-
question_answer1)
Incorrect statement about \[P{{H}_{3}}\] is:
A)
It is produced by hydrolysis of \[C{{a}_{2}}{{P}_{2}}\]. done
clear
B)
It gives black ppt. \[(C{{u}_{3}}{{P}_{2}})\] with \[CuS{{O}_{4}}\]solution. done
clear
C)
Spontaneously burns in presence of \[{{P}_{2}}{{H}_{2}}\]. done
clear
D)
It does not react with \[{{B}_{2}}{{H}_{6}}\]. done
clear
View Solution play_arrow
-
question_answer2)
The number of \[P-OH\] bonds and the oxidation state of phosphorus atom in pyrophosphoric acid \[({{H}_{4}}{{P}_{2}}{{O}_{7}})\] respectively are:
A)
four and four done
clear
B)
five and four done
clear
C)
five and five done
clear
D)
four and five done
clear
View Solution play_arrow
-
question_answer3)
\[{{P}_{2}}{{H}_{4}}\] can be removed from phosphme containing traces of it
A)
by passing impure \[P{{H}_{3}}\] gas through a freezing mixture. done
clear
B)
by passing the impure \[P{{H}_{3}}\] gas through HI and then its treatment with KOH (aq). done
clear
C)
by both [a] and [b] done
clear
D)
by none of these. done
clear
View Solution play_arrow
-
question_answer4)
\[N{{H}_{4}}Cl{{O}_{4}}+HN{{O}_{3}}\left( dil. \right)\xrightarrow{{}}HCl{{O}_{4}}+\left[ X \right]\] \[[X]\xrightarrow{\Delta }Y(g)\] [X] and [Y] are respectively-
A)
\[N{{H}_{4}}N{{O}_{3}}\And {{N}_{2}}O\] done
clear
B)
\[N{{H}_{4}}N{{O}_{2}}\And {{N}_{2}}\] done
clear
C)
\[HN{{O}_{4}}\And {{O}_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer5)
For \[{{H}_{3}}P{{O}_{3}}\] and \[{{H}_{3}}P{{O}_{4}}\], the correct choice is
A)
\[{{H}_{3}}P{{O}_{3}}\] is dibasic and reducing done
clear
B)
\[{{H}_{3}}P{{O}_{4}}\] is diabasic and non-reducing done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\] is tribasic and reducing done
clear
D)
\[{{H}_{3}}P{{O}_{4}}\] is tribasic and non-reducing done
clear
View Solution play_arrow
-
question_answer6)
One mole of calcium phosphide on reaction with excess of water gives
A)
one mole of phosphine. done
clear
B)
two moles of phosphoric acid. done
clear
C)
two moles of phosphine. done
clear
D)
one mole of phosphorus oxide. done
clear
View Solution play_arrow
-
question_answer7)
In nitrogen family, the H-M-H bond angle in the hydrides gradually becomes closer to \[90{}^\circ \] on going from N to Sb. This shows that gradually
A)
the basic strength of the hydrides increases. done
clear
B)
almost pure p-orbitals are used for M-H bonding. done
clear
C)
the bond energies of M-H bonds increases. done
clear
D)
the bond pairs of electrons become nearer to the central atom. done
clear
View Solution play_arrow
-
question_answer8)
Brown colour in \[HN{{O}_{3}}\] can be removed by
A)
adding Mg powder. done
clear
B)
boiling the acid. done
clear
C)
passing \[N{{H}_{3}}\] through acid. done
clear
D)
passing air through warm acid. done
clear
View Solution play_arrow
-
question_answer9)
In nitroprusside ion, the iron and NO exist as \[F{{e}^{2+}}\] and \[N{{O}^{+}}\] rather than \[F{{e}^{3+}}\] and NO. These forms can be differentiated by
A)
estimating the concentration of iron. done
clear
B)
measuring the concentration of \[C{{N}^{-}}\] done
clear
C)
measuring the solid state magnetic moment. done
clear
D)
thermally decomposing the compound. done
clear
View Solution play_arrow
-
question_answer10)
A deep brown gas is formed by mixing two colourless gases which are
A)
\[N{{O}_{2}}\] and \[{{O}_{2}}\] done
clear
B)
\[{{N}_{2}}O\] and NO done
clear
C)
NO and \[{{O}_{2}}\] done
clear
D)
\[N{{H}_{3}}\] and \[HCl\] done
clear
View Solution play_arrow
-
question_answer11)
Which of the following compounds does not exist?
