In a hydrogen atom, the binding energy of the electron in the ground state is \[{{E}_{1}}.\]Then the frequency of revolution of the electron in the nth orbit is
A)
\[\frac{2{{E}_{1}}}{{{n}^{3}}h}\]
done
clear
B)
\[\frac{2{{E}_{1}}{{n}^{3}}}{h}\]
done
clear
C)
\[\sqrt{\frac{2m{{E}_{1}}}{{{n}^{3}}h}}\]
done
clear
D)
\[\frac{{{E}_{1}}{{n}^{2}}}{h}\]
done
clear
View Answer play_arrow
A radioactive isotope is being produced at a constant rate X. Half-life of the radioactive substance is Y. After some time, the number of radioactive nuclei become constant. The value of this constant is
A)
\[\frac{XY}{\ell n(2)}\]
done
clear
B)
\[XY\]
done
clear
C)
\[XY\ln (2)\]
done
clear
D)
\[\frac{X}{Y}\]
done
clear
View Answer play_arrow
A proton and an \[\alpha -\]particle are accelerated through the same potential differences. The ratio of their de-Broglie wavelengths is
A)
\[\sqrt{2}\]
done
clear
B)
\[\frac{1}{2}\sqrt{2}\]
done
clear
C)
\[2\sqrt{2}\]
done
clear
D)
2
done
clear
View Answer play_arrow
Radiations of frequency v are incident on a photo sensitive metal. The maximum kinetic energy of the photo electrons is E. When the frequency of the incident radiations is doubled, what is the maximum kinetic energy of the photoelectrons?
A)
2E
done
clear
B)
4E
done
clear
C)
\[E+hv\]
done
clear
D)
\[E-hv\]
done
clear
View Answer play_arrow
The escape velocity of a body projected vertically upwards from the surface of the earth is d. If the body is projected at an angle of \[30{}^\circ \] with the horizontal, the escape velocity would be
A)
\[\frac{\upsilon }{2}\]
done
clear
B)
\[\sqrt{3}\frac{\upsilon }{2}\]
done
clear
C)
\[2\upsilon \]
done
clear
D)
\[\upsilon \]
done
clear
View Answer play_arrow
When a force is applied at one end of an elastic wire, it produces a strain £ in the wire. If Y is the Young's modulus of the material of the wire, the amount of energy stored per unit volume of the wire is given by
A)
\[Y\times e\]
done
clear
B)
\[\frac{1}{2}(Y\times \varepsilon )\]
done
clear
C)
\[Y\times {{\varepsilon }^{2}}\]
done
clear
D)
\[\frac{1}{2}(Y\times {{\varepsilon }^{2}})\]
done
clear
View Answer play_arrow
The wavelength \[\lambda \] of the \[{{K}_{\alpha }}\]X-ray line with an anticathode element of atomic number Z is nearly proportional to:
A)
\[{{Z}^{2}}\]
done
clear
B)
\[{{(Z-1)}^{2}}\]
done
clear
C)
\[\frac{1}{(z-1)}\]
done
clear
D)
\[\frac{1}{{{(z-1)}^{2}}}\]
done
clear
View Answer play_arrow
An open and a closed pipe have same length. The ratio of frequencies of their nth overtone is:
A)
\[\frac{n+1}{(2n+1)}\]
done
clear
B)
\[\frac{2(n+1)}{(2n+1)}\]
done
clear
C)
\[\frac{n}{(2n+1)}\]
done
clear
D)
\[\frac{(n+1)}{2n}\]
done
clear
View Answer play_arrow
Two trains, one is coming towards and another is going away from an observer both at 4 m/s produce a whistle simultaneously of frequency 300 Hz. Find the number of beats produced:
A)
5
done
clear
B)
6
done
clear
C)
7
done
clear
D)
12
done
clear
View Answer play_arrow
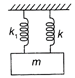
A mass is suspended separately by two springs of spring constant\[{{k}_{1}}\]and \[{{k}_{2}}\]in successive order. The time periods of oscillations in the two cases are \[{{T}_{1}}\] and \[{{T}_{2}},\]respectively. If the same mass be suspended by connecting two springs in parallel (as shown in the figure) then the time period of the oscillation is T. The correct relation is:
A)
\[{{T}^{2}}=T_{1}^{2}+T_{2}^{2}\]
done
clear
B)
\[{{T}^{-2}}=T_{1}^{-2}+T_{2}^{-2}\]
done
clear
C)
\[{{T}^{-1}}={{T}_{1}}^{-1}+{{T}_{2}}^{-1}\]
done
clear
D)
\[T={{T}_{1}}+{{T}_{2}}\]
done
clear
View Answer play_arrow
The potential energy of a simple harmonic oscillator when the particle is half way to its end point is (where E is the total energy):
A)
\[\left( \frac{1}{8} \right)E\]
done
clear
B)
\[\left( \frac{1}{4} \right)E\]
done
clear
C)
\[\left( \frac{1}{2} \right)E\]
done
clear
D)
\[\left( \frac{2}{3} \right)E\]
done
clear
View Answer play_arrow
A pendulum clock having copper rod keeps correct time at \[20{}^\circ C\]. It gains 15 seconds per day if cooled to \[0{}^\circ C\]. The coefficient of linear expansion of copper is:
A)
\[1.7\times {{10}^{-4}}/{{\,}^{o}}C\]
done
clear
B)
\[1.7\times {{10}^{-5}}/{{\,}^{o}}C\]
done
clear
C)
\[3.4\times {{10}^{-4}}/{{\,}^{o}}C\]
done
clear
D)
\[3.4\times {{10}^{-5}}/{{\,}^{o}}C\]
done
clear
View Answer play_arrow
Three stars A, B, and C appear as green, red, and blue, respectively. The star having minimum temperature is
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
all
done
clear
View Answer play_arrow
If one mole of a monatomic gas \[\gamma =\frac{5}{3}\]is mixed with one mole of a diatomic gas \[\gamma =\frac{7}{5},\]the value of \[\gamma \]for the mixture is
A)
1.40
done
clear
B)
1.50
done
clear
C)
1.53
done
clear
D)
3.07
done
clear
View Answer play_arrow
During an adiabatic process, the pressure of a gas is proportional to the cube of its absolute temperature. The value of\[\frac{{{C}_{p}}}{{{C}_{v}}}\]for the gas is:
A)
\[\frac{3}{5}\]
done
clear
B)
\[\frac{4}{3}\]
done
clear
C)
\[\frac{5}{3}\]
done
clear
D)
\[\frac{3}{2}\]
done
clear
View Answer play_arrow
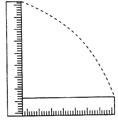
A metre stick is held vertically with one end on the floor and is then allowed to fall. If the end touching the floor is not allowed to slip, the other end will hit the ground with a velocity of (Take\[~g=9.8\text{ }m/{{s}^{2}}\])
A)
3.2 m/s
done
clear
B)
5.4 m/s
done
clear
C)
7.6 m/s
done
clear
D)
9.2 m/s
done
clear
View Answer play_arrow
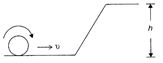
A solid sphere is rolling on a frictionless surface shown in the figure with a translational velocity \[\upsilon \,m/s.\]If the sphere climbs up to a height h then value of \[\upsilon \]should be
A)
\[\ge \sqrt{\frac{10}{7}gh}\]
done
clear
B)
\[\ge \sqrt{2gh}\]
done
clear
C)
\[\sqrt{2gha}\]
done
clear
D)
\[\frac{10}{7}gh\]
done
clear
View Answer play_arrow
In a series LCR circuit the voltage across the resistance, capacitance and inductance is 10 V each. If the capacitance is short circuited, the voltage across the inductance will be:
A)
10 V
done
clear
B)
\[10\sqrt{2}\,V\]
done
clear
C)
\[\left( \frac{10}{\sqrt{2}} \right)V\]
done
clear
D)
20 V
done
clear
View Answer play_arrow
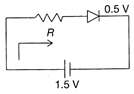
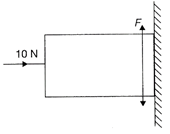
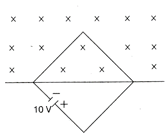
The diode used in the circuit shown in the figure has a constant voltage drop of 0.5 V at all currents and a maximum power rating of 100 milliwatts. What should be the value of the resistor R, connected in series with the diode, for obtaining maximum current?
