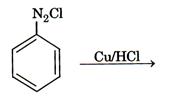
 1
1
 1
(ii)
1
(ii)
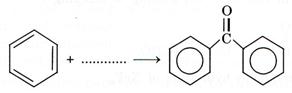 1
(b)
How will you obtain following:
(i)
Benzoic acid from ethyl benzene. 1
(ii)
Benzaldehyde from Toluene. 1
1
(b)
How will you obtain following:
(i)
Benzoic acid from ethyl benzene. 1
(ii)
Benzaldehyde from Toluene. 1
You need to login to perform this action.
You will be redirected in
3 sec
