary oocyte
done
clear
B)
Pollen grain of Angiosperm
done
clear
C)
Yeast cell during bud formation
done
clear
D)
All
done
clear
View Answer play_arrow
In context of reproductive health, STD stands for
A)
Sexually Terminal Disease
done
clear
B)
Sexually Transmitted Disease
done
clear
C)
Sexually Transformed Disease
done
clear
D)
(s) Sexually Transducted Disease
done
clear
View Answer play_arrow
?Pills' have to be taken for contraception continuously for a period of ____ days.
A)
22
done
clear
B)
21
done
clear
C)
24
done
clear
D)
25
done
clear
View Answer play_arrow
Under ZIFT procedure, zygote or embryo with up to 8 blastomeres can be transferred into the
A)
Uterus
done
clear
B)
Placenta
done
clear
C)
Fallopian tube
done
clear
D)
Cervix
done
clear
View Answer play_arrow
Every _____ person in the world is an Indian.
A)
4th
done
clear
B)
6th
done
clear
C)
8th
done
clear
D)
10th
done
clear
View Answer play_arrow
Which of the following infection can be transmitted by sharing of injection needles, surgical instruments, etc., with infected persons; through transfusion of blood or from an infected mother to the foetus?
A)
AIDS
done
clear
B)
Hepatitis B
done
clear
C)
Genital herpes
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
Government of India legalized MTP in the year
A)
1970
done
clear
B)
1971
done
clear
C)
1972
done
clear
D)
1973
done
clear
View Answer play_arrow
Mendel is famous for his work on
A)
Pisum
done
clear
B)
Drosophila
done
clear
C)
Neurospora
done
clear
D)
Oenothera
done
clear
View Answer play_arrow
Which genotype characterizes an organism that is heterozygous for two genes?
A)
RRYy
done
clear
B)
RrYY
done
clear
C)
RRYY
done
clear
D)
RrYy
done
clear
View Answer play_arrow
Cross between \[{{F}_{1}}\]plant and recessive female plant is called
A)
Back cross
done
clear
B)
Test cross
done
clear
C)
Out cross
done
clear
D)
Mutation
done
clear
View Answer play_arrow
Crossing over in diploid organism is responsible for
A)
Dominance of genes
done
clear
B)
Linkage between genes
done
clear
C)
Segregation of alleles
done
clear
D)
Recombination of linked allele
done
clear
View Answer play_arrow
Predict the characteristics from the following chart.
A)
Character is dominant and carried by \[x\] chromosome
done
clear
B)
Character is carried by y chromosome
done
clear
C)
Character is sex-linked recessive
done
clear
D)
Character is autosomal recessive
done
clear
View Answer play_arrow
The failure of segregation of chromatids during cell division cycles results in the gain or loss of a chromosome(s) is called
A)
Aneuploidy
done
clear
B)
Polyploidy
done
clear
C)
Trisomy
done
clear
D)
Nullisomy
done
clear
View Answer play_arrow
If a certain patient with blood group B requires immediate blood transfusion, the following type can be given to him
A)
O and B
done
clear
B)
O and AB
done
clear
C)
A and AB
done
clear
D)
B and AB
done
clear
View Answer play_arrow
The process of making RNA from DNA is termed as
A)
Transaction
done
clear
B)
Transformation
done
clear
C)
Transcription
done
clear
D)
Transduction
done
clear
View Answer play_arrow
Uracil is associated with which sugar?
A)
Generally deoxyribose
done
clear
B)
Generally ribose
done
clear
C)
Only ribose
done
clear
D)
Hexose
done
clear
View Answer play_arrow
In a nucleosome, histones are organized in which structure?
A)
Octamer
done
clear
B)
Hexamer
done
clear
C)
Decamer
done
clear
D)
Septamer
done
clear
View Answer play_arrow
Taylor and his colleagues used which radioactive probe to detect the distribution of newly synthesized DNA in chromosomes?
A)
Thymine.
done
clear
B)
Adenine
done
clear
C)
Guanine
done
clear
D)
Cytosine
done
clear
View Answer play_arrow
In bacteria, in context of mRNA which of the following is not true?
A)
Transcription and translocation takes place in different component.
done
clear
B)
Post-transcriptional modifications are required.
done
clear
C)
Translation occurs after transcription only.
done
clear
D)
None of these
done
clear
View Answer play_arrow
The codon AUG has dual function as it codes for _____ and also act as a _____ codon.
