-
question_answer1)
What is the angular velocity \[\left( \omega \right)\] of an electron occupying second orbit of \[L{{i}^{2+}}\]ion?
A)
\[\frac{8{{\pi }^{3}}m{{e}^{4}}}{{{h}^{3}}}{{K}^{2}}\] done
clear
B)
\[\frac{8{{\pi }^{3}}m{{e}^{4}}}{9{{h}^{3}}}{{K}^{2}}\] done
clear
C)
\[\frac{64}{9}\times \frac{{{\pi }^{3}}m{{e}^{4}}}{{{h}^{3}}}{{K}^{2}}\] done
clear
D)
\[\frac{9{{\pi }^{3}}m{{e}^{4}}}{{{h}^{3}}}{{K}^{2}}\] done
clear
View Solution play_arrow
-
question_answer2)
In a collection of H-atoms, all the electrons jump from n = 5 to ground level finally (directly or indirectly), without emitting any line in Balmer series. The number of possible different radiations is:
A)
10 done
clear
B)
8 done
clear
C)
7 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer3)
The wavelength of \[{{H}_{\alpha }}\] line of Balmer series is X \[\overset{\text{o}}{\mathop{\text{A}}}\,\]. What is the X of\[{{H}_{\beta }}\] line of Balmer series.
A)
\[X\frac{108}{80}\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
B)
\[X\frac{80}{108}\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
C)
\[\frac{1}{X}\frac{80}{108}\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
D)
\[\frac{1}{X}\frac{108}{80}\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
View Solution play_arrow
-
question_answer4)
The number of photons of light having wave number 'x' in 10 J of energy source is:
A)
\[10hcx\] done
clear
B)
\[\frac{hc}{10x}\] done
clear
C)
\[\frac{10}{hcx}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer5)
The values of Planck's constant is \[6.63\times {{10}^{-34}}\] Js. The velocity of light is\[3.0\times {{10}^{8}}m{{s}^{-1}}\]. Which value is closest to the wavelength in nanometres of a quantum of light with frequency of \[8\times {{10}^{15}}{{s}^{-1}}\]?
A)
\[5\times {{10}^{-18}}\] done
clear
B)
\[4\times {{10}^{1}}\] done
clear
C)
\[3\times {{10}^{7}}\] done
clear
D)
\[2\times {{10}^{-25}}\] done
clear
View Solution play_arrow
-
question_answer6)
According to law of photochemical equivalence the energy absorbed (in ergs/mole) is given as \[(h=6.62\times {{10}^{-27}}\]ergs, \[c=3\times {{10}^{10}}cm\text{ }{{s}^{-1}}\], \[{{N}_{A}}=6.02\times {{10}^{23}}mo{{l}^{-1}}\])
A)
\[\frac{1.956\times {{10}^{16}}}{\lambda }\] done
clear
B)
\[\frac{1.19\times {{10}^{8}}}{\lambda }\] done
clear
C)
\[\frac{2.859\times {{10}^{5}}}{\lambda }\] done
clear
D)
\[\frac{2.859\times {{10}^{16}}}{\lambda }\] done
clear
View Solution play_arrow
-
question_answer7)
The energy of an electron in first Bohr orbit of Pi- atom is \[-13.6\text{ }eV\]. The energy value of electron in the excited state of \[L{{i}^{2+}}\] is:
A)
\[-27.2\text{ }eV\] done
clear
B)
\[30.6\text{ }eV\] done
clear
C)
\[-30.6eV\] done
clear
D)
\[27.2\text{ }eV\] done
clear
View Solution play_arrow
-
question_answer8)
Based on the equation: \[\Delta E=-2.0\times {{10}^{-18}}J\left( \frac{1}{n_{2}^{2}}-\frac{1}{n_{1}^{2}} \right)\] the wavelength of the light that must be absorbed to excite hydrogen electron from level n = 1 to level \[n=2\] will be: (\[h=6.625\times {{10}^{-34}}Js\],\[C=3\times {{10}^{8}}m{{s}^{-1}}\])
A)
\[1.325\times {{10}^{-7}}m\] done
clear
B)
\[1.325\times {{10}^{-10}}m\] done
clear
C)
\[2.650\times {{10}^{-7}}m\] done
clear
D)
\[5.300\times {{10}^{-10}}m\] done
clear
View Solution play_arrow
-
question_answer9)
In an atom, an electron is moving with a speed of 600 m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (\[h=6.6\times {{10}^{-34}}kg\,{{m}^{2}}{{s}^{-1}}\], mass of electron,\[{{e}_{m}}=9.1\times {{10}^{-31}}kg\])
A)
\[5.10\times {{10}^{-3}}m\] done
clear
B)
\[1.92\times {{10}^{-3}}m\] done
clear
C)
\[3.84\times {{10}^{-3}}m\] done
clear
D)
\[1.52\times {{10}^{-4}}m\] done
clear
View Solution play_arrow
-
question_answer10)
Which of the following is not permissible arrangement of electrons in an atom?
