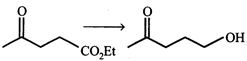-
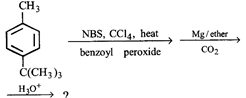
question_answer1)

Product [B] is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer2)
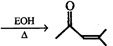 |
| \[(Q)\xrightarrow[^{\Delta }]{EOH}Ph-C{{H}_{2}}-OH+Ph-C{{O}_{2}}\] |
| \[(R)\xrightarrow{{{O}_{3}}}P+Q\], Structure of(R) is: |
A)
\[Ph-CH=CH-C{{H}_{3}}\] done
clear
B)
C)
\[Ph-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,=CH-C{{H}_{3}}\] done
clear
D)
\[Ph-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
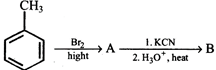
question_answer3)
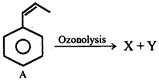
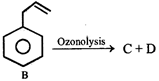
From the ozonolysis products, the two isomers A and B can be distinguished with the help of
A)
Fehling solution done
clear
B)
Tollen's reagent done
clear
C)
Haloformtest done
clear
D)
only spectroscopy done
clear
View Solution play_arrow
-
question_answer4)
The correct set of reagents to carry out the following conversion
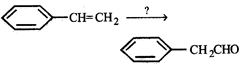
A)
\[{{B}_{2}}{{H}_{6}}/diglyme,{{H}_{2}}{{O}_{2}}/NaOH,\]\[PCC/C{{H}_{2}}C{{l}_{2}}\] done
clear
B)
\[B{{r}_{2}},NaN{{H}_{2}},HgS{{O}_{4}}/{{H}_{2}}S{{O}_{4}}\] done
clear
C)
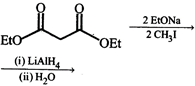
\[{{H}_{2}}O/{{H}_{2}}S{{O}_{4}},Cr{{O}_{3}}/{{H}_{2}}S{{O}_{4}}\] done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer5)
Which of the following gives ethanal \[C{{H}_{3}}CHO\]on acid hydrolysis?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer6)
The following reaction

is known as
A)
Perkin reaction done
clear
B)
Gattermann aldehyde synthesis done
clear
C)
Kolbe's reaction done
clear
D)
Gattermann-Koch reaction done
clear
View Solution play_arrow
-
question_answer7)
What is X in the following conversion?
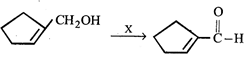
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}},{{H}_{2}}S{{O}_{4}}/{{H}_{2}}O\] done
clear
B)
\[HI{{O}_{4}}\] done
clear
C)
\[PCC/C{{H}_{2}}C{{l}_{2}}\] done
clear
D)
\[Os{{O}_{4}},{{\left( C{{H}_{3}} \right)}_{3}}C.COOH,{{\left( C{{H}_{3}} \right)}_{3}}COH,O{{H}^{-}}\] done
clear
View Solution play_arrow
-
question_answer8)
What is the product of the following reaction?
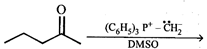
A)
2 - methyl -1 - pentene done
clear
B)
4 - methyl -1 - pentene done
clear
C)
2 - methyl - 2- propyloxirane done
clear
D)
1 - pentene done
clear
View Solution play_arrow
-
question_answer9)
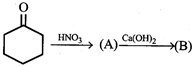
Product [B] in this reaction is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer10)
Which of the following pairs of reactants is most effective in forming an enamine?
A)
\[C{{H}_{3}}C{{H}_{2}}\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,H+{{[{{(C{{H}_{3}})}_{2}}CH]}_{2}}NH\] done
clear
B)
C)
\[{{(C{{H}_{3}})}_{3}}C\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,H+{{(C{{H}_{3}})}_{2}}NH\] done
clear
D)
None of these form an enamine. done
clear
View Solution play_arrow
-
question_answer11)
In the reaction sequence \[2C{{H}_{3}}CHO\xrightarrow{O{{H}^{-}}}A\xrightarrow{\Delta }B;\] the product B is:
A)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-OH\] done
clear
B)
\[C{{H}_{3}}-CH=CH-CHO\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer12)
Which one of the following reactions will not result in the formation of carbon-carbon bond?
A)
Reimer-Tieman reaction done
clear
B)
Friedel Craft's acylation done
clear
C)
Wurtz reaction done
clear
D)
Cannizzaro reaction done
clear
View Solution play_arrow
-
question_answer13)
Which is major product formed when acetone is heated with iodine and potassium hydroxide?
A)
lodoacetone done
clear
B)
Acetic acid done
clear
C)
lodoform done
clear
D)
Acetophenone done
clear
View Solution play_arrow
-
question_answer14)