A)
Phenylalanine, Initiator
done
clear
B)
Methionine, Terminator
done
clear
C)
Methionine, Regulator
done
clear
D)
Methionine, Initiator
done
clear
View Answer play_arrow
Millar simulated the early earth conditions in a laboratory by passing electric discharge through a closed flask raising its temperature to \[800{}^\circ C\] and containing
A)
\[C{{H}_{4}}\] and\[{{H}_{2}}\]
done
clear
B)
\[~N{{H}_{3}}\]
done
clear
C)
Water vapour
done
clear
D)
All
done
clear
View Answer play_arrow
All these facts are true about the Neanderthal man except
A)
Brain size is 1400 cc.
done
clear
B)
Use hides to protect their bodies.
done
clear
C)
Lived in east and central Asia between 1,00,000 to 40,000 years ago.
done
clear
D)
Their fossils were discovered in Java in 1891.
done
clear
View Answer play_arrow
Match the column:
Column I Column II (A) Invertebrates (1) 500 Mya (B) Jawless fish (2) 320 Mya (C) Seaweeds (3) 350 Mya (D) Dryopithecus and Ramapithecus (4) 15 Mya
A)
A: 2, B: 1, C: 4, D: 3
done
clear
B)
A: 1, B: 3, C: 2, D: 4
done
clear
C)
A: 1, B: 4, C: 2, D: 3
done
clear
D)
A: 4, B: 3, C: 1, D: 2
done
clear
View Answer play_arrow
Life came from outer space. This theory is called
A)
Spore theory
done
clear
B)
Naturalistic theory
done
clear
C)
Special creation theory
done
clear
D)
Spontaneous generation
done
clear
View Answer play_arrow
Analogy is found between
A)
Wings of birds and bats
done
clear
B)
Wings of bat and butterfly
done
clear
C)
Hands of man and flippers of whale
done
clear
D)
Hands of man and forelimbs of horse
done
clear
View Answer play_arrow
If you want to see a dinosaur, it would be best to set the controls of your time machine for the
A)
Mesozoic era
done
clear
B)
Precambrian era
done
clear
C)
Palaeozoic era
done
clear
D)
Pleistocene period
done
clear
View Answer play_arrow
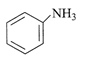
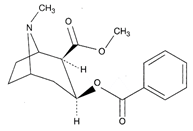
What is shown in the figure?
A)
Cannabinoid molecule
done
clear
B)
Morphine
done
clear
C)
Nicotine
done
clear
D)
Lobeline
done
clear
View Answer play_arrow
The vector of plasmodium is
A)
Male anopheles mosquito
done
clear
B)
Female anopheles mosquito
done
clear
C)
Aedes Aegypti
done
clear
D)
Sand worm
done
clear
View Answer play_arrow
Wuchereria causes
A)
Acute inflammation
done
clear
B)
Chronic inflammation
done
clear
C)
Bacterial inflammation
done
clear
D)
Viral inflammation
done
clear
View Answer play_arrow
Principle of _____ is based on the property of 'memory' of the immune system.
A)
Passive immunity
done
clear
B)
Antigen antibody
done
clear
C)
Immunization
done
clear
D)
Agglutination
done
clear
View Answer play_arrow
Which structure of the lymph system keeps degenerating from birth to puberty?
A)
Spleen
done
clear
B)
Tonsils
done
clear
C)
Peyer's patches
done
clear
D)
Thymus
done
clear
View Answer play_arrow
Tumour is an abnormal mass of
A)
Muscles
done
clear
B)
Cells
done
clear
C)
Inorganic salts
done
clear
D)
Cartilaginous membrane
done
clear
View Answer play_arrow
In India agriculture accounts for approximate _____ % of India's GDP and employs nearly ____ % of the population.
A)
33, 62
done
clear
B)
62, 33
done
clear
C)
50, 50
done
clear
D)
60, 40
done
clear
View Answer play_arrow
In which plant the resistance to yellow mosaic virus and powdery mildew were induced by mutation?
A)
Flat bean
done
clear
B)
Abelmoschus esculentus
done
clear
C)
Mung bean
done
clear
D)
Pusa A-4
done
clear
View Answer play_arrow
Which brassica variety is resistant to white rust?
A)
Pusa sawani
done
clear
B)
Pusa komal
done
clear
C)
Pusa swarnim
done
clear
D)
Pusa sadabahar
done
clear
View Answer play_arrow
The sequential steps for breeding are
(A) Screening germ plasm for resistance source (B) Selection and evaluation of hybrid (C) Hybridization of selected parents (D) Testing and release of new varieties
A)
D, C, B, A
done
clear
B)
A, B, C, D
done
clear
C)
A, C, B, D
done
clear
D)
A, C, D, B
done
clear
View Answer play_arrow
Match the columns.
Column I Column II (1) White revolution (A) Increase in milk production (2) Green revolution (B) Increase in crop production (3) Blue revolution (C) Increase in fish production
A)
A-1, B-2, C-3
done
clear
B)
A-2, B-3, C-1
done
clear
C)
A-3, B-2, C-1
done
clear
D)
A-1, B-3, C-2
done
clear
View Answer play_arrow
Which of the following is a variety of rice?