A)
\[n=5,\,\,l=3,\text{ }m=0,\text{ }s=+\,1\text{/}2\] done
clear
B)
\[n=3,\text{ }l=2,\text{ m}=-\,3,\text{ }s=-1/2\] done
clear
C)
\[n=3,\text{ }l=2,\text{ m}=-\,2,\text{ }s=-1/2\] done
clear
D)
\[n=4,\text{ }l=0,\text{ m}=0,\text{ }s=-1/2\] done
clear
View Solution play_arrow
-
question_answer11)
A dye absorbs a photon of wavelength \[\lambda \] and re-emits the same energy into two photons of wavelength \[{{\lambda }_{1}}\] and \[{{\lambda }_{2}}\] respectively. The wavelength \[\lambda \] is related with \[{{\lambda }_{1}}\] and \[{{\lambda }_{2}}\] as:
A)
\[\lambda =\frac{{{\lambda }_{1}}+{{\lambda }_{2}}}{{{\lambda }_{1}}{{\lambda }_{2}}}\] done
clear
B)
\[\lambda =\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\] done
clear
C)
\[\lambda =\frac{{{\lambda }_{1}}^{2}{{\lambda }_{2}}^{2}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\] done
clear
D)
\[\lambda =\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{({{\lambda }_{1}}+{{\lambda }_{2}})}^{2}}}\] done
clear
View Solution play_arrow
-
question_answer12)
The Bohr's energy equation for H atom reveals that the energy level of a shell is given by \[E=-13.58/{{n}^{2}}eV\]. The smallest amount that an H atom will absorb if in ground state is
A)
\[1.0\text{ }eV\] done
clear
B)
\[3.39\text{ }eV\] done
clear
C)
\[6.79\text{ }eV\] done
clear
D)
\[10.19\text{ }eV\] done
clear
View Solution play_arrow
-
question_answer13)
If m and e are the mass and charge of the revolving electron in the orbit of radius r for hydrogen atom, the total energy of the revolving electron will be:
A)
\[\frac{1}{2}\frac{{{e}^{2}}}{r}\] done
clear
B)
\[-\frac{{{e}^{2}}}{r}\] done
clear
C)
\[\frac{m{{e}^{2}}}{r}\] done
clear
D)
\[-\frac{1}{2}\frac{{{e}^{2}}}{r}\] done
clear
View Solution play_arrow
-
question_answer14)
Excited hydrogen atom emits light in the ultraviolet region at\[2.47\times {{10}^{15}}Hz\]. With this frequency, the energy of a single photon is: \[(h=6.63\times {{10}^{-34}}Js)\]
A)
\[8.041\times {{10}^{-40}}J\] done
clear
B)
\[2.680\times {{10}^{-19}}J\] done
clear
C)
\[1.640\times {{10}^{-18}}J\] done
clear
D)
\[6.111\times {{10}^{-17}}J\] done
clear
View Solution play_arrow
-
question_answer15)
The orbital diagram in which the Aufbau principle is violated is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
Calculate the velocity of ejected electron from the metal surface when light of frequency \[2\times {{10}^{15}}Hz\] fall on the metal surface and the threshold frequency is \[~7\times {{10}^{14}}Hz\] for metal?
