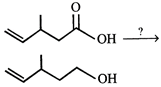
question_answer 1)
Which one of the following statements is not correct?
A)
done
clear
B)
done
clear
C)
done
clear
D)
View Solution play_arrow
question_answer 2)
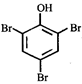
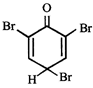
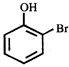
What is the structure of the major product when phenol is treated with bromine water?
A)
B)
C)
D)
View Solution play_arrow
question_answer 3)
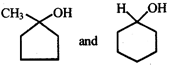
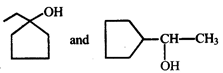
In the given pair of alcohol, in which pair second alcohol is more reactive than first towards hydrogen bromide?
A)
B)
C)
done
clear
D)
done
clear
View Solution play_arrow
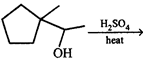
question_answer 4)
\[\xrightarrow{\begin{smallmatrix} (1)\,MeMgBr \\ (2)\,{{H}_{3}}{{O}^{+}} \end{smallmatrix}}(C)\xrightarrow{NaB{{H}_{4}}.EtOH}(D)\] Product [D] in above reaction is:
A)
B)
C)
D)
View Solution play_arrow
question_answer 5)
Which of the following reagents would carry out the following transformation? \[(D{{=}^{2}}H)\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
In the Victor-Meyer's test, the colour given by \[1{}^\circ ,2{}^\circ \] and \[3{}^\circ \] alcohols are respectively:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 7)
Rate of dehydration of alcohols follows the order:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
In Williamson synthesis of mixed ether having a primary and a tertiary alkyl group if tertiary halide is used, then:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
An ether [A], \[{{C}_{5}}{{H}_{12}}O\], when heated with excess of hot concentrated HI produced two alkyl halides which when treated with \[NaOH\] yielded compounds [B] and [C]. Oxidation of [B] and [C] gave a propanone and an ethanoic acid respectively. The IUPAC name of the ether [A] is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
Allyl phenyl ether can be prepared by heating:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
Amongst the following alcohols which would react fastest with cone. \[HCl\] and \[ZnC{{l}_{2}}\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
Reagent used to convert allyl alcohol to acrolein is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
Which are not cleaved by \[HI{{O}_{4}}\]?
I. glycerol
II. glycol
III. 1, 3 propenediol
IV. methoxy-2-propanol
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 14)
The reaction of phenol with benzoyl chloride to give phenyl benzoate is known as:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
Product [B] of the above reaction is:
A)
B)
C)
D)
View Solution play_arrow
question_answer 16)
Which one of the following compounds will not be soluble in sodium bicarbonate?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 17)
Which one of the following substituents at para- position is most effective in stabilizing the Product of the reaction is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
Product of the reaction is:
A)
B)
C)
D)
View Solution play_arrow
question_answer 19)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 20)
Williamson synthesis of ether is an example of:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 21)
Phenol on heating with \[CHC{{l}_{3}}\] and \[NaOH\] gives salicylaldehyde. The reaction is called:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 22)
In which of the following reactions hydrogen gas will not be evolved?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 23)
Which is the best reagent to convert isopropyl alcohol to isopropyl bromide? \[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-OH\xrightarrow{?}C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-Br\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 24)
How many isomers of \[{{C}_{5}}{{H}_{11}}OH\] will be primary alcohols?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 25)
On reaction with sodium, 1 mol of a compound X gives 1 mol of \[{{H}_{2}}\] Which one of the following compounds might be X?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 26)
Which of the following reactions would convert 2-butanol into deuterated compound \[C{{H}_{3}}-C{{H}_{2}}-\underset{D}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\] ?
