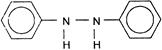A uniform circular disc has radius r. A square portion of diagonal a is cut from it. The centre of mass of the remaining portion from the centre is:
A)
\[\frac{a}{\pi -2}\]
done
clear
B)
\[\frac{a}{2\pi -2}\]
done
clear
C)
\[\frac{a}{4\pi -2}\]
done
clear
D)
\[\frac{a}{4\pi -2}\]
done
clear
View Answer play_arrow
A bullet of mass \[20g\] travelling horizontaly with a speed of \[500m/s\] passes through a wooden block of mass 80kg initially at rest on a level surface. The bullet emerges with a speed of \[100m/s\] and the block slides 20cm on the surface before coming to rest. The coefficient of friction between the block and the surface is \[(g=10m{{s}^{-2}})\]
A)
\[0.4\]
done
clear
B)
\[0.25\]
done
clear
C)
\[0.2\]
done
clear
D)
\[0.1\]
done
clear
View Answer play_arrow
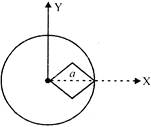
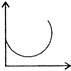
The figure show how the displacement of a particle discribing SUM varies with time. The incorrect statement is:
A)
the force is zero at time \[\frac{3T}{4}\]
done
clear
B)
the velocity is maximum at time \[T/2\]
done
clear
C)
the acceleration is maximum at time T
done
clear
D)
the potential energy = total energy of time \[T/2\]
done
clear
View Answer play_arrow
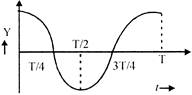
A container has two immiscrible liquid of densities \[{{P}_{1}}\] and \[{{P}_{2}}(>{{P}_{1}})\]. A capillary tube of radius r is inserted in the liquid so that its bottom reaches up to the denser liquid. The denser liquid rises in the capillary and attains a height h from the interface of the liquids, which is equal to the column length of the lighter liquid. Assuming angle of contact to be zero, the surface tension of havier liquid is:
A)
\[2\pi r{{p}_{2}}gh\]
done
clear
B)
\[\frac{{{p}_{2}}rgh}{2}\]
done
clear
C)
\[\frac{r}{2}\,({{p}_{2}}-{{p}_{1}})gh\]
done
clear
D)
\[2\pi r\,({{p}_{2}}-{{p}_{1}})gh\]
done
clear
View Answer play_arrow
The time period of artificial satellite in a circular orbit of radius R is T. The radius of the orbit in which time period is 8T is:
A)
2R
done
clear
B)
3R
done
clear
C)
4R
done
clear
D)
5R
done
clear
View Answer play_arrow
A stone is projected at time, \[t=0\] with a speed \[{{V}_{0}}\]at an angle \[\theta \] with the horizontal in a uniform gravitational field. The rate of work done (p) by the gravitational force plotted against time (t) will be as:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A man holds a ball of mass \[1/2\text{ }kg\] in his hand. He throws it vertically upward. During this process his hand moves up by 40cm the ball leaves his hand with an upward velocity of \[4m{{s}^{-1}},\] the constant force with which the man pushes the ball is:
A)
2N
done
clear
B)
10N
done
clear
C)
15N
done
clear
D)
7N
done
clear
View Answer play_arrow
A particle is projected horizontally from the top of a cliff of height H with a speed\[\sqrt{2gh}\] . The radius of curvature of the trajectory at the instant of projection will.
A)
\[H/2\]
done
clear
B)
\[H\]
done
clear
C)
\[2H\]
done
clear
D)
\[\propto \]
done
clear
View Answer play_arrow
Energy due to position of a particles is given, \[U=\frac{\alpha \sqrt{V}}{Y+\beta },\] where \[\alpha \] and \[\beta \] are constants, Y is distance. The dimensions of \[(\alpha \times \beta )\] are
A)
\[[{{M}^{3}}L{{T}^{0}}]\]
done
clear
B)
\[[{{M}^{1/2}}{{L}^{3/2}}{{T}^{-2}}]\]
done
clear
C)
\[[{{M}^{0}}{{L}^{-7/2}}{{T}^{0}}]\]
done
clear
D)
\[[M{{L}^{7/2}}{{T}^{-2}}]\]
done
clear
View Answer play_arrow
The diameter of a wire is measured with a screw gauge having 50 divisions on circular scale and by one complete rotation of circular scale, main scale moves\[0.5mm\]. If rolling of screw gauge is\[0.250cm\]. The minimum percentage error in the reading will be:
A)
\[0.4\]
done
clear
B)
\[0.8\]
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
For a spring block system is SHM, during one time period the kinetic energy of the block (starting from mean position)
A)
is always contant
done
clear
B)
initially increases and then decreases
done
clear
C)
first decreases and then increases
done
clear
D)
keeps on changing
done
clear
View Answer play_arrow
A simple harmonic motion has an amplitude A and time period T. The time required by it to travel from \[X-A\] to \[X=A/2\] is:
A)
\[T/6\]
done
clear
B)
\[T/4\]
done
clear
C)
\[T/3\]
done
clear
D)
\[T/2\]
done
clear
View Answer play_arrow
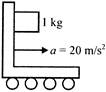
A block of mass 1kg just remains in equilibrium with the verticle wall of a cart accelerating uniformly with \[20m/{{s}^{2}}\]as shown. The coefficient of friction between block and wall is \[(g=10m/{{s}^{2}})\]:
A)
\[0.1\]
done
clear
B)
\[0.2\]
done
clear
C)
\[0.5\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
A wire of length L, Cross-sectional area A is made of a material of Young's modulas Y. The wire is stretched by an amount X, which lies well within the elastic limit. The work done(W) by the force is:
A)
\[W=\frac{Y{{X}^{2}}}{{{L}^{2}}}\]
done
clear
B)
\[W=\frac{Y{{X}^{2}}}{2{{L}^{2}}}\]
done
clear
C)
\[W=\frac{1}{2}\frac{YA{{X}^{2}}}{{{L}^{2}}}\]
done
clear
D)
\[W=\frac{YA{{X}^{2}}}{L}\]
done
clear
View Answer play_arrow
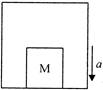
With what acceleration a should box of figure descend so that the block of mass m exerts a force \[mg/g\] on the floor for the box?