A)
\[~1.37\times {{10}^{6}}\] done
clear
B)
\[~1.26\times {{10}^{6}}\] done
clear
C)
\[1.45\times {{10}^{7}}\] done
clear
D)
\[~1.37 \times {{10}^{7}}\] done
clear
View Solution play_arrow
-
question_answer17)
An electron, \[{{e}_{1}}\] is moving in the fifth stationary state, and another electro \[{{e}_{2}}\] is moving in the fourth stationary state. The radius of orbit of electron \[{{e}_{1}}\] is five times the radius of orbit of electron, \[{{e}_{2}}\] calculate the ratio of velocity of electron \[{{e}_{1}}({{v}_{1}})\] to the velocity of electron \[{{e}_{2}}({{v}_{2}})\]
A)
5 : 1 done
clear
B)
4 : 1 done
clear
C)
1 : 5 done
clear
D)
1 : 4 done
clear
View Solution play_arrow
-
question_answer18)
Among the following groupings which represents the collection of isoelectronic species?
A)
\[N{{O}^{+}},C_{2}^{2-},O_{2}^{-},CO\] done
clear
B)
\[{{N}_{2}},C_{2}^{2-},CO,NO\] done
clear
C)
\[CO,N{{O}^{+}},C{{N}^{-}},C_{2}^{2-}\] done
clear
D)
\[NO,C{{N}^{-}},{{N}_{2}},O_{2}^{-}\] done
clear
View Solution play_arrow
-
question_answer19)
The wave number of the first emission line in the Balmer series of H-Spectrum is: (R = Rydberg constant):
A)
\[\frac{5}{36}R\] done
clear
B)
\[\frac{9}{400}R\] done
clear
C)
\[\frac{7}{6}R\] done
clear
D)
\[\frac{3}{4}R\] done
clear
View Solution play_arrow
-
question_answer20)
What is the maximum wavelength line in the Lyman series of He+ ion?
A)
\[3R\] done
clear
B)
\[\frac{1}{3R}\] done
clear
C)
\[\frac{4}{4R}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer21)
The position of both, an electron and a helium atom is known within 1.0 nm. Further the momentum of the electron is known within \[5.0\times {{10}^{-26}}kg\,m{{s}^{-1}}\]. The minimum uncertainty in the measurement of the momentum of the helium atom is
A)
\[50kgm{{s}^{-1}}\] done
clear
B)
\[80kgm{{s}^{-1}}\] done
clear
C)
\[8.0\times {{10}^{-26}}kg\text{ }m{{s}^{-1}}\] done
clear
D)
\[5.0\times {{10}^{-26}}kg\text{ }m{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer22)
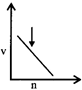
What will be the difference between electromagnetic radiation shown in A and B respectively?
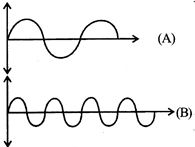
| (i) Velocity |
| (ii) Wavelength |
| (iii) Frequency |
| (iv) Energy |
A)
(ii) only done
clear
B)
(ii) and (iv) done
clear
C)
(ii), (iii) and (iv) done
clear
D)
(iv) only done
clear
View Solution play_arrow
-
question_answer23)
Arrange the electromagnetic radiations a, b, c, d and e in increasing order of energy. Frequencies of a, b and c are \[{{10}^{15}},{{10}^{14}}\] and \[~{{10}^{17}}\] respectively whereas wavelength of d and e are 350 nm and 100 nm respectively?