A)
\[AsC{{l}_{5}}\] done
clear
B)
\[SbC{{l}_{3}}\] done
clear
C)
\[BiC{{l}_{5}}\] done
clear
D)
\[SbC{{l}_{5}}\] done
clear
View Solution play_arrow
-
question_answer12)
The brown ring test for \[NO_{2}^{-}\] and \[NO_{3}^{-}\] is due to the formation of complex ion with a formula
A)
\[{{[Fe{{({{H}_{2}}O)}_{6}}]}^{2+}}\] done
clear
B)
\[{{[Fe(NO){{(CN)}_{5}}]}^{2+}}\] done
clear
C)
\[{{[Fe{{({{H}_{2}}O)}_{5}}NO]}^{2+}}\] done
clear
D)
\[{{[Fe({{H}_{2}}O){{(NO)}_{5}}]}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer13)
Which of the following is a cyclic phosphate?
A)
\[{{H}_{3}}{{P}_{3}}{{O}_{10}}\] done
clear
B)
\[{{H}_{6}}{{P}_{4}}{{O}_{13}}\] done
clear
C)
\[{{H}_{5}}{{P}_{5}}{{O}_{15}}\] done
clear
D)
\[{{H}_{7}}{{P}_{5}}{{O}_{16}}\] done
clear
View Solution play_arrow
-
question_answer14)
An element (X) forms compounds of the formula \[XC{{l}_{3}},{{X}_{2}}{{\text{O}}_{5}}\] and \[C{{a}_{3}}{{X}_{2}}\] but does not form\[XC{{l}_{5}}\]. Which of the following is the element X?
A)
B done
clear
B)
Al done
clear
C)
N done
clear
D)
P done
clear
View Solution play_arrow
-
question_answer15)
\[{{P}_{4}}{{O}_{10}}\] is not used to dry \[N{{H}_{3}}\] gas because
A)
\[{{P}_{4}}{{O}_{10}}\] reacts with moisture in \[N{{H}_{3}}\]. done
clear
B)
\[{{P}_{4}}{{O}_{10}}\] is not a drying agent. done
clear
C)
\[{{P}_{4}}{{O}_{10}}\] is acidic and \[N{{H}_{3}}\] is basic. done
clear
D)
\[{{P}_{4}}{{O}_{10}}\] is basic and \[N{{H}_{3}}\] is acidic. done
clear
View Solution play_arrow
-
question_answer16)
Pick out the wrong statement.
A)
Nitrogen has the ability to form \[p\pi -p\pi \] bonds with itself done
clear
B)
Bismuth forms metallic bonds in elemental state. done
clear
C)
Catenation tendency is higher in nitrogen when compared with other elements of the same group. done
clear
D)
Nitrogen has higher first ionisation enthalpy when compared with other elements of the same group. done
clear
View Solution play_arrow
-
question_answer17)
Which of the following is incorrect for white and red phosphorus?
A)
They are both soluble in \[C{{S}_{2}}\]. done
clear
B)
They can be oxidised by heating in air. done
clear
C)
They consist of the same kind of atoms. done
clear
D)
They can be converted into one another. done
clear
View Solution play_arrow
-
question_answer18)
\[BC{{l}_{3}}\] is a planar molecule whereas \[NC{{l}_{3}}\] is pyramidal because
A)
\[BC{{l}_{3}}\] has no lone pair of electrons but \[NC{{l}_{3}}\] has a lone pair of electrons. done
clear
B)
\[B-Cl\] bond is more polar than \[N-Cl\] bond. done
clear
C)
nitrogen atom is smaller than boron atom. done
clear
D)
\[N-Cl\] bond is more covalent than \[B-Cl\] bond. done
clear
View Solution play_arrow
-
question_answer19)
A metal X on heating in nitrogen gas gives Y. Y on treatment with \[{{H}_{2}}O\] gives a colourless gas which when passed through \[CuS{{O}_{4}}\] solution gives a blue colour. Y is
A)
\[Mg{{(N{{O}_{3}})}_{2}}\] done
clear
B)
\[M{{g}_{3}}{{N}_{2}}\] done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
\[MgO\] done
clear
View Solution play_arrow
-
question_answer20)
Which of the following oxy-acids has the maximum number of hydrogens directly attached to phosphorus?
A)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
B)
\[{{H}_{3}}P{{O}_{2}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{3}}\] done
clear
D)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer21)
Number of sigma bonds in \[{{P}_{4}}{{O}_{10}}\] is
A)
6 done
clear
B)
7 done
clear
C)
17 done
clear
D)
16 done
clear
View Solution play_arrow
-
question_answer22)
Which of the following compound has a P-P bond?