A)
\[g/4\]
done
clear
B)
\[g/2\]
done
clear
C)
\[3g/4\]
done
clear
D)
\[4g\]
done
clear
View Answer play_arrow
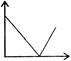
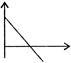
Time-displacement \[(t-x)\]graph of two objects A and B is shown in figure. The ratio of their speeds \[({{V}_{A}}/{{V}_{B}})\] is \[(\tan {{37}^{o}}=3/4)\]
A)
\[\frac{\sqrt{3}}{4}\]
done
clear
B)
\[\frac{4}{\sqrt{3}}\]
done
clear
C)
\[\frac{4}{3\sqrt{3}}\]
done
clear
D)
\[\frac{3\sqrt{3}}{4}\]
done
clear
View Answer play_arrow
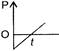
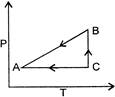
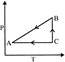
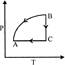
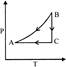
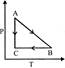
P-T curve of the following process will be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A stretched wire of some length under a tension is vibrating with its fundamental frequency. Its length is decreased by 45%, and tension is increased by 21%. Now its fundamental frequency (assuming linear mass density remains the same):
A)
increases by 50%
done
clear
B)
increases by 100%
done
clear
C)
decreases by 50%
done
clear
D)
decreases by 25%
done
clear
View Answer play_arrow
A wave is represented by the equation\[Y=0.1\,\sin \,(100nt-KX)\]. If wave velocity is \[100m/s,\] its waven number is equal to:
A)
\[1{{m}^{-1}}\]
done
clear
B)
\[2{{m}^{-1}}\]
done
clear
C)
\[\pi {{m}^{-1}}\]
done
clear
D)
\[2\pi {{m}^{-1}}\]
done
clear
View Answer play_arrow
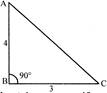
There is a flat uniform triangular plate ABC such that \[AB=4cm,\text{ }ABC=3cm\] and \[\angle ABC={{90}^{o}},\]as shown in figure. The moment of inertia of the plate about AB, BC and CA is axis is respectively \[{{l}_{1}}{{l}_{2}}\] and \[{{l}_{3}}\]. The incorrect statement is:
A)
\[{{l}_{3}}<{{l}_{2}}\]
done
clear
B)
\[{{l}_{2}}>{{l}_{1}}\]
done
clear
C)
\[{{l}_{3}}<{{l}_{1}}\]
done
clear
D)
\[{{l}_{3}}>{{l}_{2}}\]
done
clear
View Answer play_arrow
A magnetic needle is kept in a non-uniform magnetic field. If experiences
A)
a torque but not a force
done
clear
B)
neither a force non a torque
done
clear
C)
a force and a torque
done
clear
D)
a force but not a torque
done
clear
View Answer play_arrow
A length of wire carries a steady current. It is bent first to form circular coil of one turn. The same length is now bent more sharply to give a double loop of smaller radius. The magnetic field at the centre caused by the same current is:
A)
a quarter of its first value
done
clear
B)
unaltered
done
clear
C)
fourtimes of its first value
done
clear
D)
half of its first value
done
clear
View Answer play_arrow
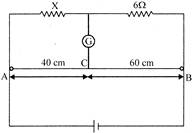
In the circuit shown, a meter bridge is in its balanced state. The meter bridge wire has resistance\[0.1\Omega /cm\]. The value of unknown resistance X and the current drawn from the battery of negligible resistance is:
A)
\[6\Omega ,\,5A\]
done
clear
B)
\[4\Omega ,\,0.1A\]
done
clear
C)
\[4\Omega ,\,1.0A\]
done
clear
D)
\[12\Omega ,\,0.5A\]
done
clear
View Answer play_arrow
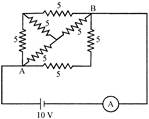
The ammeter will read the value of current:
A)
\[3A\]
done
clear
B)
\[\frac{10}{3}A\]
done
clear
C)
\[30A\]
done
clear
D)
\[\frac{100}{3}A\]
done
clear
View Answer play_arrow
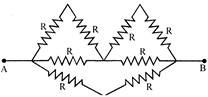
What is the equivalent resistance between A and B (each resistor has resistance R):
A)
\[\frac{4R}{3}\]
done
clear
B)
\[\frac{5R}{3}\]
done
clear
C)
\[\frac{4R}{5}\]
done
clear
D)
\[\frac{3R}{5}\]
done
clear
View Answer play_arrow
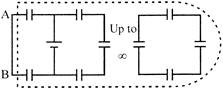
The equivalent capacitance of the network (with all capacitors having the same capacitance c) is:
A)
\[\alpha \]
done
clear
B)
zero
done
clear
C)
\[C\left( \frac{\sqrt{3}-1}{2} \right)\]
done
clear
D)
\[C\left( \frac{\sqrt{3}+1}{2} \right)\]
done
clear
View Answer play_arrow
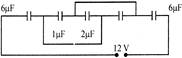
Figure shown in five capacitors connected across a 12V power supply. What is the potential drop across the \[2\mu f\] capicitor?
A)
2V
done
clear
B)
4V
done
clear
C)
8V
done
clear
D)
10V
done
clear
View Answer play_arrow
a particle A has charge \[+q\] and a particle B has charge \[+4q\] with each of them having the same mass m. When allowed to fall from rest through the same electric potential difference, the ratio of their speed \[\frac{{{V}_{A}}}{{{V}_{B}}}\] will become:
A)
\[2:1\]
done
clear
B)
\[1:2\]
done
clear
C)
\[1:4\]
done
clear
D)
\[4:1\]
done
clear
View Answer play_arrow
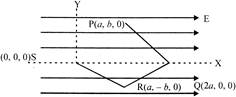
A point charge q moves from point P to point S along the path PQRS in a uniform electric field E pointing parallel to the positive direction of the x-axis, the coordinate of the points P,Q,R and S are \[(a,b,0)\,\,(2a,0,0),\,(a-b,0)\] and \[(0,0,0)\] respectively. The work done by the field in the above process is given by the expression:
A)
\[qaE\]
done
clear
B)
\[-qaE\]
done
clear
C)
\[q\sqrt{({{a}^{2}}+{{b}^{2}})}E\]
done
clear
D)
\[3qE\sqrt{{{a}^{2}}+{{b}^{2}}}\]
done
clear
View Answer play_arrow
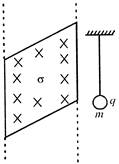
A small charged particle of mass m and charge q is suspended by an insulated thread in front of a very large sheet of charge density a. The angle made by the treat with the vertical is equilibrium is:
A)
\[{{\tan }^{-1}}\left( \frac{\sigma q}{2{{\varepsilon }_{0}}mg} \right)\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{\sigma q}{{{\varepsilon }_{0}}mg} \right)\]
done
clear
C)
\[\tan \left( \frac{2\sigma q}{{{\varepsilon }_{0}}mg} \right)\]
done
clear
D)
zero
done
clear
View Answer play_arrow
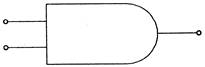
Which logic gate is represented by following diagram?
