Answer:
(a) \[{{C}_{2}}{{H}_{5}}\underset{\centerdot \,\,\centerdot }{\overset{\centerdot \,\,\centerdot }{\mathop{O}}}\,H+{{H}_{2}}S{{O}_{4}}\,\xrightarrow{{{0}^{\text{o}}}C}\,\underset{\,\,\text{Ethyloxonium}\,\text{hydrogensulphate}}{\mathop{({{C}_{2}}{{H}_{5}}\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\oplus }{\mathop{O}}}\,H)HSO_{4}^{-}}}\,\]
(b)\[{{C}_{2}}{{H}_{5}}OH+{{H}_{2}}S{{O}_{4}}\,\xrightarrow[\text{temperature}]{\text{Room}}\,\underset{\,\,\text{Ethylhydrogensulphate}}{\mathop{{{C}_{2}}{{H}_{5}}HSO_{4}^{{}}+{{H}_{2}}O}}\,\]
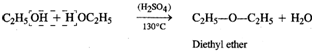
(c)
 (d) \[H-\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-H\xrightarrow[{{180}^{\text{o}}}]{({{H}_{2}}S{{O}_{4}})}\underset{\begin{smallmatrix} \\ \\ \text{Ethylene} \end{smallmatrix}}{\mathop{C{{H}_{2}}=C{{H}_{2}}+{{H}_{2}}O}}\,\]
(d) \[H-\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-H\xrightarrow[{{180}^{\text{o}}}]{({{H}_{2}}S{{O}_{4}})}\underset{\begin{smallmatrix} \\ \\ \text{Ethylene} \end{smallmatrix}}{\mathop{C{{H}_{2}}=C{{H}_{2}}+{{H}_{2}}O}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