A)
\[{{H}_{4}}{{P}_{2}}{{O}_{5}}\] done
clear
B)
\[{{(HP{{O}_{3}})}_{3}}\] done
clear
C)
\[{{H}_{4}}{{P}_{2}}{{O}_{6}}\] done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer23)
Which one of the following does not have a pyramidal shape?
A)
\[{{(C{{H}_{3}})}_{3}}N\] done
clear
B)
\[{{(Si{{H}_{3}})}_{3}}N\] done
clear
C)
\[P{{\left( C{{H}_{3}} \right)}_{3}}\] done
clear
D)
\[P{{\left( Si{{H}_{3}} \right)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer24)
Consider the following sequence of reaction. \[Na+N{{H}_{3}}\left( g \right)\xrightarrow{{}}\left[ X \right]\xrightarrow{{{N}_{2}}O}\] \[\left[ Y \right]\xrightarrow{Heat}\underset{Gas\,Pure}{\mathop{\left[ Z \right]}}\,\] Identify (Z) gas:
A)
\[{{N}_{2}}\] done
clear
B)
\[N{{H}_{3}}\] done
clear
C)
\[{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer25)
Among the following compounds, which on heating do not produce \[{{N}_{2}}\]?
A)
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{\text{O}}_{7}}\] done
clear
B)
\[N{{H}_{4}}Cl+NaN{{O}_{2}}\] done
clear
C)
\[N{{H}_{4}}Cl+CaO\] done
clear
D)
\[Ba{{({{N}_{3}})}_{2}}\] done
clear
View Solution play_arrow
-
question_answer26)
\[A+{{H}_{2}}O\xrightarrow{{}}B+HCl\] \[B+{{H}_{2}}O\xrightarrow{{}}C+HCl\] Compound (A), (B) and (C) will be respectively:
A)
\[PC{{l}_{5}},POC{{l}_{3}},{{H}_{3}}P{{O}_{3}}\] done
clear
B)
\[PC{{l}_{5}},POC{{l}_{3}},{{H}_{3}}P{{O}_{4}}\] done
clear
C)
\[SOC{{l}_{2}},POC{{l}_{3}},{{H}_{3}}P{{O}_{3}}\] done
clear
D)
\[PC{{l}_{3}},POC{{l}_{3}},{{H}_{3}}P{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer27)
A white precipitate is obtained on hydrolysis of
A)
\[PC{{l}_{5}}\] done
clear
B)
\[NC{{l}_{3}}\] done
clear
C)
\[BiC{{l}_{3}}\] done
clear
D)
\[AsC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer28)
Superphosphate of lime is obtained from the reaction of:
A)
Bones with gypsum. done
clear
B)
Calcium phosphate with sulphuric acid. done
clear
C)
Calcium phosphate with \[HCl\]. done
clear
D)
Calcium carbonate with phosphoric acid. done
clear
View Solution play_arrow
-
question_answer29)
Phosphine is not obtained by which of the following reaction?
A)
White P is heated with\[NaOH\]. done
clear
B)
Red P is heated with\[NaOH\]. done
clear
C)
\[C{{a}_{3}}{{P}_{2}}\] reacts with water. done
clear
D)
Phosphorus trioxide is boiled with water. done
clear
View Solution play_arrow
-
question_answer30)
Which of the following statements is not true?
A)
\[N{{O}_{2}}\] can be prepared by heating\[Pb{{\left( N{{O}_{3}} \right)}_{2}}\]. done
clear
B)
\[N{{O}_{2}}\] is red - brown gas. done
clear
C)
\[N{{O}_{2}}\] is diamagnetic. done
clear
D)
\[N{{O}_{2}}\] readily dimerises to \[{{N}_{2}}{{O}_{4}}\]. done
clear
View Solution play_arrow
-
question_answer31)
The deep blue colour produced on adding excess of ammonia to copper sulphate is due to presence of
A)
\[C{{u}^{2+}}\] done
clear
B)
\[{{\left[ Cu{{\left( N{{H}_{3}} \right)}_{4}} \right]}^{2+}}\] done
clear
C)
\[{{\left[ Cu{{\left( N{{H}_{3}} \right)}_{6}} \right]}^{2+}}\] done
clear
D)
\[{{\left[ Cu{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer32)
Cone. \[HN{{O}_{3}}\] is heated with \[{{P}_{2}}{{O}_{5}}\] to form:
A)
\[N{{O}_{2}}\] done
clear
B)
NO done
clear
C)
\[{{N}_{2}}{{O}_{^{5}}}\] done
clear
D)
\[{{N}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer33)
From the following information \[X+{{H}_{2}}S{{O}_{4}}\to Y\] (a colourless arid irritating gas) \[Y+{{K}_{2}}C{{r}_{2}}+{{H}_{2}}S{{O}_{4}}\to \] (green coloured solution) Identify the pair X and Y.