A)
AND
done
clear
B)
OR
done
clear
C)
NOR
done
clear
D)
XOR
done
clear
View Answer play_arrow
In the hydrogen atomspectrum, \[{{\lambda }_{3}}=1\] and \[{{\lambda }_{2}}=1\]represent wavelengths emitted due to transition from second and first excited states to the ground state respectively. The value of \[\frac{{{\lambda }_{3-1}}}{{{\lambda }_{2-1}}}\] is:
A)
\[\frac{27}{32}\]
done
clear
B)
\[\frac{32}{27}\]
done
clear
C)
\[\frac{4}{9}\]
done
clear
D)
\[\frac{9}{4}\]
done
clear
View Answer play_arrow
In a transistor, connected in the \[C\varepsilon \] confugarion, the collection current changes by 1mA when the base current changes by \[10\mu A,\] in the operating condition. Then the value of a for the transistor will be:
A)
\[0.88\]
done
clear
B)
\[0.99\]
done
clear
C)
\[0.72\]
done
clear
D)
\[0.8\]
done
clear
View Answer play_arrow
The speed of an electron having a wavelenth of the order of \[1\overset{o}{\mathop{A}}\,\] will be:
A)
\[7.25\times {{10}^{6}}m/s\]
done
clear
B)
\[6.26\times {{10}^{6}}m/s\]
done
clear
C)
\[5.25\times {{10}^{6}}m/s\]
done
clear
D)
\[4.25\times {{10}^{6}}\]
done
clear
View Answer play_arrow
In a photo electric experiment, it was found that the stopping potential decreases from 1.85 V to 0.82 V as the wavelength of the incident light is varies from 300 nm to 400 nm. The value of the Planck constant from these data is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In an ore containg uranium, the ratio of
to
nuclei is 3. The age of the ore, assuming that all the lead present life of
to be
In
in
)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
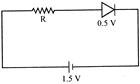
A diode used in the circuit shown has constant voltage drop of \[0.5V\]at all currents and a maximum power rating of \[100mW\]. What should be the value of the resistor R, connected in series with the diode to obtain in maximum current?
A)
\[5\Omega \]
done
clear
B)
\[5.6\Omega \]
done
clear
C)
\[6.76\Omega \]
done
clear
D)
\[20\Omega \]
done
clear
View Answer play_arrow
A concave lens of focal length 10cm and a convex lens of focal length 20cm are placed certain distance apart. If parallel rays incident on the lens becomes converging after passing through other lens, then the separation between the lenses must be greater than.
A)
zero
done
clear
B)
\[5cm\]
done
clear
C)
\[10cm\]
done
clear
D)
\[9cm\]
done
clear
View Answer play_arrow
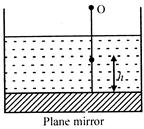
A plane mirror's placed at the bottom of a tank containing a liquid of refractive index \[\mu ,\] P is a small object at a height h above the plane mirror. An observer O, vertically above \[{{P}_{app}}\] outside the liquid. Observer, P and its image in the mirror. The apparent distance between object and its image and will be:
A)
\[2\mu h\]
done
clear
B)
\[\frac{2h}{\mu }\]
done
clear
C)
\[\frac{2h}{\mu -1}\]
done
clear
D)
\[h\left( \frac{1}{\mu } \right)\]
done
clear
View Answer play_arrow
If a graph is drawn between the separation of slits and bandwidth in Young's double slit experiment, the graph will be:
A)
a straight line having positive slope
done
clear
B)
a straight line having negative slope
done
clear
C)
a rectangular hyperbola
done
clear
D)
a parabola
done
clear
View Answer play_arrow
An astronical telescope has magnifying power 10. The focal length of the eyepiece is 20cm. The focal length of the objective is:
A)
\[1/200\,cm\]
done
clear
B)
\[1/2\,cm\]
done
clear
C)
\[2\,cm\]
done
clear
D)
\[200\,cm\]
done
clear
View Answer play_arrow
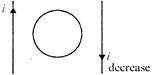
What is direction of induced current in the coil as shown in the figure? (if rate of increase of current is equal to the rate of decrease)
A)
Clockwise
done
clear
B)
Anti-clockwise
done
clear
C)
Zero
done
clear
D)
Not defined with same rate
done
clear
View Answer play_arrow
A semicircular conducting ring is placed in Y-Z plane in a uniform magnetic field directed along positive Z-direction. An induced emf will be developed in the ring, if it is moved along:
A)
position X-direction
done
clear
B)
position Y-direction
done
clear
C)
position Z-direction
done
clear
D)
None of those
done
clear
View Answer play_arrow
If the flux of magnetic through a coil of resistance R and having n turns changes from \[{{\phi }_{1}}\] to \[{{\phi }_{2}},\] then the magnitude of the charge that possess through the coil is:
A)
\[\frac{\left( {{\phi }_{2}}-{{\phi }_{1}} \right)}{R}\]
done
clear
B)
\[\frac{n\,\left( {{\phi }_{2}}-{{\phi }_{1}} \right)}{R}\]
done
clear
C)
\[\frac{\,\left( {{\phi }_{2}}-{{\phi }_{1}} \right)}{nR}\]
done
clear
D)
\[\frac{nR}{\,\left( {{\phi }_{2}}-{{\phi }_{1}} \right)}\]
done
clear
View Answer play_arrow
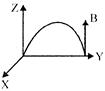
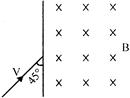
Magnetic inductance B exists as shown in the figure. On the other side of line, a charged particle of charge q, mass m enters the magnetic field at\[{{45}^{o}}\]. The displacement of the particle when it emerges out of the magnetic field will be (velocity of the particles is V)
A)
\[\frac{\sqrt{2}m\,\,V}{qB}\]
done
clear
B)
\[\left( \frac{mV}{qe} \right)\]
done
clear
C)
\[\frac{m\upsilon }{\sqrt{2q\,B}}\]
done
clear
D)
\[\frac{\sqrt{3m\,V}}{qB}\]
done
clear
View Answer play_arrow
At \[{{25}^{o}}C\] the total pressure of an ideal solution obtained by mixing 3 moles of A and 2 moles of B is 184 torr. What is the vapour pressure (in torr) of pure B at the same temperature? (Vapour pressure of pure A at \[{{25}^{o}}C\] is\[200\text{ }torr\]).
A)
180
done
clear
B)
160
done
clear
C)
16
done
clear
D)
100
done
clear
View Answer play_arrow
Relative lowering of vapour pressure of a dilute solution is \[0.2\]. What is the mole fraction of the non-volatile solute?
A)
\[0.8\]
done
clear
B)
\[0.5\]
done
clear
C)
\[0.3\]
done
clear
D)
\[0.2\]
done
clear
View Answer play_arrow
What is the cell reaction occuring in Daniel cell (Galvanic cell)?
