-
question_answer1)
The correct increasing bond angles order is
A)
\[B{{F}_{3}}<N{{F}_{3}}<P{{F}_{3}}<Cl{{F}_{3}}\] done
clear
B)
\[Cl{{F}_{3}}<P{{F}_{3}}<N{{F}_{3}}<B{{F}_{3}}\] done
clear
C)
\[B{{F}_{3}}=N{{F}_{3}}<P{{F}_{3}}<Cl{{F}_{3}}\] done
clear
D)
\[B{{F}_{3}}<N{{F}_{3}}<P{{F}_{3}}<Cl{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer2)
Arrange the following in decreasing order of their bond length, \[{{O}_{2}},{{O}_{2}}^{+},{{O}_{2}}^{-}\] and \[O_{2}^{2-}\]
A)
\[O_{2}^{2-}<O_{2}^{-}<{{O}_{2}}+O_{2}^{+}\] done
clear
B)
\[{{O}_{2}}<O_{2}^{2-}<O_{2}^{-}<O_{2}^{+}\] done
clear
C)
\[O_{2}^{+}<O_{2}^{-}<O_{2}^{2-}<{{O}_{2}}\] done
clear
D)
\[O_{2}^{+}<{{O}_{2}}<O_{2}^{-}<O_{2}^{2-}\] done
clear
View Solution play_arrow
-
question_answer3)
The hybridization of atomic orbital's of nitrogen in \[NO_{2}^{+},NO_{2}^{-}\] and \[NH_{4}^{+}\]are
A)
\[s{{p}^{2}},\text{ }s{{p}^{3}}\] and \[s{{p}^{2}}\] respectively done
clear
B)
\[sp,\text{ }s{{p}^{2}}\] and \[s{{p}^{3}}\] respectively done
clear
C)
\[s{{p}^{2}},\text{ }sp\] and \[s{{p}^{3}}\] respectively done
clear
D)
\[s{{p}^{2}},\text{ }s{{p}^{3}}\] and sp respectively done
clear
View Solution play_arrow
-
question_answer4)
Which of the following has been arranged in order of increasing covalent character?
A)
\[KCl<CaC{{l}_{2}}<AlC{{l}_{3}}<SnC{{l}_{4}}\] done
clear
B)
\[SnC{{l}_{4}}<AlC{{l}_{3}}<CaC{{l}_{2}}<KCl\] done
clear
C)
\[AlC{{l}_{3}}<CaC{{l}_{2}}<KCl<SnC{{l}_{4}}\] done
clear
D)
\[CaC{{l}_{2}}<SnC{{l}_{4}}<KCl<AlC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer5)
In \[I{{F}_{3}}\] the bond angle of F - I - F is
A)
equal to \[{{90}^{o}}\] done
clear
B)
less than \[{{90}^{o}}\] done
clear
C)
greater than \[{{90}^{o}}\] done
clear
D)
equal to \[{{90}^{o}}\] done
clear
View Solution play_arrow
-
question_answer6)
Hydrogen of 'N' in \[N(Si{{H}_{3}})\] is
A)
\[s{{p}^{2}}\] done
clear
B)
\[s{{p}^{3}}\] done
clear
C)
sp done
clear
D)
Nis unhybridised done
clear
View Solution play_arrow
-
question_answer7)
Among the following species identify the isostructural pairs: \[N{{F}_{3}},NO_{3}^{-},B{{F}_{3}}{{H}_{3}}{{O}^{+}},H{{N}_{3}}\]
A)
\[[N{{F}_{3}},NO_{3}^{-}\,]\] and \[[B{{F}_{3}},\,\,{{H}_{3}}{{O}^{+}}]\] done
clear
B)
\[[N{{F}_{3}},H{{N}_{3}}]\] and \[[N{{O}_{3}}^{-},B{{F}_{3}}]\] done
clear
C)
\[[N{{F}_{3}},{{H}_{3}}{{O}^{+}}]\] and \[[N{{O}_{3}}^{-},B{{F}_{3}}]\] done
clear
D)
\[[N{{F}_{3}},{{H}_{3}}{{O}^{+}}]\] and \[[H{{N}_{3}},B{{F}_{3}}]\] done
clear
View Solution play_arrow
-
question_answer8)
Which one of the following arrangements of molecules is correct on the basis of their dipole moments?