A)
\[1.5\,\Omega \]
done
clear
B)
\[5\,\Omega \]
done
clear
C)
\[6.67\,\Omega \]
done
clear
D)
\[200\Omega \]
done
clear
View Answer play_arrow
A body at rest breaks up into 3 parts. If 2 parts having equal masses fly off perpendicularly after explosion, each with a velocity of 12 m/s, then the velocity of the third part, which has 3 times the mass of each of the other two parts is
A)
\[4\sqrt{2}\,m/s\]at an angle of \[45{}^\circ \] from each body
done
clear
B)
\[24\sqrt{2}\,m/s\] at an angle of \[135{}^\circ \] from each body
done
clear
C)
\[6\sqrt{2}\,m/s\] at \[135{}^\circ \] from each body
done
clear
D)
\[4\sqrt{2}\,m/s\] at \[135{}^\circ \] from each body
done
clear
View Answer play_arrow
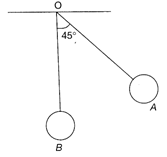
The bob A of a simple pendulum is released when the string makes an angle of \[45{}^\circ \] with the vertical. It collides with another bob B of the same material and same mass kept at rest on a table. If the collision is elastic
A)
Both A and B rise to same height
done
clear
B)
Both A and B come to rest at B
done
clear
C)
Both A and B move with the same velocity of A
done
clear
D)
A comes to rest and B moves with the velocity of A
done
clear
View Answer play_arrow
A particle of mass m at rest is acted upon by a force F for a time t. Its Kinetic energy after an interval t is
A)
\[\frac{{{F}^{2}}{{t}^{2}}}{m}+6\]
done
clear
B)
\[\frac{{{F}^{2}}{{t}^{2}}}{2m}\]
done
clear
C)
\[\frac{{{F}^{2}}{{t}^{2}}}{3m}\]
done
clear
D)
\[\frac{Ft}{2m}\]
done
clear
View Answer play_arrow
What are the dimensions of latent heat?
A)
\[M{{L}^{2}}{{T}^{-2}}\]
done
clear
B)
\[M{{L}^{-2}}{{T}^{-2}}\]
done
clear
C)
\[{{M}^{0}}L{{T}^{-2}}\]
done
clear
D)
\[{{M}^{0}}{{L}^{2}}{{T}^{-2}}\]
done
clear
View Answer play_arrow
A body travels uniformly a distance of \[(13.8\,\pm \,0.2)m\]in a time\[(4.0\,\pm \,0.3)s.\]The velocity of the body within error limits is
A)
\[(3.45\pm 0.2)m{{s}^{-1}}\]
done
clear
B)
\[(3.45\pm 0.3)m{{s}^{-1}}\]
done
clear
C)
\[(3.45\pm 0.4)m{{s}^{-1}}\]
done
clear
D)
\[(3.45\pm 0.5)m{{s}^{-1}}\]
done
clear
View Answer play_arrow
A simple pendulum with a bob of mass m swings with an angular amplitude of \[60{}^\circ \] , when its angular displacement is \[30{}^\circ \], the tension of string would be
A)
\[3\sqrt{3}\,mg\]
done
clear
B)
\[\frac{1}{2}\,mg(3\sqrt{3}-2)\]
done
clear
C)
\[\frac{1}{2}\,mg(3\sqrt{3}+2)\]
done
clear
D)
\[\frac{1}{2}\,mg(3-\sqrt{2})\]
done
clear
View Answer play_arrow
A long horizontal rod has a bead, which can slide along its length, and initially placed at a distance L from one end A of the rod. The rod is set in angular motion about A with constant angular acceleration. If the coefficient of friction between the rod and the bead is, and gravity is neglected, then the time after, which the bead starts slipping is
A)
\[\sqrt{\frac{\mu }{\alpha }}\]
done
clear
B)
\[\frac{\mu }{\alpha }\]
done
clear
C)
\[\frac{1}{\sqrt{\mu \alpha }}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A horizontal force of 10 N is necessary to just hold a block stationary against a wall. The coefficient of friction between the block and the wall is 0.2. The weight of the block is:
A)
20 N
done
clear
B)
50 N
done
clear
C)
100 N
done
clear
D)
2 N
done
clear
View Answer play_arrow
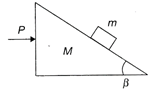
Two wooden blocks are moving on a smooth horizontal surface, such that the mass m remains stationary with respect to block of mass M as shown in the figure. The magnitude of force P is
A)
\[g\tan \,\beta \]
done
clear
B)
\[mg\,\cos \beta \]
done
clear
C)
\[(M+m)\cos ec\beta \]
done
clear
D)
\[(M+m)g\tan \beta \]
done
clear
View Answer play_arrow
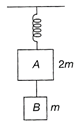
Two blocks A and B of masses 2 m and m, respectively are connected by a massless and in extensible string. The whole system is suspended by a mass-less spring as shown in the figure. The magnitude of acceleration of A and B immediately after the string is cut are respectively
A)
\[\frac{g.g}{2}\]
done
clear
B)
\[\frac{g}{2},g\]
done
clear
C)
\[g.g\]
done
clear
D)
\[\frac{g}{2},\frac{g}{2}\]
done
clear
View Answer play_arrow
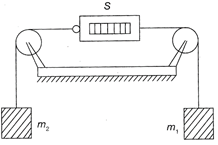
In the arrangement shown, the pulleys are fixed and ideal, the strings are light, \[{{m}_{1}}>{{m}_{2}},\]and S is a spring balance, which is itself massless. The reading of S (in units of mass) is
A)
\[{{m}_{1}}-{{m}_{2}}\]
done
clear
B)
\[\frac{1}{2}({{m}_{1}}+{{m}_{2}})\]
done
clear
C)
\[\frac{{{m}_{1}}{{m}_{2}}}{{{m}_{1}}+{{m}_{2}}}\]
done
clear
D)
\[\frac{2{{m}_{1}}{{m}_{2}}}{{{m}_{1}}+{{m}_{2}}}\]
done
clear
View Answer play_arrow
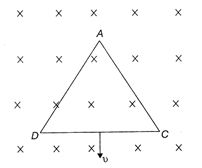
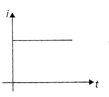
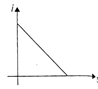
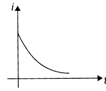
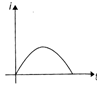
An equilateral triangular loop ADC having some resistance is pulled with a constant velocity v out of a uniform magnetic field directed into the paper. At time t = 0, side DC of the loop is the edge of the magnetic field. The induced current (i) versus time\[(t)\] graph will be as
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A square loop of side 1 m is placed in a perpendicular magnetic field. Half of the area of the loop lies inside the magnetic field. A battery of emf 10 V and negligible internal resistance is connected in the loop. The magnetic field changes with time according to the relation \[B=(0.01-2t)\]Tesla. The net emf in the circuit will be
A)
1 V
done
clear
B)
11 V
done
clear
C)
9 V
done
clear
D)
10 V
done
clear
View Answer play_arrow
A real image is formed by a convex lens. If we put it in contact with a concave lens and the combination again forms a real image, which of the following is true for the new image from the combination?
A)
Shifts towards the lens system
done
clear
B)
Shifts away from the lens system
done
clear
C)
Remains at the original position
done
clear
D)
No image is formed
done
clear
View Answer play_arrow
A convex lens of focal length f is placed somewhere in between an object and a screen so that an image is formed on the screen. The distance between the object and the screen is\[x.\]If the magnitude of magnification produced by the lens is m, the focal length of the lens is:
A)
\[\frac{mx}{{{(m+1)}^{2}}}\]
done
clear
B)
\[\frac{mx}{{{(m-1)}^{2}}}\]
done
clear
C)
\[\frac{{{(m+1)}^{2}}x}{m}\]
done
clear
D)
\[\frac{{{(m-1)}^{2}}x}{m}\]
done
clear
View Answer play_arrow
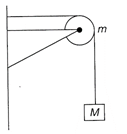
A string of negligible mass going over a clamped pulley of mass m supports a block of mass M as shown in the figure. The force on the pulley by the clamp is given by
A)
\[\sqrt{2}mg\]
done
clear
B)
\[\sqrt{2}mg\]
done
clear
C)
\[[\sqrt{{{(M+m)}^{2}}+{{m}^{2}}}]g\]
done
clear
D)
\[[\sqrt{{{(M+m)}^{2}}+{{M}^{2}}}]g\]
done
clear
View Answer play_arrow
A block of mass 4 kg is placed on a rough horizontal plane. A time dependent force \[F=k{{t}^{2}}\]acts on the block, where \[k=2\text{ }N/{{s}^{2}}.\]Coefficient of friction\[\mu =0.8.\] Force of friction between the block and the plane at t = 2 s is
A)
32 N
done
clear
B)
4 N
done
clear
C)
2 N
done
clear
D)
8 N
done
clear
View Answer play_arrow
A particle of mass m moving towards the east with speed \[\upsilon \]collides with another particle of same mass and same speed\[\upsilon \]moving towards north. If two particles stick to each other, the new particle of mass 2 m will have a speed of
A)
\[\upsilon \]
done
clear
B)
\[\frac{\upsilon }{2}\]
done
clear
C)
\[\frac{\upsilon }{\sqrt{2}}\]
done
clear
D)
\[\frac{\upsilon }{\sqrt{2}}\]
done
clear
View Answer play_arrow
Two vectors\[\vec{A}\]and\[\vec{B}\] are such that \[|\vec{A}+\vec{B}|=|\vec{A}-\vec{B}|.\] Then angle between the vectors A and B is
A)
\[0{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[180{}^\circ \]
done
clear
View Answer play_arrow
A charge q is placed at the centre of the joining the equal charges Q. The system of three charges will be in equilibrium if q is equal to
A)
\[\frac{-Q}{2}\]
done
clear
B)
\[\frac{-Q}{4}\]
done
clear
C)
\[-4Q\]
done
clear
D)
\[\frac{+Q}{2}\]
done
clear
View Answer play_arrow
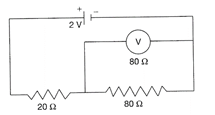
In the figure emf of the cell is 2 V and internal resistance is negligible. The resistance of the voltmeter is 80 ohm. The reading of voltmeter will be
A)
2.00 volt
done
clear
B)
1.33 volt
done
clear
C)
1.60 volt
done
clear
D)
0.80
done
clear
View Answer play_arrow
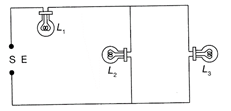
The figure shows three similar lamps \[{{L}_{1}},{{L}_{2}},\]and \[{{L}_{3}}\]connected across a power supply. If the lamp \[{{L}_{3}}\]fuse, how will the light emitted by \[{{L}_{1}}\]and\[{{L}_{2}}\]charge?