A)
Atlas-66
done
clear
B)
Himgiri
done
clear
C)
Sonalika
done
clear
D)
Ratna
done
clear
View Answer play_arrow
Choose the correct option with regard to statement A and B.
A: Lactic Acid Bacteria (LAB) are responsible for converting milk into curd. B: LAB creates acidic medium necessary to coagulate and fully digest the milk proteins.
A)
Statement A and B are correct.
done
clear
B)
Statement A and B are incorrect.
done
clear
C)
Statement A is correct but B is incorrect.
done
clear
D)
Statement B is correct and A is incorrect.
done
clear
View Answer play_arrow
The rod shaped bacteria showing flagella are magnified at _____.
A)
\[5\times {{10}^{3}}\]
done
clear
B)
\[5\times {{10}^{4}}\]
done
clear
C)
\[1.5\times {{10}^{3}}\]
done
clear
D)
\[15\times {{10}^{4}}\]
done
clear
View Answer play_arrow
Sewage is treated in _____.
A)
STD
done
clear
B)
STP
done
clear
C)
SPT
done
clear
D)
SDT
done
clear
View Answer play_arrow
BOD can be defined as the
A)
Amount of \[C{{O}_{2}}\]consumed if all the inorganic matter in one litre of water is oxidized by bacteria.
done
clear
B)
Amount of\[{{O}_{2}}\]consumed if all the organic matter in one decilitre of water is reduced by bacteria.
done
clear
C)
Amount of \[{{O}_{2}}\]consumed if all the organic matter in one litre of water is oxidized by bacteria.
done
clear
D)
Amount of \[{{O}_{2}}\] consumed if all the inorganic matter in one litre of water is oxidized by bacteria.
done
clear
View Answer play_arrow
In an anaerobic sludge digester, during digestion process produces gaseous mixture contain following gases.
A)
\[C{{H}_{4}},{{H}_{2}}O,{{O}_{2}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}},{{H}_{2}}S,C{{O}_{2}}\]
done
clear
C)
\[C{{H}_{4}},{{H}_{2}}{{O}_{2}},C{{O}_{2}}\]
done
clear
D)
\[C{{H}_{4}},{{H}_{2}}S,C{{O}_{2}}\]
done
clear
View Answer play_arrow
Methanogens produce certain gases by growing on cellulosic material. What kind of organisms are they?
A)
Fungi
done
clear
B)
Algae
done
clear
C)
Bacteria
done
clear
D)
Lichen
done
clear
View Answer play_arrow
Select the false statement from the following:
A)
Genetic engineering include techniques to alter the chemistry of genetic material (DNA and RNA).
done
clear
B)
Sexual reproduction is more advantageous than asexual reproduction.
done
clear
C)
Genetic engineering allows us to introduce desirable set of gene without undesirable gene into the target organism.
done
clear
D)
Plasmid is autonomously replicating linear extra chromosomal DNA.
done
clear
View Answer play_arrow
The way to introduce alien DNA into host cell includes
A)
Disarmed pathogens
done
clear
B)
Biolistics or gene gun
done
clear
C)
Microinjection
done
clear
D)
All the above
done
clear
View Answer play_arrow
PCR includes
A)
Denaturation
done
clear
B)
Annealing
done
clear
C)
Extension
done
clear
D)
All the above
done
clear
View Answer play_arrow
Broken ends of two DNA strands are joined by
A)
Exonuclease
done
clear
B)
Endonuclease
done
clear
C)
DNA ligase
done
clear
D)
Gyrases
done
clear
View Answer play_arrow
Transgenic plants are the ones
A)
Grown in artificial medium after hybridization in the field.
done
clear
B)
Produced by a somatic embryo in artificial medium.
done
clear
C)
Generated by introducing foreign DNA into a cell and regenerating a plant from the cell.
done
clear
D)
Produced after protoplast fusion in artificial medium.
done
clear
View Answer play_arrow
DNA or RNA segment tagged with radioactive molecules is called
A)
Vector
done
clear
B)
Probe
done
clear
C)
Clone
done
clear
D)
Plasmid
done
clear
View Answer play_arrow
Food production can be increased by
A)
Agro chemical based agriculture
done
clear
B)
Organic agriculture
done
clear
C)
Genetic engineering based agriculture
done
clear
D)
All the above
done
clear
View Answer play_arrow
Hepatitis B vaccine is a
A)
First generation vaccine
done
clear
B)
Third generation vaccine
done
clear
C)
Product of biotechnology
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
Which of the following is the source of complementary strand in mRNA silencing?