A)
\[B{{F}_{3}}>N{{H}_{3}}>N{{F}_{3}}\] done
clear
B)
\[B{{F}_{3}}>N{{F}_{3}}>N{{H}_{3}}\] done
clear
C)
\[N{{H}_{3}}>B{{F}_{3}}>N{{F}_{3}}\] done
clear
D)
\[N{{H}_{3}}>N{{F}_{3}}>B{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer9)
Among \[K{{O}_{2}},AlO_{2}^{-},Ba{{O}_{2}}\] and \[NO_{2}^{+}\] unpaired electron is present in
A)
\[NO_{2}^{+}\] and \[Ba{{O}_{2}}\] done
clear
B)
\[K{{O}_{2}}\]and \[AlO_{2}^{-}\] done
clear
C)
\[K{{O}_{2}}\] only done
clear
D)
\[Ba{{O}_{2}}\] only done
clear
View Solution play_arrow
-
question_answer10)
Correctly match is:
A)
\[{{d}_{{{x}^{2}}-{{y}^{2}}}}\]atomic orbital - One nodal plane done
clear
B)
\[{{p}_{y}}\] atomic orbital - Two nodal planes done
clear
C)
\[{{\sigma }_{{{p}_{x}}}}\] molecular orbital - \[\Psi \](gerade) done
clear
D)
\[\pi _{{{p}_{y}}}^{*}\] molecularorbital - \[\Psi \](ungerade) done
clear
View Solution play_arrow
-
question_answer11)
Choose the correct order of bond angle of the following
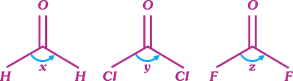
A)
x > z > y done
clear
B)
x > y > z done
clear
C)
z > y > x done
clear
D)
y > x > z done
clear
View Solution play_arrow
-
question_answer12)
Which of the following is not formed?
A)
\[BeF_{4}^{2-}\] done
clear
B)
\[BeF_{3}^{-}\] done
clear
C)
\[BF_{4}^{-}\] done
clear
D)
\[BF_{6}^{3-}\] done
clear
View Solution play_arrow
-
question_answer13)
Which one shows maximum hydrogen bonding?
A)
\[{{H}_{2}}O\] done
clear
B)
\[{{H}_{2}}Se\] done
clear
C)
\[{{H}_{2}}S\] done
clear
D)
HF done
clear
View Solution play_arrow
-
question_answer14)
Number of lone pairs around phosphorus in \[PC{{l}_{5}},PCl_{4}^{+}\] and \[PCl_{6}^{-}\] are respectively
A)
0, 1, 2 done
clear
B)
0, 0, 0 done
clear
C)
1, 2, 3 done
clear
D)
0, 2, 1 done
clear
View Solution play_arrow
-
question_answer15)
During steam distillation of a mixture of o-nitrophenol and p-nitrophenol
A)
vapours of o-nitrophenol are carried by steam because of its lower boiling point due to chelation. done
clear
B)
vapours of o-nitrophenols are carried by steam because of its lower boiling point and solubility in steam. done
clear
C)
vapours of o-nitrophenol are carried by steam because its boiling point is reduced by steam. done
clear
D)
vapours of p-nitrophenol are carried by steam because of its lower boiling point. done
clear
View Solution play_arrow
-
question_answer16)
The bond between carbon atom (1) and carbon atom (2) in compound \[N\equiv \overset{1}{\mathop{C}}\,-\overset{2}{\mathop{C}}\,H=C{{H}_{2}}\] involves the hybrid as
A)
\[s{{p}^{2}}\] and \[s{{p}^{2}}\] done
clear
B)
\[s{{p}^{3}}\] and sp done
clear
C)
sp and \[s{{p}^{2}}\] done
clear
D)
sp and sp done
clear
View Solution play_arrow
-
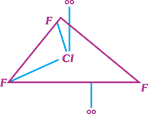
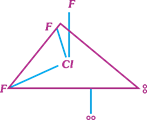
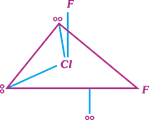
question_answer17)
Which of the following structure of \[Cl{{F}_{3}}\] is most stable?