A)
\[C{{u}_{(s)}}+ZnS{{O}_{4(aq)}}\xrightarrow{{}}CuS{{O}_{4(aq)}}+Z{{n}_{(s)}}\]
done
clear
B)
\[Z{{n}_{(s)}}+CuS{{O}_{4}}(aq)\xrightarrow{{}}C{{u}_{(s)}}+ZnS{{O}_{4}}_{(aq)}\]
done
clear
C)
\[N{{i}_{(s)}}+ZnS{{O}_{4}}(aq)\xrightarrow{{}}NiS{{O}_{4}}_{(aq)}+Z{{n}_{(s)}}\]
done
clear
D)
\[2N{{a}_{(s)}}+CdS{{O}_{4}}(aq)\xrightarrow{{}}N{{a}_{2}}S{{O}_{4}}_{(aq)}+C{{d}_{(s)}}\]
done
clear
View Answer play_arrow
The magnetic moment (in BM) of \[Z{{n}^{2+}}\] ion according to spin-only formula is
A)
zero
done
clear
B)
\[1.73\]
done
clear
C)
\[2.84\]
done
clear
D)
\[3.87\]
done
clear
View Answer play_arrow
Which of the following statements (or equation) is correct?
A)
The units of cell emf are \[V\,\,c{{m}^{-1}}\]
done
clear
B)
\[\Delta G=\frac{nF}{{{E}_{cell}}}\]
done
clear
C)
In Galvanic cell, chemical energy is transformed into electrical energy
done
clear
D)
Oxidation state of \[Mn\] in potassium permanganate is \[+6\]
done
clear
View Answer play_arrow
The gold number of some colloidal solutions are given below
Colloidal Solution Gold Number A 0.01 B 2.5 C 20
The protective nature of these colloidal solutions follow the order:
A)
\[C>B>A\]
done
clear
B)
\[A>B>C\]
done
clear
C)
\[A=B=C\]
done
clear
D)
\[B>A>C\]
done
clear
View Answer play_arrow
What is the energy (in eV) required to excite the electron from \[n=1\] to \[n=2\] state in hydrogen atom?
A)
\[13.6\]
done
clear
B)
\[3.4\]
done
clear
C)
\[17.0\]
done
clear
D)
\[10.2\]
done
clear
View Answer play_arrow
The 3d- block element that exhibit maximum number of oxidation states in
A)
\[Sc\]
done
clear
B)
\[Ti\]
done
clear
C)
\[Mn\]
done
clear
D)
\[Zn\]
done
clear
View Answer play_arrow
The activity of a radioactive nuclide is \[2\times {{10}^{7}}\] disintegrations per minute. After \[23.03\text{ }min,\]its activity is reduced to\[2\times {{10}^{6}}dpm\]. What is the average life of the nuclide?
A)
100 min
done
clear
B)
10 min
done
clear
C)
50 min
done
clear
D)
5 min
done
clear
View Answer play_arrow
The equilibrium reaction that is not influenced by volume change at constant temperature is:
A)
\[{{H}_{2(g)}}+{{I}_{2(g)}}2H{{I}_{(g)}}\]
done
clear
B)
\[{{N}_{2(g)}}+3{{H}_{2(g)}}2N{{H}_{3(g)}}\]
done
clear
C)
\[{{N}_{2}}{{O}_{4(g)}}2N{{O}_{2(g)}}\]
done
clear
D)
\[2N{{O}_{(g)}}+{{O}_{2}}2N{{O}_{2(g)}}\]
done
clear
View Answer play_arrow
Consider the following solutions of equal concentration
\[A=N{{H}_{4}}Cl\] \[B=C{{H}_{3}}COONA\] \[C=N{{H}_{4}}OH\] \[D=C{{H}_{3}}COOH\]
A buffer solution can be obtained by mixing equal volumes of:
A)
C and D
done
clear
B)
A and B
done
clear
C)
A and C
done
clear
D)
C and D
done
clear
View Answer play_arrow
Which of the following is a Lewis acid?
A)
\[AlC{{l}_{3}}\]
done
clear
B)
\[C{{l}^{-}}\]
done
clear
C)
\[Co\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
View Answer play_arrow
The reason for almost doubling the rate of reaction on increasing the temperature of the reaction system by \[{{10}^{o}}C\] is:
A)
the value of threshold energy increases
done
clear
B)
collision frequency increases
done
clear
C)
the fraction of the molecule having energy equal to threshold energy or more increases
done
clear
D)
activation energy decreases
done
clear
View Answer play_arrow
Which of the following is incorrect?
A)
Relative lowering of vapour pressure is independent of the nature of the solute and the solvent.
done
clear
B)
The vapour pressure is a colligative property.
done
clear
C)
Vapour pressure of a solution is lower than the vapour pressure of the solvent.
done
clear
D)
The relative lowering of vapour pressure is directly proportional to the original pressure.
done
clear
View Answer play_arrow
In a solid lattice, the cation has left a lattice site and is located at interstitial position, the lattice defect is:
A)
interstitial defect
done
clear
B)
vacancy defect
done
clear
C)
Frenkel defect
done
clear
D)
schottky defect
done
clear
View Answer play_arrow
The Ti-bonded organo metallic compound which has ethene as one of its component is:
A)
Zeise's salt
done
clear
B)
ferro cene
done
clear
C)
tetraethyltin
done
clear
D)
dibenzene chromium
done
clear
View Answer play_arrow
The correct set of the four quantum numbers of a 4d electrons is:
A)
\[4,2,1,-\frac{1}{2}\]
done
clear
B)
\[4,2,1,0\]
done
clear
C)
\[4,3,2,+\frac{1}{2}\]
done
clear
D)
\[4,3,-2,+\frac{1}{2}\]
done
clear
View Answer play_arrow
\[C{{H}_{3}}Br+O{{H}^{-}}\xrightarrow{{}}C{{H}_{3}}OH+B{{r}^{-}}\] reaction proceeds by \[{{S}_{N}}2\] mechanism. Its rate is dependent on the concentration of:
A)
\[C{{H}_{3}}Br,\,C{{H}_{3}}OH\]
done
clear
B)
\[C{{H}_{3}}Br,\,O{{H}^{-}}\]
done
clear
C)
\[C{{H}_{3}}Br\] only
done
clear
D)
\[O{{H}^{-}}\] only
done
clear
View Answer play_arrow
Which of the following molecule in its valence shell has three bond pairs of electrons and one lone pair of electrons?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[B{{F}_{3}}\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
The complex which does not give a precipitate with \[AgN{{O}_{3}}\] solution:
A)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}]\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]
done
clear
C)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
D)
\[[Cr{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl\]
done
clear
View Answer play_arrow
The metal ion in complex A has EAN identical to the atomic number of krypton. A is: (Atomic number of \[Cr=24,\text{ }Fe=26\] \[Pd=46\])
A)
\[[Pd{{(N{{H}_{3}})}_{6}}]C{{l}_{4}}\]
done
clear
B)
\[[Cr{{(N{{H}_{3}})}_{5}}Cl]S{{O}_{4}}\]
done
clear
C)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
D)
\[[Cr{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl\]
done
clear
View Answer play_arrow
The radius ratio \[\left( \frac{{{r}^{+}}}{{{r}^{-}}} \right)\] of an ionic solid \[({{A}^{+}}{{B}^{-}})\] is\[0.69\]. What is the co-ordination number of \[{{B}^{-}}\]?
