A) 125 J
B) -10 J
C) 10 J
D) 55 J
Correct Answer: A
Solution :
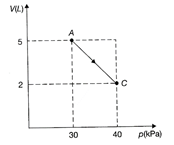
Here, V - P curve is given and not P - V. \[W=-\frac{1}{2}[30+40\times {{10}^{3}}]\times 3\times {{10}^{-3}}=-105\,J\] Work is negative because the volume is decreasing From\[dQ=dU+dW\] \[\Rightarrow \]\[dU=(20+105)J=125\,J\] Hence, the correction option is [a].You need to login to perform this action.
You will be redirected in
3 sec
