A parallel plate condenser with a dielectric of constant K between the plates has a capacity C and is charged to a potential V volt. The dielectric slab is slowly removed from between the plates and then reinserted. The network done by the system is this process is:
A)
0
done
clear
B)
\[\frac{1}{2}(K-1)C{{V}^{2}}\]
done
clear
C)
\[\frac{C{{V}^{2}}(K-1)}{K}\]
done
clear
D)
\[(K-1)C{{V}^{2}}\]
done
clear
View Answer play_arrow
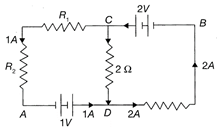
In the circuit shown in the figure. If the potential at point A is taken to be 0, the potential at point B is
A)
\[-1V\]
done
clear
B)
\[+2V\]
done
clear
C)
\[-2V\]
done
clear
D)
+1V
done
clear
View Answer play_arrow
An electric dipole is placed at the centre of a hollow conducting sphere. Which of the following is correct?
A)
Electric field is 0 at every point of sphere.
done
clear
B)
Electric field is non-zero anywhere on the sphere.
done
clear
C)
The flux of electric field is 0 through the sphere.
done
clear
D)
All are true.
done
clear
View Answer play_arrow
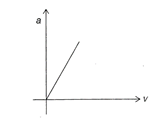
Acceleration-velocity graph of a particle moving in a straight line is as shown in figure. Then slope of velocity displacement graph
A)
increases linearly.
done
clear
B)
decreases linearly.
done
clear
C)
is constant.
done
clear
D)
increases parabolically.
done
clear
View Answer play_arrow
A particle is projected at an angle of \[60{}^\circ \] above the horizontal with a speed of 10 m/s. After some time the direction of its velocity makes an angle of \[30{}^\circ \] above the horizontal. The speed of the particle at this instant is
A)
\[\frac{5}{\sqrt{3}}m/s\]
done
clear
B)
\[5\sqrt{3}\,m/s\]
done
clear
C)
\[5\,m/s\]
done
clear
D)
\[\frac{10}{\sqrt{3}}\,m/s\]
done
clear
View Answer play_arrow
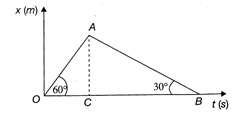
On the displacement-time graph, two straight lines make angles \[60{}^\circ \] and \[30{}^\circ \], with time axis as shown in the figure. The ratio of the velocities represented by them is
A)
1 : 2
done
clear
B)
1 : 3
done
clear
C)
2 : 1
done
clear
D)
3 : 1
done
clear
View Answer play_arrow
Three blocks of masses\[{{m}_{1}},{{m}_{2}}\]and\[{{m}_{3}}\]are connected by massless string as shown in the figure on a friction- less table. They are pulled with a force\[{{T}_{3}}=40\,N.\]If \[{{m}_{1}}=10\,kg,\,{{m}_{2}}=6kg\]and \[{{m}_{3}}=4\,kg,\]tension \[{{T}_{2}}=\]will be
A)
20 N
done
clear
B)
40 N
done
clear
C)
10 N
done
clear
D)
32 N
done
clear
View Answer play_arrow
A conducting bar is pulled with a constant speed U on a smooth conducting rail. The region has a steady magnetic field of induction B as shown in the figure. If the speed of the bar is doubled then the rate of heat dissipation will:
A)
remain constant.
done
clear
B)
become quarter of the initial value.
done
clear
C)
become four fold.
done
clear
D)
get doubled.
done
clear
View Answer play_arrow
A conducting loop of resistance R and radius r has its centre at the origin of the coordinate system in a magnetic field of induction B. When it is rotated about F-axis through \[90{}^\circ \], the net charge flown in the loop is directly proportional to:
A)
\[{{B}^{-1}}\]
done
clear
B)
R
done
clear
C)
\[{{r}^{2}}\]
done
clear
D)
r
done
clear
View Answer play_arrow
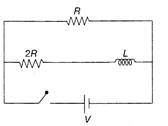
The ratio of time constants during current growth and current decay of the circuit shown in the figure is
A)
1 : 1
done
clear
B)
3 : 2
done
clear
C)
2 : 3
done
clear
D)
1 : 3
done
clear
View Answer play_arrow
A cubical block of wood of specific gravity 0.5 and a chunk of concrete of specific gravity 2.5 are fastener together. The ratio of the mass of wood to the mass of concrete which makes the combination to float with its entire volume submerged under water is:
A)
\[\frac{1}{5}\]
done
clear
B)
\[\frac{1}{3}\]
done
clear
C)
\[\frac{3}{5}\]
done
clear
D)
\[\frac{2}{5}\]
done
clear
View Answer play_arrow
The surface tension of a soap solution is\[2\times {{10}^{-2}}N/m.\]it To blow a bubble of radius 1 cm, the work done is
A)
\[4\pi \times {{10}^{-6}}J\]
done
clear
B)
\[8\pi \times {{10}^{-6}}J\]
done
clear
C)
\[12\pi \times {{10}^{-6}}J\]
done
clear
D)
\[16\pi \times {{10}^{-6}}J\]
done
clear
View Answer play_arrow
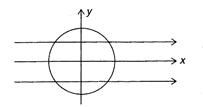
In a beaker, there is a hole at the bottom and a hole of same area is at the top and water starts flowing through it into the beaker with speed U, then:
A)
water level will oscillate about a height of\[{{U}^{2}}/2g\] is reached and remains constant
done
clear
B)
water level will rise till a height of \[{{U}^{2}}/2g\]reached and remains constant there after
done
clear
C)
no water will remains in the beaker
done
clear
D)
none of the above
done
clear
View Answer play_arrow
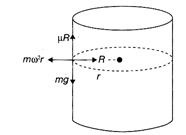
A hollow cylinder drum or radius r is placed with its axis vertical. It is rotated about an axis passing through its centre and perpendicular to the base and a body is placed on the inside surface of the drum. If the coefficient of friction is , the minimum frequency of revolution so that the body does not fall down is:
A)
\[\frac{1}{2\pi }\sqrt{\frac{\mu g}{r}}\]
done
clear
B)
\[\frac{1}{2\pi }\sqrt{\frac{g}{\mu r}}\]
done
clear
C)
\[\frac{1}{2\pi }\sqrt{\frac{g}{2\mu r}}\]
done
clear
D)
\[\frac{1}{2\pi }\sqrt{\frac{2\mu g}{r}}\]
done
clear
View Answer play_arrow
A uniform rod of mass m and length \[l\] rotates in a horizontal plane with an angular velocity co about a vertical axis passing through one end. The tension in the rod at a distance \[x\]from the axis is
A)
\[\frac{1}{2}m{{\omega }^{2}}x\]
done
clear
B)
\[\frac{1}{2}m{{\omega }^{2}}\frac{{{x}^{2}}}{\ell }\]
done
clear
C)
\[\frac{1}{2}m{{\omega }^{2}}\ell \left( l-\frac{x}{l} \right)\]
done
clear
D)
\[\frac{1}{2}\frac{m{{\omega }^{2}}}{l}({{l}^{2}}-{{x}^{2}})\]
done
clear
View Answer play_arrow
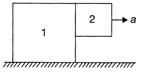
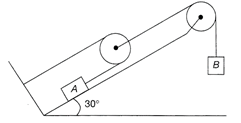
The coefficient of static friction between the two blocks is 0.363, what is the minimum acceleration of block 1 so that block 2 does not fall?