A)
B)
C)
D)
View Solution play_arrow
question_answer 27)
To prepare 3-ethylpentan-3-ol, the reagents needed are:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 28)
What is X in the following reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 29)
What is the major product 6f the following reaction? \[C{{H}_{3}}-\overset{OH}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-C{{H}_{2}}-OH\] \[\xrightarrow[\operatorname{Pyridine}\,cold]{Cr{{O}_{3}}}\Pr oduct\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 30)
A compound of the formula \[{{C}_{4}}{{H}_{10}}O\] reacts with sodium and undergoes oxidation to give a carbonyl compound which does not reduce Tollen's reagent, the original compound is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 31)
What amount of bromine will be required to convert 2g of phenol into 2,4,6-tribromophenol?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 32)
Identify X in the following sequence: \[\underset{(X)}{\mathop{{{C}_{3}}{{H}_{8}}O}}\,\xrightarrow[{{H}_{2}}S{{O}_{4}}]{{{K}_{2}}C{{r}_{2}}{{O}_{7}}}{{C}_{3}}{{H}_{6}}O\xrightarrow[{{I}_{2}}+NaOH]{warn}CH{{I}_{3}}\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 33)
tert-Butyl ethyl ether can't be prepared by which reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 34)
Succinic acid \[\xrightarrow{\Delta }(A)\xrightarrow[\Delta ]{N{{H}_{3}}}(B)\xrightarrow[KOH]{B{{r}_{2}}}(C);\] Product [C] will be:
A)
B)
C)
D)
View Solution play_arrow
question_answer 35)
Find missing reagents.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 36)
Diethyl ether reacts, inspite of its usual inert nature, with:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 37)
Give the best conditions for this transformation:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 38)
The order of reactivity of the following alcohols towards cone. \[HCl\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 39)
The end product of the following sequence of reactions
A)
B)
C)
D)
View Solution play_arrow
question_answer 40)
An optically active alcohol of formula \[{{C}_{9}}{{H}_{12}}{{O}_{2}}\] produced the following compound when refluxed with \[KMn{{O}_{4}}\]
The original compound showed these properties also:
What is structure of (A) ?
A)
B)
C)
D)
done
clear
View Solution play_arrow
question_answer 41)
The product of the following reaction,
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 42)
An aromatic ether is not cleaved by HI even at 525 K. The compound is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 43)
Which of the following reactions will not result in the formation of anisole?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 44)
Which of the following is not true in case of reaction with heated copper at \[300{}^\circ C\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 45)
Which of the following synthesis gives 3 -methyl. 1- hexanol?
A)
done
clear
B)
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 46)
What is the product of the following sequence of reactions? \[{{\left( C{{H}_{3}} \right)}_{2}}C=CH.C{{H}_{2}}C{{H}_{3}}~\xrightarrow[(ii)\,{{H}_{2}}{{O}_{2}},O{{H}^{-}}]{\left( i \right)B{{H}_{3}}/THF}\] \[\xrightarrow[C{{H}_{2}}C{{l}_{2}}]{PCC}\,\,\xrightarrow[(ii)\,{{H}_{3}}{{O}^{+}}]{(i)\,C{{H}_{3}}MgBr}\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 47)
Which of the following are intermediates in the reaction of excess of \[C{{H}_{3}}MgBr\] with \[{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}\] to make 2-phenyl - 2-propanol?
A. \[{{C}_{6}}{{H}_{5}}-\underset{C{{H}_{3}}}{\overset{OMgBr}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-OC{{H}_{2}}C{{H}_{3}}\]
B. \[{{C}_{6}}{{H}_{5}}-\underset{{}}{\overset{O}{\mathop{\underset{{}}{\overset{||}{\mathop{C}}}\,}}}\,-C{{H}_{3}}\]
C. \[{{C}_{6}}{{H}_{5}}-\underset{C{{H}_{3}}}{\overset{OMgBr}{\mathop{\underset{|}{\overset{||}{\mathop{C}}}\,}}}\,-C{{H}_{3}}\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 48)
Which of the following compound can react with hydroxylamine?