A)
\[\overline{C}r,HCl\] done
clear
B)
\[SO_{3}^{2-},S{{\text{O}}_{2}}\] done
clear
C)
\[{{S}^{2-}},{{H}_{2}}S\] done
clear
D)
\[CO_{3}^{2-},C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer34)
\[{{H}_{2}}S{{O}_{4}}\] and \[{{H}_{2}}S{{O}_{3}}\] can be distinguished by the addition of
A)
magnesium powder done
clear
B)
\[NaHS{{O}_{4}}\] solution done
clear
C)
\[FeC{{l}_{3}}\] solution done
clear
D)
litmus solution done
clear
View Solution play_arrow
-
question_answer35)
Which of the following is the key step in the manufacture of sulphuric acid?
A)
Burning of sulphur or sulphide ores in air to generate \[S{{O}_{2}}\]. done
clear
B)
Conversion of \[S{{O}_{2}}\] to \[S{{O}_{3}}\] by the reaction with oxygen in presence of catalyst. done
clear
C)
Absorption of \[S{{O}_{3}}\] in \[{{H}_{2}}S{{O}_{4}}\] to give oleum. done
clear
D)
Both [b] and [c] done
clear
View Solution play_arrow
-
question_answer36)
\[FeC{{l}_{3}}\] solution on reaction with \[S{{O}_{2}}\] changes to
A)
\[FeC{{l}_{2}}\] done
clear
B)
\[F{{e}_{2}}{{(S{{O}_{4}})}_{3}}\] done
clear
C)
\[F{{e}_{2}}{{(S{{O}_{3}})}_{3}}\] done
clear
D)
\[FeS{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer37)
Hydrolysis of one mole of peroxodisulphuric acid produces
A)
two moles of sulphuric acid. done
clear
B)
two moles of peroxomonosulphuric acid. done
clear
C)
one mole of sulphuric acid and one mole of peroxomonosulphuric acid. done
clear
D)
one mole of sulphuric acid, one mole of peroxomonosulphuric acid and one mole of hydrogen peroxide. done
clear
View Solution play_arrow
-
question_answer38)
Concentrated \[{{H}_{2}}S{{O}_{4}}\] is not used to prepare \[HBr\] from KBr because it
A)
oxidizes \[HBr\]. done
clear
B)
reduces \[HBr\]. done
clear
C)
causes disproportionation of \[HBr\]. done
clear
D)
reacts too slowly with KBr. done
clear
View Solution play_arrow
-
question_answer39)
Which reaction represents the oxidizing behavior of \[{{H}_{2}}S{{O}_{4}}\]?
A)
\[2PC{{l}_{5}}+{{H}_{2}}S{{O}_{4}}\to 2POC{{l}_{3}}+2HCl+S{{O}_{2}}C{{l}_{2}}\] done
clear
B)
\[2NaOH+{{H}_{2}}S{{O}_{4}}\to N{{a}_{2}}S{{O}_{4}}+2{{H}_{2}}O\] done
clear
C)
\[NaCl+{{H}_{2}}S{{O}_{4}}\to NaHS{{O}_{4}}\text{+}HCl\] done
clear
D)
\[2HI+{{H}_{2}}S{{O}_{4}}\to {{I}_{2}}+S{{O}_{2}}+2{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer40)
Which of the following is not correctly matched?
A)
\[S{{F}_{4}}\]- gas done
clear
B)
\[Se{{F}_{4}}\]- liquid done
clear
C)
\[Te{{F}_{4}}\]- solid done
clear
D)
\[S{{F}_{6}}\]- solid done
clear
View Solution play_arrow
-
question_answer41)
Which of the following on thermal decomposition gives oxygen gas?