A)
\[~6\text{ }m{{s}^{-2}}\]
done
clear
B)
\[~12\text{ }m{{s}^{-2}}\]
done
clear
C)
\[~~18\text{ }m{{s}^{-2}}\]
done
clear
D)
\[~27\text{ }m{{s}^{-2}}\]
done
clear
View Answer play_arrow
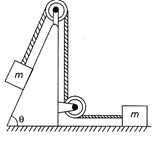
For the system shown in the figure, the pulleys are light and frictionless. The tension in the string will be:
A)
\[\frac{2}{3}mg\,\sin \theta \]
done
clear
B)
\[\frac{3}{2}mg\,\sin \theta \]
done
clear
C)
\[\frac{1}{2}mg\,\sin \theta \]
done
clear
D)
\[2mg\,\sin \theta \]
done
clear
View Answer play_arrow
The area of the parallelogram determined by\[\vec{A}=2\hat{i}+\hat{j}-2\hat{k}\]and\[\vec{B}=12\hat{j}-2\hat{k}\]is:
A)
42
done
clear
B)
56
done
clear
C)
38
done
clear
D)
74
done
clear
View Answer play_arrow
In the system in figure \[{{m}_{B}}=4\,kg\]and \[{{m}_{A}}=2\,kg.\]The pulleys are massless and friction is absent every- where. The acceleration of block A is\[(g=10\,m{{s}^{-2}})\]
A)
\[2\,m/{{s}^{2}}\]
done
clear
B)
\[4\,m/{{s}^{2}}\]
done
clear
C)
\[\frac{10}{3}m/{{s}^{2}}\]
done
clear
D)
\[\frac{20}{3}\,m/{{s}^{2}}\]
done
clear
View Answer play_arrow
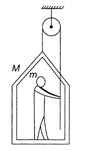
A man of mass m stands on a frame of mass M. He pulls on a light rope, which passes over a pulley. The other end of the rope is attached to the frame. For the system to be in equilibrium, what force must the man exert on the rope?
A)
\[\frac{1}{2}(M+m)g\]
done
clear
B)
\[(M+m)g\]
done
clear
C)
\[(M-m)g\]
done
clear
D)
\[(M+2m)g\]
done
clear
View Answer play_arrow
litres of a liquid with specific heat \[0.2\,cal/g{{m}^{-0}}C\]has the same thermal capacity as that of 20 litres of liquid with specific heat 0.3, find the ratio of their densities:
A)
3 : 1
done
clear
B)
1 : 3
done
clear
C)
1 : 6
done
clear
D)
6 : 1
done
clear
View Answer play_arrow
P-V diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will be:
A)
4 R
done
clear
B)
2.5 R
done
clear
C)
3R
done
clear
D)
\[4\frac{R}{3}\]
done
clear
View Answer play_arrow
The internal energy of an ideal gas during isothermal expansion:
A)
increases
done
clear
B)
decreases
done
clear
C)
remains constant
done
clear
D)
becomes zero
done
clear
View Answer play_arrow
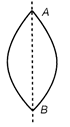
The equi-convex lens shown in the figure has focal length\[f.\] What will be the focal length of each half if the lens is cut along AB?
A)
\[\frac{f}{2}\]
done
clear
B)
\[f\]
done
clear
C)
\[\frac{3f}{2}\]
done
clear
D)
\[2f\]
done
clear
View Answer play_arrow
The mass and volume of a body are found to be 5.00 ± 0.05 kg and\[1.00\text{ }\pm \text{ }0.05\text{ }{{m}^{3}},\] respectively. Then the maximum possible percentage error in its density is
A)
6%
done
clear
B)
3%
done
clear
C)
7%
done
clear
D)
5%
done
clear
View Answer play_arrow
A body is moved along a straight line by a machine delivering constant power. The distance moved by the body in time t is proportional to
A)
\[{{t}^{\frac{1}{2}}}\]
done
clear
B)
\[{{t}^{\frac{3}{4}}}\]
done
clear
C)
\[{{t}^{\frac{3}{2}}}\]
done
clear
D)
\[{{t}^{2}}\]
done
clear
View Answer play_arrow
If the coefficient of restitution between a ball and the floor is 0.5, what is the percentage loss of energy on each rebounding of a ball dropped from a height?
A)
12.5%
done
clear
B)
25%
done
clear
C)
50%
done
clear
D)
75%
done
clear
View Answer play_arrow
When a sphere rolls without slipping, the ratio of its kinetic energy of translation to its total kinetic energy is:
A)
1 : 7
done
clear
B)
1 : 2
done
clear
C)
1 : 1
done
clear
D)
5 : 7
done
clear
View Answer play_arrow
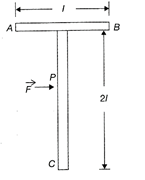
A T-shaped object with dimensions shown in the figure, is lying on a smooth floor. A force \[\vec{F}\]is applied at the point P parallel to AB, such that the object has only the translational motion without rotation. Find the location of P with respect to C
A)
\[\frac{4\ell }{3}\]
done
clear
B)
\[\ell \]
done
clear
C)
\[\frac{2\ell }{3}\]
done
clear
D)
\[\frac{3\ell }{2}\]
done
clear
View Answer play_arrow
Two satellites A and B go round a planet in circular orbits having radii 4R and R, respectively. If the speed of satellite A is 3v, then the speed of satellite B is
A)
\[\frac{3v}{2}\]
done
clear
B)
\[\frac{4v}{2}\]
done
clear
C)
\[6v\]
done
clear
D)
\[12v\]
done
clear
View Answer play_arrow
The electrical conductivity of a semiconductor increases when em radiation of wavelength shorter than 2480 nm is incident on it. The band gap (in eV) the semiconductor is:
A)
0.9
done
clear
B)
0.7
done
clear
C)
0.5
done
clear
D)
0.1
done
clear
View Answer play_arrow
The electric current in a circuit is given by: \[i=3t.\]Here t is in second and current in ampere. The rms current for the period t = 0 to t = 1 sec is:
A)
3 A
done
clear
B)
9 A
done
clear
C)
\[\sqrt{3A}\]
done
clear
D)
\[\sqrt[3]{3A}\]
done
clear
View Answer play_arrow
Light of two different frequencies whose photons have energies 1 and 2.5 eV, respectively, successively illuminate a metal whose work function is 0.5 eV. The ratio of the maximum speeds of the emitted electrons will be:
A)
1 : 5
done
clear
B)
1 : 4
done
clear
C)
1 : 2
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
lionization potential of hydrogen atom is 13.6 Hydrogen atom in the ground state is excited by monochromatic radiation of photons of energy 12.1 eV. The number of spectral lines emitted by the hydrogen atom, according to Bohr's theory, will be
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
One of the line spectra emitted from the X-ray tube is \[10\overset{\text{o}}{\mathop{\text{A}}}\,.\] What is the energy level difference in the emitting atom that is responsible for X-rays?