A)
a, b, c, d, e done
clear
B)
a, b, d, e, c done
clear
C)
a, d, b, e, c done
clear
D)
b, a, d, e, c done
clear
View Solution play_arrow
-
question_answer24)
When electronic transition occurs from higher energy state to lower energy state with energy difference equal to \[\Delta E\] electron volts, the wavelength of the line emitted is approximately equal to
A)
\[\frac{12395}{\Delta E}\times {{10}^{-10}}m\] done
clear
B)
\[\frac{12395}{\Delta E}\times {{10}^{10}}m\] done
clear
C)
\[\frac{12395}{\Delta E}\times {{10}^{-10}}cm\] done
clear
D)
\[\frac{12395}{\Delta E}\times {{10}^{10}}cm\] done
clear
View Solution play_arrow
-
question_answer25)
Which of the following should be the number of electrons present in \[{{X}^{2+}}\]on the basis of electronic configuration, if the ion \[{{X}^{3-}}\]has 14 protons?
A)
12 done
clear
B)
14 done
clear
C)
16 done
clear
D)
18 done
clear
View Solution play_arrow
-
question_answer26)
In a hydrogen atom, if energy of an electron in ground state is 13.6. ev, then that in the 2nd excited state is
A)
\[1.51\text{ }eV\] done
clear
B)
\[3.4\,eV\] done
clear
C)
\[6.04\,eV\] done
clear
D)
\[13.6\text{ }eV\] done
clear
View Solution play_arrow
-
question_answer27)
At temperature T, the average kinetic energy of any particle is \[\frac{3}{2}KT.\] The de Broglie wavelength follows the order:
A)
Visible photon > Thermal neutron > Thermal electron done
clear
B)
Thermal proton > Thermal electron > Visible photon done
clear
C)
Thermal proton > Visible photon > Thermal electron done
clear
D)
Visible photon > Thermal electron > Thermal neutron done
clear
View Solution play_arrow
-
question_answer28)
Which one of the following set of quantum numbers is not possible for \[4p\] electron?
A)
\[n=4,l=1,m=-1,{{m}_{s}}=+\frac{1}{2}\] done
clear
B)
\[n=4,l=1,m=0,{{m}_{s}}=+\frac{1}{2}\] done
clear
C)
\[n=4,l=1,m=2,{{m}_{s}}=+\frac{1}{2}\] done
clear
D)
\[n=4,l=1,m=-1,{{m}_{s}}=-\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer29)
What is the maximum number of electrons that can be accomodated in an atom in which the highest principal quantum number value is 4?
A)
10 done
clear
B)
18 done
clear
C)
36 done
clear
D)
54 done
clear
View Solution play_arrow
-
question_answer30)
The de Broglie wavelength of a car of mass 1000 kg and velocity 36 km/hr is:
A)
\[6.626\times {{10}^{-34}}m\] done
clear
B)
\[6.626\times {{10}^{-38}}m\] done
clear
C)
\[6.626\times {{10}^{-31}}m\] done
clear
D)
\[6.626\times {{10}^{-30}}m\] done
clear
View Solution play_arrow
-
question_answer31)
If the radius of first orbit of H atom is \[{{a}_{0}}\]the de- Broglie wavelength of an electron in the third orbit is
A)
\[4\pi {{a}_{0}}\] done
clear
B)
\[8\pi {{a}_{0}}\] done
clear
C)
\[6\pi {{a}_{0}}\] done
clear
D)
\[2\pi {{a}_{0}}\] done
clear
View Solution play_arrow
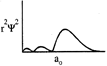
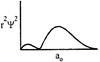
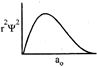
-
question_answer32)
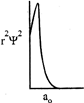
Which of the following radial distribution graphs correspond to\[l=2\] for the H atom?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer33)
In ground state of \[C{{u}^{+}}\]. The no. of shells occupied, subshells, filled orbitals and unpaired electrons respectively are:
A)
4, 8, 15, 0 done
clear
B)
3, 6, 15, 1 done
clear
C)
3, 6, 14, 0 done
clear
D)
4, 7, 14, 2 done
clear
View Solution play_arrow
-
question_answer34)
The \[L{{i}^{2+}}\] ion is moving in the third stationary state, and its linear momentum is \[7.3\times {{10}^{-34}}gm{{s}^{-1}}\]. Angular momentum is.