A)
\[A{{g}_{2}}O\] done
clear
B)
\[P{{b}_{3}}{{O}_{4}}\] done
clear
C)
\[Pb{{O}_{2}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer42)
When sugar is treated with cone. sulphuric acid, the sugar is charred. In this process, sugar is
A)
reduced done
clear
B)
oxidized done
clear
C)
sulphonated done
clear
D)
dehydrated done
clear
View Solution play_arrow
-
question_answer43)
The number of \[S-S\] bonds in \[S{{O}_{3}},{{S}_{2}}O_{3}^{2-},{{S}_{2}}O_{6}^{2-}\] and \[{{S}_{2}}O_{8}^{2-}\] respectively are
A)
1,0,0,1 done
clear
B)
1,0,1,0 done
clear
C)
0,1,1,0 done
clear
D)
0,1,0,1 done
clear
View Solution play_arrow
-
question_answer44)
Correct order of \[O-O\] bond length (increasing) in \[{{O}_{2}},\,\,{{H}_{2}}{{O}_{2}}\] and \[{{O}_{3}}\] is
A)
\[{{H}_{2}}{{O}_{2}}<{{O}_{3}}<{{O}_{2}}\] done
clear
B)
\[{{O}_{2}}<{{O}_{3}}<{{H}_{2}}{{O}_{2}}\] done
clear
C)
\[{{O}_{3}}<{{O}_{2}}<{{H}_{2}}{{O}_{2}}\] done
clear
D)
\[{{O}_{3}}<{{H}_{2}}{{O}_{2}}<{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer45)
Iron sulphide is heated in air to form A, an oxide of sulphur. A is dissolved in water to give an acid. The basicity of this acid is
A)
2 done
clear
B)
3 done
clear
C)
1 done
clear
D)
zero done
clear
View Solution play_arrow
-
question_answer46)
The shape of the molecule \[S{{F}_{3}}C{{l}_{3}}\] is
A)
trigonal bipyramidal done
clear
B)
cubic done
clear
C)
octahedral done
clear
D)
tetrahedral done
clear
View Solution play_arrow
-
question_answer47)
What are the products formed when chlorine is passed through aqueous hypo solution?
A)
\[N{{a}_{2}}S{{O}_{3}}+HCl+S\] done
clear
B)
\[N{{a}_{2}}S{{O}_{3}}+S{{O}_{3}}+HCl\] done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}+HCl+S\] done
clear
D)
\[N{{a}_{2}}S{{O}_{4}}+HCl+S{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer48)
Which of the following is incorrect?
A)
M.p of monoclinic sulphur > m.p. of rhombic sulphur. done
clear
B)
Specific gravity of rhombic sulphur > specific gravity of monoclinic sulphur. done
clear
C)
Monoclinic sulphur is stable below 369 K. done
clear
D)
Both rhombic sulphur and monoclinic sulphur have \[{{S}_{8}}\] molecules. done
clear
View Solution play_arrow
-
question_answer49)
Which of the following form of the sulphur shows paramagnetic behaviour?
A)
\[{{S}_{8}}\] done
clear
B)
\[{{S}_{6}}\] done
clear
C)
\[{{S}_{2}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer50)
Which of the following has \[p\pi -d\pi \] bonding?
A)
\[NO_{3}^{-}\] done
clear
B)
\[SO_{3}^{2-}\] done
clear
C)
\[BO_{3}^{3-}\] done
clear
D)
\[CO_{3}^{2-}\] done
clear
View Solution play_arrow
-
question_answer51)
Sulphuric acid reacts with \[PC{{l}_{5}}\] to give
A)
thionyl chloride done
clear
B)
sulphur monochloride done
clear
C)
sulphuryl chloride done
clear
D)
sulphur tetrachloride done
clear
View Solution play_arrow
-
question_answer52)
By which of the following \[S{{O}_{2}}\] is formed?
A)
Reaction of dil. \[{{H}_{2}}S{{O}_{4}}\] with \[{{O}_{2}}\] done
clear
B)
Hydrolysis of dil. \[{{H}_{2}}S{{O}_{4}}\] done
clear
C)
Reaction of cone. \[{{H}_{2}}S{{O}_{4}}\] with Cu done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer53)
The acid which has a peroxy linkage is
A)
Sulphurous acid done
clear
B)
Pyrosulphuric acid done
clear
C)
Dithionic acid done
clear
D)
Caro's acid done
clear
View Solution play_arrow
-
question_answer54)
Excess of KI reacts with \[CuS{{O}_{4}}\] solution and then \[N{{a}_{2}}{{S}_{2}}{{O}_{3}}\] solution is added to it. Which of the statements is incorrect for this reaction?