\[\xrightarrow{LAH}\left( C \right);\] product (C) of the reaction is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer15)
End product of the following sequence of reactions are:
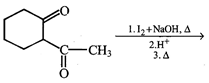
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
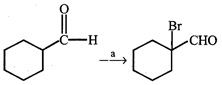 |
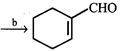 |
| Identify appropriate reagents for the above reaction: |
A)
\[a=B{{r}_{2}}/CC{{l}_{4}},\] \[b=aq.KOH\] done
clear
B)
\[a=B{{r}_{2}}/{{H}^{+}},\] \[b=aq.KOH\] done
clear
C)
\[a=B{{r}_{2}}/{{H}^{+}},\] \[b=alc.KOH\] done
clear
D)
\[a=B{{r}_{2}}/H{{O}^{-}},\] \[b=aq.KOH\] done
clear
View Solution play_arrow
-
question_answer17)
The starting compounds needed to make the following compound?
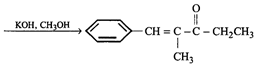
A)
benzaldehyde and 3-pentanone done
clear
B)
acetophenone and 2-butanone done
clear
C)
acetophenone and butanal done
clear
D)
benzaldehyde and 2-pentanone done
clear
View Solution play_arrow
-
question_answer18)
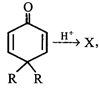
X, most likely the compound X is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer19)
The reagents employed to carry the following transformation
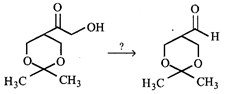
A)
\[LiAl{{H}_{4}},{{H}_{2}}S{{O}_{4}}/heat\] done
clear
B)
\[PCC/C{{H}_{2}}C{{l}_{2}}\] followed by \[HI{{O}_{4}}\] done
clear
C)
\[NaB{{H}_{4}}/C{{H}_{3}}OH\] followed by \[HI{{O}_{4}}\] done
clear
D)
\[{{O}_{3}}\] followed by \[{{(C{{H}_{3}})}_{2}}S\] done
clear
View Solution play_arrow
-
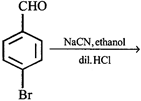
question_answer20)
What is the product of the following reaction?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer21)
Arrange the following in order of decreasing acidity
A)
C>A>B done
clear
B)
C>B>A done
clear
C)
A>C>B done
clear
D)
A>B>C done
clear
View Solution play_arrow
-
question_answer22)
What is the product of the following reaction?
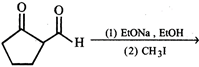
A)
B)
C)
D)
View Solution play_arrow
-
question_answer23)
Identify Z in the sequence \[C{{H}_{3}}COON{{H}_{4}}\xrightarrow{\Delta }X\xrightarrow[\Delta ]{{{P}_{2}}{{O}_{5}}}Y\xrightarrow{{{H}_{2}}O/{{H}^{+}}}Z\]
A)
\[C{{H}_{3}}C{{H}_{2}}CON{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}CN\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
\[{{(C{{H}_{3}}CO)}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer24)
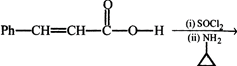
[A]; Product [A] of the reaction is:
A)
B)
C)
\[Ph-CH=CH-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-H\] done
clear
D)
View Solution play_arrow
-
question_answer25)
\[(X){{C}_{4}}{{H}_{7}}OCl\xrightarrow{N{{H}_{3}}}{{C}_{4}}{{H}_{9}}OH\] \[\xrightarrow[KOH]{B{{r}_{2}}}C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}};\] compound (X) is:
A)
B)
C)
D)
View Solution play_arrow
-
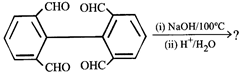
question_answer26)