A)
B)
C)
D)
View Solution play_arrow
question_answer 49)
Acetic anhydride reacts with diethyl ether in the presence of anhydrous \[AlC{{l}_{3}}\] to give
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 50)
In the following reaction. \[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}+4H\xrightarrow{Red\,P+HI}2X+{{H}_{2}}O;\] X is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 51)
n-propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 52)
An organic compound A reacts with methyl magnesium iodide to form an addition product which on hydrolysis forms the compound B. Compound B gives blue colour salt in Victor Meyer's test. The compounds A and B are respectively
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 53)
Identify the major product,
A)
B)
C)
D)
View Solution play_arrow
question_answer 54)
When Grignard reagent reacts with [B] product [D] will obtained. Reactant [A] of the above reaction is:
A)
B)
C)
D)
View Solution play_arrow
question_answer 55)
Diols (I-IV) which react with \[Cr{{O}_{3}}\] in aqueous \[{{H}_{2}}S{{O}_{4}}\] and yield products that readily under go dercarboxylation on heating, are:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 56)
\[C{{H}_{3}}\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-CH=C{{H}_{2}}\] \[\xrightarrow[(ii)\,{{H}_{2}}{{O}_{2}}/O{{H}^{-}}]{(i)\,{{B}_{2}}{{H}_{6}}}X\xrightarrow[140{}^\circ C]{{{H}_{2}}S{{O}_{4}}}Y.\] What is Y?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 57)
Identify Z in the sequence: \[C{{H}_{3}}C{{H}_{2}}CH=C{{H}_{2}}\xrightarrow{HBr/{{H}_{2}}{{O}_{2}}}\] \[Y\xrightarrow{{{C}_{2}}{{H}_{5}}ONa}Z\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 58)
\[C{{H}_{3}}-CH=C{{H}_{2}}\xrightarrow[(ii)\,NaB{{H}_{4}}]{(i)\,Hg{{(OAc)}_{2}}/{{H}_{2}}O}X+Na\] \[\xrightarrow{{}}Y+C{{H}_{3}}Cl\xrightarrow{{}}Z+HI\xrightarrow{0{}^\circ C}A+B\] What are A and B?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 59)
\[{{H}_{2}}C=CH-C{{H}_{2}}-C{{H}_{2}}-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\] \[\xrightarrow[Pyridine]{SOC{{l}_{2}}}(A)\xrightarrow[({{H}_{2}}O)]{{{O}_{3}}/Zn}\underset{{{C}_{5}}{{H}_{9}}ClO}{\mathop{(B)}}\,\xrightarrow{NaB{{H}_{4}}}(C)\] Compound (C) is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 60)
\[\underset{\left( {{C}_{5}}{{H}_{10}}O \right)}{\mathop{\left( X \right)}}\,~\xrightarrow{{{H}_{3}}{{O}^{+}}}Y+Z\] (Y and Z both give the lodoform test). The compound X is -
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 61)
What is the major product of the following reaction?
A)
B)
C)
D)
View Solution play_arrow
question_answer 62)
Dehydration of alcohols by conc. \[{{H}_{2}}S{{O}_{4}}\] takes place according to following steps:
The slowest and fastest steps in the above reaction are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 63)
In order to get 2-hydroxybenzaldehyde from phenol, which of the following reagents is required?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 64)
The structure of the product formed in the reaction given below is:
A)
B)
C)
D)
View Solution play_arrow
question_answer 65)
The most probable product in the reaction given below is
A)
B)
C)
D)
View Solution play_arrow
question_answer 66)
Which of the following alcohols is dehydrated most readily with cone.\[{{H}_{2}}S{{O}_{4}}\]?
A)
B)
C)
D)
View Solution play_arrow
question_answer 67)
Identify the nature of product in the following reaction
A)
B)
C)
D)
View Solution play_arrow
question_answer 68)
Which one of the following compounds has the most acidic nature?
A)
B)
C)
D)
View Solution play_arrow
question_answer 69)
The major product P in the following reaction is \[{{\left( C{{H}_{3}} \right)}_{3}}COH+{{C}_{2}}{{H}_{5}}OH\xrightarrow{H+}P\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 70)
Correct increasing order of acidity is as follows
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 71)
Which one/ones of the following reactions will yield 2-propanol?
