A) It gives mixture of \[{{F}_{2}},\] \[C{{l}_{2}}\] and \[B{{r}_{2}}\]
B) It gives chlorine
C) It gives bromine
D) None of these
Correct Answer: D
Solution :
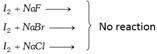 Because \[{{I}_{2}}\] is least electronegative among halogens.
Because \[{{I}_{2}}\] is least electronegative among halogens.
You need to login to perform this action.
You will be redirected in
3 sec