A)
24.8 eV
done
clear
B)
12.4 eV
done
clear
C)
6.2 eV
done
clear
D)
10.2 eVs
done
clear
View Answer play_arrow
Two radioactive materials X and X have decay constants 10/1 and A., respectively. If initially they have the same number of nuclei, then the ratio of the number of nuclei of \[{{X}_{1}}\]to that of \[{{X}_{2}}\]will be \[1/e\] after a time
A)
\[\frac{1}{(10\lambda )}\]
done
clear
B)
\[\frac{1}{(11\lambda )}\]
done
clear
C)
\[\frac{11}{(10\lambda )}\]
done
clear
D)
\[\frac{1}{(9\lambda )}\]
done
clear
View Answer play_arrow
Equations of two progressive waves at a certain point in a medium are given by \[{{y}_{1}}=a\sin (\omega t+{{\phi }_{1}}),\]and\[{{y}_{2}}=a\sin (\omega t+{{\phi }_{2}}).\]If amplitude and time period of resultant wave formed by the superposition of two waves is same as that of both the waves, then\[({{\phi }_{1}}-{{\phi }_{2}})\]is:
A)
\[\frac{\pi }{3}\]
done
clear
B)
\[\frac{2\pi }{3}\]
done
clear
C)
\[\frac{\pi }{6}\]
done
clear
D)
\[\frac{\pi }{4}\]
done
clear
View Answer play_arrow
It the ratio of intensities of two waves causing interference be 9:4, then the ratio of the resultant maximum and minimum intensities will be:
A)
9 : 4
done
clear
B)
3 : 2
done
clear
C)
25 : 1
done
clear
D)
5 : 1
done
clear
View Answer play_arrow
A convex lens of focal length 40 cm is in contact with a concave lens of focal length 25 cm. The power of the combination is
A)
-1.5 dioptre
done
clear
B)
-6.5 dioptre
done
clear
C)
+6.5 dioptre
done
clear
D)
+6.67 dioptre
done
clear
View Answer play_arrow
If two SHMs are represented by equations \[{{y}_{1}}=10\]\[\sin (3\pi t+\pi /4)\] and \[{{y}_{2}}=5[\sin (3\pi t)+\]\[\sqrt{3}\cos (3\pi t)]\]the ratio of their amplitudes is:
A)
2 : 1
done
clear
B)
1 : 2
done
clear
C)
1 : 1
done
clear
D)
\[1:\sqrt{2}\]
done
clear
View Answer play_arrow
The plane of a rectangular loop of wire with sides 0.05 m and 0.08 m is parallel to a uniform magnetic field of induction \[1.5\times {{10}^{-2}}\]Tesla. A current of 10.0 ampere flows through the loop. If the side of length 0.08 m is normal and the side of length 0.05 m is parallel to the lines of induction, then the torque acting on it is:
A)
\[~6000\text{ Newton }\!\!\times\!\!\text{ metre}\]
done
clear
B)
\[\text{0 Newton}\,\text{ }\!\!\times\!\!\text{ }\,\text{metre}\]
done
clear
C)
\[1.2\times {{10}^{-2}}\,\text{Newton}\,\text{ }\!\!\times\!\!\text{ }\,\text{metre}\]
done
clear
D)
\[6\times {{10}^{-4}}\,\text{Newton }\!\!\times\!\!\text{ metre}\]
done
clear
View Answer play_arrow
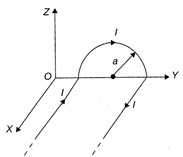
A long wire bent as shown in the figure carries I. If the radius of the semi-circular portion is a, the magnetic field at the centre of the semi-circle is:
A)
\[\frac{{{\mu }_{0}}I}{4a}\]
done
clear
B)
\[\frac{{{\mu }_{0}}I}{4\pi a}\sqrt{{{\pi }^{2}}+4}\]
done
clear
C)
\[\frac{{{\mu }_{0}}I}{4a}+\frac{{{\mu }_{0}}I}{2\pi a}\]
done
clear
D)
\[\frac{{{\mu }_{0}}I}{4\pi a}\sqrt{{{\pi }^{2}}-4}\]
done
clear
View Answer play_arrow
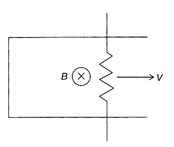
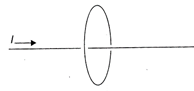
The long, straight wire as shown in the figure, carries a current i to the right that increases with time. What is the direction of the induced current in the circular wire loop, which lies in a plane perpendicular to the wire?
A)
Clockwise as viewed from the right.
done
clear
B)
Counterclockwise as viewed from the right.
done
clear
C)
There is no induced current.
done
clear
D)
The induced current is alternating.
done
clear
View Answer play_arrow
Which of the following quantities is 0 on an average for the molecules of an ideal gas in equilibrium
A)
Kinetic Energy
done
clear
B)
Momentum
done
clear
C)
Density
done
clear
D)
Speed
done
clear
View Answer play_arrow
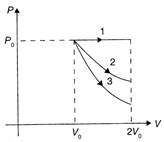
A gas is expanded from volume \[{{V}_{0}}\]to \[2{{V}_{0}}\] under three different processes. Process 1 is isobaric, Process 2 is isothermal, and Process 3 is adiabatic. Let \[\Delta {{U}_{1}},\] \[\Delta {{U}_{2}},\]and \[\Delta {{U}_{3}}\] be the change in internal energy of the gas in these three processes. Then
A)
\[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
done
clear
B)
\[\Delta {{U}_{1}}<\Delta {{U}_{2}}<\Delta {{U}_{3}}\]
done
clear
C)
\[\Delta {{U}_{2}}<\Delta {{U}_{1}}<\Delta {{U}_{3}}\]
done
clear
D)
\[\Delta {{U}_{2}}<\Delta {{U}_{3}}<\Delta {{U}_{1}}\]
done
clear
View Answer play_arrow
The least volatile hydrogen halide is
A)
\[HF\]
done
clear
B)
\[HCl\]
done
clear
C)
\[HI\]
done
clear
D)
\[HBr\]
done
clear
View Answer play_arrow
Bromine water reacts with \[S{{O}_{2}}\]to form
A)
\[{{H}_{2}}O\]and \[HBr\]
done
clear
B)
\[HBr\] and S
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]and \[HBr\]
done
clear
D)
S and\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
The chemical formula of phosgene is
A)
\[CaOC{{l}_{2}}\]
done
clear
B)
\[COCl\]
done
clear
C)
\[CaC{{O}_{3}}\]
done
clear
D)
\[COC{{l}_{2}}\]
done
clear
View Answer play_arrow
Which of the following molecules has trigonal planar geometry?
A)
\[N{{H}_{3}}\]
done
clear
B)
\[B{{F}_{3}}\]
done
clear
C)
\[I{{F}_{3}}\]
done
clear
D)
\[P{{H}_{3}}\]
done
clear
View Answer play_arrow
Highest oxidation state of Mn is present in
A)
\[M{{n}_{2}}{{O}_{3}}\]
done
clear
B)
\[Mn{{O}_{2}}\]
done
clear
C)
\[KMn{{O}_{4}}\]
done
clear
D)
\[{{K}_{2}}Mn{{O}_{4}}\]
done
clear
View Answer play_arrow
In the following reaction, the product 'R' is \[Ca{{C}_{2}}\xrightarrow{{{H}_{2}}O}P\xrightarrow[\text{Tube}]{\text{Hot}\,\text{iron}}Q\xrightarrow[\text{AlC}{{\text{l}}_{\text{3}}}]{\text{C}{{\text{H}}_{\text{3}}}\text{Cl}}R\]
A)
Toluene
done
clear
B)
n-propylbenzene
done
clear
C)
Ethylbenzene
done
clear
D)
Benzene
done
clear
View Answer play_arrow
Ammonia on reaction with excess of chlorine gives
A)
\[{{N}_{4}}\]and\[N{{H}_{4}}Cl\]
done
clear
B)
\[NC{{l}_{3}}\]and\[HCl\]
done
clear
C)
\[{{N}_{2}}\]and\[HCl\]
done
clear
D)
\[NC{{l}_{3}}\]and\[N{{H}_{4}}Cl\]
done
clear
View Answer play_arrow
Deuterium nucleus contains
A)
\[2p+0n\]
done
clear
B)
\[1p+1n\]
done
clear
C)
\[2p+2n\]
done
clear
D)
\[1p+1{{e}^{-}}\]
done
clear
View Answer play_arrow
In which process, fused sodium hydroxide is electro- lysed for extraction of sodium?