A)
\[1.158\times {{10}^{-45}}kg{{m}^{2}}{{s}^{-1}}\] done
clear
B)
\[1.158\times {{10}^{-48}}kg{{m}^{2}}{{s}^{-1}}\] done
clear
C)
\[1.158\times {{10}^{-47}}kg{{m}^{2}}{{s}^{-1}}\] done
clear
D)
\[12\times {{10}^{-45}}kg{{m}^{2}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer35)
The de Broglie wavelength of a tennis ball of mass 60 g moving with a velocity of 10 metres per second is approximately
A)
\[{{10}^{-31}}\] done
clear
B)
\[{{10}^{-16}}\] done
clear
C)
\[{{10}^{-25}}\] done
clear
D)
\[{{10}^{-33}}\] done
clear
View Solution play_arrow
-
question_answer36)
In an atom how many orbital(s) will have the quantum numbers;\[n=3,l=2\] and \[{{m}_{1}}=+2\]?
A)
5 done
clear
B)
3 done
clear
C)
1 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer37)
According to Bohr's theory, the angular momentum of an electron in 5th orbit is
A)
\[10h/\pi \] done
clear
B)
\[2.5h/\pi \] done
clear
C)
\[25h/\pi \] done
clear
D)
\[1.0h/\pi \] done
clear
View Solution play_arrow
-
question_answer38)
The energy of electron in first energy level is -\[21.79\times {{10}^{-12}}\] erg per atom. The energy of electron in second energy level is:
A)
\[-54.47\times {{10}^{-12}}erg\text{ }ato{{m}^{-1}}\] done
clear
B)
\[-5.447\times {{10}^{-12}}erg\text{ }ato{{m}^{-1}}\] done
clear
C)
\[-0.5447\times {{10}^{-12}}erg\text{ }ato{{m}^{-1}}\] done
clear
D)
\[-0.05447\times {{10}^{-12}}erg\text{ }ato{{m}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer39)
Which of the following electronic configuration of d-orbital will have highest affinity for gaining an electron?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer40)
If the de-Broglie wavelength of a particle mass m is 100 times its velocity, then its value in terms of its mass (m) and Planck's constant (h) is
A)
\[\frac{1}{10}\sqrt{\frac{m}{h}}\] done
clear
B)
\[10\sqrt{\frac{h}{m}}\] done
clear
C)
\[\frac{1}{10}\sqrt{\frac{h}{m}}\] done
clear
D)
\[10\sqrt{\frac{m}{h}}\] done
clear
View Solution play_arrow
-
question_answer41)
An electron in an atom jumps in such a way that its kinetic energy changes from x to\[\frac{x}{4}\]. The change in potential energy will be:
A)
\[+\frac{3}{2}x\] done
clear
B)
\[-\frac{3}{8}x\] done
clear
C)
\[+\frac{3}{4}x\] done
clear
D)
\[-\frac{3}{4}x\] done
clear
View Solution play_arrow
-
question_answer42)
The kinetic and potential energy (in eV) of an electron present in third Bohr's orbit of hydrogen atom are respectively:
A)
\[-1.51.-3.02\] done
clear
B)
\[1.51,-3.02\] done
clear
C)
\[-3.02,1.51\] done
clear
D)
\[1.51,-1.51\] done
clear
View Solution play_arrow
-
question_answer43)
What atomic number of an element "X" would have to become so that the 4th orbit around X would fit inside the I Bohr orbit of H atom?