Product. The product formed in the reaction is-
A)
B)
\[Me-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-Ph\] done
clear
C)
\[Me-\underset{Ph}{\mathop{\underset{|}{\mathop{\overset{OH}{\mathop{\overset{|}{\mathop{C}}\,}}\,}}\,}}\,-COOH\] done
clear
D)
\[Ph-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-O-Me\] done
clear
View Solution play_arrow
-
question_answer27)
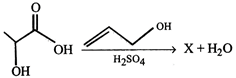
Product (X) of the reaction is-
A)
B)
C)
D)
View Solution play_arrow
-
question_answer28)
In a set of reactions, acetic acid yielded a product D. \[C{{H}_{3}}COOH\xrightarrow{SOC{{l}_{2}}}\left( A \right)\xrightarrow[AlC{{l}_{3}}]{Benzene}\left( B \right)\] \[\xrightarrow{HCN}(C)\xrightarrow{HOH}(D)\] The structure of [D] would be -
A)
B)
C)
D)
View Solution play_arrow
-
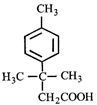
question_answer29)
Calculate number of molecules of Grignard reagent consumed by 1 molecule of following compound.
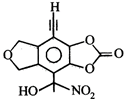
A)
5 done
clear
B)
2 done
clear
C)
3 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer30)
Which of the following will not give butyl acetate when treated with 1-butanol
A)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-Cl\left( pyridene \right)\] done
clear
B)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-OH\left( {{H}_{2}}S{{O}_{4}} \right)\] done
clear
C)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-O-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\left( pyridene \right)\] done
clear
D)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-H\left( {{H}_{2}}S{{O}_{4}} \right)\] done
clear
View Solution play_arrow
-
question_answer31)
Tischenko reaction is a modification of:
A)
Aldol condensation done
clear
B)
Claisen condensation done
clear
C)
Cannizzaro reaction done
clear
D)
Pinacol-pinacolon reaction done
clear
View Solution play_arrow
-
question_answer32)
Formaldehyde can be distinguished from acetaldehyde by the use of:
A)
Schiffs reagent done
clear
B)
Tollen's reagent done
clear
C)
\[{{I}_{2}}\]/Alkali done
clear
D)
Fehling's solution done
clear
View Solution play_arrow
-
question_answer33)
Clemmensen reduction of a ketone is carried out in the presence of:
A)
\[LiAl{{H}_{4}}\] done
clear
B)
\[Zn-Hg\,with\,HCl\] done
clear
C)
Glycol with KOH done
clear
D)
\[{{H}_{2}}\] with Pt as catalyst done
clear
View Solution play_arrow
-
question_answer34)
Which of the following is the product of aldol condensation?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer35)
Cannizaro?s reaction is not given by:
A)
B)
C)
\[C{{H}_{3}}CHO\] done
clear
D)
\[HCHO\] done
clear
View Solution play_arrow
-
question_answer36)
A)
B)
C)
D)
View Solution play_arrow
-
question_answer37)
What is the product of the following reaction.
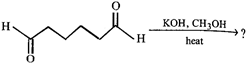
A)
B)
C)
D)
View Solution play_arrow
-
question_answer38)
Compound 'A' (molecular formula \[{{C}_{3}}{{H}_{8}}O\]) is treated with acidified potassium dichromate to form a product 'B' (molecular formula \[{{C}_{3}}{{H}_{6}}O\]). 'B' forms a shining silver mirror on warming with ammonical silver nitrate. 'B' when treated with an aqueous solution of \[{{H}_{2}}NCONHN{{H}_{2}}.HCl\] and sodium acetate gives a product 'C'. Identify the structure of 'C'?
A)
\[C{{H}_{3}}C{{H}_{2}}CH=NNHCON{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,=NNHCON{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,=NCONHN{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CH=NCONHN{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer39)
The most reactive compound towards formation of cyanohydrin on treatment with KCN followed by acidification is
A)
benzaldehyde done
clear
B)
p-nitrobenzaldehyde done
clear
C)
phenylacetaldehyde done
clear
D)
p-hydroxybenzaldehyde done
clear
View Solution play_arrow
-
question_answer40)
An organic compound 'A' has the molecular formula \[{{C}_{3}}{{H}_{6}}O\]. It undergoes iodoform test. When staturated with \[HCl\] it gives 'B' of molecula formula \[{{C}_{9}}{{H}_{14}}O\]. 'A' and 'B' respectively are
A)
Propanal and mesitylene done
clear
B)
Propanone and mesityl oxide done
clear
C)
Propanone and 2,6-dimethyl-2,5-heptadien- 4-one done
clear
D)
Propanone and mesitylene oxide done
clear
View Solution play_arrow
-
question_answer41)
What is D in the following sequence of reactions?
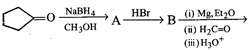
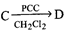
A)
B)
C)
D)
View Solution play_arrow
-
question_answer42)
The major product in the above reaction is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer43)
Identify the missing reagent in the reaction given below
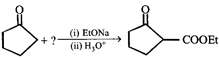
A)
Diethyl carbonate \[{{\left( EtO \right)}_{2}}C=O\] done
clear
B)
Ethyl acetate \[C{{H}_{3}}COOEt\] done
clear
C)
Ethyl formate \[HCOOEt\] done
clear
D)
Diethyl oxalate \[Et.OOC.COOEt\] done
clear
View Solution play_arrow
-
question_answer44)
The final product of the following sequence of reactions is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer45)
Identify Z in the following reaction sequence. \[C{{H}_{3}}CHO\xrightarrow[Dil.{{H}_{2}}S{{O}_{4}}]{MnO_{4}^{-}}X\xrightarrow{SOC{{l}_{2}}}Y\xrightarrow[\Delta ]{C{{H}_{3}}COONa}Z\]
A)
\[C{{H}_{3}}COC{{H}_{2}}COONa\] done
clear
B)
\[{{(C{{H}_{3}}CO)}_{2}}O\] done
clear
C)
\[C{{H}_{3}}CO-O-COC{{H}_{2}}Cl\] done
clear
D)
\[C{{H}_{3}}CO-OCOCHC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer46)
In the following sequence of reactions, what is D?
\[A\xrightarrow{SOC{{l}_{2}}}B\xrightarrow{Na{{N}_{3}}}C\xrightarrow{heat}D\]
A)
Primary amine done
clear
B)
An amide done
clear
C)
Phenyl isocyanate done
clear
D)
A higher hydrocarbon done
clear
View Solution play_arrow
-
question_answer47)
Which one of the following compounds undergoes decarboxylation upon heating?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer48)
Which lactone is formed by heating the following hydroxy acid?
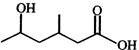
A)
B)
C)
D)
View Solution play_arrow
-
question_answer49)
The correct product of the following sequence of reactions
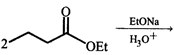
\[\xrightarrow[{{H}_{3}}{{O}^{+}}]{NaOH.{{H}_{2}}O,\Delta }\xrightarrow{\Delta }?\]
A)
4 - heptanone done
clear
B)
4 - methyl -3 - hexanone done
clear
C)
2 - ethyl pentanoic acid done
clear
D)
2 - propyl butanoic acid done
clear
View Solution play_arrow
-
question_answer50)
The correct product of the following reactions
A)
2, 2-dimethyl propane diol done
clear
B)
2-methyl-1-propanol done
clear
C)
2, 2-dimethyl propanedioic acid done
clear
D)
2-methyl propanoic acid done
clear
View Solution play_arrow
-
question_answer51)
Arrange the following carbonyl compounds in decreasing order of their reactivity in nucleophilic addition reaction.

