-
question_answer1)
There are three isotopes of carbon which are named as C-12, C-13 and C-14 out of which C-12 is the most abundant isotope. In the given structures of three isotopes, what will be the composition of the nucleus?
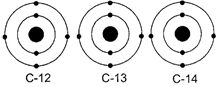
A)
C -12 : 6p + 6n, C - 13 : 7p + 6n, C - 14 : 8p + 6n done
clear
B)
C - 12 : 6p + 6n, C -13 : 6p + 7n, C - 14 : 6p + 8n done
clear
C)
C - 12 : 6p + 6n, C - 13 : 5p + 8n, C - 14 : 7p + 7n done
clear
D)
C - 12 : 6p + 6n, C - 13 : 12p +1n, C - 14 : 5p + 9n done
clear
View Solution play_arrow
-
question_answer2)
A thin sheet of gold foil is bombarded with \[\alpha \text{-particles}\]as in Rutherford's experiment. Which of the given descriptions most accurately represents the path of\[\alpha \text{-particles}\]?
| | Undeflected | No. of particles deflected through a small angle | No. of Particles deflected through a large angle |
| I. | All | None | None |
| II. | Most | A few | None |
| III. | Most | A few | A few |
| IV. | A few | Most | A few |
A)
I done
clear
B)
II done
clear
C)
III done
clear
D)
IV done
clear
View Solution play_arrow
-
question_answer3)
Three invisible radiations X, Y and Z are passed through an electric field. X goes straight, Y deviates towards the positive end while Z deviates towards the negative end. The particles present in X, Y and Z and the elm ratio for X are e/m ratio for X are
A)
B)
C)
D)
View Solution play_arrow
-
question_answer4)
Identify the \[A{{l}^{3+}}ion\] from the given structures.
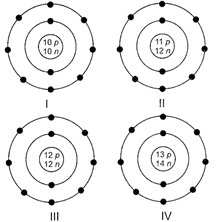
A)
I done
clear
B)
II done
clear
C)
III done
clear
D)
IV done
clear
View Solution play_arrow
-
question_answer5)
There are two atomic species X and Y, such that
| Atomic species | X | Y |
| Protons | 8 | 8 |
| Neutrons | 8 | 10 |
Which of the following statements is true about X and Y?
A)
X and Y are isobars. done
clear
B)
X and Y have different chemical properties. done
clear
C)
X and Y have different physical properties. done
clear
D)
All are correct. done
clear
View Solution play_arrow
-
question_answer6)
Match column I with column II and select the correct option from the codes given below.
| Column I | Column II |
| (a) Ions | (i) Same mass number, different atomic numbers |
| (b) Isobars | (ii) Same number of neutrons, different atomic numbers, different mass numbers |
| (c) Isotopes | (iii) Formed by loss or gain of electrons |
| (d) Isotones | (iv) Combining capacity of an atom |
| (e) Valency | (v) Same atomic number, different mass numbers. |
A)
(a)-(i), (b)-(iii), (c)-(v), (d)-(ii), (e)-(iv) done
clear
B)
(a)-(iii), (b)-(i), (c)-(v), (d)-(ii), (e)-(iv) done
clear
C)
(a)-(iii), (b)-(v), (c)-(i), (d)-(ii), (e)-(iv) done
clear
D)
(a)-(i), (b)-(ii), (c)-(iii), (d)-(iv), (e)-(v) done
clear
View Solution play_arrow
-
question_answer7)
A brief information about two atoms
|
X and Y is given: X: Atomic number = 7, Mass number = 14
|
|
Y: Atomic number = 7, Mass number = 15
|
Which of the following is correct about these two atoms?
A)
Electronic configuration of X is 2, 8, 4 while that of Y is 2, 8, 5. done
clear
B)
Both X and Y contain 7 neutrons. done
clear
C)
X has 2 electron shells while Y has 3 electron shells. done
clear
D)
Both X and Y have 5 valence electrons. done
clear
View Solution play_arrow
-
question_answer8)
|
An electron jumps from an orbit P to Q and loses energy. The same electron when jumps from R to Q, gains energy.
|
|
The increasing order of distance of these orbits from the nucleus is
|
A)
P < Q < R done
clear
B)
R < P < Q done
clear
C)
R < Q < P done
clear
D)
P < R < Q done
clear
View Solution play_arrow
-
question_answer9)
|
Few statements regarding the distribution of electrons in the energy shells are given.
|
|
Which of the following statements is not correct?
|
A)
For fourth energy shell (N), the maximum number of electrons is 32 according to \[2{{n}^{2}}\] rule. done
clear
B)
When N is the outermost shell of an atom, it can accommodate a maximum of 8 electrons only. done
clear
C)
The filling of the fourth shell begins even before the third shell is completely filled. done
clear
D)
Fifth shell can accommodate 50 electrons according to \[2{{n}^{2}}\] rule. done
clear
View Solution play_arrow
-
question_answer10)
Which of the following are isobars?