A)
No charge
done
clear
B)
Brilliance of \[{{L}_{1}}\]decreases and that of \[{{L}_{2}}\]increases
done
clear
C)
Brilliance of both \[{{L}_{1}}\]and \[{{L}_{2}}\]increases
done
clear
D)
Brilliance of both \[{{L}_{1}}\]and \[{{L}_{2}}\]decreases
done
clear
View Answer play_arrow
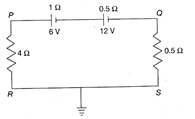
Which of the following statements is correct about the circuit shown in the figure, where \[1\Omega \]and\[0.5\Omega \]are the internal resistances of the 6 V and 12 V batteries, respectively?
A)
The potential at point P is 6 V
done
clear
B)
The potential at point Q is +0.5 V
done
clear
C)
If a voltmeter is connected across the 6 V battery, it will read 7 V
done
clear
D)
If a voltmeter is connected across the 6 V battery, it will read 5 V
done
clear
View Answer play_arrow
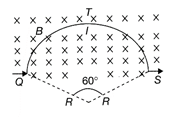
A circular arc QTS is kept in an external magnetic field \[{{\vec{B}}_{0}}\]as shown in the figure. The arc carries a current L. The magnetic field is directed normal and into the page. The force acting on the arc is
A)
\[2I{{B}_{0}}R\hat{k}\]
done
clear
B)
\[I{{B}_{0}}R\hat{k}\]
done
clear
C)
\[-2I{{B}_{0}}R\hat{k}\]
done
clear
D)
\[-I{{B}_{0}}R\hat{k}\]
done
clear
View Answer play_arrow
An equilateral triangular loop is made up of uniform wire as shown in the figure. A current/enters through one of the vertices of triangle and exits from other vertices of the triangle of side 'a' as shown. The magnitude of magnetic field at the centre of the equilateral triangle loop due to itself is
A)
\[\frac{3{{\mu }_{0}}\ell }{2\pi a}\]
done
clear
B)
\[\frac{9{{\mu }_{0}}\ell }{2\pi a}\]
done
clear
C)
\[\frac{3\sqrt{3}{{\mu }_{0}}\ell }{2\pi a}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The magnifying power of a telescope is 9. When it is adjusted for parallel rays the distance between the objective and eyepiece is 20 cm. The focal length of lenses are
A)
10 cm, 10 cm
done
clear
B)
15 cm, 5 cm
done
clear
C)
18 cm, 2 cm
done
clear
D)
11 cm, 9 cm
done
clear
View Answer play_arrow
When the electron of a hydrogen atom jumps from \[n=4\] to \[n=1\] state, the number of spectral lines emitted is
A)
9
done
clear
B)
3
done
clear
C)
6
done
clear
D)
15
done
clear
View Answer play_arrow
Consider the decomposition of \[{{N}_{2}}{{O}_{5}}\] as \[{{N}_{2}}{{O}_{5}}\to 2N{{O}_{2}}+\frac{1}{2}{{O}_{2}}\] Choose the correct option.
A)
\[4{{k}_{1}}=2k_{1}^{'}=k_{1}^{''}\]
done
clear
B)
\[4{{k}_{1}}=k_{1}^{'}=2k_{1}^{''}\]
done
clear
C)
\[{{k}_{1}}=2k_{1}^{'}=k_{1}^{''}\]
done
clear
D)
\[2{{k}_{1}}=2k_{1}^{'}=4k_{1}^{''}\]
done
clear
View Answer play_arrow
The logarithm of the equilibrium constant of the cell reaction corresponding to the cell \[X(s)|{{X}^{2+}}(aq)||{{Y}^{+}}(aq)|Y(s)\]with standard cell potential,\[E_{cell}^{o}=1.2\,V\] is given by
A)
40.2
done
clear
B)
47.2
done
clear
C)
12.5
done
clear
D)
21.5
done
clear
View Answer play_arrow
If the half-cell reactions are given as
(i)\[F{{e}^{2+}}+2e\to Fe(s);{{E}^{o}}=-0.44\,V.\] (ii)\[2{{H}^{+}}(aq)+\frac{1}{2}{{O}_{2}}(g)+2e\to {{H}_{2}}O(l);{{E}^{o}}=+1.23\,V.\]
The \[E{}^\circ \] for the reaction \[Fe\,(s)+2{{H}^{+}}+\frac{1}{2}{{O}_{2}}(g)\to F{{e}^{2+}}(aq)+{{H}_{2}}O(l)\]
A)
\[-0.79\,V\]
done
clear
B)
\[-1.67\,V\]
done
clear
C)
\[1.67\,V\]
done
clear
D)
\[0.79\,V\]
done
clear
View Answer play_arrow
The most adsorbed gas on activated charcoal is
A)
\[{{N}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[{{H}_{2}}\]
done
clear
View Answer play_arrow
Which of the following represents the arrangement in increasing order of bonds and bond dissociation energy?
A)
\[O_{2}^{2-}<O_{2}^{-}<{{O}_{2}}<O_{2}^{+}\]
done
clear
B)
\[O_{2}^{+}<O_{2}^{2-}<O_{2}^{-}<{{O}_{2}}\]
done
clear
C)
\[O_{2}^{2-}<O_{2}^{-}<O_{2}^{+}<{{O}_{2}}\]
done
clear
D)
\[{{O}_{2}}<O_{2}^{+}<O_{2}^{2-}<O_{2}^{-}\]
done
clear
View Answer play_arrow
The change of energy on freezing 1.00 kg of liquid water at \[0{}^\circ C\] and 1 atm is
A)
\[-333.4\,kJ\,k{{g}^{-1}}\]
done
clear
B)
\[-236.7\,kJ\,k{{g}^{-1}}\]
done
clear
C)
\[236.7\,kJ\,k{{g}^{-1}}\]
done
clear
D)
\[333.4\,kJ\,k{{g}^{-1}}\]
done
clear
View Answer play_arrow
\[0.1\,M\,NaCl\]and \[0.05\,M\,BaC{{l}_{2}}\] solutions are separated by a semi-permeable membrane in the container. For this system, choose the correct answer.
A)
Water flows from\[BaC{{l}_{2}}\]solution towards NaCl solution.
done
clear
B)
There is no movement of any solution across the membrane.
done
clear
C)
Osmotic pressure of 0.1 M NaCl is lower than the osmotic pressure of \[BaC{{l}_{2}}\](Assume complete dissociation).
done
clear
D)
Water flows from NaCl solution towards \[BaC{{l}_{2}}\]solution.
done
clear
View Answer play_arrow
If w is the amount of work done by the system and q is the amount of heat supplied to the system. Identify the type of the system.
A)
Open system
done
clear
B)
Isolated system
done
clear
C)
System with thermally conducing walls
done
clear
D)
Closed system
done
clear
View Answer play_arrow
the charge balance equation of species in 0.100 m acetic acid solution is given by
A)
\[[{{H}^{+}}]=[C{{H}_{3}}COO]\]
done
clear
B)
\[[{{H}^{+}}]=[O{{H}^{-}}]\]
done
clear
C)
\[2[{{H}^{+}}]=[OH]+[C{{H}_{3}}CO{{O}^{-}}]\]
done
clear
D)
\[[{{H}^{+}}]=[O{{H}^{-}}]+[C{{H}_{3}}CO{{O}^{-}}]\]
done
clear
View Answer play_arrow
The coordination number value in Hexagonal close packing is?