A)
(ii)>(iii)>(i)>(iv) done
clear
B)
(ii)>(i)>(iv)>(iii) done
clear
C)
(iii)>(ii)>(i)>(iv) done
clear
D)
(iii)>(i)>(iv)>(ii) done
clear
View Solution play_arrow
-
question_answer52)

In the above reaction, product is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer53)
A)
I>II>III done
clear
B)
II>I>III done
clear
C)
II>III>I done
clear
D)
I>III>II done
clear
View Solution play_arrow
-
question_answer54)
What is the final product (B) of this sequence?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer55)
1-Phenylethanol can be prepared by reaction of benzaldehyde with
A)
methyl bromide done
clear
B)
ethyl iodide and magnesium done
clear
C)
methyl bromide and aluminium bromide done
clear
D)
methyl iodide and magnesium done
clear
View Solution play_arrow
-
question_answer56)
Oxidation of acetaldehyde with selenium dioxide produces
A)
ethanoicacid done
clear
B)
methanoic acid done
clear
C)
glyoxal done
clear
D)
oxalic acid done
clear
View Solution play_arrow
-
question_answer57)
Which of the following will respond to Cannizzaro's reaction?
A)
2,2-Dimethylpropanal done
clear
B)
Acetaldehyde done
clear
C)
Propionaldehyde done
clear
D)
Cinnamaldehyde done
clear
View Solution play_arrow
-
question_answer58)
If 3-hexanone is reacted with \[NaB{{H}_{4}}\] followed by hydrolysis with\[{{D}_{2}}O\], the product will be
A)
\[C{{H}_{3}}C{{H}_{2}}CH(OH)C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}CD\left( OH \right)C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CH\left( OD \right)C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CD\left( OD \right)C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer59)
Propanal on reaction with dilute \[NaOH\] forms
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}CHO\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}CH(OH)C{{H}_{2}}C{{H}_{2}}CHO\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CH(OH)C{{H}_{2}}CHO\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CH(OH)CH{{(CH)}_{3}}CHO\] done
clear
View Solution play_arrow
-
question_answer60)
Which is not true about acetophenone?
A)
It reacts with 2, 4-dinitrophenylhydrazine to form 2,4-dinitrophenylhydrazone done
clear
B)
It reacts with Tollen 's reagent to form silver mirror done
clear
C)
It reacts with \[{{I}_{2}}/NaOH\] to form iodoform done
clear
D)
On oxidation with alkaline \[KMn{{O}_{4}}\] followed by hydrolysis it gives benzoic acid done
clear
View Solution play_arrow
-
question_answer61)
The order of decreasing ease of reaction with ammonia is
A)
anhydrides, esters, ethers done
clear
B)
anhydrides, ethers, esters done
clear
C)
ethers, anhydrides, esters done
clear
D)
esters, ethers, anhydrides done
clear
View Solution play_arrow
-
question_answer62)
\[C{{H}_{3}}C{{H}_{2}}COOH\xrightarrow[\operatorname{Red}\,P]{B{{r}_{2}}}X\xrightarrow{N{{H}_{3}}(alc.)}Y\] Y in the above reactions is
A)
lactic acid done
clear
B)
ethyiamine done
clear
C)
propylamine done
clear
D)
alanine done
clear
View Solution play_arrow
-
question_answer63)
In the mechanism of Hoffinann reaction, which intermediate rearranges to alkyl isocyanate?
A)
Bromamide done
clear
B)
Nitrene done
clear
C)
Nitroso done
clear
D)
Amide done
clear
View Solution play_arrow
-
question_answer64)
A colourless liquid, at room temperature, reacts with soda-lime to form sodium salt of a carboxylic acid and ammonia gas. The liquid is
A)
propanoic acid done
clear
B)
formamide done
clear
C)
propanamide done
clear
D)
methyl enthanoate done
clear
View Solution play_arrow
-
question_answer65)
Which of the following on oxidation followed by hydrolysis gives pyruvic acid?
A)
Acetaldehyde cyanohydrin done
clear
B)
Acetone cyanohydrin done
clear
C)
Formaldehyde cyanohydrin done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer66)
Identify the correct order of boiling points of the following compounds:
| \[\underset{1}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH}}\,\] |
| \[\underset{2}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHO}}\,\] |