A)
B)
C)
D)
All the three have equivalent stability done
clear
View Solution play_arrow
-
question_answer18)
Which compound contains both ionic and covalent bonds?
A)
Sodium nitrate done
clear
B)
Ammonia done
clear
C)
Methane done
clear
D)
Potassium chloride done
clear
View Solution play_arrow
-
question_answer19)
Which of following statement is true?
A)
Absolutely pure water does not contain any ions done
clear
B)
London forces is directly proportional to more number of loosely held electron done
clear
C)
Allene is polar due to its non-planar structure done
clear
D)
\[C{{H}_{3}}OC{{H}_{3}}\] is less volatile than \[{{C}_{2}}{{H}_{5}}OH\] due to higher molecular weight. done
clear
View Solution play_arrow
-
question_answer20)
Correct order of bond energy is
A)
\[{{N}_{2}}>N_{2}^{+}>N_{2}^{-}>N_{2}^{2-}\] done
clear
B)
\[N_{2}^{+}>N_{2}^{-}>N_{2}^{2-}>{{N}_{2}}\] done
clear
C)
\[{{N}_{2}}>N_{2}^{+}=N_{2}^{-}>N_{2}^{2-}\] done
clear
D)
\[N_{2}^{-}>N_{2}^{{}}=N_{2}^{+}>N_{2}^{2-}\] done
clear
View Solution play_arrow
-
question_answer21)
Which of the following molecule is not having perfect geometrical shape.
A)
\[PC{{l}_{3}}{{F}_{2}}\] done
clear
B)
\[C{{F}_{4}}\] done
clear
C)
\[B{{F}_{2}}Cl\] done
clear
D)
\[{{[Si{{F}_{6}}]}^{2-}}\] done
clear
View Solution play_arrow
-
question_answer22)
The orbital picture of a triplet carbene is :
A)
B)
C)
D)
View Solution play_arrow
-
question_answer23)
The molecule that will have minimum dipole moment is
A)
\[C{{H}_{3}}Cl\] done
clear
B)
\[C{{H}_{2}}Cl\] done
clear
C)
\[CHC{{l}_{3}}\] done
clear
D)
\[CFC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer24)
| Which of the following option is correct about \[N{{O}_{2}}\And Cl{{O}_{2}}?\] |
| (i) Both are paramagnetic species |
| (ii) Both compounds dimerised readily |
| (iii) Both have \[s{{p}^{2}}\] hybridisation |
| (iv) Both have Bent shape. |
A)
(i), (ii) done
clear
B)
(i), (iii), (iv) done
clear
C)
(ii), (iii), (iv) done
clear
D)
only (i) done
clear
View Solution play_arrow
-
question_answer25)
The structure of \[I{{F}_{7}}\] is
A)
pentagonal bipyramid done
clear
B)
square pyramid done
clear
C)
trigonal bipyramid done
clear
D)
octahedral done
clear
View Solution play_arrow
-
question_answer26)
Which of the following is correct regarding ionization potential?
