![]()
![]() and OH?
and OH?
![]() reaction of 1°, 2° and 3° alkyl
halides?
reaction of 1°, 2° and 3° alkyl
halides?
![]() reaction.
(a)
reaction.
(a) ![]() or
or ![]() (b)
(b)
![]() or
or 
![]() reactivity.
reactivity.
![]()
![]() (ii)
(ii)![]() (iii)
(iii)![]()
![]() which lives only one monochlorination
product.
which lives only one monochlorination
product.
![]() reaction?
reaction?
![]() has lowest boiling point?
has lowest boiling point?
 (b)
(b)![]()

![]() in
in ![]()
![]() reaction very fast?
reaction very fast?
![]() elimination?
Explain with example.
elimination?
Explain with example.
![]() and
and ![]() reactions?
reactions?
![]() and
and ![]() reactions?
reactions?
![]() reactions. Explain.
reactions. Explain.
![]() and
and![]() .
.
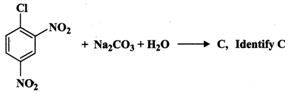 (ii) Predict the product of the reaction.
(ii) Predict the product of the reaction. 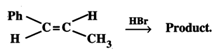
You need to login to perform this action.
You will be redirected in
3 sec