A)
An infection by viruses having RNA genome
done
clear
B)
Mobile genetic elements (Transposons)
done
clear
C)
Both
done
clear
D)
Proteins
done
clear
View Answer play_arrow
The Bt Toxin is non-toxic to human beings because
A)
The pro Bt Toxin inactivation requires temperature above human body temperature.
done
clear
B)
The Bt Toxin recognizes only insect specific target.
done
clear
C)
The Bt Toxin formation from pro Bt Toxin requires pH lower than that present in the human stomach.
done
clear
D)
Conversion of pro Bt Toxin to Bt Toxin takes place only in highly alkaline conditions.
done
clear
View Answer play_arrow
First clinical gene therapy was given to a ____ year old girl.
A)
2 year
done
clear
B)
6 year
done
clear
C)
4 year
done
clear
D)
8 year
done
clear
View Answer play_arrow
Which gene does not appear in photographic film in autoradiography?
A)
Housekeeping gene
done
clear
B)
Structural gene
done
clear
C)
Mutated gene
done
clear
D)
Transcriptionally active gene
done
clear
View Answer play_arrow
Quercus species are dominant component in
A)
Alpine forests
done
clear
B)
Scrub forests
done
clear
C)
Temperate deciduous forests
done
clear
D)
Tropical rain forests
done
clear
View Answer play_arrow
The salinity of inland waters is _____.
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
2
done
clear
View Answer play_arrow
The activates of animals like foraging, reproductive and migratory depend on
A)
Temperature
done
clear
B)
Water
done
clear
C)
Light
done
clear
D)
Air
done
clear
View Answer play_arrow
Adaptation includes ____ attribute of an organism.
A)
Behavioural
done
clear
B)
Physiological
done
clear
C)
Morphological
done
clear
D)
All the above
done
clear
View Answer play_arrow
The symptoms of altitude sickness are
A)
Nausea
done
clear
B)
Heart palpitations
done
clear
C)
Fatigue
done
clear
D)
All the above
done
clear
View Answer play_arrow
After poaching 3 lions in certain forest of Gujarat only 15 are left in a year. Determine the death rate per capita.
A)
5.000
done
clear
B)
0.200
done
clear
C)
0.166
done
clear
D)
0.800
done
clear
View Answer play_arrow
Which of the following occupy top vertical strata of forest ecosystem?
A)
Shrubs
done
clear
B)
Herbs and grasses
done
clear
C)
Tree
done
clear
D)
Phytoplankton
done
clear
View Answer play_arrow
Ocean occupy _____ % of the surface of ear but the productivity of ocean is _____ billion tons.
A)
70, 45
done
clear
B)
60, 55
done
clear
C)
70, 55
done
clear
D)
60, 65
done
clear
View Answer play_arrow
Plant capture how much of PAR?
A)
2 to 10%
done
clear
B)
10 to 20%
done
clear
C)
50%
done
clear
D)
30%
done
clear
View Answer play_arrow
If 20 kcal of energy are available at producer level, then how much energy will be transferred to the lion in the food chain: producer\[\to \]deer \[\to \]lion?
A)
0.2 Kcal
done
clear
B)
0.02 Kcal
done
clear
C)
0.002 Kcal
done
clear
D)
2 Kcal
done
clear
View Answer play_arrow
How many genetically different strain of rice are found in India?
A)
50,000
done
clear
B)
5,000
done
clear
C)
2,000
done
clear
D)
1,000
done
clear
View Answer play_arrow
Tropical environments, unlike temperate ones are
A)
Less seasonal
done
clear
B)
Relatively more constant
done
clear
C)
More predictable
done
clear
D)
All the above
done
clear
View Answer play_arrow
Loss of biodiversity in a region may lead to
A)
Decline in plants production.
done
clear
B)
Lowered resistance to environmental perturbations such as draught.
done
clear
C)
Increased variability in certain ecosystem process such as plants productivity, water use and pest and disease cycle.
done
clear
D)
All the above
done
clear
View Answer play_arrow
Eutrophication occurs because of
A)
\[Ca\] and \[Mg\]
done
clear
B)
Nitrogen and phosphorous
done
clear
C)
Carbon and oxygen
done
clear
D)
\[Hg\] and DDT
done
clear
View Answer play_arrow
National forest policy (1988) of India has recommended _____ per cent forest cover for the plains and _____ per cent for the hills.
A)
60, 40
done
clear
B)
33, 67
done
clear
C)
40, 60
done
clear
D)
67, 33
done
clear
View Answer play_arrow
Desertification is a major problem nowadays, mainly due to
A)
Over grazing
done
clear
B)
Over-cultivation
done
clear
C)
Increased urbanization
done
clear
D)
Water logging
done
clear
View Answer play_arrow
Which international treaty was signed at Montreal (Canada) in 1987 (effective in 1989) to control the emission of ozone depleting substances?
A)
Kyoto Protocol
done
clear
B)
Earth Summit
done
clear
C)
Montreal Protocol
done
clear
D)
All the above
done
clear
View Answer play_arrow