A) 5
B) 6
C) 3
D) 4
Correct Answer: A
Solution :
\[F{{e}^{3+}}\]\[(z=26)\]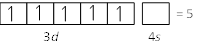 \[F{{e}^{3+}}=[Ar]\,3{{d}^{5}}4{{s}^{0}}\] Total no. of unpaired electron=5
\[F{{e}^{3+}}=[Ar]\,3{{d}^{5}}4{{s}^{0}}\] Total no. of unpaired electron=5
You need to login to perform this action.
You will be redirected in
3 sec