A)
4
done
clear
B)
6
done
clear
C)
8
done
clear
D)
12
done
clear
View Answer play_arrow
Four solutions of\[{{K}_{2}}S{{O}_{4}}\]with the following concentration 0.1 m, 0.01 m, 0.001 m and 0.0001 m are available. The maximum value of Van't Hoff factor i, corresponds to
A)
0.001 m solution
done
clear
B)
0.0001 m solution
done
clear
C)
0.1 m solution
done
clear
D)
0.01 m solution
done
clear
View Answer play_arrow
The pH of a solution prepared by mixing 2.0 ml of HCl solution of pH 3.0 and 3.0 ml of NaOH of pH 10.0 is
A)
3.5
done
clear
B)
2.5
done
clear
C)
6.5
done
clear
D)
5.5
done
clear
View Answer play_arrow
Find the following sets of quantum numbers the represents the highest energy of an atom.
A)
\[n=3,\,l=2,\,m=1,\,s=+\frac{1}{2}\]
done
clear
B)
\[n=4,\,l=0,\,m=0,\,s=+\frac{1}{2}\]
done
clear
C)
\[n=3,\,l=0,\,m=0,\,s=+\frac{1}{2}\]
done
clear
D)
\[n=3,\,l=1,\,m=1,\,s=+\frac{1}{2}\]
done
clear
View Answer play_arrow
Which of the following sets of quantum numbers represents the 19th electron in chromium?\[[Z=24\,for\,Cr]\]
A)
\[4,1,-1,\frac{1}{2}\]
done
clear
B)
\[4,0,0,\frac{1}{2}\]
done
clear
C)
\[3,2,-2,\frac{1}{2}\]
done
clear
D)
\[3,\,2,\,2,\frac{1}{2}\]
done
clear
View Answer play_arrow
The number of molecules in 100 ml of\[0.02\,N{{H}_{2}}S{{O}_{4}}\]is
A)
\[6.02\times {{10}^{20}}\]
done
clear
B)
\[6.02\times {{10}^{16}}\]
done
clear
C)
\[6.02\times {{10}^{22}}\]
done
clear
D)
\[6.02\times {{10}^{21}}\]
done
clear
View Answer play_arrow
During the conversion of\[N{{H}_{2}}O\to {{N}_{2}}O,\]the equivalent weight of \[N{{H}_{2}}OH\](molecular weight of \[N{{H}_{2}}OH\]is
A)
\[\frac{M}{4}\]
done
clear
B)
\[\frac{M}{5}\]
done
clear
C)
\[\frac{M}{2}\]
done
clear
D)
M
done
clear
View Answer play_arrow
The number of\[\beta -\]particles emitted during the transformation of \[_{y}^{x}A\]to \[_{n}^{m}B.\]
A)
\[n+\frac{x-m}{2}+y\]
done
clear
B)
\[\frac{x-m}{4}\]
done
clear
C)
\[2y-n+x-m\]
done
clear
D)
\[n+\frac{x-m}{4}-y\]
done
clear
View Answer play_arrow
In the disintegration process \[A\xrightarrow{\alpha }B\xrightarrow{-\beta }C\xrightarrow{-\beta }D\] The correct statement is
A)
A and C are isotones
done
clear
B)
A and B are isobars
done
clear
C)
A and D are isotopes
done
clear
D)
A and B are isotopes
done
clear
View Answer play_arrow
Which of the following compounds shows both Frenkel and Schottky defects?
A)
AgCl
done
clear
B)
NaCl
done
clear
C)
KCl
done
clear
D)
AgBr
done
clear
View Answer play_arrow
The pH of a solution obtained by mixing equal volumes\[\frac{N}{10}NaOH\] and \[\frac{N}{20}\,HCl\]is?
A)
12.4
done
clear
B)
1.6
done
clear
C)
2.2
done
clear
D)
13.4
done
clear
View Answer play_arrow
For the reaction \[AB\,(g)A(g)+B(g).AB\]AB is 33% dissociated at a total pressure of P. Therefore P is related to\[{{K}_{p}}\]by one of the following options
A)
\[P=3{{K}_{p}}\]
done
clear
B)
\[P={{K}_{p}}\]
done
clear
C)
\[P=8{{K}_{p}}\]
done
clear
D)
\[P=4{{K}_{p}}\]
done
clear
View Answer play_arrow
The strongest Lewis acid among boron halides is
A)
\[B{{I}_{3}}\]
done
clear
B)
\[BC{{l}_{3}}\]
done
clear
C)
\[B{{F}_{3}}\]
done
clear
D)
\[BB{{r}_{3}}\]
done
clear
View Answer play_arrow
The correct order of increasing hydration energy of the following conjugate bases of oxoacids of chlorine is
A)
\[Cl{{O}_{4}}^{-}<Cl{{O}_{3}}^{-}<Cl{{O}_{2}}^{-}<Cl{{O}^{-}}\]
done
clear
B)
\[Cl{{O}^{-}}<Cl{{O}_{2}}^{-}<Cl{{O}_{3}}^{-}<Cl{{O}_{4}}^{-}\]
done
clear
C)
\[Cl{{O}_{3}}^{-}<Cl{{O}_{4}}^{-}<Cl{{O}_{2}}^{-}<Cl{{O}^{-}}\]
done
clear
D)
\[Cl{{O}_{4}}^{-}<Cl{{O}_{3}}^{-}<Cl{{O}^{-}}<Cl{{O}_{2}}^{-}\]
done
clear
View Answer play_arrow
A solid is formed by two elements P and Q The element Q forms cubic close packing and the atoms of P occupy one third of the tetrahedral voids The formula of the compound is
A)
\[{{P}_{3}}Q\]
done
clear
B)
\[{{P}_{2}}{{Q}_{3}}\]
done
clear
C)
\[{{P}_{3}}{{Q}_{2}}\]
done
clear
D)
\[P{{Q}_{3}}\]
done
clear
View Answer play_arrow
The compound exhibiting maximum value of equivalent conductance in a fused state is
A)
\[SrC{{l}_{2}}\]
done
clear
B)
\[MgC{{l}_{2}}\]
done
clear
C)
\[BeC{{l}_{2}}\]
done
clear
D)
\[CaC{{l}_{2}}\]
done
clear
View Answer play_arrow
Which of the following have been arranged in decreasing order of oxidation number of sulphur?
A)
\[{{H}_{2}}S{{O}_{4}}>S{{O}_{2}}>{{H}_{2}}S>{{H}_{2}}{{S}_{2}}{{O}_{8}}\]
done
clear
B)
\[N{{a}_{2}}{{S}_{4}}{{O}_{6}}>{{H}_{2}}{{S}_{2}}{{O}_{7}}>N{{a}_{2}}{{S}_{2}}{{O}_{3}}>{{S}_{8}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{5}}>{{H}_{2}}S{{O}_{3}}>SC{{l}_{2}}>{{H}_{2}}S\]
done
clear
D)
\[S{{O}_{2}}^{2+}>S{{O}_{4}}^{2-}>S{{O}_{3}}^{2-}>HS{{O}_{4}}^{-}\]
done
clear
View Answer play_arrow
Thermodynamically the most stable form of carbon is
A)
Graphite
done
clear
B)
Fullerenes
done
clear
C)
Coal
done
clear
D)
Diamond
done
clear
View Answer play_arrow
The estimation of available chlorine in bleaching powder is done by
A)
Permanganometric titration
done
clear
B)
Acid base titration
done
clear
C)
Iodometric titration
done
clear
D)
Iodimetric titration
done
clear
View Answer play_arrow
The degree of hardness of water is usually expressed in terms of
A)
g/L of \[CaC{{O}_{3}}\]and\[MgC{{O}_{3}}\]present.
done
clear
B)
ppm by weight of\[MgS{{O}_{4}}.\]
done
clear
C)
ppm of\[CaC{{O}_{3}}\]actually present in water.
done
clear
D)
ppm by weight of\[CaC{{O}_{3}}\]irrespective of whether it is actually present.
done
clear
View Answer play_arrow
AgCl is dissolved in excess of each of \[\text{N}{{\text{H}}_{\text{3}}}\text{, KCN}\]and\[N{{a}_{2}}{{S}_{2}}{{O}_{3}}.\]The complex ions produced in each case are
A)
\[{{[Ag{{(N{{H}_{3}})}_{2}}]}^{2+}},{{[Ag{{(CN)}_{2}}]}^{3-}}\]and\[{{[Ag{{({{S}_{2}}{{O}_{3}})}_{2}}]}^{2-}}\]
done
clear
B)
\[{{[Ag{{(N{{H}_{3}})}_{2}}]}^{+}},{{[Ag{{(CN)}_{2}}]}^{3-}}\]and\[{{[Ag{{({{S}_{2}}{{O}_{3}})}_{2}}]}^{2-}}\]
done
clear
C)
\[{{[Ag{{(N{{H}_{3}})}_{2}}]}^{+}},{{[Ag{{(CN)}_{2}}]}^{-}}\]and\[{{[Ag{{({{S}_{2}}{{O}_{3}})}_{2}}]}^{3-}}\]
done
clear
D)
\[{{[Ag{{(N{{H}_{3}})}_{4}}]}^{2+}},{{[Ag{{(CN)}_{2}}]}^{3-}}\]and\[{{[A{{g}_{2}}{{({{S}_{2}}{{O}_{3}})}_{2}}]}^{2-}}\]
done
clear
View Answer play_arrow
The most stable complex among the following is
A)
\[[Fe{{(CO)}_{5}}]\]
done
clear
B)
\[{{[Pd{{(CN)}_{4}}]}^{4-}}\]
done
clear
C)
\[{{[Ni{{(CN)}_{4}}]}^{3-}}\]
done
clear
D)
\[{{[Ni{{(CN)}_{4}}]}^{4-}}\]
done
clear
View Answer play_arrow
The maximum number of P-H bonds is contained in which of the following molecules?