| \[\underset{3}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COOH}}\,\] |
A)
1>2>3 done
clear
B)
3>1>2 done
clear
C)
1>3>2 done
clear
D)
3>2>1 done
clear
View Solution play_arrow
-
question_answer67)
Carboxylic acids are more acidic than phenol and alcohol because of
A)
intermolecular hydrogen bonding done
clear
B)
formation of dimers done
clear
C)
highly acidic hydrogen done
clear
D)
resonance stabilization of their conjugate base done
clear
View Solution play_arrow
-
question_answer68)
Which of the following compounds will react with \[NaHC{{O}_{3}}\] solution to give sodium salt and carbon dioxide?
A)
Acetic acid done
clear
B)
n-Hexanol done
clear
C)
Phenol done
clear
D)
Both [b] and [c] done
clear
View Solution play_arrow
-
question_answer69)
Which of the following acids has the smallest dissociation constant?
A)
\[C{{H}_{3}}CH\left( F \right)COOH\] done
clear
B)
\[FC{{H}_{2}}C{{H}_{2}}COOH\] done
clear
C)
\[BrC{{H}_{2}}C{{H}_{2}}COOH\] done
clear
D)
\[C{{H}_{3}}CH(Br)COOH\] done
clear
View Solution play_arrow
-
question_answer70)
Which of the following orders of relative strengths of acids is correct?
A)
\[FC{{H}_{2}}COOH>ClC{{H}_{2}}COOH>BrC{{H}_{2}}COOH\] done
clear
B)
\[ClC{{H}_{2}}COOH>BrC{{H}_{2}}COOH>FC{{H}_{2}}COOH\] done
clear
C)
\[BrC{{H}_{2}}COOH>ClC{{H}_{2}}COOH>FC{{H}_{2}}COOH\] done
clear
D)
\[ClC{{H}_{2}}C{{O}_{2}}H>FC{{H}_{2}}COOH>BrC{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer71)
Acetic acid on heating in presence of \[{{P}_{2}}{{O}_{5}}\] gives
A)
acetic anhydride done
clear
B)
acetylene done
clear
C)
peracid done
clear
D)
no reaction done
clear
View Solution play_arrow
-
question_answer72)
Among the given compounds, the most susceptible to nucleophilic attack at the carbonyl group is
A)
\[MeCOCl\] done
clear
B)
\[MeCHO\] done
clear
C)
\[MeCOOMe\] done
clear
D)
\[MeCOOCOMe\] done
clear
View Solution play_arrow
-
question_answer73)
Ethyl acetate is obtained when methylmagnesium iodide reacts with
A)
ethyl formate done
clear
B)
ethyl chloroformate done
clear
C)
acetyl chloride done
clear
D)
carbon dioxide done
clear
View Solution play_arrow
-
question_answer74)
An ester is boiled with KOH. The product is cooled and acidified with concentrated\[HCl\]. A white crystalline acid separates. The ester is
A)
methyl acetate done
clear
B)
ethyl acetate done
clear
C)
ethyl formate done
clear
D)
ethyl benzoate done
clear
View Solution play_arrow
-
question_answer75)
Hydrolsis of an ester gives a carboxylic acid which on Kolbe's electrolysis yields ethane. The ester is
A)
ethyl methanoate done
clear
B)
methyl ethanoate done
clear
C)
methyl methanoate done
clear
D)
methyl propanoate done
clear
View Solution play_arrow
-
question_answer76)
What is the missing reagent in the synthesis shown below
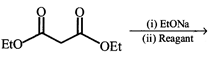
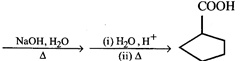
A)
bromocyclopentane done
clear
B)
1,5- dibromo pentane done
clear
C)
1,4-dibromo butane done
clear
D)
1, 1- dibromo cyclopentane done
clear
View Solution play_arrow
-
question_answer77)
The correct product of the following sequence of reactions \[{{(C{{H}_{3}})}_{2}}CHCOOH\text{ }\xrightarrow[(ii)\,{{H}_{2}}O]{(i)\,LiAl{{H}_{4}}}\,\,\,\xrightarrow{PB{{r}_{3}}}\] \[\xrightarrow[DMSO]{KCN}\,\,\,\,\xrightarrow[\Delta ]{{{H}_{2}}{{O}^{+}},\,{{H}^{+}}}\]
A)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHCHBr.COOH\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}CHC{{H}_{2}}COOH\] done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHC{{H}_{2}}C{{H}_{2}}N{{H}_{2}}\] done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{2}}C=CHCOOH\] done
clear
View Solution play_arrow
-
question_answer78)
Carboxylic acid group does not give the usual addition and elimination reactions of aldehydes and ketones because
A)
B)
carboxylate ion gets ionized done
clear
C)
carboxylate ion gets stabilised by resonance done
clear
D)
it exists as \[-COOH\] and there is no carbonyl group done
clear
View Solution play_arrow
-
question_answer79)
Which one of the following orders of acid strength is correct?