A)
6
done
clear
B)
8
done
clear
C)
2
done
clear
D)
10
done
clear
View Answer play_arrow
German silver is an alloy of
A)
\[Cu,\text{ }Zn\]
done
clear
B)
\[Cu,\text{ }Ni,\text{ }Zn\]
done
clear
C)
\[Cu,\text{ }Sn,\text{ }Zn\]
done
clear
D)
\[Cu,\text{ }Zn\]
done
clear
View Answer play_arrow
Which of the following is used as indelible ink?
A)
Aqueous \[CuS{{O}_{4}}\] solution
done
clear
B)
Aqueous \[AgN{{O}_{3}}\] solution
done
clear
C)
Aqueous \[NaCl\] solution
done
clear
D)
Aqueous \[NaOH\] solution
done
clear
View Answer play_arrow
Which of the following ores is an ore of copper?
A)
Argentite
done
clear
B)
Haematite
done
clear
C)
Malachite
done
clear
D)
Calamine
done
clear
View Answer play_arrow
Consider the following abbreviations for hydrated alkali ions:
\[X={{[Li{{({{H}_{2}}O)}_{n}}]}^{+}}\] \[Y={{[K{{({{H}_{2}}O)}_{n}}]}^{+}}\] \[Z={{[Cs{{({{H}_{2}}O)}_{n}}]}^{+}}\]
What is the correct order of size of these hydrated alkali ions?
A)
\[Z>Y>X\]
done
clear
B)
\[Z>X>Y\]
done
clear
C)
\[X=Y=Z\]
done
clear
D)
\[X>Y>Z\]
done
clear
View Answer play_arrow
\[KMn{{O}_{4}}\] (molecular weight = 158) oxidizes oxalic acid in acid medium to \[C{{O}_{2}}\] and water as follows: \[5{{C}_{2}}O_{4}^{2-}+2MnO_{4}^{-}+16{{H}^{+}}\xrightarrow{{}}10C{{O}_{2}}+2M{{n}^{2+}}+8{{H}_{2}}O\]. What is the equivalent weight of \[KMn{{O}_{4}}\]?
A)
\[158\]
done
clear
B)
\[31.6\]
done
clear
C)
\[39.5\]
done
clear
D)
\[79\]
done
clear
View Answer play_arrow
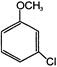
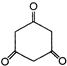
In the reaction
The major product A is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Tautomerism is not exhibited by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Ethylamine reacts with nitrous acid to form
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH,{{N}_{2}},{{H}_{2}}O\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}N_{2}^{+}C{{l}^{-}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}NHOH,\,N{{H}_{3}}\]
done
clear
View Answer play_arrow
Coupling the diazonium salts of following takes place in the order.
A)
\[IV<II<III<I\]
done
clear
B)
\[IV>III<II<I\]
done
clear
C)
\[II<IV<I<III\]
done
clear
D)
\[I<II<III<IV\]
done
clear
View Answer play_arrow
Which of the following pentafluorides cannot be formed?
A)
\[P{{F}_{5}}\]
done
clear
B)
\[As{{F}_{5}}\]
done
clear
C)
\[Sb{{F}_{5}}\]
done
clear
D)
\[Bi{{F}_{5}}\]
done
clear
View Answer play_arrow
Choose the correct statement:
A)
Transition elements have low melting points
done
clear
B)
Transition elements do not have catalytic activity
done
clear
C)
Transition elements exhibit variable oxidation states
done
clear
D)
Transition elements show inert pair effect.
done
clear
View Answer play_arrow
The (R) and (S) enatiomers of an optically active compound differ in
A)
their reactivity
done
clear
B)
their optical rotation of plane polarised light
done
clear
C)
their melting point
done
clear
D)
their solubility in achiral reagents
done
clear
View Answer play_arrow
Chemical A is used for water softening to remove temporary hardness. A reacts with sodium carbonate to generate caustic soda. When \[C{{O}_{2}}\] is bubbled through A, it turns cloudy. What is A?
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[CaO\]
done
clear
C)
\[Ca{{(OH)}_{2}}\]
done
clear
D)
\[Ca{{(HC{{O}_{3}})}_{2}}\]
done
clear
View Answer play_arrow
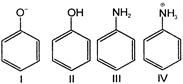
Which of the following species could be expected to exhibit aromatic character?
Select the correct answer from the following
A)
I and TV
done
clear
B)
II and IV
done
clear
C)
I and III
done
clear
D)
II and III
done
clear
View Answer play_arrow
Chlorination of toluene in the presence of light and heat followed by treatment with aqueous \[NaOH\] solution gives:
A)
O-cresol
done
clear
B)
P-cresol
done
clear
C)
benzoic acid
done
clear
D)
2, 4-dihydroxytolucne
done
clear
View Answer play_arrow
Reaction oft- bytylbromide with sodium methoxide produces:
A)
isobutane
done
clear
B)
isobutylene
done
clear
C)
sodium t-butoxide
done
clear
D)
24,-butylmethylether
done
clear
View Answer play_arrow
The space in the dry cell is filled with:
A)
Paste of \[KOH\] and \[ZnO\]
done
clear
B)
\[Mn{{O}_{2}},ZnC{{l}_{2}};\] a filter
done
clear
C)
\[Mn{{O}_{2}},ZnC{{l}_{2}},N{{H}_{4}}Cl\] and a filter
done
clear
D)
\[MnC{{l}_{2}},ZnC{{l}_{2}},N{{H}_{4}}Cl\] and a filter
done
clear
View Answer play_arrow
Which reagent can be used for the alkglation of aromatic nitro compounds?
A)
Methyl sulfinyl carbanion
done
clear
B)
Methyl iodide
done
clear
C)
Dimethyl sulphate
done
clear
D)
Methyl lithium
done
clear
View Answer play_arrow
Which of the following compounds is used as broad spectrum antibiotics?
A)
Ampicillin
done
clear
B)
Penicillin G
done
clear
C)
Penicillin K
done
clear
D)
Tetrycycline
done
clear
View Answer play_arrow
Which is not present in chlorophyll?
A)
Carbon
done
clear
B)
Calcium
done
clear
C)
Magnesium
done
clear
D)
Hydrogen
done
clear
View Answer play_arrow
The monomer used to produce orlon is
A)
\[C{{H}_{2}}=CHF\]
done
clear
B)
\[C{{H}_{2}}=CC{{l}_{2}}\]
done
clear
C)
\[C{{H}_{2}}=CHCl\]
done
clear
D)
\[C{{H}_{2}}=CH-CN\]
done
clear
View Answer play_arrow
The structure of the compound formed, when nitrobcnzene is reduced by lithium aluminium hydride is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is the most stable carbocation?
A)
\[P{{h}_{3}}\overset{+}{\mathop{C}}\,\]
done
clear
B)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,H\]
done
clear
C)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
D)
\[P{{h}_{2}}\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
View Answer play_arrow
Predation is just not act of capturing, killing and eating. Many important functions are related to it, except
A)
Maintains species diversity of community
done
clear
B)
Biological control
done
clear
C)
Conduit of energy level across trophic levels
done
clear
D)
Increase competition between prey population
done
clear
View Answer play_arrow
If the number of pre-reproductive individuals is almost similar to reproductive individuals, then what type of pyramid is formed?