A)
3 done
clear
B)
4 done
clear
C)
16 done
clear
D)
25 done
clear
View Solution play_arrow
-
question_answer44)
Which combinations of quantum numbers, \[n,l,m\] and s for the electron in an atom does not provide a permissible solution of the wave equation?
A)
\[3,2,1,+\frac{1}{2}\] done
clear
B)
\[3,1,1,-\frac{1}{2}\] done
clear
C)
\[~3,3,1-\frac{1}{2}\] done
clear
D)
\[3,2,-2,+\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer45)
If the nitrogen atom had electronic configuration \[1{{s}^{7}}\] it would have energy lower than that of the normal ground state configuration \[1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}\] because the electrons would be closer to the nucleus. Yet \[1{{s}^{7}}\] is not observed. It violates
A)
Heisenberg's uncertainty principle done
clear
B)
Hund's rule done
clear
C)
Pauli exclusion principle done
clear
D)
Bohr postulate of stationary orbits done
clear
View Solution play_arrow
-
question_answer46)
The position of both, an electron and a helium atom is known within 1.0 nm. Further the momentum of the electron is known within \[5.0\times {{10}^{-26}}kg\text{ }m{{s}^{-1}}\]. The minimum uncertainty in the measurement of the momentum of the helium atom is
A)
\[50kgm{{s}^{-1}}\] done
clear
B)
\[80kgm{{s}^{-1}}\] done
clear
C)
\[8.0\times {{10}^{-26}}kgm{{s}^{-1}}\] done
clear
D)
\[5.0\times {{10}^{-26}}kgm{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer47)
Two fast moving particles X and Y are associated with de Broglie wavelengths 1 nm and 4 nm respectively. If mass of X is nine times the mass of Y, the ratio of kinetic energies of X and Y would
A)
3 : 1 done
clear
B)
9 : 1 done
clear
C)
5 : 12 done
clear
D)
16 : 9 done
clear
View Solution play_arrow
-
question_answer48)
In a multi-electron atom, which of the following orbitals described by the three quantum members will have the same energy in the absence of magnetic and electric fields?
| [A] \[n=1,l=0,m=0\] |
| [B] \[n=2,l=0,m=0\] |
| [C] \[n=2,l=1,m=1\] |
| [D] \[n=3,l=2,m=1\] |
| [E] \[n=3,l=2,m=0\] |
A)
[D] and [E] done
clear
B)
[C] and [D] done
clear
C)
[B] and [C] done
clear
D)
[A] and [B] done
clear
View Solution play_arrow
-
question_answer49)
A system irradiated for 10 min. is found to absorb \[3\times {{10}^{18}}\]quanta per sec. If the amount of substance decomposed is\[~3\times {{10}^{-3}}mol\] \[({{N}_{A}}=6\times {{10}^{23}})\]. The quantum efficiency of the reaction is
A)
2.5 done
clear
B)
2 done
clear
C)
1.5 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer50)
The radii of maximum probability for 3s, 3p and 3d electrons are in the order
A)
\[{{({{r}_{max}})}_{3d}}>{{({{r}_{max}})}_{3p}}>{{({{r}_{max}})}_{3s}}\] done
clear
B)
\[{{({{r}_{max}})}_{3d}}>{{({{r}_{max}})}_{3s}}>{{({{r}_{max}})}_{3p}}\] done
clear
C)
\[{{({{r}_{max}})}_{3s}}>{{({{r}_{max}})}_{3p}}>{{({{r}_{max}})}_{3d}}\] done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer51)
If in Bohr's model, for unielectronic atom, time period of revolution is represented as \[{{T}_{n,z}}\] where n represents shell no. and Z represents atomic number then the value of\[{{T}_{1,2}}:{{T}_{2,1}}\], will be:
A)
8 : 1 done
clear
B)
1 : 8 done
clear
C)
1 : 1 done
clear
D)
1 : 32 done
clear
View Solution play_arrow
-
question_answer52)
Find the value of wave number \[~\left( \overline{v} \right)\] in terms of Rydberg's constant, when transition of electron takes place between two levels of \[H{{e}^{+}}\] ion whose sum is 4 and difference is 2.