A)
Cyanide process
done
clear
B)
Castner process
done
clear
C)
Down's process
done
clear
D)
Both (a) and (c)
done
clear
View Answer play_arrow
Number of atoms present in 4.25 g of \[N{{H}_{3}}\]is
A)
\[4\times 6.023\times {{10}^{23}}\]
done
clear
B)
\[6.023\times {{10}^{23}}\]
done
clear
C)
\[4.25\times 6.023\times {{10}^{23}}\]
done
clear
D)
\[1.7\times {{10}^{24}}\]
done
clear
View Answer play_arrow
If 1 ml of water contains 20 drops. Then number of molecules in a drop of water is
A)
\[4.346\times {{10}^{20}}\]molecules
done
clear
B)
\[1.344\times {{10}^{18}}\]molecules
done
clear
C)
\[1.376\times {{10}^{26}}\]molecules
done
clear
D)
\[6.023\times {{10}^{23}}\]molecules
done
clear
View Answer play_arrow
When adding 100 ml of \[HCl+35\,\,ml\] of \[NaOH\], the colour of methyl orange in the solution will be
A)
Yellow
done
clear
B)
Red
done
clear
C)
Methyl orange is not a suitable indicator
done
clear
D)
Can't be predicted
done
clear
View Answer play_arrow
A developer used in photography is
A)
A weak base
done
clear
B)
A weak acid
done
clear
C)
An oxidizing agent
done
clear
D)
A mild reducing agent
done
clear
View Answer play_arrow
Given, \[N{{H}_{3}}(g)+3C{{l}_{2}}(g)\]
\[NC{{l}_{3}}(g)+3HCl(g);-\Delta {{H}_{1}}\] (1)
\[{{N}_{2}}(g)+3{{H}_{2}}(g)2N{{H}_{3}}(g);-\Delta {{H}_{2}}\] (2)
\[{{H}_{2}}(g)+C{{l}_{2}}(g)\rightleftharpoons 2HCl(g);\Delta {{H }_{3}}\] (3)
The heat formation of \[NC{{l}_{3}}\,(g)\]in terms of \[\Delta {{H}_{1}},\,\Delta {{H}_{2}}\]and \[\Delta {{H}_{3}}\]is
A)
\[\Delta {{H}_{f}}=\Delta {{H}_{1}}+\frac{\Delta {{H}_{2}}}{2}-\frac{3}{2}\Delta {{H}_{3}}\]
done
clear
B)
\[\Delta {{H}_{f}}=-\Delta {{H}_{1}}+\frac{\Delta {{H}_{2}}}{2}-\frac{3}{2}\Delta {{H}_{3}}\]
done
clear
C)
\[\Delta {{H}_{f}}=\Delta {{H}_{1}}-\frac{\Delta {{H}_{2}}}{2}-\frac{3}{2}\Delta {{H}_{3}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The electronegativity of the following elements increases in the order
A)
N, Si, C, P
done
clear
B)
C, N, Si, P
done
clear
C)
P, Si, N, C
done
clear
D)
Si, P, C, N
done
clear
View Answer play_arrow
Which of the following compounds on reaction with \[NaOH\] and \[N{{a}_{2}}{{O}_{2}}\]gives yellow colour?
A)
\[Al{{(OH)}_{3}}\]
done
clear
B)
\[Zn{{(OH)}_{2}}\]
done
clear
C)
\[CaC{{O}_{3}}\]
done
clear
D)
\[Cr{{(OH)}_{3}}\]
done
clear
View Answer play_arrow
At \[18{}^\circ C\], the conductance of\[{{H}^{+}}\] and \[C{{H}_{3}}CO{{O}^{-}}\]at an infinite dilution is 315 and 35 mho\[c{{m}^{2}}\,e{{q}^{-1}}\]respectively. The equivalent conductivity of\[C{{H}_{3}}COOH\] at infinite dilution is __ mho \[e{{m}^{2}}\,e{{q}^{-1}}\]
A)
280
done
clear
B)
350
done
clear
C)
315
done
clear
D)
30
done
clear
View Answer play_arrow
Permanent hardness of water can be removed by adding.
A)
K
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[C{{l}_{2}}\]
done
clear
D)
\[Ca(OCl)Cl\]
done
clear
View Answer play_arrow
In the complex ion \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}},\] the\[N{{H}_{3}}\] molecules are linked to the central metal ion by
A)
Coordinate bonds
done
clear
B)
Hydrogen bonds
done
clear
C)
Covalent bonds
done
clear
D)
Ionic bonds
done
clear
View Answer play_arrow
How many moles of iodine are liberated when 1 mole of potassium dichromate reacts with potassium iodide?
A)
4
done
clear
B)
2
done
clear
C)
3
done
clear
D)
1
done
clear
View Answer play_arrow
Pure benzene freezes at \[5.3{}^\circ C\]. A solution of 0.223 g of phenylacetic acid \[({{C}_{6}}{{H}_{5}}C{{H}_{2}}COOH)\] in 4.4 g of benzene \[({{K}_{f}}=5.12\,K\,kg\,mo{{l}^{-1}})\] freezes at \[4.47{}^\circ C\]. From this observation, one can conclude that
A)
Phenylacetic acid undergoes partial ionization in benzene.
done
clear
B)
Phenylacetic acid exists as such in benzene.
done
clear
C)
Phenylacetic acid dimerizes in benzene.
done
clear
D)
Phenylacetic acid undergoes complete ionization in benzene.
done
clear
View Answer play_arrow
The reaction \[{{N}_{2}}{{O}_{5}}\]in\[CC{{l}_{4}}(solution)\to 2N{{O}_{2}}(solution)+\frac{1}{2}{{O}_{2}}(g)\] is of first order in\[{{N}_{2}}{{O}_{5}}\] with a rate constant of \[6.2\times {{10}^{-4}}{{s}^{-1}}.\]What is the value of rate of reaction when\[[{{N}_{2}}{{O}_{5}}]=1.25\,\text{mole}\,{{\text{L}}^{-1}}?\]
A)
\[6.35\times {{10}^{-3}}\,\text{mol}\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
B)
\[5.15\times {{10}^{-5}}\,\text{mol}\,{{\text{L}}^{-1}}{{s}^{-1}}\]
done
clear
C)
\[3.85\times {{10}^{-4}}\,\text{mol}\,{{\text{L}}^{-1}}\,{{s}^{-1}}\]
done
clear
D)
\[7.75\times {{10}^{-4}}\,\text{mol}\,{{\text{L}}^{-1}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
If 1 mole of an ideal gas expands isothermally at \[37{}^\circ C\] from 15 litres to 25 litres, the maximum work obtained is
A)
6.43 J
done
clear
B)
12.87 J
done
clear
C)
2.92 J
done
clear
D)
8.57 J
done
clear
View Answer play_arrow
Hydrolysis of trichloromethane with aqueous \[KOH\] gives
A)
Acetic acid
done
clear
B)
Methanol
done
clear
C)
Formic acid
done
clear
D)
Ethanol
done
clear
View Answer play_arrow
Which of the following transition metal ions is not coloured?
