-
What is meant by 'disproportionate' ? Give one example of disproportionate
reaction in aqueous solution,
[1]
View Answer play_arrow
-
Which metal in the first series of transition metals
exhibits +1 oxidation state most frequently and why?
[1]
View Answer play_arrow
-
Which of the following is most effective in coagulation of
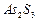
sol?
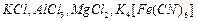
[1]
-
How will the pH of brine (aq. NaCl solution) be affected
when it is electrolyzed? [1]
View Answer play_arrow
-
Why is the use of aspartame limited to cold foods and
drinks?
[1]
View Answer play_arrow
-
Arrange the following compounds in increasing order of
their reactivity in nucleophilic addition reactions.
Ethanal, propanal, propanone, butanone [1]
View Answer play_arrow
-
What products are formed when D-glucose is treated with bromine
water? [1]
View Answer play_arrow
-
Write the cell reaction for a lead storage battery when it
is discharged. How does the density of the electrolyte change when the battery
is
discharged?
[1]
View Answer play_arrow
-
How can we enhance the reactivity of aryl
halides? [2]
Or
(i) How can you obtain iodoethane from ethanol when no
other iodine containing reagent except Nal is available in the laboratory?
(ii) Write a test to detect the presence of double bond in
a molecule, [1+1]
View Answer play_arrow
-
In what respect do prontosil and salvarsan resemble. Is
these any resemblance between azo dye and protosil?
Explain. [2]
View Answer play_arrow
-
p-dichloro benzene has higher melting point and lower
solubility than those of o-and m-isomers.
Discuss.
[2]
View Answer play_arrow
-
Write the mechanism of acid dehydration of ethanol to
yield ethene. [2]
View Answer play_arrow
-
(i) Explain the Kolbe's reaction with an example,
[1+1]
(ii) Give the structures and IUPAC names of monohydric
phenols of molecular formula
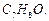
-
Explain the following terms with suitable examples.
(i) Schottky defect (ii) Frenkel
defect [1+1]
View Answer play_arrow
-
Explain why does conductivity of germanium crystals
increase on doping with gallium? [2]
View Answer play_arrow
-
Write short notes on the following.
(i) Carbylamines reaction (ii) Coupling
reaction [1+1]
View Answer play_arrow
-
If
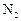
gas is
bubbled through water at 293 K, how many millimoles of
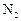
gas
would dissolve in 1 litre of water. Assume that
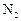
exerts
a partial pressure of 0.987 bar.
Given that Henry's law constant for N3 at 293K is 76.48 K
bar. [2]
-
Arrange the following
(i) In increasing order of basic strength aniline, p-nitro
aniline and p-toluidine.
(ii) In increasing order of solubility in water.
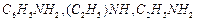
[1+1]
-
Determine the amount of
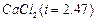
dissolved
in 2.5 litre of water such that its osmotic pressure is 0.75 atm at 27°C. (R
=0.0821 L atm
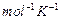
)
[3]
Or
Determine the osmotic pressure of a solution prepared by
dissolving 25 mg of
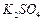
in
2 litre of water at 25°C, assuming that it is completely dissociated.
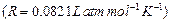
[3]
-
Miss Fleming was asked to synthesized alcohol by acidic
hydration of 1-butene. She was unaware of the fact that the vessel she used had
some coating of a metal, and in addition to alcohol (b.p. 373 K), compound-Y
(b.p. 353 K) was also isolated. X forms bisulphite compound as well as 2,4-dinitrophenyl
hydrazone. Separation of alcohol could be made by physical as well as chemical
methods.
Answer the following questions, based on above passage.
(a) How are alcohol acid X
formed? [1]
(b) Can alcohol acid X give iodo form
test? [1]
(c) Give different methods of
separation. [1]
View Answer play_arrow
-
How much electricity is required in Coulomb for the
oxidation of
(i) 1 mole of
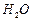
to
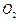
? (ii)
1 mole of
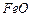
to
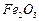
? (iii)
1 mole of
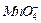
to
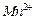
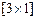
-
Give reasons for the following.