A)
\[RCOOH>HOH>ROH>HC\equiv CH\] done
clear
B)
\[RCOOH>HOH>HC\equiv CH>ROH\] done
clear
C)
\[RCOOH>HC\equiv CH>HOH>ROH\] done
clear
D)
\[RCOOH>ROH>HOH>HC\equiv CH\] done
clear
View Solution play_arrow
-
question_answer80)
Which of the following is incorrect?
A)
\[FeC{{l}_{3}}\] is used in detection of phenols. done
clear
B)
Fehling solution is used in detection of glucose. done
clear
C)
Tollen's reagent is used in detection of unsaturation. done
clear
D)
\[NaHS{{O}_{3}}\] is used in detection of carbonyl compounds. done
clear
View Solution play_arrow
-
question_answer81)
\[{{(C{{H}_{3}})}_{2}}CO\xrightarrow[(HCl)]{NaCN}A\xrightarrow[\Delta ]{{{H}_{3}}{{O}^{+}}}B\] In the above sequence of reactions A and B are
A)
\[{{(C{{H}_{3}})}_{2}}C(OH)CN,{{(C{{H}_{3}})}_{2}}C(OH)COOH\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C(OH)CN,{{(C{{H}_{3}})}_{2}}C{{(OH)}_{2}}\] done
clear
C)
\[{{(C{{H}_{3}})}_{2}}C(OH)CN,{{(C{{H}_{3}})}_{2}}CHCOOH\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}C(OH)CN,{{(C{{H}_{3}})}_{2}}C=O\] done
clear
View Solution play_arrow
-
question_answer82)
Acetophenone when reacted with a base, \[{{C}_{2}}{{H}_{5}}ONa,\] yields a stable compound which has the structure.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer83)
In Cannizzaro reaction given below \[2PhCHO\xrightarrow{:O{{H}^{-}}}PhC{{H}_{2}}OH+PhC\overset{..}{\mathop{O}}\,_{2}^{-}\] the slowest step is:
A)
the transfer of proton to the carbonyl group done
clear
B)
the abstraction of proton from the carboxylic group done
clear
C)
the deprotonation of \[PhC{{H}_{2}}OH\] done
clear
D)
the attack of: \[\overline{O}H\] at the carboxyl group done
clear
View Solution play_arrow
-
question_answer84)
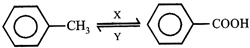
In the above sequence of reaction X and Y are respectively
A)
\[{{H}_{2}}/Pt;B{{r}_{2}}\] done
clear
B)
\[KMn{{O}_{4}};{{H}_{2}}/Pt\] done
clear
C)
\[KMn{{O}_{4}}\left( aq \right);HI/P\] done
clear
D)
\[N{{H}_{2}}-N{{H}_{2}}/KOH,HI/P\] done
clear
View Solution play_arrow
-
question_answer85)
Consider the following transformations: \[C{{H}_{3}}COOH\xrightarrow{CaC{{O}_{3}}}A\xrightarrow{heat}B\xrightarrow[NaOH]{{{I}_{2}}}C\] The molecular formula of C is
A)
\[C{{H}_{3}}-\underset{I}{\overset{OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-C{{H}_{3}}\] done
clear
B)
\[IC{{H}_{2}}-COC{{H}_{3}}\] done
clear
C)
\[CH{{I}_{3}}\] done
clear
D)
\[C{{H}_{3}}I\] done
clear
View Solution play_arrow
-
question_answer86)
Silver benzoate will react with bromine in \[CC{{l}_{4}}\] to form:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer87)
The compound that undergoes decarboxylation most readily under mild condition is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer88)
Which of the following reaction can produce \[R-CO-Ar?\]
A)
\[ArCOCl+H-Ar\xrightarrow{AlC{{l}_{3}}}\] done
clear
B)
\[RCOCl+ArMgX\xrightarrow{{}}\] done
clear
C)
\[ArCOCl+RMgX\xrightarrow{\text{ }}\] done
clear
D)
\[RCOCl+H-Ar\xrightarrow{Alc{{l}_{3}}}\] done
clear
View Solution play_arrow
-
question_answer89)
The end product (C) in the following sequence of reactions is \[HC\equiv CH\xrightarrow[20(percent){{H}_{2}}S{{O}_{4}}]{1(percent)HgS{{O}_{4}}}A\xrightarrow[{{H}_{2}}O]{C{{H}_{3}}MgX}B\xrightarrow{(O)}(C)\]
A)
acetic acid done
clear
B)
isopropyi alcohol done
clear
C)
acetone done
clear
D)
ethanol done
clear
View Solution play_arrow
-
question_answer90)
Predict the product 'B' in the sequence of reactions \[HC\equiv CH\xrightarrow[(percent) HgS{{O}_{4}}]{30(percent){{H}_{2}}S{{O}_{4}}}A\xrightarrow[{}]{NaOH}B\]
A)
\[C{{H}_{3}}COONa\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}CHO\] done
clear
D)
\[C{{H}_{3}}-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-CHO\] done
clear
View Solution play_arrow
-
question_answer91)
A substance \[{{C}_{4}}{{H}_{10}}O\] yields on oxidation a compound, \[{{C}_{4}}{{H}_{8}}O\] which gives an oxime and a positive iodoform test. The original substance on treatment with cone. \[{{H}_{2}}S{{O}_{4}}\] gives \[{{C}_{2}}{{H}_{8}}\] The structure of the compound is