A)
\[{{V}^{3+}}\]
done
clear
B)
\[C{{u}^{+}}\]
done
clear
C)
\[N{{i}^{2+}}\]
done
clear
D)
\[C{{o}^{2+}}\]
done
clear
View Answer play_arrow
The shape of \[I{{F}_{7}}\]molecule is
A)
Pentagonal bipyramidal
done
clear
B)
Tetrahedral
done
clear
C)
Trigonal bipyramidal
done
clear
D)
Octahedral
done
clear
View Answer play_arrow
The compound containing coordinate bond is
A)
\[{{O}_{3}}\]
done
clear
B)
\[S{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
Maximum number of H bonds in one molecule of water is
A)
1
done
clear
B)
3
done
clear
C)
2
done
clear
D)
4
done
clear
View Answer play_arrow
Among the given compounds, the one which can be distinguished by\[AgN{{O}_{3}}\]is
A)
Ethylene
done
clear
B)
Ethane
done
clear
C)
Diethyl ether
done
clear
D)
Acetylene
done
clear
View Answer play_arrow
What will be the partial pressure of He and \[{{O}_{2}}\]respectively, if 200 ml of He at 0.66 atm and 400 ml of \[{{O}_{2}}\]at 0.52 atm pressure are mixed in 400 ml vessel at \[20{}^\circ C\]?
A)
0.33 and 0.52
done
clear
B)
0.33 and 0.56
done
clear
C)
0.25 and 0.45
done
clear
D)
0.38 and 0.52
done
clear
View Answer play_arrow
Which of the following carbonates is the most stable?
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[MgC{{O}_{3}}\]
done
clear
C)
\[~BaC{{O}_{3}}\]
done
clear
D)
\[~SrC{{O}_{3}}\]
done
clear
View Answer play_arrow
At room temperature, the eclipsed and staggered forms of ethane cannot be isolated because
A)
Both the conformers are equally stable.
done
clear
B)
They interconvert rapidly.
done
clear
C)
There is a large energy barrier of rotation around the o bond.
done
clear
D)
The energy difference between the conformers is large.
done
clear
View Answer play_arrow
In an experiment, 4 g of\[{{M}_{2}}{{O}_{x}}\]oxide was reduced to 2.8 g of the metal. If the atomic mass of the metal is \[56\,g\,mo{{l}^{-1}},\] the number of 0 atoms in the oxide is
A)
2
done
clear
B)
1
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
If the supply of oxygen is limited, \[{{H}_{2}}S\]reacts with \[{{O}_{2}}\]to form
A)
\[{{H}_{2}}O+S\]
done
clear
B)
\[{{H}_{2}}O+S{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}O+S{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{4}}+S\]
done
clear
View Answer play_arrow
Primary secondary and tertiary amines can be distinguished by
A)
Fehling's test
done
clear
B)
SchifFs test
done
clear
C)
Hinsberg test
done
clear
D)
Tollen's test
done
clear
View Answer play_arrow
Which of the following has the highest electron affinity?
A)
\[{{O}^{-}}\]
done
clear
B)
\[{{F}^{-}}\]
done
clear
C)
Na
done
clear
D)
O
done
clear
View Answer play_arrow
In the equation,\[4M+8C{{N}^{-}}+2{{H}_{2}}O+{{O}_{2}}\to \]\[4{{[M{{(CN)}_{2}}]}^{-}}+4O{{H}^{-}}\] Identify the metal M. It is
A)
Iron
done
clear
B)
Copper
done
clear
C)
Zinc
done
clear
D)
Gold
done
clear
View Answer play_arrow
IUPAC nomenclature of \[{{H}_{3}}C-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-CH=\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,-C{{H}_{3}}\]
A)
2, 4, 4-Trimethylpent-3-ene
done
clear
B)
2, 4, 4-Trimethylpent-2-ene
done
clear
C)
2, 2, 4-Trimethylpent-2-ene
done
clear
D)
2, 2, 4-Trimethylpent-3-ene
done
clear
View Answer play_arrow
Number of acyclic isomers of the compound having the molecular formula \[{{C}_{4}}{{H}_{10}}O\]is
A)
4
done
clear
B)
7
done
clear
C)
6
done
clear
D)
5
done
clear
View Answer play_arrow
Violet coloured complex obtained in the detection of sulphur is
A)
\[N{{a}_{3}}[Fe(ONSNa){{(CN)}_{5}}]\]
done
clear
B)
\[N{{a}_{2}}[Fe(NO){{(CN)}_{5}}]\]
done
clear
C)
\[N{{a}_{4}}[Fe{{(CN)}_{5}}NOS]\]
done
clear
D)
Both (a) and (c)
done
clear
View Answer play_arrow
Hydrolysis of sucrose is called
A)
Esterification
done
clear
B)
Inversion
done
clear
C)
Saponification
done
clear
D)
Hydration
done
clear
View Answer play_arrow
The heat liberated when 1.89 g of benzoic acid is burnt in a bomb calorimeter at \[25{}^\circ C\] increases the temperature of 18.94 kg of water by \[0.632{}^\circ C\]. If the specific heat of water at \[25{}^\circ C\] is 0.998 cal/g-deg, the value of the heat of combustion of benzoic acid is
A)
771.4 kcal
done
clear
B)
881.1 kcal
done
clear
C)
871.2 kcal
done
clear
D)
981.1 kcal
done
clear
View Answer play_arrow
In an organic compound, phosphorus is estimated as
A)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
B)
\[{{P}_{2}}{{O}_{5}}\]
done
clear
C)
\[M{{g}_{2}}{{P}_{2}}{{O}_{7}}\]
done
clear
D)
\[M{{g}_{3}}{{(P{{O}_{4}})}_{2}}\]
done
clear
View Answer play_arrow
If 1,3-Dibromopropane reacts with zinc and Nal, the product obtained is
A)
Propane
done
clear
B)
Propene
done
clear
C)
Hexane
done
clear
D)
Cyclopropane
done
clear
View Answer play_arrow
Chemical formula of Prussian blue is
A)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
B)
\[N{{a}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
C)
\[F{{e}_{4}}{{[Fe{{(CN)}_{6}}]}_{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Oxalic acid when heated with concentrated \[{{H}_{2}}S{{O}_{4}}\]gives
A)
\[C{{O}_{2}}+{{H}_{2}}O\]
done
clear
B)
\[CO+C{{O}_{2}}+{{H}_{2}}O\]
done
clear
C)
CO only
done
clear
D)
\[C{{O}_{2}}\]only
done
clear
View Answer play_arrow
\[Pb+\] concentrated\[HN{{O}_{3}}\]gives
A)
\[Pb{{(N{{O}_{3}})}_{4}}+N{{O}_{3}}\]
done
clear
B)
\[Pb{{(N{{O}_{3}})}_{3}}{{N}_{2}}O\]
done
clear
C)
\[Pb{{(N{{O}_{3}})}_{2}}+N{{O}_{2}}\]
done
clear
D)
\[~PbN{{O}_{3}}+NO\]
done
clear
View Answer play_arrow
When ethanol is treated with Fehling's solution, it gives a precipitate of
A)
Cu
done
clear
B)
\[C{{u}_{2}}O\]
done
clear
C)
\[CuO\]
done
clear
D)
\[C{{u}_{3}}O\]
done
clear
View Answer play_arrow
The major organic product formed from the following reaction
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[{{N}_{2}}\]gas will not be evolved upon reaction of\[HN{{O}_{2}}\]with which of the following amines
A)
\[{{1}^{o}}\]
done
clear
B)
\[{{3}^{o}}\]
done
clear
C)
\[{{2}^{o}}\]
done
clear
D)
Bothe (b) and (c)
done
clear
View Answer play_arrow
Binomial nomenclature consists of two words
A)
Genus and species
done
clear
B)
Order and family
done
clear
C)
Family and genus
done
clear
D)
Species and variety
done
clear
View Answer play_arrow
Reverse transcriptase is a/an
A)
RNA dependent RNA polymerase
done
clear
B)
DNA dependent RNA polymerase
done
clear
C)
DNA dependent DNA polymerase
done
clear
D)
RNA dependent DNA polymerase
done
clear
View Answer play_arrow
Tobacco Mosaic Virus (TMV) genes are
A)
Double-stranded RNA
done
clear
B)
Single-stranded RNA
done
clear
C)
Polyribonucleotides
done
clear
D)
Proteinaceous
done
clear
View Answer play_arrow
Which antibiotic inhabits interaction between tRNA and mRNA during bacterial protein synthesis?