A)
\[\frac{8R}{9}\] done
clear
B)
\[\frac{32R}{9}\] done
clear
C)
\[\frac{3R}{9}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer53)
The uncertainty in the position of an electron \[(mass=9.1\times {{10}^{28}}g)\] moving with a velocity of \[3.0\times {{10}^{4}}cm\text{ }{{s}^{-1}}\] accurate up to 0.011 % will be
A)
1.92 cm done
clear
B)
7.68 cm done
clear
C)
0.175 cm done
clear
D)
3.84 cm done
clear
View Solution play_arrow
-
question_answer54)
The energy of \[{{e}^{-}}\] in first orbit of \[H{{e}^{+}}\]is \[-871.6\times {{10}^{-20}}J.\] The energy of \[{{e}^{-}}\]in first orbit of H is:
A)
\[-871.6\times {{10}^{-20}}\] done
clear
B)
\[-435.8\times {{10}^{-20}}J\] done
clear
C)
\[-217.9\times {{10}^{-20}}J\] done
clear
D)
\[-108.9\times {{10}^{-20}}\] done
clear
View Solution play_arrow
-
question_answer55)
If a proton and \[\alpha \]-particle are accelerated through the same potential difference, the ratio of de-Broglie wavelengths \[{{\lambda }_{p}}\] and \[{{\lambda }_{\alpha }}\] is
A)
3 done
clear
B)
\[2\sqrt{2}\] done
clear
C)
1 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer56)
The velocity of particle A is \[0.1\text{ }m{{s}^{-1}}\] and that of particle B is\[~0.05\,m{{s}^{-1}}\]. If the mass of particle B is five times that of particle A, then the ratio of de- Broglie wavelengths associated with the particles A and B is
A)
2 : 5 done
clear
B)
3 : 4 done
clear
C)
6 : 4 done
clear
D)
5 : 2 done
clear
View Solution play_arrow
-
question_answer57)
Let \[{{m}_{p}}\]be the mass of a proton, \[{{m}_{n}}\]that of a neutron, \[{{M}_{1}}\]that of a \[_{10}^{20}Ne\] nucleus and \[{{M}_{2}}\] that of a \[_{20}^{40}Ca\] nucleus. Then
A)
\[{{M}_{2}}=2{{M}_{1}}\] done
clear
B)
\[{{M}_{1}}<10({{m}_{p}}+{{m}_{n}})\] done
clear
C)
\[{{M}_{2}}>2{{M}_{1}}\] done
clear
D)
\[{{M}_{1}}={{M}_{2}}\] done
clear
View Solution play_arrow
-
question_answer58)
If radius of second stationary orbit (in Bohr's atom) is R then radius of third orbit will be:
A)
R/3 done
clear
B)
9 R done
clear
C)
R/9 done
clear
D)
2.25 R done
clear
View Solution play_arrow
-
question_answer59)
The dissociation energy of \[{{H}_{2}}\] is \[430.53\text{ }kJmo{{l}^{-1}}\]. If hydrogen is dissociated by illumination with radiation of wavelength 253.7 nm the fraction of the radiant energy which will be converted into kinetic energy is given by
A)
100% done
clear
B)
8.82% done
clear
C)
2.22% done
clear
D)
1.22% done
clear
View Solution play_arrow
-
question_answer60)
The threshold frequency of a metal is \[1\times {{10}^{15}}{{s}^{-1}}.\] The ratio of the maximum kinetic energies of the photoelectrons when the metal is irradiated with radiations of frequencies \[1.5\times {{10}^{15}}{{s}^{-1}}\] and \[2.0\times {{10}^{15}}{{s}^{-1}}\] respectively would be
A)
4 : 3 done
clear
B)
1 : 2 done
clear
C)
2 : 1 done
clear
D)
3 : 4 done
clear
View Solution play_arrow
-
question_answer61)
The wave number of electromagnetic radiation emitted during the transition in between two energy levels of \[L{{i}^{2+}}\] ion whose principal quantum number sum is 4 and difference is 2, is
