Which of the following statements are correct? (i) Smaller the gold number of lyophilic colloid, the larger will be its protective power. (ii) Lyophilic sols, in contrast to lyophobic sols can easily coagulate on addition of small amount of electrolyte. (iii) Ferric chloride solution is used to stop bleeding from a fresh cut because it coagulates the blood. (iv)The flocculation value of arsenious sulphide sol is independent of the charge of anion of the coagulating electrolyte.
A)
(i), (ii) and (iii)
done
clear
B)
(i), (iii) and (iv)
done
clear
C)
(ii), (iii) and (iv)
done
clear
D)
(i), (ii) and (iv)
done
clear
View Answer play_arrow
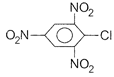
Which chloro derivative of benzene among the following would undergo hydrolysis most readily with aqueous sodium hydroxide to furnish the corresponding hydroxyl derivative?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
When vapours of X were gradually heated from \[25{}^\circ C\]at atmospheric pressure, the colour was found to deepen at first and then fades as the temperature was raised above \[{{160}^{o}}C\]. At \[{{600}^{o}}C\],the vapours were almost colourless, but the colour deepened When the pressure was raised at this temperature. Identify X.
A)
Bromine.
done
clear
B)
A mixture of nitrogen dioxide and dinitrogen tetroxide.
done
clear
C)
Pure nitrogen dioxide.
done
clear
D)
Pure dinitrogen tetroxide.
done
clear
View Answer play_arrow
Observe the given reactions : 1.\[FeC{{l}_{3}}\,\,\,Solution+Zn\to product\,\,X\] 2. \[FeC{{l}_{3}}\,\,\,Solution+{{H}_{2}}\,\,gas\to product\,\,Y\] \[FeC{{l}_{3}}\] solution gives blue colour with \[{{K}_{4}}[Fe{{(CN)}_{6}}]\], hence
A)
X also gives blue colour with \[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
B)
Y also gives blue colour with \[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
C)
Both X and Y give blue colour with \[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
D)
None gives colour with \[{{K}_{4}}[Fe{{(CN)}_{6}}]\].
done
clear
View Answer play_arrow
The equivalent conductance at infinite dilution of an acid like HF
A)
Can be determined by extrapolation of measurements of dilute solutions of HCI, HBr and HI
done
clear
B)
Can be determined by measurement of very dilute HF solution
done
clear
C)
Can best be determined from measurements of concentrated solutions of NaF, NaCI and HCI
done
clear
D)
Can best be determined from measurements of dilute solutions of NaF, NaCI and HCI.
done
clear
View Answer play_arrow
Which has maximum percentage of Cl?
A)
PVC
done
clear
B)
BHC
done
clear
C)
Neoprene
done
clear
D)
DDT
done
clear
View Answer play_arrow
A mole of complex compound \[Co{{(N{{H}_{3}})}_{5}}C{{l}_{3}}\]gives 3 mole of ions, when dissolved in water. One mole of the same complex reacts with two moles of \[AgN{{O}_{3}}\] solution to form two moles of AgCI(s). The structure of complex is
A)
\[[Co{{(N{{H}_{3}})}_{5}}Cl]C{{l}_{2}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}].2N{{H}_{3}}\]
done
clear
C)
\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl.N{{H}_{3}}\]
done
clear
D)
\[[Co{{(N{{H}_{3}})}_{4}}Cl]C{{l}_{2}}.N{{H}_{3}}\]
done
clear
View Answer play_arrow
In a cubic lattice, each edge of the unit cell is 400 pm. Atomic weight of the element is 60 and its density is \[6.25g/c.c.\]Avogadro number\[=60\times \,{{10}^{23}}\]. The crystal lattice is
A)
Face-centred
done
clear
B)
Primitive
done
clear
C)
Body-centred
done
clear
D)
End-centred.
done
clear
View Answer play_arrow
The rate of change of concentration of P for reaction : \[P\to Q\,\,is\,\,given\,\,by\frac{-d[P]}{dt}=k{{[P]}^{1/3}}\] The half-life period of the reaction will be
A)
\[\frac{3{{[{{P}_{0}}]}^{2/3}}{{[{{(2)}^{2/3}}-1]}^{2}}}{{{(2)}^{5/3}}\,\,\,K}\]
done
clear
B)
\[\frac{\frac{3}{2}{{[{{P}_{0}}]}^{2/3}}[{{(2)}^{2/3}}-1]}{\,K}\]
done
clear
C)
\[\frac{3{{[{{P}_{0}}]}^{2/3}}[{{(2)}^{2/3}}-1]}{{{(2)}^{5/3}}\,\,\,K}\]
done
clear
D)
\[\frac{\frac{2}{3}{{[{{P}_{0}}]}^{3/2}}[{{(2)}^{2/3}}-1]}{\,K}\]
done
clear
View Answer play_arrow
At isoelectric point, solubility of amino acids in water is minimum because
A)
They are organic acids
done
clear
B)
Molecules show intramolecular hydrogen bonding
done
clear
C)
Concentration of cations and anions becomes equal
done
clear
D)
Concentration of zwitter ions is maximum.
done
clear
View Answer play_arrow
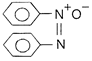
In the reduction of nitrobenzene into aniline, the intermediate is
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of these.
done
clear
View Answer play_arrow
To a solution containing \[F{{e}^{3+}}\]and \[T{{i}^{4+}}\] ions when excess of NH40H is added, a ppt. (X) is produced. The ppt. (X) contains
A)
\[TiO.n{{H}_{2}}O\,\,\,\,and\,\,\,Fe\,\,{{\left( OH \right)}_{3}}\]
done
clear
B)
\[Ti{{O}_{2}}n{{H}_{2}}O\,\,\,\,nad\,\,\,Fe\,\,{{\left( OH \right)}_{3}}\]
done
clear
C)
\[Only\,\,Ti{{O}_{2}}.n{{H}_{2}}O\]
done
clear
D)
\[Only\,\,Fe{{\left( HO \right)}_{3}}\]
done
clear
View Answer play_arrow
Which is not the correct statement?
A)
Cassiterite, chromite and pitchblende are concentrated by hydraulic washing (Tabling).
done
clear
B)
Pure \[Al{{O}_{2}}\] is obtained from the bauxite ore by leaching in the Baeyer's process.
done
clear
C)
Sulphide ore is concentrated by calcination method.
done
clear
D)
Roasting can convert sulphide into oxide or sulphate and part of sulphide may also act as a reducing agent.
done
clear
View Answer play_arrow