A)
Neomycin
done
clear
B)
Streptomycin
done
clear
C)
Tetracycline
done
clear
D)
Erythromycin
done
clear
View Answer play_arrow
Which pair of the following belongs to basidiomycetes?
A)
Puffballs and claviceps
done
clear
B)
Peziza and stinkhorns
done
clear
C)
Morchella and mushrooms
done
clear
D)
Birds nest fungi and puffballs
done
clear
View Answer play_arrow
The plant group that produces spores and embryo but lacks vascular tissues and seeds is
A)
Pteridophyta
done
clear
B)
Rhodophyta
done
clear
C)
Bryophyta
done
clear
D)
Phaeophyta
done
clear
View Answer play_arrow
Multicellular branched rhizoids and leafy gametophytes are characteristics of
A)
All bryophytes
done
clear
B)
Some bryophytes
done
clear
C)
All pteridophytes
done
clear
D)
Some pteridophytes
done
clear
View Answer play_arrow
Polyribosomes are aggregates of
A)
Ribosomes and rRNA
done
clear
B)
Only rRNA
done
clear
C)
Peroxisomes
done
clear
D)
Several ribosomes held together by string of mRNA
done
clear
View Answer play_arrow
Membranous bag with hydrolytic enzymes which is used for controlling intracellular digestion of macro- molecules is
A)
Endoplasmic reticulum
done
clear
B)
Nucleosome
done
clear
C)
Lysosome
done
clear
D)
Phagosome
done
clear
View Answer play_arrow
Ribosomes are produced in
A)
Nucleolus
done
clear
B)
Cytoplasm
done
clear
C)
Mitochondria
done
clear
D)
Golgi body
done
clear
View Answer play_arrow
Which one of the following is not a constituent of cell membrane?
A)
Glycolipids
done
clear
B)
Proline
done
clear
C)
Phospholipids
done
clear
D)
Cholesterol
done
clear
View Answer play_arrow
A family of five daughters only is expecting their sixth issue. The chance of its being a son is
A)
Zero
done
clear
B)
25 per cent
done
clear
C)
50 per cent
done
clear
D)
100 per cent
done
clear
View Answer play_arrow
In a genetic cross having recessive epistasis, the F2 phenotypic ratio would be
A)
9 : 6 : 1
done
clear
B)
15 : 1
done
clear
C)
9 : 3 : 4
done
clear
D)
12 : 3 : 1
done
clear
View Answer play_arrow
Down's syndrome is due to
A)
Crossing over
done
clear
B)
Linkage
done
clear
C)
Sex-linked inheritance
done
clear
D)
Nondisjunction of chromosomes
done
clear
View Answer play_arrow
In a dihybrid cross \[\text{AABB }\!\!\times\!\!\text{ aabb, F2}\]progeny of AABB, AABb, AaBB and AaBb occurs in the ratio of
A)
1 : 1 : 1 : 1
done
clear
B)
9 : 3 : 3 : 1
done
clear
C)
1 : 2 : 2 : 1
done
clear
D)
1 : 2 : 2 : 4
done
clear
View Answer play_arrow
The genes controlling the seven pea characters studied by Mendel are now known to be located on how many different chromosomes?
A)
Four
done
clear
B)
Seven
done
clear
C)
Six
done
clear
D)
Five
done
clear
View Answer play_arrow
Who proved that DNA is a basic genetic material?
A)
Griffith
done
clear
B)
Watson
done
clear
C)
Boveri and Sutton
done
clear
D)
Hershey and chase
done
clear
View Answer play_arrow
The telomeres of eukaryotic chromosomes consist of short sequences of
A)
Thymine rich repeats
done
clear
B)
Cytosine rich repeats
done
clear
C)
Adenine rich repeats
done
clear
D)
Guanine rich repeats
done
clear
View Answer play_arrow
Genetic engineering is possible because
A)
We can see the DNA by electron microscope.
done
clear
B)
We can cut the DNA at specific sites by endonucleases like DNase I.
done
clear
C)
Restriction endonucleases purified from bacteria can be used in vitro.
done
clear
D)
The phenomenon of transduction in bacteria is well understood.
done
clear
View Answer play_arrow
Germination of seed within fruit is
A)
Ovipary
done
clear
B)
Vivipary
done
clear
C)
Hypogeal
done
clear
D)
Epigeal
done
clear
View Answer play_arrow
Aril represents the edible part of
A)
Banana
done
clear
B)
Litchi
done
clear
C)
Mango
done
clear
D)
Apple
done
clear
View Answer play_arrow
What type of placentation is seen in sweet pea?
A)
Axile
done
clear
B)
Free central
done
clear
C)
Marginal
done
clear
D)
Basal
done
clear
View Answer play_arrow
Four radial vascular bundles are found in
A)
Dicot root
done
clear
B)
Monocot root
done
clear
C)
Dicot stem
done
clear
D)
Monocot stem
done
clear
View Answer play_arrow
Passage cells are thin walled cells found in
A)
Phloem elements that serve as entry points for substance for transport of other plant parts.
done
clear
B)
Testa of seeds to enable emergence of growing embryonic axis during seed germination.
done
clear
C)
Central region of style through which the pollen tube grows towards the ovary.
done
clear
D)
Endodermis of roots facilitating the rapid transport of water from cortex to pericycle.
done
clear
View Answer play_arrow
Amino acids are mostly synthesized from
A)
Mineral salts
done
clear
B)
Fatty acids
done
clear
C)
Volatile acids
done
clear
D)
\[\alpha -\]Ketoglutaric acid
done
clear
View Answer play_arrow
Which one of the following mineral elements plays an important role in biological nitrogen fixation?
A)
Molybdenum
done
clear
B)
Copper
done
clear
C)
Manganese
done
clear
D)
Zinc
done
clear
View Answer play_arrow
Stomata of a plant is open due to
A)
Influx of calcium ions
done
clear
B)
Influx of potassium ions
done
clear
C)
Efflux of potassium ions
done
clear
D)
Influx of hydrogen ions
done
clear
View Answer play_arrow
The first carbon dioxide acceptor in \[{{C}_{4}}\] plants is
A)
Phosphoenolpyruvate
done
clear
B)
Ribulose-1, 5-bisphosphate
done
clear
C)
Oxaloacetic acid
done
clear
D)
Phosphoglyceric acid
done
clear
View Answer play_arrow
Which one is \[a-{{C}_{4}}\]plant?
A)
Papaya
done
clear
B)
Pea
done
clear
C)
Potato
done
clear
D)
Maize/Corn
done
clear
View Answer play_arrow
Which pigment system is inactivated in red drop?