A)
\[N{{a}_{2}}{{S}_{2}}{{O}_{3}}\] is oxidized done
clear
B)
\[Cu{{I}_{2}}\] is formed done
clear
C)
\[C{{u}_{2}}{{I}_{2}}\] is formed done
clear
D)
Evolved \[{{I}_{2}}\] is reduced done
clear
View Solution play_arrow
-
question_answer55)
Of the interhalogen \[A{{X}_{3}}\] compounds, \[Cl{{F}_{3}}\] is most reactive but \[Br{{F}_{3}}\] has higher conductance in liquid state. This is because
A)
\[Br{{F}_{3}}\] has higher molecular mass. done
clear
B)
\[Cl{{F}_{3}}\] is more volatile. done
clear
C)
\[Br{{F}_{3}}\] dissociates into \[BrF_{2}^{+}\] and \[BrF_{4}^{-}\] most easily. done
clear
D)
Electrical conductance does not depend on concentration. done
clear
View Solution play_arrow
-
question_answer56)
Which of the following is the best description for the behaviour of bromine in the reaction given below? \[{{H}_{2}}O+B{{r}_{2}}\to HOBr+HBr\]
A)
Proton acceptor only done
clear
B)
Both oxidized and reduced done
clear
C)
Oxidized only done
clear
D)
Reduced only done
clear
View Solution play_arrow
-
question_answer57)
Oxidation of thiosulphate with iodine gives
A)
sulphate ion done
clear
B)
sulphite ion done
clear
C)
tetrathionate ion done
clear
D)
sulphide ion done
clear
View Solution play_arrow
-
question_answer58)
Which of the following is correct about the reaction? \[3NaClO\xrightarrow{heat}NaCl{{O}_{3}}+2NaCl\]
A)
It is disproportionation reaction done
clear
B)
Oxidation number of Cl decreases as well as increases in this reaction done
clear
C)
This reaction is used for the manufacture of halates done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer59)
The reaction of \[KMn{{O}_{4}}\] and \[HCl\] results in
A)
oxidation of Mn in\[KMn{{O}_{4}}\] and production of\[C{{l}_{2}}\]. done
clear
B)
reduction of Mn in \[KMn{{O}_{4}}\] and production of \[{{H}_{2}}\]. done
clear
C)
oxidation of Mn in \[KMn{{O}_{4}}\] and production of \[{{H}_{2}}\]. done
clear
D)
reduction of Mn in \[KMn{{O}_{4}}\] and production of\[C{{l}_{2}}\]. done
clear
View Solution play_arrow
-
question_answer60)
The correct order of reactivity of halogens with alkalies is
A)
\[F>Cl>Br>I\] done
clear
B)
\[F<Cl>Br<I\] done
clear
C)
\[F<Cl<Br<I\] done
clear
D)
\[F<Cl<Br>I\] done
clear
View Solution play_arrow
-
question_answer61)
One mole of fluorine is reacted with two moles of hot and concentrated \[KOH\]. The products formed are \[KF,{{H}_{2}}O\] and\[{{O}_{2}}\] . The molar ratio of \[KF,{{H}_{2}}O\] and \[{{O}_{2}}\] respectively is
A)
1:1:2 done
clear
B)
2:1:0.5 done
clear
C)
1:2:1 done
clear
D)
2:1:2 done
clear
View Solution play_arrow
-
question_answer62)
Chlorine cannot displace
A)
fluorine from NaF done
clear
B)
iodine from Nal done
clear
C)
bromine from NaBr done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer63)
When chlorine reacts with cold and dilute solution of sodium hydroxide, the products obtained are
A)
\[C{{l}^{-}}+Cl{{O}^{-}}\] done
clear
B)
\[C{{l}^{-}}+ClO_{2}^{-}\] done
clear
C)
\[C{{l}^{-}}+ClO_{3}^{-}\] done
clear
D)
\[C{{l}^{-}}+ClO_{4}^{-}\] done
clear
View Solution play_arrow
-
question_answer64)
A greenish yellow gas reacts with an alkali metal hydroxide to form a halate which can be used in fireworks safety matches. The gas and halate respectively are
A)
\[B{{r}_{2}},\,\,KBr{{O}_{3}}\] done
clear
B)
\[C{{l}_{2}},\,\,KCl{{O}_{3}}\] done
clear
C)
\[{{I}_{2}},NaI{{O}_{3}}\] done
clear
D)
\[C{{l}_{2}},NaCl{{\text{O}}_{3}}\] done
clear
View Solution play_arrow
-
question_answer65)
In the reaction \[HN{{O}_{3}}+HF\xrightarrow{{}}{{H}_{2}}\overset{+}{\mathop{N}}\,{{O}_{3}}+{{F}^{-}}\] base is
A)
HF done
clear
B)
\[HN{{O}_{3}}\] done
clear
C)
HF and \[HN{{O}_{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer66)
The aqueous solution of which of the following has maximum pH?