A)
\[O_{2}^{2+}>NO\] done
clear
B)
\[O_{2}^{2+}<NO\] done
clear
C)
\[O_{2}^{{}}<NO\] done
clear
D)
\[O_{2}^{2+}=NO\] done
clear
View Solution play_arrow
-
question_answer27)
In which of the following sets, each member has linear shape:
A)
\[N_{3}^{-},\text{ }I_{3}^{+},\text{ }Xe{{F}_{2}}\] done
clear
B)
\[NO_{2}^{+},\text{ }O_{3}^{{}},\text{ }{{C}_{2}}{{H}_{2}}\] done
clear
C)
\[N_{3}^{-},\text{ }I_{3}^{-},\text{ }NO_{2}^{+}\] done
clear
D)
\[BeC{{l}_{2}},\text{ }Xe{{F}_{2}},\text{ }ClO_{2}^{-}\] done
clear
View Solution play_arrow
-
question_answer28)
What is not true about ice?
A)
It has open cage like structure done
clear
B)
It has less density than water done
clear
C)
Each O atom is surrounded by 4 H atoms done
clear
D)
Each O atom has four H-bonds around it done
clear
View Solution play_arrow
-
question_answer29)
Arrange the following compounds in increasing order of their ionic character: \[SnC{{l}_{2}},SnC{{l}_{4}},SiC{{l}_{4}},Sn{{F}_{4}},Sn{{F}_{2}}\]
A)
\[Sn{{F}_{2}}<SnC{{l}_{2}}<Sn{{F}_{4}}<SnC{{l}_{4}}<SiC{{l}_{4}}\] done
clear
B)
\[Sn{{F}_{2}}<SnC{{l}_{2}}<Sn{{F}_{4}}<SiC{{l}_{4}}<SnC{{l}_{4}}\] done
clear
C)
\[SiC{{l}_{4}}<SnC{{l}_{4}}<Sn{{F}_{4}}<SnC{{l}_{2}}<Sn{{F}_{2}}\] done
clear
D)
\[SnC{{l}_{4}}<Sn{{F}_{4}}<SnC{{l}_{2}}<Sn{{F}_{2}}<SiC{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer30)
Bond length of ethane (I), ethene (II), acetylene (III), and benzene (IV) follows the order:
A)
\[I>II>III>IV\] done
clear
B)
\[I>II>IV>III\] done
clear
C)
\[I>IV>II>III\] done
clear
D)
\[III>IV>II>I\] done
clear
View Solution play_arrow
-
question_answer31)
Amongst the following, the total number of species which does/do not exist is : \[S{{F}_{6}},B{{F}_{6}}^{3-},S{{F}_{4}},O{{F}_{4}},Al{{F}_{6}}^{3-},P{{H}_{5}},PC{{l}_{5}},NC{{l}_{5}},SC{{l}_{6}}\]
A)
9 done
clear
B)
5 done
clear
C)
6 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer32)
Most favorable condition for ionic bonding are:
A)
low charge on ions, large cations, small anions done
clear
B)
low charge on ions, large cations, large anions done
clear
C)
high charge on ions, small cations, large anions done
clear
D)
high charge on ions, large cations, small anions done
clear
View Solution play_arrow
-
question_answer33)
In which of the following mixtures, the London dispersion force acts as major intermolecular force of attraction?
A)
Sodium chloride and water. done
clear
B)
Cyciohexane and carbontetrachloride. done
clear
C)
Water and ethyl alcohol. done
clear
D)
Benzene and acetone. done
clear
View Solution play_arrow
-
question_answer34)
Which of the following is incorrect?
A)
One of the favourable conditions for the formation of an anion is high negative value of electron gain enthalpy of an element. done
clear
B)
Ionic bonds are non-directional whereas covalent bonds are directional in nature. done
clear
C)
One of the favourable conditions for the formation of ionic bonds is low lattice energy. done
clear
D)
Ionic compounds have high melting and boiling points and are soluble in polar solvents. done
clear
View Solution play_arrow
-
question_answer35)
Which combination is best explained by the coordinate covalent bond?
A)
\[{{H}^{+}}+{{H}_{2}}O\] done
clear
B)
\[Cl+Cl\] done
clear
C)
\[Mg+\frac{1}{2}{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}+{{I}_{2}}\] done
clear
View Solution play_arrow

 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