A)
PS-I and PS-II
done
clear
B)
PS-I
done
clear
C)
PS-II
done
clear
D)
None of these
done
clear
View Answer play_arrow
In animal cells, the first stage of glucose breakdown is
A)
Krebs cycle
done
clear
B)
Glycolysis
done
clear
C)
Oxidative phosphorylation
done
clear
D)
ETC
done
clear
View Answer play_arrow
During the stage in the complete oxidation of glucose is the greatest number of ATP molecules formed from ADP
A)
Glycolysis
done
clear
B)
Krebs cycle
done
clear
C)
Conversion of pyruvic acid to acetyl-CoA
done
clear
D)
Electron transport chain
done
clear
View Answer play_arrow
Total number of meiotic division required for forming 100 zygotes/100 grains of wheat is
A)
100
done
clear
B)
75
done
clear
C)
125
done
clear
D)
50
done
clear
View Answer play_arrow
Fertilization involving carrying of male gametes by pollen tube is
A)
Porogamy
done
clear
B)
Siphonogamy
done
clear
C)
Chalazogamy
done
clear
D)
Syngonogamy
done
clear
View Answer play_arrow
In angiosperms pollen tubes liberate their male gametes into the
A)
Central cell
done
clear
B)
Antipodal cell
done
clear
C)
Egg cell
done
clear
D)
Synergids
done
clear
View Answer play_arrow
The hormone responsible for apical dominance
A)
IAA
done
clear
B)
GA
done
clear
C)
ABA
done
clear
D)
Florigen
done
clear
View Answer play_arrow
An enzyme that can stimulate germination of barley seeds is
A)
Lipase
done
clear
B)
Protease
done
clear
C)
Invertase
done
clear
D)
\[\alpha -\]Amylase
done
clear
View Answer play_arrow
Treatment of seed at low temperature under moist conditions to break its dormancy is called
A)
Verbalization
done
clear
B)
Chelation
done
clear
C)
Stratification
done
clear
D)
Scarification
done
clear
View Answer play_arrow
Keystone species in an ecosystem are those
A)
Present in maximum number
done
clear
B)
That are most frequent
done
clear
C)
Attaining a large biomass
done
clear
D)
Contributing to ecosystem properties
done
clear
View Answer play_arrow
What is BOD?
A)
Amount of \[{{O}_{2}}\]utilized by organisms in water.
done
clear
B)
Amount of\[{{O}_{2}}\]utilized by microorganism's for biological oxidation.
done
clear
C)
Total amount of \[{{O}_{2}}\]present in water.
done
clear
D)
All the above.
done
clear
View Answer play_arrow
Drawback of DDT as pesticide is
A)
It becomes ineffective after sometime.
done
clear
B)
It is less effective than others.
done
clear
C)
It is not easily/rapidly degraded in nature.
done
clear
D)
It is highly expensive.
done
clear
View Answer play_arrow
Sonalika and Kalyan Sona are high yielding verities of
A)
Sugarcane
done
clear
B)
Rice
done
clear
C)
Wheat
done
clear
D)
Maize
done
clear
View Answer play_arrow
In maize, the hybrid vigour is exploited by
A)
Crossing of two inbred parental lines.
done
clear
B)
Harvesting seed from the most productive plants.
done
clear
C)
Inducing mutations.
done
clear
D)
Bombarding the seeds with DNA.
done
clear
View Answer play_arrow
LSD is
A)
Hallucinogenic
done
clear
B)
Sedative
done
clear
C)
Stimulant
done
clear
D)
Tranquilizer
done
clear
View Answer play_arrow
Parthenocarpic tomato fruits can be produced by
A)
Treating the plants with low concentrations of gibberellic acid and auxins.
done
clear
B)
Raising the plants from vernalized seeds.
done
clear
C)
Treating the plants with phenylmercuric acetate.
done
clear
D)
Removing androecium of flowers before the pollen grains are released.
done
clear
View Answer play_arrow
A nucleotide is formed of
A)
Purine, pyrimidine and phosphate
done
clear
B)
Purine, sugar and phosphate
done
clear
C)
Nitrogen base, sugar and phosphate
done
clear
D)
Pyrimidine, sugar and phosphate
done
clear
View Answer play_arrow
Which one contains four pyrimidine bases?
A)
GATCAATGC
done
clear
B)
GCUAGACAA
done
clear
C)
UAGCGGUAA
done
clear
D)
TGCCTAACG
done
clear
View Answer play_arrow
Most diverse macromolecules, found in the cell both physically and chemically are
A)
Proteins
done
clear
B)
Carbohydrates
done
clear
C)
Nucleic acids
done
clear
D)
Lipids
done
clear
View Answer play_arrow
Which of the following is not a reducing sugar?
A)
Galactose
done
clear
B)
Glucose
done
clear
C)
Maltose
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
Antibodies in our body are complex
A)
Steroids
done
clear
B)
Prostaglandins
done
clear
C)
Glycoproteins
done
clear
D)
Lipoproteins
done
clear
View Answer play_arrow
Which is not a true amphibian animal?
A)
Salamander
done
clear
B)
Toad
done
clear
C)
Tortoise
done
clear
D)
Frog
done
clear
View Answer play_arrow
Malpighian tubules are
A)
Excretory organs of insects
done
clear
B)
Excretory organs of annelids
done
clear
C)
Respiratory organs of insects
done
clear
D)
Respiratory organs of annelids
done
clear
View Answer play_arrow
Pneumatic bones are expected to be found in
A)
Pigeon
done
clear
B)
House lizard
done
clear
C)
Frog's tadpole
done
clear
D)
Flying fish
done
clear
View Answer play_arrow
One of the following is a very unique feature of the mammalian body:
A)
Homeothermy
done
clear
B)
Presence of diaphragm
done
clear
C)
Four chambered heart
done
clear
D)
Rib cage
done
clear
View Answer play_arrow
In which one of the following sets of animals do all the four give birth to young ones?
A)
Platypus, penguin, bat, hippopotamus
done
clear
B)
Shrew, bat, cat, kiwi
done
clear
C)
Kangaroo, hedgehog, dolphin, loris
done
clear
D)
Lion, bat, whale, ostrich
done
clear
View Answer play_arrow
Metameric segmentation is the characteristic of
A)
Echinodermata and Annelida
done
clear
B)
Annelida and Arthropoda
done
clear
C)
Mollusca and Chordata
done
clear
D)
Platyhelminthes and Arthropoda
done
clear
View Answer play_arrow
Which animal has become extinct fromIndia?
A)
Snow Leopard
done
clear
B)
Hippopotamus
done
clear
C)
Wolf
done
clear
D)
Cheetah
done
clear
View Answer play_arrow
The functional unit of contractile system in striated muscle is
A)
Myofibril
done
clear
B)
Sarcomere
done
clear
C)
Z-band
done
clear
D)
Cross bridges
done
clear
View Answer play_arrow
ATPase enzyme needed for muscle contraction is located in
A)
Actinin
done
clear
B)
Troponin
done
clear
C)
Myosin
done
clear
D)
Actin
done
clear
View Answer play_arrow
The contraction of gall bladder is due to
A)
Gastrin
done
clear
B)
Cholecystokinin
done
clear
C)
Secretin
done
clear
D)
Enterogastrone
done
clear
View Answer play_arrow
Angiotensinogen is a protein produced and secreted by
A)
Endothelial cells (lining the blood vessels)
done
clear
B)
Liver cells
done
clear
C)
Juxtaglomerular (JG) cells
done
clear
D)
Macula densa cells
done
clear
View Answer play_arrow
In alveoli of the lungs, the air at the site of gas exchange is separated from the blood by
A)
Alveolar epithelium only.
done
clear
B)
Alveolar epithelium and capillary endothelium.
done
clear
C)
Alveolar epithelium, capillary endothelium and tunica adventitia.
done
clear
D)
Alveolar epithelium, capillary endothelium, a thin layer of tunica media and tunica adventitia.