A)
\[NaClO\] done
clear
B)
\[NaCl{{O}_{2}}\] done
clear
C)
\[NaCl{{O}_{3}}\] done
clear
D)
\[NaCl{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer67)
Which one of the following oxides of chlorine is obtained by passing dry chlorine over silver chlorate at \[90{}^\circ C\]?
A)
\[C{{l}_{2}}O\] done
clear
B)
\[Cl{{O}_{3}}\] done
clear
C)
\[Cl{{O}_{2}}\] done
clear
D)
\[CI{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer68)
Identify the incorrect statement among the following
A)
\[B{{r}_{2}}\] reacts with hot and strong \[NaOH\]solution to give \[NaBr\] and\[{{H}_{2}}O\]. done
clear
B)
Ozone reacts with \[S{{O}_{2}}\] to give \[S{{O}_{3}}\]. done
clear
C)
Silicon reacts with \[NaOH\left( aq \right)\] in the presence of air to give \[N{{a}_{2}}Si{{O}_{3}}\] and\[{{H}_{2}}O\] . done
clear
D)
\[C{{l}_{2}}\] reacts with excess of \[N{{H}_{3}}\] to give \[{{N}_{2}}\] and\[HCl\]. done
clear
View Solution play_arrow
-
question_answer69)
The least number of oxyacids are formed by:
A)
Chlorine done
clear
B)
Nitrogen done
clear
C)
Fluorine done
clear
D)
Sulphur done
clear
View Solution play_arrow
-
question_answer70)
Chlorine water on standing loses its colour and forms:
A)
\[HCl\] only done
clear
B)
\[HCl\] and \[HCl{{O}_{2}}\] done
clear
C)
\[HCl\] and HOCl done
clear
D)
HOCl and \[HCl{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer71)
Shapes of certain interhalogen compounds are stated below. Which one of them is not correctly stated?
A)
\[I{{F}_{7}}\]: pentagonal bipyramidal done
clear
B)
\[Br{{F}_{5}}\]: trigonal bipyramidal done
clear
C)
\[Br{{F}_{3}}\]: planar T-shaped done
clear
D)
\[IC{{I}_{3}}\]: planar dimeric done
clear
View Solution play_arrow
-
question_answer72)
Electron gain enthalpy with negative sign of fluorine is less than that of chlorine due to:
A)
High ionization enthalpy of fluorine done
clear
B)
Smaller size of chlorine atom done
clear
C)
Smaller size of fluorine atom done
clear
D)
Bigger size of 2p orbital of fluorine done
clear
View Solution play_arrow
-
question_answer73)
Which of the following xenon-oxo compounds may not be obtained by hydrolysis of xenon fluorides?
A)
\[Xe{{O}_{2}}{{F}_{2}}\] done
clear
B)
\[XeO{{F}_{4}}\] done
clear
C)
\[Xe{{O}_{3}}\] done
clear
D)
\[Xe{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer74)
Trigonal bipyramidal geometry is shown by:
A)
\[Xe{{O}_{3}}\] done
clear
B)
\[Xe{{O}_{3}}{{F}_{2}}\] done
clear
C)
\[FXeOS{{O}_{2}}F\] done
clear
D)
\[{{\left[ Xe{{F}_{8}} \right]}^{2-}}\] done
clear
View Solution play_arrow
-
question_answer75)
\[Xe{{O}_{4}}\] molecule is tetrahedral having:
A)
Two \[p\pi -d\pi \] bonds done
clear
B)
One \[p\pi -d\pi \] bonds done
clear
C)
Four \[p\pi -d\pi \] bonds done
clear
D)
Three \[p\pi -d\pi \] bonds done
clear
View Solution play_arrow
-
question_answer76)
The compound of xenon with zero dipole moment is
A)
\[Xe{{O}_{3}}\] done
clear
B)
\[Xe{{F}_{4}}\] done
clear
C)
\[XeO{{F}_{4}}\] done
clear
D)
\[Xe{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer77)
\[Xe{{F}_{6}}\] dissolves in anhydrous HF to give a good conducting solution which contains:
A)
\[{{H}^{+}}\] and \[XeF_{7}^{-}\] ion done
clear
B)
\[HF_{2}^{-}\] and \[XeF_{5}^{+}\] ions done
clear
C)
\[HXeF_{6}^{+}\] and \[{{F}^{-}}\] ions done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer78)
Incorrect statement regarding following reactions is:
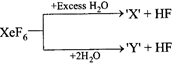
A)
'X' is explosive done
clear
B)
'Y' is an oxyacid of xenon done
clear
C)
Both are example of non-redox reaction done
clear
D)
\[Xe{{F}_{6}}\] can undergo partial hydrolysis. done
clear
View Solution play_arrow
-
question_answer79)
In the clathrates of xenon with water, the nature of bonding between xenon and water molecule is
A)
covalent done
clear
B)
hydrogen bonding done
clear
C)
co-ordinate done
clear
D)
dipole-induced dipole interaction done
clear
View Solution play_arrow
-
question_answer80)
\[Xe{{F}_{4}}\] on partial hydrolysis produces
A)
\[Xe{{F}_{4}}\] done
clear
B)
\[XeO{{F}_{2}}\] done
clear
C)
\[XeO{{F}_{4}}\] done
clear
D)
\[Xe{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer81)
Which one of the following is correct pair with respect to molecular formula of xenon compound and hybridization state of xenon in it?