(i) Zinc oxide can be reduced to the metal by heating with
carbon but not
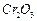
(ii) Alumina is dissolved in cryolite for electrolysis
instead of being electrolyzed directly.
Extraction of copper directly from sulphide ores is less
favourable than that from its oxide ore through reduction. [3
x 1]
-
State reasons for the following.
(i)
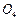
the d4
species,
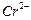
is
strongly reducing while manganese (III) is strongly oxidising.
(ii) The d configuration is very unstable in
ions.
(iii) Cobalt (II) is sable in aqueous solution but in the
presence of complexing reagents it is easily oxidised.
[
]
-
Explain the following terms giving a suitable example in
each case.
(i)Ambident ligand (ii) Denticity
of a ligand
(iii) Crystal field splitting in an octahedral field [3
x 1]
View Answer play_arrow
-
(a) What is meant by peptisation? Explain.
(b) What is the effect of temperature on chemisorption?
Explain.
(c) What are assoicated colloids? Explain with an
example. [3
x 1]
View Answer play_arrow
-
(i) Explain the difference between Buna-N and Buna-S.
(ii) Arrange the following polymers in increasing order of
their intermolecular forces.
(a) Nylon-66, buna-S, polythene (b)
Nylon-6, neoprene, polyvinyl chloride [2 - 1]
View Answer play_arrow
-
How would you account for the following.
(i)
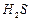
is more
acidic than
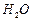
.
(ii) The N?0 bond in N0^ is shorter than the N?0 bond in
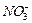
.
(iii) Both
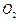
and
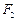
stabilize
high oxidation states but the ability of oxygen to stabilize the higher
oxidation state exceeds that of fluorine. [3
x 1]
-
(a) What do you understand by the rate law and rate
constant of a reaction?
Identify the order of reaction if the units of its rate
constant are
(i)
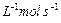
(ii)
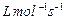
(b) The thermal decomposition of HCOOH is a first order
reaction with a rate constant of
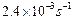
at a
certain temperature. Calculate how long will it take for three-fourths of
initial quantity of HCOOH to decompose (log 0.25
=-0.6021). [2+3]
Or
(a) For the reaction,
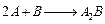
the
rate
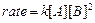
with
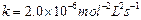
.
Calculate the initial rate of reaction when
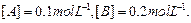
Calculate
the rate of reaction after [A] is reduced to 0.06
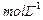
.
(b) The rate constant for the decomposition of
hydrocarbons is
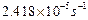
at
546 K. If the energy of activation is 179.9 kJ mol"1 what will
be the value of pre exponential
factor? [2 + 3]
-
(a) Draw the structures of the following molecules.
(i)
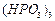
(ii)
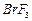
(b) Complete the following chemical equations.
(i)
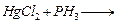
(ii)
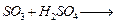
(iii)
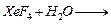
[2+3]
Or
(a) What happens when
(i) chlorine gas is passed through a hot concentrated
solution of NaOH?
(ii) sulphur dioxide gas is passed through an aqueous
solution of a Fe (III) salt?
(b) Answer the following.
(i) What is the basicity ofH3P03 and why?
(ii) Why does fluorine not play the role of a central atom
in interhalogen compounds?
(iii) Why do noble gases have very low boiling
points? [2
+3]
-
(a) How can the following conversions be carried out?
(i) Ethanol to acetone (ii) Benzene to acetophenone
(iii) Benzoic acid to benzaldehyde
(b) Describe the following giving a suitable example in each case.
(i) Decarboxylation (ii)
Cannizzaro's reaction [3+2]
Or
(a) An organic compound 'A' contains 69.77% carbon, 11.63%
hydrogen and rest is oxygen. The molecular mass of'A' is 86. It does not reduce
Tollen's reagent but forms an addition compound with sodium hydrogen sulphite. 'A'
gives a positive idoform test. On vigorous oxidation 'A' gives ethanoic and
propanoic acids. Deduce the possible structure of 'A'.
(b) State reasons for the following situations.
(i) Monochloro ethanoic acid is a weaker acid than
dichloro ethanoic acid.
(ii) Benzoic acid is a stronger acid than ethanoic acid. [3
+ 2]
View Answer play_arrow