A)
Expanding
done
clear
B)
Triangular
done
clear
C)
Stable
done
clear
D)
Urn shape
done
clear
View Answer play_arrow
Out of a population of 40 fruit flies in a laboratory culture, 4 died during a specified time interval, i.e., for a week, the death rate in population during the period is
A)
\[10\]
done
clear
B)
\[0.1\]
done
clear
C)
\[0.01\]
done
clear
D)
\[100\]
done
clear
View Answer play_arrow
Species which face competition might evolve mechanism of changing feeding time or foraging patterns show
A)
Coexistence
done
clear
B)
Resource partitioing
done
clear
C)
Interference
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
The weed calotropis growing in abundoned fields produces highly poisonous
A)
Cardiac glycosides
done
clear
B)
Muscular glycosides
done
clear
C)
Epithelial glycosides
done
clear
D)
Nervous glycosides
done
clear
View Answer play_arrow
Select the incorrect match
A)
Brood parasitism - Cuckoo
done
clear
B)
Cardiac glycosides - Calotropis
done
clear
C)
Allelopathic plant - Black walnut
done
clear
D)
Sexual deceit - Ficus
done
clear
View Answer play_arrow
Select the incorrect match
A)
Tropical rain forest - Diptcrocarpus
done
clear
B)
Tundra- Betula
done
clear
C)
Temperate broad leaf forest - Cedrus
done
clear
D)
Savanna grasslands - Zizyhus
done
clear
View Answer play_arrow
Phytoplanktons are abundant in _____zone of lakes.
A)
Benthic
done
clear
B)
Profundal
done
clear
C)
Littroal
done
clear
D)
Limnetic
done
clear
View Answer play_arrow
Paramecium Aurelia when kept in a water beaker along with P. Caudatum, will kill P. Caudatum. This represents
A)
Competitive release
done
clear
B)
Competitive exclusion
done
clear
C)
Coexistence
done
clear
D)
Resource - partitioning
done
clear
View Answer play_arrow
All are delights of Keoladeo National Park except
A)
Flamingoes.
done
clear
B)
Siberian Cranes
done
clear
C)
Mohawks
done
clear
D)
Purple herons
done
clear
View Answer play_arrow
Pyramid of number in a tree ecosystem is
A)
Always inverted
done
clear
B)
Upright
done
clear
C)
Always spindle - shaped
done
clear
D)
Either inverted or spindle - shaped
done
clear
View Answer play_arrow
PAR is ______ of total incident radiation
A)
50%
done
clear
B)
10%
done
clear
C)
1-5%.
done
clear
D)
20%
done
clear
View Answer play_arrow
Approximate what percent of incident solar radiation is captured by producers in their photosynthetic activity?
A)
1 - 5%
done
clear
B)
2 - 10%
done
clear
C)
20%
done
clear
D)
90%
done
clear
View Answer play_arrow
In a food chain 10 kg of wheat will produce _______ kg of biomass in goat
A)
1
done
clear
B)
9
done
clear
C)
10
done
clear
D)
5
done
clear
View Answer play_arrow
Pyramid of biomass for aquatic ecosystem is
A)
Upright
done
clear
B)
Spindle - shaped
done
clear
C)
Inverted
done
clear
D)
Booth [a] and [c]
done
clear
View Answer play_arrow
Trophic level of a man eating curd is
A)
\[{{T}_{1}}\]
done
clear
B)
\[{{T}_{2}}\]
done
clear
C)
\[{{T}_{4}}\]
done
clear
D)
\[{{T}_{3}}\]
done
clear
View Answer play_arrow
Mark the incorrect option (with respect to humus)
A)
Dark coloured amorphous matter
done
clear
B)
It is colloidal
done
clear
C)
Function as reservoir of nutrients
done
clear
D)
Slightly alkaline
done
clear
View Answer play_arrow
A very high number of amphibian species are endemic to
A)
Western Ghat
done
clear
B)
Eastern Ghat
done
clear
C)
Eastern Himalayas
done
clear
D)
Sundarban
done
clear
View Answer play_arrow
How many hot spots are present in India?
A)
25
done
clear
B)
6
done
clear
C)
34
done
clear
D)
3
done
clear
View Answer play_arrow
The elephant is the most important protected species in
A)
Manas Sanctuary
done
clear
B)
Kaziranga National Park
done
clear
C)
Ranthambhore National Park
done
clear
D)
More the one are correct
done
clear
View Answer play_arrow
Which one is incorrect?
A)
World forest day is 21st March
done
clear
B)
Tapioca is called energy crop
done
clear
C)
Grassland in India cover 30% of total land
done
clear
D)
Biogas is mixture of 60% methods include protective
done
clear
View Answer play_arrow
Which one is incorrect with respect to classical smog?
A)
It occurs at low temperature
done
clear
B)
Contain sulphur gases
done
clear
C)
It is dark brown or opaque
done
clear
D)
Secondary pollutants are present
done
clear
View Answer play_arrow
Acid rain is caused due to
A)
\[S{{O}_{2}}\] pollution
done
clear
B)
\[C{{O}_{2}}\] pollution
done
clear
C)
Negative pollution
done
clear
D)
Pesticide pollution
done
clear
View Answer play_arrow
Select the correct match
A)
Green muffler - noise pollution
done
clear
B)
Chemical weed - CFC
done
clear
C)
National pollution prevention day - 6th August
done
clear
D)
Byssinosis - Iron mill
done
clear
View Answer play_arrow
Which is not an effect of increasing greenhouse gasses?
A)
Global warming
done
clear
B)
El Nino effect
done
clear
C)
Cooling of stratosphere
done
clear
D)
Decreased sea level
done
clear
View Answer play_arrow
Which one of the following is/are not the example of cross-breed?
A)
Hisardle
done
clear
B)
Sunandini
done
clear
C)
Karan fries
done
clear
D)
Red Sindhi
done
clear
View Answer play_arrow
Inbreeding depression is overcome by
A)
Repeated inbreeding continuously
done
clear
B)
Cross breeding with different breeds
done
clear
C)
Out breeding with same breed
done
clear
D)
Inter specific hybridization
done
clear
View Answer play_arrow
Shagreen is obtained from
A)
Bony fishes
done
clear
B)
Cartilaginous fishes
done
clear
C)
Buffaloes milk
done
clear
D)
Cow milk
done
clear
View Answer play_arrow
Cross-breeding occurs in all except
A)
Cattle
done
clear
B)
Dog
done
clear
C)
Poultry
done
clear
D)
Fishes
done
clear
View Answer play_arrow
Which of the following cow of general utility breed has been crossed with Holstein Friesian breed to raise Karan fries by NDRI?