A)
\[3.5{{R}_{H}}\] done
clear
B)
\[4{{R}_{H}}\] done
clear
C)
\[8{{R}_{H}}\] done
clear
D)
\[\frac{8}{9}{{R}_{H}}\] done
clear
View Solution play_arrow
-
question_answer62)
An electron travels with a velocity of \[xm{{s}^{-1}}.\] For a proton to have the same de-Broglie wavelength, the velocity will be approximately:
A)
\[\frac{1840}{x}\] done
clear
B)
\[\frac{x}{1840}\] done
clear
C)
\[1840x\] done
clear
D)
\[x\] done
clear
View Solution play_arrow
-
question_answer63)
The total spin and magnetic moment for the atom with atomic number 24 are:
A)
\[\pm 3,\sqrt{48}BM\] done
clear
B)
\[\pm 3,\sqrt{35}BM\] done
clear
C)
\[\pm \frac{3}{2},\sqrt{48}BM\] done
clear
D)
\[\pm \frac{3}{2},\sqrt{35}BM\] done
clear
View Solution play_arrow
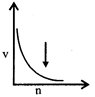
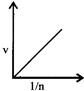
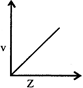
-
question_answer64)
Select the incorrect graph for velocity of \[{{e}^{-}}\]in an orbit vs. \[Z,\frac{1}{n}\] and n :
A)
B)
C)
D)
View Solution play_arrow
-
question_answer65)
An electron has been excited from the first to the fourth energy state in an atom. Which of the following transitions are possible when the electron comes back to the ground state?
A)
\[4\to 1\] done
clear
B)
\[4\to 2,2\to 1\] done
clear
C)
\[4\to 3,3\to 2,2\to 1\] done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer66)
What should be the kinetic energy and total energy of the electron present in hydrogen atom, if its potential energy is \[-5.02\text{ }eV\]?
A)
\[+2.51eV,-2.51eV\] done
clear
B)
\[+0.251\text{ }eV,-0.251eV\] done
clear
C)
\[-2.51eV,-2.51eV\] done
clear
D)
\[+2.51eV,+2.51eV\] done
clear
View Solution play_arrow
-
question_answer67)
Ionization potential of hydrogen atom is 13. 6eV. Hydrogen atom in ground state are excited by monochromatic light of energy 12.1 eV. The spectral lines emitted by hydrogen according to Bohr's theory will be
A)
one done
clear
B)
two done
clear
C)
three done
clear
D)
four done
clear
View Solution play_arrow
-
question_answer68)
If the subsidiary quantum number of a subenergy level is 4, the maximum and minimum values of the spin multiplicities are:
A)
9, 1 done
clear
B)
10, 1 done
clear
C)
10, 2 done
clear
D)
4,- 4 done
clear
View Solution play_arrow
-
question_answer69)
If the wavelength of the electromagnetic radiation is increased to thrice the digital value, then what will be the percent change in the value of frequency of the electromagnetic radiation.
A)
Increases by 33% done
clear
B)
Decreases by 33% done
clear
C)
Increases by 66% done
clear
D)
Decreases by 66% done
clear
View Solution play_arrow
-
question_answer70)
According to Bohr's theory the energy required for the transition of H-atom from\[~n=6\text{ }to\text{ }n=8\] state is
A)
equal to the energy required for the transition from \[n=5\text{ }to\text{ }n=6\]state done
clear
B)
larger than in [a] done
clear
C)
less than in [a] done
clear
D)
equal to the energy required for the transition from \[n=7\text{ }to\text{ }n=9\]state. done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
