A) 1 atm
B) 4/5 atm
C) 4/3 atm
D) 3/2 atm
Correct Answer: C
Solution :
Initial pressure, \[{{p}_{1}}={{p}_{2}}={{p}_{0}}=\frac{nR{{T}_{0}}}{{{V}_{0}}}\] when \[{{p}_{0}}=1\]atm. \[{{T}_{0}}=300K.\]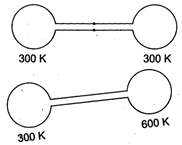 Finally, let common pressure is p. So, \[\frac{p{{V}_{0}}}{R\times 2{{T}_{0}}}={{n}_{1}}\]and \[\frac{p{{V}_{0}}}{R{{T}_{0}}}={{n}_{2}}\]and \[{{n}_{1}}+{{n}_{2}}=2n\] \[\frac{p{{V}_{0}}}{2R{{T}_{0}}}+\frac{p{{V}_{0}}}{R{{T}_{0}}}=2\left( \frac{{{p}_{0}}{{V}_{0}}}{R{{T}_{0}}} \right)\] \[\Rightarrow \] \[p=\frac{4}{3}{{p}_{0}}=\frac{4}{3}\text{atm}\]
Finally, let common pressure is p. So, \[\frac{p{{V}_{0}}}{R\times 2{{T}_{0}}}={{n}_{1}}\]and \[\frac{p{{V}_{0}}}{R{{T}_{0}}}={{n}_{2}}\]and \[{{n}_{1}}+{{n}_{2}}=2n\] \[\frac{p{{V}_{0}}}{2R{{T}_{0}}}+\frac{p{{V}_{0}}}{R{{T}_{0}}}=2\left( \frac{{{p}_{0}}{{V}_{0}}}{R{{T}_{0}}} \right)\] \[\Rightarrow \] \[p=\frac{4}{3}{{p}_{0}}=\frac{4}{3}\text{atm}\]
You need to login to perform this action.
You will be redirected in
3 sec