done
clear
View Answer play_arrow
Child death may occur in the marriage between
A)
\[R{{h}^{+}}\]man and \[R{{h}^{+}}\]woman
done
clear
B)
\[R{{h}^{+}}\] man and\[R{{h}^{-}}\]woman
done
clear
C)
\[R{{h}^{-}}\]man and \[R{{h}^{-}}\]woman
done
clear
D)
\[R{{h}^{-}}\] man and\[R{{h}^{-}}\]woman
done
clear
View Answer play_arrow
Pace maker of heart is
A)
AV node
done
clear
B)
Bundle of His
done
clear
C)
SA node
done
clear
D)
Purkinje fibres
done
clear
View Answer play_arrow
Bundle of His is a network of
A)
Nerve fibres found throughout the heart.
done
clear
B)
Muscle fibres distributed throughout the heart walls.
done
clear
C)
Muscle fibres found only in the ventricle wall.
done
clear
D)
Nerve fibres distributed in ventricles.
done
clear
View Answer play_arrow
Nitrogenous waste products are eliminated mainly as
A)
Urea in tadpole and ammonia in adult frog.
done
clear
B)
Ammonia in tadpole and urea in adult frog.
done
clear
C)
Urea in both tadpole and adult frog.
done
clear
D)
Urea in tadpole and uric acid in adult frog.
done
clear
View Answer play_arrow
Lysozyme that is present in perspiration and tears destroys
A)
Certain types of bacteria
done
clear
B)
All viruses
done
clear
C)
Most virus-infected cells
done
clear
D)
Certain fungi
done
clear
View Answer play_arrow
Which statement is correct for muscle contraction?
A)
Length of H-line decreases
done
clear
B)
Length of A-band remains constant
done
clear
C)
Length of I-band increases
done
clear
D)
Length of two Z-lines increases
done
clear
View Answer play_arrow
Respiratory centre is situated in
A)
Cerebellum
done
clear
B)
Medulla oblongata
done
clear
C)
Hypothalamus
done
clear
D)
Cerebrum
done
clear
View Answer play_arrow
In the chemistry of vision in mammals, the photo- sensitive substance is called
A)
Sclerotin
done
clear
B)
Retinol
done
clear
C)
Rhodopsin
done
clear
D)
Melanin
done
clear
View Answer play_arrow
Which hormone possesses anti-insulin effect?
A)
Cortisol
done
clear
B)
Calcitonin
done
clear
C)
Oxytocin
done
clear
D)
Aldosterone
done
clear
View Answer play_arrow
Which one of the following pairs matches a hormone with a disease resulting from its deficiency?
A)
Insulin - Diabetes insipidus
done
clear
B)
Relaxin - Gigantism
done
clear
C)
Prolactin - Cretinism
done
clear
D)
Parathyroid hormone - Tetany
done
clear
View Answer play_arrow
The correct sequence in the process of development of human embryo is
A)
Fertilization - zygote - cleavage - morula - blastula - gastrula
done
clear
B)
Fertilization -cleavage-morula-zygote-blastula-gastrula
done
clear
C)
Fertilization - zygote - blastula - morula - cleavage - gastrula
done
clear
D)
Cleavage - zygote - Fertilization - morula - blastula - gastrula
done
clear
View Answer play_arrow
What is true for cleavage?
A)
Size of embryo increases
done
clear
B)
Size of cells decrease
done
clear
C)
Size of cells increase
done
clear
D)
Size of embryo decreases
done
clear
View Answer play_arrow
The gland that regresses with age is
A)
Adrenal
done
clear
B)
Gonad
done
clear
C)
Thymus
done
clear
D)
Thyroid
done
clear
View Answer play_arrow
Barr body in human female is formed by
A)
Inactivation of paternal X chromosome
done
clear
B)
Inactivation of maternal X chromosome
done
clear
C)
Inactivation of both X chromosomes
done
clear
D)
Inactivation of either the paternal or maternal X chromosome
done
clear
View Answer play_arrow
Which of the following is an example of pleiotropic effect?
A)
Haemophilia
done
clear
B)
Thalassemia
done
clear
C)
Sickle cell anaemia
done
clear
D)
Colour blindness
done
clear
View Answer play_arrow
Which one of the following is an example of polygenic inheritance?
A)
Production of male honey bee
done
clear
B)
Pod shape in garden pea
done
clear
C)
Skin colour in humans
done
clear
D)
Flower colour in Mirabilis jalapa
done
clear
View Answer play_arrow
The homologous organs are those that show similarity in
A)
Size
done
clear
B)
Origin
done
clear
C)
Function
done
clear
D)
Appearance
done
clear
View Answer play_arrow
Extremities, tail and ear are relatively shorter in animals living in cooler regions as compared to those inhabiting warmer zones. This is
A)
Bergman's rule
done
clear
B)
Jordan's rule
done
clear
C)
Gloger's rule
done
clear
D)
Alien's rule
done
clear
View Answer play_arrow
Hugo de Vries gave his mutation theory on organic evolution while working on
A)
Pisum sativum
done
clear
B)
Drosophila melanogaster
done
clear
C)
Oenothera lamarckiana
done
clear
D)
Althea rosea
done
clear
View Answer play_arrow
Which is the correct order of increasing geological time scale for a hypothetical vertebrate evolution?
A)
Cenozoic, Mesozoic, Palaeozoic, Precambrian
done
clear
B)
Cenozoic, Palaeozoic, Mesozoic, Precambrian
done
clear
C)
Precambrian, Cenozoic, Palaeozoic, Mesozoic
done
clear
D)
Precambrian, Palaeozoic, Mesozoic, Cenozoic
done
clear
View Answer play_arrow
Forelimbs of cat and lizard is used in walking; fore- limbs of whale is used in swimming and forelimbs of bats is used in flying are an example of
A)
Analogous organs
done
clear
B)
Adaptive radiation
done
clear
C)
Homologous organs
done
clear
D)
Convergent evolution
done
clear
View Answer play_arrow
The earliest animal to have been domesticated by man was most likely the
A)
Horse
done
clear
B)
Cow
done
clear
C)
Dog
done
clear
D)
Pig
done
clear
View Answer play_arrow
The human immunodeficiency virus (HIV) has a protein coat and a genetic material which is a
A)
Single-stranded DNA
done
clear
B)
Double-stranded DNA
done
clear
C)
Single-stranded RNA
done
clear
D)
Double-stranded RNA
done
clear
View Answer play_arrow
The formula for exponential population growth is
A)
\[\frac{dN}{dt}=rN\]
done
clear
B)
\[\frac{rN}{dN}=dt\]
done
clear
C)
\[\frac{dN}{dt}=rN\]
done
clear
D)
\[\frac{dt}{dN}=rN\]
done
clear
View Answer play_arrow
The wild type E. coli cells are growing in normal medium with glucose. They are transferred to a medium containing only lactose as sugar. Which of the following changes takes place?
A)
The lac operon is repressed
done
clear
B)
All operons are induced
done
clear
C)
The lac operon is induced
done
clear
D)
E. coli cells stop dividing
done
clear
View Answer play_arrow
In the genetic code dictionary, how many codons are used to code for all the 20 essential amino acids?
A)
60
done
clear
B)
20
done
clear
C)
64
done
clear
D)
61
done
clear
View Answer play_arrow
The Montreal Protocol which serves as an appropriate action to protect the ozone layer from human activities was passed in the year.
A)
1987
done
clear
B)
1988
done
clear
C)
1985
done
clear
D)
1986
done
clear
View Answer play_arrow
Oxygen dissociation curve of haemoglobin is
A)
Sigmoid
done
clear
B)
Hyperbolic
done
clear
C)
Linear
done
clear
D)
Parabolic
done
clear
View Answer play_arrow