Answer:
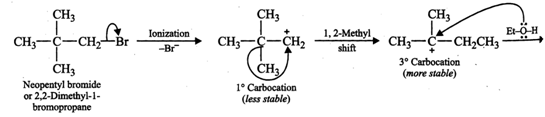
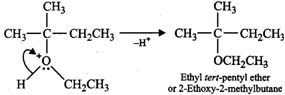
In presence of a weak nucleophile such as ethanol, neopentyl bromide ionizes to form first a \[1{}^\circ \] carbocation which rearranges to form the more stable \[3{}^\circ \]carbocation. This is then attacked by weak nucleophile ethanol followed by loss of a proton to yield ethyl tert-pentyl ether.
You need to login to perform this action.
You will be redirected in
3 sec
