![]() write the expression fo1
write the expression fo1 ![]() to indicate its base strength.
to indicate its base strength.
![]()

![]()
![]()
![]() Why?
Why?
![]() order
order
![]() in aqueous solution.
in aqueous solution.
![]() and
and![]() .
.
![]() (ii)
(ii)![]() (iii)
(iii)![]()
![]() (ii)
(ii)![]()
![]()
![]() water to give 2,4,6-tribromoaniline Why?
water to give 2,4,6-tribromoaniline Why?
![]() and cone.
and cone. ![]() at 298 K?
at 298 K?
![]()
![]() and
and![]()
![]() (ii)
(ii)
![]()
![]()
![]() whereas in aqueous solution
whereas in aqueous solution ![]() Why?
Why?
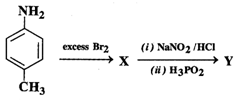
You need to login to perform this action.
You will be redirected in
3 sec