A)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
C)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\]
done
clear
D)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
View Answer play_arrow
The bond order of the N-O bonds in \[N{{O}_{3}}^{-}\]ion is
A)
1.33
done
clear
B)
1.50
done
clear
C)
1.00
done
clear
D)
0.33
done
clear
View Answer play_arrow
Which of the following fluoride of xenon has zero dipole moment?
A)
\[Xe{{F}_{3}}\]
done
clear
B)
\[Xe{{F}_{4}}\]
done
clear
C)
\[Xe{{F}_{6}}\]
done
clear
D)
\[Xe{{F}_{2}}\]
done
clear
View Answer play_arrow
Choose the correct statement.
A)
Square planar complexes are more stable than octahedral complexes.
done
clear
B)
\[{{[CO{{(N{{H}_{3}})}_{6}}]}^{2+}}\] is oxidized to diamagnetic \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\] by the oxygen in air.
done
clear
C)
The\[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] ion has a tetrahedral geometry and is diamagnetic.
done
clear
D)
\[{{[Fe{{(CN)}_{6}}]}^{3-}}\]is stable but\[{{[Fe{{F}_{6}}]}^{3-}}\] is unstable.
done
clear
View Answer play_arrow
The hydrolysis of\[NC{{l}_{3}}\]by water produces
A)
\[N{{H}_{2}}N{{H}_{2}}\]and\[HOCl\]
done
clear
B)
\[N{{H}_{2}}OH\]and\[HOCl\]
done
clear
C)
\[N{{H}_{2}}Cl\]and HOCl
done
clear
D)
\[N{{H}_{4}}OH\]and\[HOCl\]
done
clear
View Answer play_arrow
The solubilities of \[N{{a}_{2}}S{{O}_{4}},\text{ }BeS{{O}_{4}},\text{ }MgS{{O}_{4}}\]and\[BaS{{O}_{4}}\] will follow the order
A)
\[BeS{{O}_{4}}>Na2S{{O}_{4}}>MgS{{O}_{4}}>BaS{{O}_{4}}\]
done
clear
B)
\[BeS{{O}_{4}}>MgS{{O}_{4}}>N{{a}_{2}}S{{O}_{4}}>BaS{{O}_{4}}\]
done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}>BeS{{O}_{4}}>MgS{{O}_{4}}>BaS{{O}_{4}}\]
done
clear
D)
\[MgS{{O}_{4}}>BeS{{O}_{4}}>N{{a}_{2}}S{{O}_{4}}>BaS{{O}_{4}}\]
done
clear
View Answer play_arrow
Among the following, the third ionization energy is highest for
A)
Aluminium
done
clear
B)
Beryllium
done
clear
C)
Boron
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
The highest lattice energy corresponds to
A)
SrO
done
clear
B)
BaO
done
clear
C)
MgO
done
clear
D)
CaO
done
clear
View Answer play_arrow
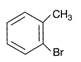
Which of the following is an appropriate set of reactants for the preparation of 1 -methoxy-4-nitrobenzene?
[a] [b]
A)
A
done
clear
B)
B
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
When subjected to acid catalysed hydration, the order of reactivity of the alkenes; \[{{(C{{H}_{3}})}_{2}}C=C{{H}_{2}}(I), C{{H}_{3}}CH=C{{H}_{2}}(II),\] and \[C{{H}_{2}}=C{{H}_{2}}\,(III)\] is
A)
I > III > II
done
clear
B)
III > II > I
done
clear
C)
II > I > III
done
clear
D)
I > II > III
done
clear
View Answer play_arrow
Name the reaction that used to bring about the following transformation; But-2-ene to ethanol.
A)
\[Cr{{O}_{2}}C{{l}_{2}}/{{H}_{3}}{{O}^{+}}\]
done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]in acidic medium
done
clear
C)
\[{{O}_{3}}/{{H}_{2}}O-Zn\]dust
done
clear
D)
PCC
done
clear
View Answer play_arrow
Arrange the following in increasing order of their basic strength:\[C{{H}_{3}}N{{H}_{2}}(I),{{(C{{H}_{3}})}_{2}}NH(II),\]\[{{(C{{H}_{3}})}_{3}}N\]\[(III),\,{{C}_{6}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}(IV)\]
A)
\[IV<III<I<II\]
done
clear
B)
\[IV<III<II<I\]
done
clear
C)
\[IV<III<II<I\]
done
clear
D)
\[I<II<III<IV\]
done
clear
View Answer play_arrow
Arrange the following in increasing order of their intermolecular forces: Nylon-6,6 (I), Buna-S (II), Polythene (III)
A)
I, II, III
done
clear
B)
II, III, I
done
clear
C)
II, I, III
done
clear
D)
III, II, I
done
clear
View Answer play_arrow
The\[p{{K}_{{{a}_{1}}}}\]and\[p{{K}_{{{a}_{2}}}}\]of an amino acid are 2.3 and 9.7 respectively. The isoelectric point of the amino acid is
A)
7.4
done
clear
B)
3.5
done
clear
C)
12.0
done
clear
D)
6.0
done
clear
View Answer play_arrow
The transfer RNA anticodon for the messenger RNA codon G-C-A is
A)
U-D-C
done
clear
B)
G-C-U
done
clear
C)
G-U-C
done
clear
D)
C-G-U
done
clear
View Answer play_arrow
In the following reaction,
The major product obtained is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
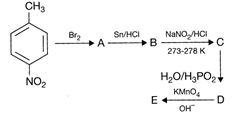
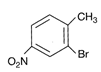
Identify the product (E) in the following sequence of reactions.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Arrange the following compounds in increasing order of their reactivity in nucleophilic addition reactions. Ethanal (I), Propanal (II), Propanone (III), Butanone (IV).
A)
II < I < III < IV
done
clear
B)
III < II < I < IV
done
clear
C)
I < II < III < IV
done
clear
D)
IV < III < II < I
done
clear
View Answer play_arrow
Select the correct statement from the following options.
A)
Increase in substrate concentration, increases the velocity of enzymatic reaction at first then it reaches maximum ant further increment does not occur.
done
clear
B)
In competitive inhibition, the inhibitor closely resembles the substrate in its molecular structure.
done
clear
C)
Competitive inhibition are often used in the control of bacterial pathogens.
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Ribosomes are the granular structure first observed under the electron microscope as dense particle by which scientist?
A)
Robert Brown (1831)
done
clear
B)
George Palade (1953)
done
clear
C)
Camillo Golgi (1898)
done
clear
D)
Singer and Nicolson (1972)
done
clear
View Answer play_arrow
Find out the total number of false statements given below.
[1] Cyanobacteria have chlorophyll 'a5 like green plants [2] Bacteria which oxidises various inorganic substance such as nitrates and ammonia and use the released energy for ATP production are chemo synthesis autotrophic bacteria. [3] Heterotrophic bacteria are less in abundance by nature. [4] Majority of heterotrophic bacteria are decomposers. [5] Chemosynthetic autotrophic bacteria play important role in recycling of nutrients like nitrogen, phosphrous iron and sulpur
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
Micro-bodies which contain enzyme for \[\beta -\]oxidation of fatty acid and glyoxylate pathway.
A)
Sphaerosome
done
clear
B)
Peroxisomes
done
clear
C)
Glyoxysomes
done
clear
D)
Vacuole
done
clear
View Answer play_arrow
S-phase not characterised by
A)
DNA duplication
done
clear
B)
No increase in chromosome number
done
clear
C)
DNA replication
done
clear
D)
Duplication of centriole in the nucleus of eukaryotic animal cell.
done
clear
View Answer play_arrow
Which of the following biomolecules is common to respiration mediated breakdown of fats, carbohydrates and proteins?