A)
'Sahiwa? Cow
done
clear
B)
TTaryana' Cow
done
clear
C)
'Kankref? Cow
done
clear
D)
Tharparkar' Cow
done
clear
View Answer play_arrow
Honey is
A)
Highly acidic
done
clear
B)
Slightly acidic
done
clear
C)
Neutral
done
clear
D)
Slightly alkaline
done
clear
View Answer play_arrow
Natural silk does not contain
A)
Fibroin
done
clear
B)
Sericin
done
clear
C)
Nitrogen
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
Silk is the secretion of
A)
Cephalic glands
done
clear
B)
Gastric glands
done
clear
C)
Buccal glands
done
clear
D)
Salivary glands
done
clear
View Answer play_arrow
In the honey-bee colony; which of the following has the longest life span?
A)
Scout bee
done
clear
B)
All workers
done
clear
C)
Drones
done
clear
D)
Queen
done
clear
View Answer play_arrow
Culturing of fresh water fishes is called as
A)
Inland fishery
done
clear
B)
Aquaculture
done
clear
C)
Pisciculture
done
clear
D)
Fishery
done
clear
View Answer play_arrow
DNA finger printing involves
A)
Southern blotting
done
clear
B)
Northern blotting
done
clear
C)
Western blotting
done
clear
D)
ELISA
done
clear
View Answer play_arrow
More productive practice in biotechnology in terms of yield is
A)
Small culture and batch culture
done
clear
B)
Continuous culture
done
clear
C)
Polyculture
done
clear
D)
Composite culture
done
clear
View Answer play_arrow
A chimaeric DNA is formed by
A)
Deleting selectable markers
done
clear
B)
Joining C-DNA with plasmid
done
clear
C)
EcoRI
done
clear
D)
Enzyme P - galatesidase
done
clear
View Answer play_arrow
Which of the following enzyme is not used in PCR for polymerisation?
A)
DNA polymerase-III
done
clear
B)
Pfn DNA polymerase
done
clear
C)
Vent DNA polymerase
done
clear
D)
Taq DNA polymerase
done
clear
View Answer play_arrow
All of the following are thermolabile, except
A)
DNA polymerase
done
clear
B)
Taq polymerase
done
clear
C)
Eco RI
done
clear
D)
Reverse transcritase
done
clear
View Answer play_arrow
Natural genetic engineer ofplants5 is
A)
Bacillus Thuringiensis
done
clear
B)
Meloidogyne incognita
done
clear
C)
Agro bacterium tumefaciens
done
clear
D)
Pseudo monas putida
done
clear
View Answer play_arrow
Functional insulin molecule is made up of
A)
Chain -A(21), B(30), C(33)
done
clear
B)
Chain -A(31),C(21)
done
clear
C)
Chain-A(21),B(30)
done
clear
D)
Chain -B(33),C(31)
done
clear
View Answer play_arrow
India is known for producing at least varieties of Basmati
A)
10
done
clear
B)
15
done
clear
C)
27
done
clear
D)
51
done
clear
View Answer play_arrow
Specific Bt toxin genes were isolated from Bacillus thuringiensis and incorporated into several crop plant. Choice of genes depends upon
A)
Crop
done
clear
B)
Toxin coded by gene
done
clear
C)
Targeted pest
done
clear
D)
All of these
done
clear
View Answer play_arrow
First transgenic crop developed by genetic modification is
A)
Tobacco
done
clear
B)
Potato
done
clear
C)
Tomato
done
clear
D)
Cotton
done
clear
View Answer play_arrow
Protein encoded by which of the following Bt toxin gene controls the cotton bollworm?
A)
Cry I Ac
done
clear
B)
Cry I Ab
done
clear
C)
Cry Il Ab
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
In India the bio safety guidelines and regulations are available from which of the following organization?
A)
IVRI
done
clear
B)
RTI
done
clear
C)
GEAC
done
clear
D)
DBT
done
clear
View Answer play_arrow
Which of the following was the first product of ancient biotechnology?
A)
Insulin
done
clear
B)
Steroid
done
clear
C)
Alcohol
done
clear
D)
Interferon
done
clear
View Answer play_arrow
First transgenic cow has been named
A)
Mary
done
clear
B)
Rosie
done
clear
C)
Polly
done
clear
D)
Molly
done
clear
View Answer play_arrow
Downstream processing includes
A)
Separation
done
clear
B)
Purification
done
clear
C)
Inactivation
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
Elbow joint is an example of
A)
Pivot joint
done
clear
B)
Hinge joint
done
clear
C)
Gliding joint
done
clear
D)
Ball and socket joint
done
clear
View Answer play_arrow
Mannitol is the stored food in
A)
Chara
done
clear
B)
Porphyra
done
clear
C)
Fucus
done
clear
D)
Gracillaria
done
clear
View Answer play_arrow
Uric acid is the chief nitrogenous compound of the excretory product of
A)
Man
done
clear
B)
Earthworm
done
clear
C)
Cockroach
done
clear
D)
Frog
done
clear
View Answer play_arrow
Stroma in the Chloroplasts of higher plant contain
A)
Light - independent reaction enzymes
done
clear
B)
Light - dependent reaction enzymes
done
clear
C)
Ribosomes
done
clear
D)
Chlorophyll
done
clear
View Answer play_arrow
Which one of the following has haplontic life cycle?
A)
Funaria
done
clear
B)
Polytrichum
done
clear
C)
Ustilago
done
clear
D)
Wheat
done
clear
View Answer play_arrow
In barley stem, vascular bundles are
A)
Open and scattered
done
clear
B)
Closed and scattered
done
clear
C)
Open and in a ring
done
clear
D)
Closed and radial
done
clear
View Answer play_arrow
There is no DNA in
A)
An enucleated ovum
done
clear
B)
Mature RBCs
done
clear
C)
A mature spermatozoan
done
clear
D)
Hair root
done
clear
View Answer play_arrow
Plasmodesmata are
A)
Lignified cemented layers between cells
done
clear
B)
Locomotary structures
done
clear
C)
Membranes connecting the nucleus with plasmalemma
done
clear
D)
Connections between adjacent cells
done
clear
View Answer play_arrow
Compared to blood, our lymph has
A)
No plasma
done
clear
B)
Plasma without proteins
done
clear
C)
More WBCs and no RBCs
done
clear
D)
More RBCs and less WBCs
done
clear
View Answer play_arrow
The floral formula
is that of
A)
Tulip
done
clear
B)
Soyabean
done
clear
C)
Sunnhemp
done
clear
D)
Tobacco
done
clear
View Answer play_arrow
Removal of introns and joining the exons in a defined order in a transcription unit is called
A)
Splicing
done
clear
B)
Tailing
done
clear
C)
Transformation
done
clear
D)
Capping
done
clear
View Answer play_arrow
Which one of the following pairs of animals comprises 'Jawless Fishes'?