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}CHOHC{{H}_{2}}C{{H}_{3}}\] done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{3}}COH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}-O-C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer92)
Which of following compound is hemiacetal?
A)
B)
C)
D)
All of these done
clear
View Solution play_arrow
-
question_answer93)
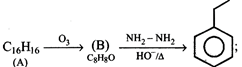
reactant [A] in this reaction is:
A)
B)
C)
D)
both [b] and [c] done
clear
View Solution play_arrow
-
question_answer94)
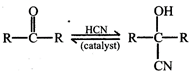
Which of following can be used as a catalyst in the above reaction?
A)
\[C{{l}^{-}}\] done
clear
B)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-{{O}^{-}}\] done
clear
C)
\[Et{{O}^{-}}\] done
clear
D)
\[HSO_{4}^{-}\] done
clear
View Solution play_arrow
-
question_answer95)
Which of the following reagent(s) used for the conversion?
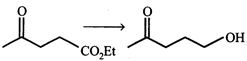
A)
\[glycol/LiAl{{H}_{4}}/{{H}_{3}}{{O}^{+}}\] done
clear
B)
\[glycol/NaH/{{H}_{3}}{{O}^{+}}\] done
clear
C)
\[LiAl{{H}_{4}}\] done
clear
D)
\[NaB{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer96)
Among the following the order of reactivity towards nucleophilic addition is
A)
\[C{{H}_{3}}CHO>C{{H}_{3}}COC{{H}_{3}}>HCHO\] done
clear
B)
\[HCHO>C{{H}_{3}}CHO>C{{H}_{3}}COC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}CHO>HCHO>C{{H}_{3}}COC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}>C{{H}_{3}}CHO>HCHO\] done
clear
View Solution play_arrow
-
question_answer97)
The reaction, \[C{{H}_{3}}CHO\xrightarrow{Zn(Hg)/Conc.HCl[H]}C{{H}_{3}}C{{H}_{3}}\] is
A)
Cannizaro's reaction done
clear
B)
Rosenmund reduction done
clear
C)
Wolf-Kishner reduction done
clear
D)
Clemmenson reduction done
clear
View Solution play_arrow
-
question_answer98)
Tollen's reagent and Fehling solutions are used to distinguish between
A)
acids and alcohols done
clear
B)
alkanes and alcohols done
clear
C)
ketones and aldehydes done
clear
D)
n-alkanes and branched alkanes done
clear
View Solution play_arrow
-
question_answer99)
In the presence of a small amount of phosphorous, aliphatic carboxylic acids react with chlorine or bromine to yield a compound in which \[\alpha \]-hydrogen has been replaced by halogen. This reaction is known as:
A)
Wolff- Kishner reaction done
clear
B)
Rosenmund reaction done
clear
C)
Etard reaction done
clear
D)
Hell - Volhard - Zelinsky reaction done
clear
View Solution play_arrow
-
question_answer100)
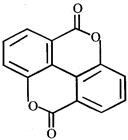
Phthalic acid reacts with resorcinol in the presence of concentrated \[{{H}_{2}}S{{O}_{4}}\] to give:
A)
Phenolphthalein done
clear
B)
Alizarin done
clear
C)
Coumarin done
clear
D)
Fluorescein done
clear
View Solution play_arrow
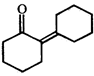
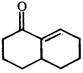
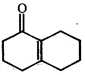
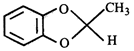
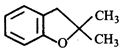
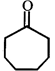
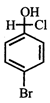
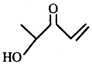
 Product [B] is
Product [B] is 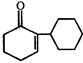 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
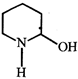
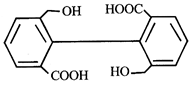
![]() is known as
is known as 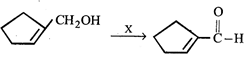
![]()
 Product [B] in this reaction is:
Product [B] in this reaction is:  done
clear
done
clear
 done
clear
done
clear
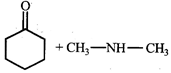 done
clear
done
clear
![]() \[\xrightarrow{LAH}\left( C \right);\] product (C) of the reaction is:
\[\xrightarrow{LAH}\left( C \right);\] product (C) of the reaction is: 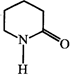 done
clear
done
clear
 done
clear
done
clear
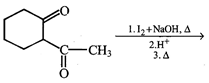
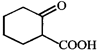 done
clear
done
clear
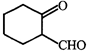 done
clear
done
clear