A)
Fructose 1,6-bisphosphate
done
clear
B)
Pyruvic acid
done
clear
C)
Acetyl-CoA
done
clear
D)
Glucose 6-phosphate
done
clear
View Answer play_arrow
A few drops of sap were collected by cutting across a plant stem by a suitable method. The sap was tested chemically. Which one of the following test results indicates that it was phloem sap?
A)
Alkaline
done
clear
B)
Low refractive index
done
clear
C)
Absence of sugar
done
clear
D)
Acidic
done
clear
View Answer play_arrow
You are given a tissue with its potential for differentiation in an artificial culture. Which of the following pairs of hormones would you add to the medium to secure shoots as well as roots?
A)
Auxin and cytokinin
done
clear
B)
Auxin and abscisic acid
done
clear
C)
Gibberellin and abscisic acid
done
clear
D)
IAA and gibberellin
done
clear
View Answer play_arrow
Enzyme nitrogenase is
A)
Mo - Fe protein
done
clear
B)
Mo - Mn protein
done
clear
C)
Mn - Fe protein
done
clear
D)
Cu - Fe protein
done
clear
View Answer play_arrow
Deficiency symptom of N, K and Mg appears in
A)
Young leaf
done
clear
B)
Meristematic tissue
done
clear
C)
Young stem
done
clear
D)
Senescent leaves
done
clear
View Answer play_arrow
Phosphorylation occurs in
A)
Mitochondria
done
clear
B)
Chloroplast
done
clear
C)
Cytoplasm
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Find the incorrect statement?
A)
Large number of gametes is released in the surrounding water in external fertilisation.
done
clear
B)
Offspring produced by external fertilisation are extremely vulnerable to predators.
done
clear
C)
External fertilisation is shown by bony fishes and frogs.
done
clear
D)
In seed plant, the motile male gametes are carried to female gamete by pollen tube.
done
clear
View Answer play_arrow
Which one of the following generate new genetic combinations leading to variation?
A)
Parthenogenesis
done
clear
B)
Sexual reproduction
done
clear
C)
Nucellar polyembrony
done
clear
D)
Vegetative reproduction
done
clear
View Answer play_arrow
Replum occur in the ovary of
A)
Pea
done
clear
B)
Lemon
done
clear
C)
Mustard
done
clear
D)
Sunflower
done
clear
View Answer play_arrow
In majority of angiosperm.
A)
Pollen grains are shed at 2-celled stage.
done
clear
B)
There are numerous egg cells.
done
clear
C)
Antipodal cells have a filiform apparatus.
done
clear
D)
Nucellus is developed into embryo sac.
done
clear
View Answer play_arrow
Pollination in water hyacinth and water lily is brought about by the agency of
A)
Insects or wind
done
clear
B)
Birds
done
clear
C)
Bats
done
clear
D)
Water
done
clear
View Answer play_arrow
Male gametophyte in angiosperm is represented by
A)
Anther
done
clear
B)
Androecium
done
clear
C)
Microsporangium
done
clear
D)
Pollen grain
done
clear
View Answer play_arrow
The experiment which provides unequivocal proof that DNA is the genetic material came from the experiment of
A)
Watson and Crick
done
clear
B)
Wilkins and Franklin
done
clear
C)
Hershey and Chase
done
clear
D)
Avery, MacLeod and Maclyn McCarty
done
clear
View Answer play_arrow
The phenomenon of having an extra chromosome segment attached to its normal homologous chromo- some so that a gene or set of genes is represented twice in the same chromosome.
A)
Duplication
done
clear
B)
Crossing over
done
clear
C)
Translocation
done
clear
D)
Inversion
done
clear
View Answer play_arrow
In eukaryotes, RNA polymerase III is responsible for synthesis of
A)
28S RNA, 18S RNA and 5.8S RNA
done
clear
B)
tRNA, hnRNA, rRNA
done
clear
C)
tRNA, 5S RNA, snRNA
done
clear
D)
hnRNA, tRNA, rRNA
done
clear
View Answer play_arrow
A true breeding plant is
A)
Produced due to cross-pollination among unrelated plants.
done
clear
B)
Near homozygous and produces offspring of its own kind.
done
clear
C)
Always homozygous recessive in this genetic constitution,
done
clear
D)
One that is able to breed on its own.
done
clear
View Answer play_arrow
Satellite DNA is a useful tool in
A)
Organ transplantation
done
clear
B)
Sex determination
done
clear
C)
Forensic science
done
clear
D)
Genetic engineering
done
clear
View Answer play_arrow
Stirred-tank bioreactors have been designed for
A)
Addition of preservatives to the product.
done
clear
B)
Availability of oxygen throughout the process.
done
clear
C)
Ensuring anaerobic conditions in the culture vessel.
done
clear
D)
Purification of the product.
done
clear
View Answer play_arrow
An analysis of chromosomal DNA using the Southern hybridisation technique does not use
A)
Electrophoresis
done
clear
B)
Blotting
done
clear
C)
Autoradiography
done
clear
D)
PCR
done
clear
View Answer play_arrow
Which of the following is not a component of downstream processing?
A)
Purification
done
clear
B)
Preservation
done
clear
C)
Expression
done
clear
D)
Separation
done
clear
View Answer play_arrow
Following restriction enzymes produces blunt ends except.
A)
Alu I
done
clear
B)
Sma I
done
clear
C)
Sea I
done
clear
D)
Hind III
done
clear
View Answer play_arrow
Which GMO/transgenic animal are used in testing the safety of polio vaccine before they are used on human?
A)
Transgenic sheep
done
clear
B)
Transgenic cow
done
clear
C)
Transgenic viruses
done
clear
D)
Transgenic mice
done
clear
View Answer play_arrow
Diapause is
A)
Adaptation to terrestrial life.
done
clear
B)
Stage of suspended development seen in unfavourable conditions in many zooplankton species in lakes and ponds.
done
clear
C)
Method of migration from stressful habitat to a more hospitable area.
done
clear
D)
It is a type of Symbiosis.
done
clear
View Answer play_arrow
Orderly and sequential changes in communities, parallel with the changes in the physical environment is known as
A)
Ecological succession
done
clear
B)
Extinction
done
clear
C)
Divergent evolution
done
clear
D)
Convergent evolution
done
clear
View Answer play_arrow
Choose the correct statement:
A)
k-selection occurs in environment in which population densities fluctuate well below the carrying capacity.
done
clear
B)
k-selection occurs in environment in which individuals are likely to fall little competition.
done
clear
C)
r-selection tends to maximise the population size and operates in populations living at a density near the limit imposed by their resources.
done
clear
D)
k-selection is also known as density dependent selection.
done
clear
View Answer play_arrow
In which of the following interaction only one of the species benefited?
A)
Mutualism
done
clear
B)
Competition
done
clear
C)
Parasitism and Predation
done
clear
D)
Amensalism
done
clear
View Answer play_arrow
Hotspots of biodiversity are
A)
Areas of earth that contain many endemic species.
done
clear
B)
Species serves as proxy for entire communities in particular areas.
done
clear
C)
Species in particular niche.
done
clear
D)
Species diversity at particular area.
done
clear
View Answer play_arrow
Red data book or red list contains how many categories of species?
A)
6
done
clear
B)
5
done
clear
C)
7
done
clear
D)
8
done
clear
View Answer play_arrow
Potato spindle tuber disease is caused by
A)
Virus
done
clear
B)
Viroids
done
clear
C)
Lichens
done
clear
D)
Fungi
done
clear
View Answer play_arrow
Methanogens belong to
A)
Archaebacteria
done
clear
B)
Dinoflagellates
done
clear
C)
Slime moulds
done
clear
D)
Eubacteria
done
clear
View Answer play_arrow
Which of the following is correct about chrysophytes?
(I) Found in fresh and marine water (II) Microscopic plankton. (Ill) Cell walls are embedded with\[CaC{{O}_{3}}.\] (IV) Most of them are photo synthetic.
A)
I and III only
done
clear
B)
I, III and IV only
done
clear
C)
I, II and IV only
done
clear
D)
I and IV only
done
clear
View Answer play_arrow
Monographs contains information of
A)
Genus
done
clear
B)
Species
done
clear
C)
Family
done
clear
D)
Any one taxon
done
clear
View Answer play_arrow
Which of the following are correct for conifers?
(I) Needle like leaves to reduce surface area. (II) Thick cuticle on leaves. (Ill) Sunken stomata to reduce water loss. (IV) The main plant body is gametophyte.
A)
II and III
done
clear
B)
IV only
done
clear
C)
I only
done
clear
D)
II, I and III
done
clear
View Answer play_arrow
Which one of the following statement is wrong?