A)
Lampreys and eels
done
clear
B)
Mackerals and Rohu
done
clear
C)
Lampreys and hag fishes
done
clear
D)
Guppies and hag fishes
done
clear
View Answer play_arrow
Use of anti-histamines and steroids give a quick relief from
A)
Allergy
done
clear
B)
Nausea
done
clear
C)
Cough
done
clear
D)
Headache
done
clear
View Answer play_arrow
What will happen if the stretch receptors of the urinary bladder wall are totally removed?
A)
Urine will not collect in the bladder
done
clear
B)
Micturition will continue
done
clear
C)
Urine will continue to collect normally in the bladder
done
clear
D)
There will be no micturition
done
clear
View Answer play_arrow
The kind of tissue that forms the supportive structure in our pinna (external ears) is also found in
A)
Vertebrae
done
clear
B)
Nails
done
clear
C)
Ear ossicles
done
clear
D)
Tip of the nose
done
clear
View Answer play_arrow
Which one of the following pairs is wrongly matched?
A)
Detergents - Lipase
done
clear
B)
Alcohol - Nitrogenase
done
clear
C)
Fruit juice - Pectinase
done
clear
D)
Textile - Amylase
done
clear
View Answer play_arrow
Which one of the following statements is correct?
A)
Patients who have undergone surgery are given cannabinoids to relieve pain
done
clear
B)
Benign tumors show the property of metastasis
done
clear
C)
Heroin accelerates body functions
done
clear
D)
Malignant tumours may exhibit metastasis
done
clear
View Answer play_arrow
The letter T in T-lymphocyte refers to
A)
Thyroid
done
clear
B)
Thalamus
done
clear
C)
Tonsil
done
clear
D)
Thymus
done
clear
View Answer play_arrow
Peripatus is a connecting link between
A)
Ctenophora and platyhelminthis
done
clear
B)
Mollusca and Echinodermata
done
clear
C)
Annelida and Arthropoda
done
clear
D)
Coelenterata and Porifera
done
clear
View Answer play_arrow
Darwin judged the fitness of an individual by
A)
Ability to defend itself
done
clear
B)
Strategy to obtain food
done
clear
C)
Number of offspring
done
clear
D)
Dominance over other individuals
done
clear
View Answer play_arrow
A horizontal underground stem is a
A)
Conn
done
clear
B)
Phylloclade
done
clear
C)
Rhizome
done
clear
D)
Rhizoid
done
clear
View Answer play_arrow
Jute fibres are obtained from the
A)
Secondary phloem
done
clear
B)
Pith
done
clear
C)
Xylem
done
clear
D)
Endodermis
done
clear
View Answer play_arrow
Which is an organic compound found in most cells?
A)
Glucose
done
clear
B)
Water
done
clear
C)
Sodium chloride
done
clear
D)
Oxygen
done
clear
View Answer play_arrow
Largest single mass of lymphatic tissue in the body is
A)
Lung
done
clear
B)
Spleen
done
clear
C)
Liver
done
clear
D)
Kidney
done
clear
View Answer play_arrow
Standing on tip toe is an example of
A)
Elevation
done
clear
B)
Flexion
done
clear
C)
Extension
done
clear
D)
Retraction
done
clear
View Answer play_arrow
If the pituitary gland of an adult rat is surgically removed, which of the following endocrine glands will be less affected?
A)
Adrenal cortex
done
clear
B)
Adrenal medulla
done
clear
C)
Thyroid
done
clear
D)
Gonads
done
clear
View Answer play_arrow
The hormone that increases the blood calcium level and decreases its excretion by kidney is
A)
Parathormonc
done
clear
B)
Calcitonin
done
clear
C)
Thyroxine
done
clear
D)
Insulin
done
clear
View Answer play_arrow
Signaling between cells usually result in the activation of protein
A)
Lipascs
done
clear
B)
Kinases
done
clear
C)
Proteases
done
clear
D)
Nucleases
done
clear
View Answer play_arrow
Ostcomalacia is due to deficiency of
A)
Vitamin A
done
clear
B)
Vitamin C
done
clear
C)
Vitamin E
done
clear
D)
Vitamin D
done
clear
View Answer play_arrow
Diploid cells have
A)
Two chromosomes
done
clear
B)
One set of chromosomes
done
clear
C)
Two pair of homologous chromosomes
done
clear
D)
Two sets of chromosomes
done
clear
View Answer play_arrow
Reproductive isolation between segments of a single population is termed
A)
Sympatry
done
clear
B)
Alloptry
done
clear
C)
Population divergence
done
clear
D)
Disruptive divergence
done
clear
View Answer play_arrow
Ampullae of lorenzini are present in
A)
Fish
done
clear
B)
Lizard
done
clear
C)
Frog
done
clear
D)
Rabbit
done
clear
View Answer play_arrow
The size of pupil is controlled by the
A)
Ciliary muscles
done
clear
B)
Suspensory ligaments
done
clear
C)
Cornea
done
clear
D)
Iris muscles
done
clear
View Answer play_arrow
The annual net primary productivity of the whole biosphere approximately is
A)
170 billion tons
done
clear
B)
17 million tons
done
clear
C)
55 million tons
done
clear
D)
55 billion tons
done
clear
View Answer play_arrow
Match the Column-I with Column-II with respect to trophic level and their example
Column-I Column-II A. Primary Producer I. Wolf B. Primary Consumer II. Cow C. Top Carnivore III. Trees D. Tertiary Consumer IV. Lion
A)
A-II, B-III, C-IV, D-I
done
clear
B)
A-III, B-II, C-IV, D-I
done
clear
C)
A-III, B-II, C-I, D-IV
done
clear
D)
A-II, B-III, C-I, D-IV
done
clear
View Answer play_arrow
How much percentage of carbon is found dissolved in oceans?
A)
71%
done
clear
B)
<1%
done
clear
C)
21%
done
clear
D)
49%
done
clear
View Answer play_arrow
Which of the following cell acts like an HIV factory?
A)
Helper T-Cells
done
clear
B)
Cytotoxic T-Cells
done
clear
C)
B-Lymphocytes
done
clear
D)
Macrophages
done
clear
View Answer play_arrow
___ is the most feared property of malignant tumours
A)
Mutation
done
clear
B)
Contact inhibition
done
clear
C)
Metastasis
done
clear
D)
Metagenesis
done
clear
View Answer play_arrow
Catharanthus roseus plant in the source of anticancer drugs which is / are
A)
Vineristin
done
clear
B)
Taxol
done
clear
C)
Vmblastin
done
clear
D)
Both [A] and [C]
done
clear
View Answer play_arrow
Excessive dosage of cocaine causes
A)
Sedation
done
clear
B)
Hallucinations
done
clear
C)
Analgesic effect
done
clear
D)
Tranquillizing effect
done
clear
View Answer play_arrow
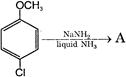 The major product A is:
The major product A is: 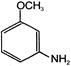
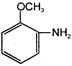
 Select the correct answer from the following
Select the correct answer from the following