A) \[F{{e}^{+2}}\]
B) \[C{{O}^{+2}}\]
C) \[N{{i}^{+2}}\]
D) \[M{{n}^{+2}}\]
Correct Answer: D
Solution :
\[M{{n}^{+2}}\] ion will have five (maximum) unpaired electrons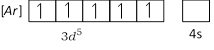
You need to login to perform this action.
You will be redirected in
3 sec