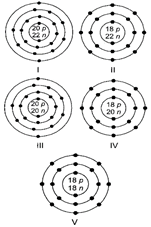
A)
I and II done
clear
B)
II and III done
clear
C)
I, II, IV and V done
clear
D)
II, III, IV and V done
clear
View Solution play_arrow
-
question_answer11)
Read the given statements and mark the correct option.
| Statement 1: Bohr's orbits are called stationary orbits. |
| Statement 2: Electrons remain stationary in these orbits for some time. |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer12)
In nature, the element boron occurs 19.9% as \[^{\text{10}}\text{B}\] and 80.1% as\[^{\text{11}}\text{B}\]. If the isotopic mass of \[^{\text{10}}\text{B}\] is 10.013 amu and that of \[^{11}\text{B}\] is 11.009 amu, the atomic mass of boron is
A)
10 done
clear
B)
11 done
clear
C)
10.8 done
clear
D)
11.8 done
clear
View Solution play_arrow
-
question_answer13)
Which of the following statements are correct regarding the elements given below?
|
\[_{\text{19}}^{\text{39}}\text{K, }_{\text{53}}^{\text{127}}\text{I,}\,\,_{\text{17}}^{\text{35}}\text{Cl,}\,\,_{\text{35}}^{\text{80}}\text{Br,}\,\,_{\text{18}}^{\text{40}}\text{Ar}\]
|
| I. The correct order of increasing proton number is \[\text{KClBrIAr}\] |
| II. The correct order of increasing relative atomic mass is \[\text{ClKArBrI}\] |
| III. There is difference in the orders of proton number and relative atomic mass. |
| IV. The number of protons is equal to number of neutrons in all the given elements |
A)
I and IV only done
clear
B)
II and IV only done
clear
C)
I and III only done
clear
D)
II and III only done
clear
View Solution play_arrow
-
question_answer14)
Hydrogen exists in three isotopic forms, \[_{\text{1}}^{\text{1}}\text{H,}\,\,_{\text{1}}^{\text{2}}\text{H,}\,\,_{\text{1}}^{\text{3}}\text{H}\] respectively known as protium, deuterium and tritium. Why are all the isotopes neutral in nature?
A)
Since neutrons are neutral in nature hence, isotopes are electrically neutral. done
clear
B)
All the isotopes have one electron and one proton, hence they are neutral. done
clear
C)
All the isotopes have one proton and one neutron, hence they are neutral. done
clear
D)
Increasing number of protons in the isotopes make them neutral. done
clear
View Solution play_arrow
-
question_answer15)
Identify the difference between particles of each given pair.
| I. \[^{\text{6}}\text{Li}\] and \[^{7}\text{Li}\] |
| II. \[^{\text{32}}\text{S}\] and \[^{\text{32}}{{\text{S}}^{2-}}\] |
| III. \[^{\text{39}}{{\text{K}}^{\text{+}}}\] and \[^{\text{40}}\text{C}{{\text{a}}^{\text{2+}}}\] |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
A compound of carbon, hydrogen and nitrogen contains these elements in the ratio 9 : 1 : 3.5. If its molecular mass is 108, what is the molecular formula?
A)
\[{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}\text{N}\] done
clear
B)
\[{{\text{C}}_{\text{3}}}{{\text{H}}_{\text{4}}}\text{N}\] done
clear
C)
\[{{\text{C}}_{\text{2}}}\text{H}{{\text{N}}_{\text{2}}}\] done
clear
D)
\[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{8}}}{{\text{N}}_{\text{2}}}\] done
clear
View Solution play_arrow
-
question_answer17)
Which of the given pairs of atoms contain(s) the same number of neutrons?
| I. \[_{\text{48}}^{\text{114}}\text{Cd}\] and \[_{50}^{\text{119}}\text{Sn}\] |
| II. \[_{\text{27}}^{\text{59}}\text{Co}\] and \[_{\text{28}}^{\text{59}}\text{Ni}\] |
| III. \[_{\text{55}}^{\text{133}}\text{Cs}\] and \[_{\text{54}}^{\text{132}}\text{Xe}\] |
| IV. \[_{\text{29}}^{\text{63}}\text{Cu}\] and \[_{\text{29}}^{\text{65}}\text{Cu}\] |
A)
I and IV only done
clear
B)
I and III only done
clear
C)
III only done
clear
D)
IV only done
clear
View Solution play_arrow
-
question_answer18)
Description of a few atoms is given in the table.
| Atom | P | Q | R | S |
| No. of protons | 17 | 11 | 17 | 9 |
| No. of neutrons | 18 | 12 | 20 | 10 |
| No. of electrons | 17 | 10 | 17 | 10 |
Identify a cation, an anion and a pair of isotopes from the given table.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer19)
|
Study the given mass spectrum of magnesium carefully.
|
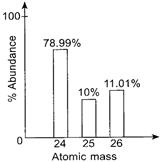
|
|
The number of protons in \[^{\text{26}}\text{Mg}\], number of neutrons in \[^{\text{25}}\text{Mg}\], and the relative atomic mass of Mg are respectively
|
A)
12, 13 and 25 done
clear
B)
16, 12 and 24 done
clear
C)
14, 13 and 25 done
clear
D)
12, 13 and 24.32 done
clear
View Solution play_arrow
-
question_answer20)
|
Electron distribution of two elements P and Q in their outermost shell is shown below.
|
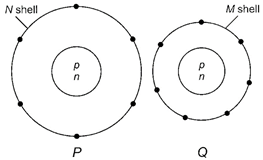
|
|
Atomic numbers of P and Q are respectively
|
A)
9, 17 done
clear
B)
15, 17 done
clear
C)
24, 17 done
clear
D)
17, 24 done
clear
View Solution play_arrow


