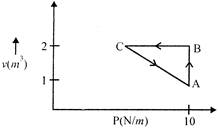
A) \[-5J\]
B) \[-10J\]
C) \[-15J\]
D) \[-20J\]
Correct Answer: A
Solution :
Given, \[{{W}_{AB}}+{{W}_{BC}}+{{W}_{CA}}=5\] \[10+0+{{W}_{CA}}=5\] (as, during BC, change volume = 0) \[{{W}_{CA}}=-5J\]You need to login to perform this action.
You will be redirected in
3 sec