A)
\[Xe{{F}_{4}},s{{p}^{3}}\] done
clear
B)
\[Xe{{F}_{2}},sp\] done
clear
C)
\[Xe{{F}_{2}},s{{p}^{3}}d\] done
clear
D)
\[Xe{{F}_{4}},s{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer82)
Which of the following noble gas is least polarisable?
A)
He done
clear
B)
Xe done
clear
C)
Ar done
clear
D)
Ne done
clear
View Solution play_arrow
-
question_answer83)
What are the products formed in the reaction of xenon hexafluoride with silicon dioxide?
A)
\[XeSi{{O}_{4}}+HF\] done
clear
B)
\[Xe{{F}_{2}}+Si{{F}_{4}}\] done
clear
C)
\[XeO{{F}_{4}}+Si{{F}_{4}}\] done
clear
D)
\[Xe{{O}_{3}}+Si{{F}_{2}}\] done
clear
View Solution play_arrow
-
question_answer84)
Which statement about noble gases is not correct?
A)
Xe forms \[Xe{{F}_{6}}\]. done
clear
B)
Ar is used in electric bulbs. done
clear
C)
Kr is obtained during radioactive disintegration. done
clear
D)
He has the lowest b.p. among all the noble gases. done
clear
View Solution play_arrow
-
question_answer85)
Density of nitrogen gas prepared from air is slightly greater than that of nitrogen prepared by chemical reaction from a compound of nitrogen due to the presence of
A)
argon done
clear
B)
carbon dioxide done
clear
C)
some \[{{N}_{3}}\] molecules analogous to \[{{O}_{3}}\]. done
clear
D)
greater amount of \[{{N}_{2}}\] molecules derived from N-15 isotope. done
clear
View Solution play_arrow
-
question_answer86)
The formation of \[O_{2}^{+}{{[Pt{{F}_{6}}]}^{-}}\] is the basis for the formation of xenon fluorides. This is because
A)
\[{{O}_{2}}\] and Xe have comparable sizes. done
clear
B)
both \[{{O}_{2}}\] and Xe are gases. done
clear
C)
\[{{O}_{2}}\] and Xe have comparable ionization energies. done
clear
D)
Both [a] and [c] done
clear
View Solution play_arrow
-
question_answer87)
The reason for not storing \[Xe{{F}_{6}}\] in a glass or a quartz vessel is that
A)
it forms an explosive having the formula\[Xe{{O}_{2}}{{F}_{2}}\]. done
clear
B)
it forms an explosive having the formula\[XeO{{F}_{4}}\]. done
clear
C)
it forms \[Xe{{O}_{2}}\] which is explosive substance. done
clear
D)
it forms \[Xe{{O}_{6}}^{4-}\] which is explosive in nature. done
clear
View Solution play_arrow
-
question_answer88)
\[Sb{{F}_{5}}\] reacts with \[Xe{{F}_{4}}\] to form an adduct. The shapes of cation and anion in the adduct are respectively:
A)
square planar, trigonal bipyramidal done
clear
B)
T-shaped, octahedral done
clear
C)
square pyramidal, octahedral done
clear
D)
square planar, octahedral done
clear
View Solution play_arrow
-
question_answer89)
Out of (i) \[Xe{{O}_{3}}\] (ii) \[XeO{{F}_{4}}\] and (iii) \[Xe{{F}_{6}}\], the molecules having same number of lone pairs on Xe are
A)
(i) and (ii) only done
clear
B)
(i) and (iii) only done
clear
C)
(ii) and (iii) only done
clear
D)
(i), (ii) and (iii) done
clear
View Solution play_arrow
-
question_answer90)
The geometry of \[XeO{{F}_{4}}\] by VSEPR theory is:
A)
pentagonal planar done
clear
B)
octahedral done
clear
C)
square pyramidal done
clear
D)
trigonal bipyramidal done
clear
View Solution play_arrow