A)
Sargassum is popularly known as gulfweed.
done
clear
B)
Fucus and luminaria are rich source of Iodine.
done
clear
C)
Chlorella and spirulina are rich in protein.
done
clear
D)
Agar-Agar is obtained from luminaria focus and sargassum.
done
clear
View Answer play_arrow
The term 'tetradynamous' is related to
A)
Corolla
done
clear
B)
Calyx
done
clear
C)
Gynoecium
done
clear
D)
Androecium
done
clear
View Answer play_arrow
Select from the following the total number of plant having non-endospermic seed. Gram, Arhar, Moong, Sesbania, Lupin, Muliathi, Soyabean, Tomato, Brinjal, Belladona, Petunia, Tobacco, Tulip, Aloe, Colchicine, Asparagus, Chilli, Ground nuts, Indigofera, Sunhemp.
A)
10
done
clear
B)
15
done
clear
C)
20
done
clear
D)
6
done
clear
View Answer play_arrow
Bilateral symmetry is found in the flowers of
A)
Mustard
done
clear
B)
Chilli
done
clear
C)
Datura
done
clear
D)
Gulmohar
done
clear
View Answer play_arrow
Select the incorrect matching from the following table
A)
Ornamental Tulip, gloriosa, lupin, sweet Pea and petunia
done
clear
B)
Medicine Muliathi, belladonna and aloe
done
clear
C)
Fodder Sesbania and trifolium
done
clear
D)
Edible oil Soybean, groundnut and colchicine
done
clear
View Answer play_arrow
Cortex is the region found between
A)
Pericycle and endodermis
done
clear
B)
Endodermis and pith
done
clear
C)
Endodermis and vascular bundle
done
clear
D)
Epidermis and stele
done
clear
View Answer play_arrow
The balloon-shaped structures called tyloses
A)
Characterises the sapwood.
done
clear
B)
Are extensions of xylem paraenchyma cells into vessels.
done
clear
C)
Are linked to the ascent of sap through xylem vessels.
done
clear
D)
Originates in the lumen of vessels.
done
clear
View Answer play_arrow
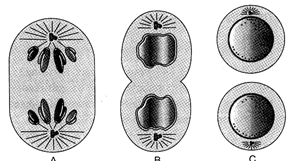
Identify A, B and C in the below diagram.
A)
A: Interphase, B: Telophase, C: Anaphase
done
clear
B)
A: Anaphase, B: Telophase, C: Interphase
done
clear
C)
A: Telophase, B: Interphase, C: Anaphase
done
clear
D)
A: Interphase, B: Anaphase, C: Telophase
done
clear
View Answer play_arrow
Which hormones do stimulate the production of pancreatic juice and bicarbonate?
A)
Gastrin and insulin
done
clear
B)
Cholecystokinin and secretin
done
clear
C)
Insulin and glucagon
done
clear
D)
Angiotensin and epinephrine
done
clear
View Answer play_arrow
Partial pressure of \[C{{O}_{2}}\]is maximum in
A)
Alveolar air
done
clear
B)
Pulmonary vein
done
clear
C)
Pulmonary artery
done
clear
D)
Aorta
done
clear
View Answer play_arrow
Choose the correct statement.
A)
Meissner's corpuscles are thermoreceptors.
done
clear
B)
Photoreceptors in the human eye are depolarized during darkness and become hyperpolarised in response to the light stimulus.
done
clear
C)
Receptors do not produce graded potentials.
done
clear
D)
Nociceptors respond to changes in pressure.
done
clear
View Answer play_arrow
Gull's disease occurs due to
A)
Hypersecretion of thyroid gland
done
clear
B)
Hyposecretion of adrenal gland
done
clear
C)
Hypersecretion of adrenal gland
done
clear
D)
Hyposecretion of thyroid gland
done
clear
View Answer play_arrow
Name the ion released from sarcoplasmic reticulum when impulse travels from motor end plate over sarcolemma and then into the T-tubule and sarcoplasmic reticulum.
A)
Magnesium
done
clear
B)
Sodium
done
clear
C)
Potassium
done
clear
D)
Calcium
done
clear
View Answer play_arrow
Name the blood cells, whose maturation is stimulated by vitamin \[{{B}_{12}}\]and folic acid.
A)
Leucocytes
done
clear
B)
Neutrophils
done
clear
C)
Thrombocytes
done
clear
D)
Erythrocytes
done
clear
View Answer play_arrow
Stress hormone among the following is
A)
Glucagon
done
clear
B)
Secretion
done
clear
C)
Gastrin
done
clear
D)
Cortisol
done
clear
View Answer play_arrow
Which is an age related disorder characterised by decreased bone mass and increased chances of fractures. Decreased levels of estrogen are a common cause.
A)
Osteoporosis
done
clear
B)
Gout
done
clear
C)
Tetany
done
clear
D)
Muscular dystrophy
done
clear
View Answer play_arrow
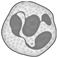
Which of the following is a true statement about this diagram?
A)
This is the most abundant cell of blood.
done
clear
B)
This cell is phagocytic in nature.
done
clear
C)
Abundance in blood is 60 to 65 per cent.
done
clear
D)
This cell secretes histamine, serotonin and heparin.
done
clear
View Answer play_arrow
Lungs do not collapse between breaths and some air always remains in the lungs which can never be expelled because
A)
There is a negative intrapleural pressure pulling at the lung walls.
done
clear
B)
There is a positive intrapleural pressure
done
clear
C)
Pressure in the lungs is higher than the atmospheric pressure.
done
clear
D)
There is a negative pressure in the lungs.
done
clear
View Answer play_arrow
A steroid hormone typically alters the activity of its target cells by
A)
Changing the membrane permeability of the cell.
done
clear
B)
Entering the cell and altering the gene expression.
done
clear
C)
Activation of \[I{{P}_{3}}.\]
done
clear
D)
Conversion of ATP to AMP
done
clear
View Answer play_arrow
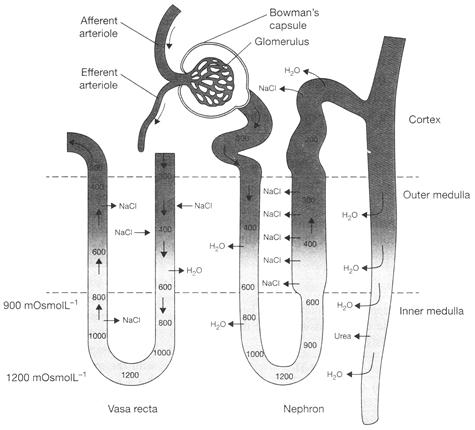
The following nephron produces how much concentrated urine?
A)
4 times
done
clear
B)
5 times
done
clear
C)
3 times
done
clear
D)
2 times
done
clear
View Answer play_arrow
Which of the following is a hormone releasing IUD?
A)
Multiload 375
done
clear
B)
Lippes loop
done
clear
C)
CuT
done
clear
D)
LNG-20
done
clear
View Answer play_arrow
Consider the statements given below regarding contraception and answer as directed thereafter.
A)
Medical Termination of Pregnancy (MTP) during the first trimester is generally safe.
done
clear
B)
Generally chances of conception are nil until mother breast-feeds the infant upto two years.
done
clear
C)
Intrauterine devices like copper-T are effective contraceptives.
done
clear
D)
Contraception pills may be taken up to one week after coitus to prevent conception.
done
clear
View Answer play_arrow
A vasectomy
A)
Prevents the production of sperm in the testes.
done
clear
B)
Prevents the production of semen.
done
clear
C)
Prevents the movement of sperm into the urethra.
done
clear
D)
Prevents a man from having an erection.
done
clear
View Answer play_arrow
Which of the following depicts the correct pathway of transport of sperms?
A)
Rete testis\[\to \]Epididymis\[\to \]Efferent ductules\[\to \]Vas deferens
done
clear
B)
Rete testis\[\to \]Vas deferens\[\to \]Efferent ductules \[\to \] Epididymis
done
clear
C)
Efferent ductules \[\to \]Rete testis\[\to \]Vas deferens\[\to \] Epididymis
done
clear
D)
Rate Testis \[\to \] Efferent ductules\[\to \] Epididymis \[\to \]Vas deferens
done
clear
View Answer play_arrow
Match the Column I with Column II and select the correct option using the codes given below:
Column I
Column II
(A) Mons pubis
(i) Embryo formation
(B) Antrum
(ii) Sperm
(C) Trophectoderm
(iii) Female external genitalia
(D) Nebenkern
(iv) Graafian follicle
A)
A-(iii) B-(iv) C-(i) D-(ii)
done
clear
B)
A-(ii) B-(i) C-(iv) D-(ii)
done
clear
C)
A-(i) B-(iv) C-(iii) D-(ii)
done
clear
D)
A-(iii) B-(iv) C-(ii) D-(i)
done
clear
View Answer play_arrow
Which part of the sperm helps in into the ovum?