I. \[C{{H}_{2}}=CH-C{{H}_{3}}+{{H}_{2}}O\xrightarrow{H+}\] II. \[C{{H}_{3}}-CHO\xrightarrow[(ii)\,{{H}_{2}}O]{(i)\,C{{H}_{3}}MgI}\] III. \[C{{H}_{2}}O\xrightarrow[(ii)\,{{H}_{2}}O]{(i)\,{{C}_{2}}{{H}_{5}}MgI}\] IV. \[C{{H}_{2}}=CH-C{{H}_{3}}\xrightarrow{Neutral\,KMn{{O}_{4}}}\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 72)
What is Z in the following sequence of reactions? \[Z\xrightarrow{PC{{l}_{5}}}X\xrightarrow{~Alc.KOH}~Y~\xrightarrow[\left( ii \right){{H}_{2}}O;\text{ }boil]{\left( i \right)Conc.{{H}_{2}}S{{O}_{4}}~}Z\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 73)
Which of the following has lowest boiling point?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 74)
Arrange the following in increasing order of their acidity? [a] o-cresol, [b] salicylic acid, [c] phenol
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 75)
Which of the following diols would cleave into two fragments with \[HI{{O}_{4}}\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 76)
In the reaction: \[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-O-C{{H}_{2}}-C{{H}_{3}}+HI\xrightarrow{Heated}\]Which of the following compounds will be formed?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 77)
The following change can be carried out with
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 78)
Product is
A)
B)
C)
done
clear
D)
View Solution play_arrow
question_answer 79)
Ethanol can be prepared more easily by which reaction?
(i) \[C{{H}_{3}}C{{H}_{2}}Br+{{H}_{2}}O\xrightarrow{{}}C{{H}_{3}}C{{H}_{2}}OH\]
(ii) \[C{{H}_{3}}C{{H}_{2}}Br+A{{g}_{2}}O\left( in\text{ }boiling\text{ }water \right)\xrightarrow{{}}\] \[C{{H}_{3}}C{{H}_{2}}OH\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 80)
3 moles of ethanol react with one mole of phosphorus tribromide to form 3 moles of bromoethane and one mole of X. Which of the following is X?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
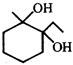 done
clear
done
clear
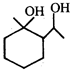 done
clear
done
clear
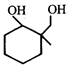
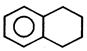
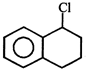
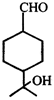
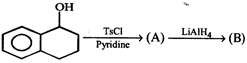 Product [B] of the above reaction is:
Product [B] of the above reaction is: 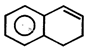 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
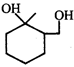
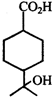
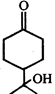
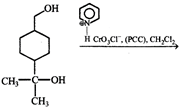 Product of the reaction is:
Product of the reaction is: 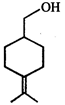 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
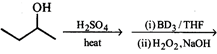 done
clear
done
clear
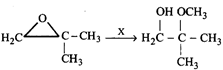
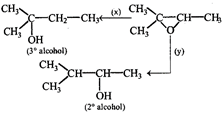 Find missing reagents.
Find missing reagents. 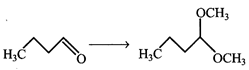
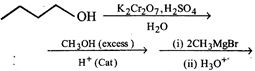
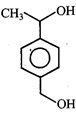 done
clear
done
clear
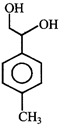 done
clear
done
clear
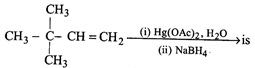
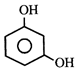 done
clear
done
clear
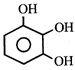 done
clear
done
clear
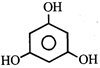 done
clear
done
clear
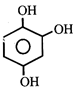 done
clear
done
clear
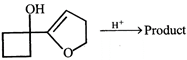
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
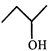 done
clear
done
clear
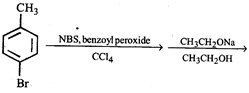
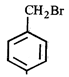
 done
clear
done
clear
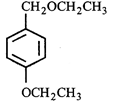 done
clear
done
clear
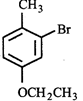 done
clear
done
clear
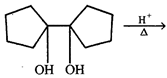
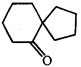 done
clear
done
clear
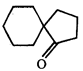 done
clear
done
clear
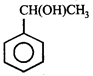 done
clear
done
clear
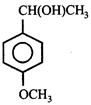 done
clear
done
clear
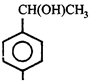
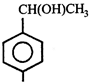
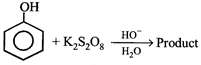
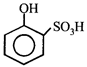 done
clear
done
clear
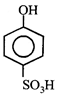 done
clear
done
clear
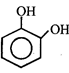 done
clear
done
clear
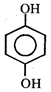 done
clear
done
clear
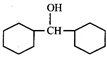 done
clear
done
clear