 X, most likely the compound X is
X, most likely the compound X is 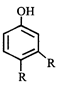 done
clear
done
clear
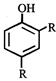 done
clear
done
clear
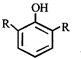 done
clear
done
clear


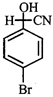 done
clear
done
clear
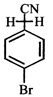 done
clear
done
clear
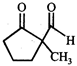
 done
clear
done
clear
![]() [B]
[B]![]() [C]
[C] ![]()

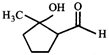
 done
clear
done
clear
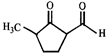
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 [A]; Product [A] of the reaction is:
[A]; Product [A] of the reaction is: 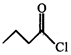 done
clear
done
clear
 done
clear
done
clear
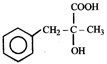
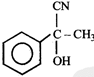
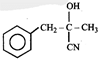
Product. The product formed in the reaction is-
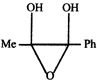 done
clear
done
clear
 Product (X) of the reaction is-
Product (X) of the reaction is- 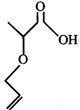 done
clear
done
clear
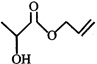 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
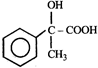 done
clear
done
clear
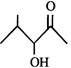 done
clear
done
clear
 done
clear
done
clear
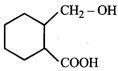 done
clear
done
clear
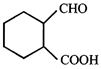 done
clear
done
clear
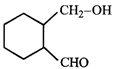 done
clear
done
clear
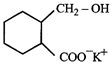 done
clear
done
clear

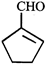 done
clear
done
clear
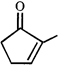 done
clear
done
clear

![]()
 The major product in the above reaction is
The major product in the above reaction is 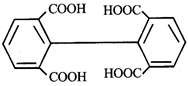 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear


 done
clear
done
clear
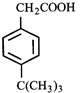 done
clear
done
clear
 done
clear
done
clear
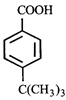 done
clear
done
clear
 done
clear
done
clear
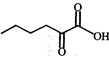 done
clear
done
clear

 done
clear
done
clear
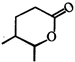 done
clear
done
clear
 done
clear
done
clear
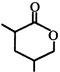 done
clear
done
clear

 In the above reaction, product is:
In the above reaction, product is:
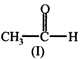
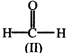
![]()

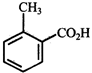 done
clear
done
clear
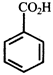 done
clear
done
clear
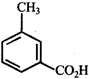 done
clear
done
clear
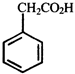 done
clear
done
clear

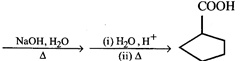
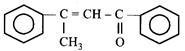 done
clear
done
clear
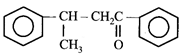 done
clear
done
clear
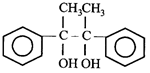 done
clear
done
clear
![]() In the above sequence of reaction X and Y are respectively
In the above sequence of reaction X and Y are respectively 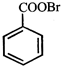 done
clear
done
clear
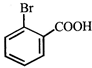 done
clear
done
clear
 done
clear
done
clear
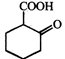 done
clear
done
clear
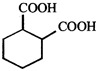 done
clear
done
clear
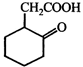 done
clear
done
clear
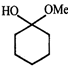 done
clear
done
clear
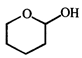 done
clear
done
clear
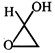 done
clear
done
clear
 reactant [A] in this reaction is:
reactant [A] in this reaction is:  done
clear
done
clear
 Which of following can be used as a catalyst in the above reaction?
Which of following can be used as a catalyst in the above reaction?