A)
Mitochondria
done
clear
B)
Acrosome
done
clear
C)
Tail
done
clear
D)
All of the above
done
clear
View Answer play_arrow
If a colour-blind man marries a woman who is homozygous for normal colour vision, the probability of their son being colour-blind is
A)
0.5
done
clear
B)
0.75
done
clear
C)
1
done
clear
D)
0
done
clear
View Answer play_arrow
Match the following concepts of evolution with List-I and List-II and select the correct answer using the codes given below the lists:
List-I
List-II
(A) Mutation
(1) Changes in population's allele frequencies due to chance alone.
(B) Gene flow
(2) Differences in survival and reproduction among variant individuals.
(C) Natural selection
(3) Immigration, emigration change allele frequencies.
(D) Genetic drift
(4) Source of new alleles.
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-4, B-2, C-3, D-1
done
clear
C)
A-3, B-1, C-4, D-2
done
clear
D)
A-4, B-3, C-2, D-1
done
clear
View Answer play_arrow
Frequency of an autosomal lethal is 0.4. The frequency of carrier in a population of 200 individual is
A)
72
done
clear
B)
96
done
clear
C)
104
done
clear
D)
36
done
clear
View Answer play_arrow
What was the most significant trend in the evolution of modern man (Homo sapiens) from his ancestors?
A)
Shortening of jaws
done
clear
B)
Binocular vision
done
clear
C)
Increasing cranial capacity
done
clear
D)
Upright posture
done
clear
View Answer play_arrow
Which of the following is the correct sequence of events in the origin of life?
I. Formation of protobionts II. Synthesis of organic monomers III. Synthesis of organic polymers IV. Formation of DNA-based genetic systems
A)
I, III, II, IV
done
clear
B)
II, III, I, IV
done
clear
C)
II, III, IV, I
done
clear
D)
I, II, III, IV
done
clear
View Answer play_arrow
A molecule that can act as a genetic material must fulfil the traits given below, except
A)
It should be able to generate its replica.
done
clear
B)
It should be unstable structurally and chemically.
done
clear
C)
It should provide the scope for slow changes that are required for evolution.
done
clear
D)
It should be able to express itself in the form of Mendelian characters.
done
clear
View Answer play_arrow
Select the two statements out of the four (A to D) given below about lac operon.
(A) Glucose or galactose may bind with the repressor and inactivate it.
(B) In the absence of lactose, the represser binds with the operator region.
(C) The z-gene codes for permease.
(D) This was elucidated by Francois Jacob and Jacques Monod.
A)
(A), (C)
done
clear
B)
(B), (D)
done
clear
C)
(B), (C)
done
clear
D)
(A), (D)
done
clear
View Answer play_arrow
IARI, New Delhi released several vegetable crops that are rich in vitamins and minerals with respect to that select the correct matching.
Column-I
Column-II
(1) Vitamin A
(A) Carrot, spinach, pumpkin
(2) Vitamin C
(B) Bitter gourd, bathua, mustard, tomato ;
(3) Fe and Ca
(C) Spinach and bathua
(4) Protein
(D) Road bean, lablab, French and garden peas
A)
A-2, B-3, C-1, D-4
done
clear
B)
A-1, B-2, C-3, D-4
done
clear
C)
A-4, B-1, C-2, D-3
done
clear
D)
A-3, B-4, C-1, D-2
done
clear
View Answer play_arrow
Which of the following is correct regarding AIDS causative agent HIV?
A)
HIV is an enveloped virus that contains two identical molecules of single-stranded RNA and two molecules of reverse transcriptase.
done
clear
B)
HIV is a non-enveloped retrovirus.
done
clear
C)
HIV does not escape but attacks the acquired immune response.
done
clear
D)
HIV is an enveloped virus containing one molecule of single-stranded RNA and one molecule of reverse transcriptase.
done
clear
View Answer play_arrow
Among the following edible fishes, which one is a marine fish having rich source of omega-3 fatty acids?
A)
Mangur
done
clear
B)
Mrigala
done
clear
C)
Mackerel
done
clear
D)
Mystus
done
clear
View Answer play_arrow
'Swiss Cheese' bears large holes due to the production of \[C{{O}_{2}}\]by which microbe?
A)
Lacto bacillus
done
clear
B)
Saccharomyces cerevisiae
done
clear
C)
Propionibacterium shermanii
done
clear
D)
Aspergillus niger
done
clear
View Answer play_arrow
Biochemical Oxygen Demand (BOD) may not be a good index for pollution of water bodies receiving effluents from
A)
Dairy industry
done
clear
B)
Petroleum industry
done
clear
C)
Sugar industry
done
clear
D)
Domestic sewage
done
clear
View Answer play_arrow
Diapause is a stage of ____.
A)
Perpetual development
done
clear
B)
Intermittent development
done
clear
C)
Suspended development
done
clear
D)
Sequential development
done
clear
View Answer play_arrow
Exotic species having been introduced in India are
A)
Lantana camara,
done
clear
B)
Water Hyacinth Water Hyacinth,
done
clear
C)
Prosopis cineraria
done
clear
D)
Lantana camara, Ficus religiosa
done
clear
View Answer play_arrow
Nile Perch, Ficus religiosa Nearly _____ % of all insects are known to be phytophagous.
A)
25
done
clear
B)
35
done
clear
C)
45
done
clear
D)
10
done
clear
View Answer play_arrow
The Earth Summit (Historic Convention on Biological Diversity) was held in
A)
Johannesburg, South Africa, 2002
done
clear
B)
Rio de Janeiro, 1992
done
clear
C)
Kyoto, Japan
done
clear
D)
Montreal, Canada
done
clear
View Answer play_arrow
Eutrophication occurs because of
A)
Ca and Mg
done
clear
B)
Nitrogen and phosphorous
done
clear
C)
Carbon and oxygen
done
clear
D)
Hg and DDT
done
clear
View Answer play_arrow
Match the column
Column I
Column II
(A) Man
(1) Order - Carnivora
(B) Mango
(2) Family - Poaceae
(C) House fly
(3) Genus - Musca
(D) Tiger
(4) Phylum - Chordata
(E) Wheat
(5) Family - Anacardiaceae
A)
A-1, B-5, C-3, D-4, E-2
done
clear
B)
A-4, B-5, C-3, D-4, E-2
done
clear
C)
A-4, B-2, C-3, D-1, E-5
done
clear
D)
A-1, B-2, C-3, D-4, E-5
done
clear
View Answer play_arrow
Which of the following is incorrect about cartilaginous fish?
A)
Its teeth are modified placoid scales which an backwardly directed.
done
clear
B)
It has 5 to 7 pair of gills for respiration but without operculum (i.e., gill cover).
done
clear
C)
The air bladder is absent so that they have to swim constantly to avoid sinking.
done
clear
D)
In males, the pelvic fins claspers are absent.
done
clear
View Answer play_arrow
Find out the incorrect statement from the following
A)
Closely-related species differ in morphological features.
done
clear
B)
Genus comprises a group of related species.
done
clear
C)
Taxonomic studies are useful in agricultural, forestry and industries.
done
clear
D)
Notochord and ventral hollow neural system are common features of phylum chordata.
done
clear
View Answer play_arrow
Fat body of cockroach is functionally analogous to which organ of vertebrate
A)
Spleen
done
clear
B)
Liver
done
clear
C)
Kidney
done
clear
D)
Lungs
done
clear
View Answer play_arrow
Which of the following is the vasoconstrictor substance secreted by mast cells?
A)
Histamine
done
clear
B)
Heparin
done
clear
C)
Serotonin
done
clear
D)
Melatonin
done
clear
View Answer play_arrow
Between which of the following stages, the GTP is formed by substrate level phosphorylation?
A)
Succinate to fumarate
done
clear
B)
Ketoglutarate to succinate
done
clear
C)
Oxalosuccinate to glutarate
done
clear
D)
Fumarate to malate
done
clear
View Answer play_arrow
Which of the following is the least likely to be involved in stabilising the three-dimensional folding of most proteins?
A)
Electrostatic interaction
done
clear
B)
Hydrophobic interaction
done
clear
C)
Ester bonds
done
clear
D)
Hydrogen bonds
done
clear
View Answer play_arrow
Which of the following combinations is correct?
A)
Metal ions loosely attached with Apoenzyme Activators
done
clear
B)
Non-protein organic part attached tightly to Apoenzyme - Prosthetic group
done
clear
C)
Non protein organic part attached loosely to Apoenzyme - Coenzyme
done
clear
D)
All of the above
done
clear
View Answer play_arrow
When the cell has a stalled DNA replication fork which checkpoint should be predominantly activated?
A)
\[{{G}_{2}}/M\]
done
clear
B)
M
done
clear
C)
Both\[{{G}_{2}}/M\]and M
done
clear
D)
\[{{G}_{1}}/S\]
done
clear
View Answer play_arrow
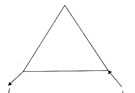
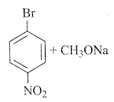
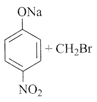
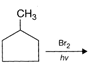 The major product obtained is
The major product obtained is