-
question_answer1)
Identify X, Y and Z in the given reaction.
|
\[\underset{\begin{smallmatrix} (Commonly\, \\ used\,in\,ba\operatorname{kery}\,products) \end{smallmatrix}}{\mathop{Salt\,X}}\,\to \underset{\begin{smallmatrix} (Common\, \\ constiuent\,of\, \\ washing\,powder) \end{smallmatrix}}{\mathop{Salt\,}}\,Y+\underset{\begin{smallmatrix} (Truns\,lime \\ water\,milky) \end{smallmatrix}}{\mathop{\,Gas\,Z}}\,\]
|
A)
B)
C)
D)
View Solution play_arrow
-
question_answer2)
Match column I (different substances) with column II (pH values) and choose the correct option from the codes given below.
|
|
Column I
|
|
Column II
|
|
P.
|
Tomato juice
|
1.
|
7.3 - 7.5
|
|
Q.
|
Gastric juice
|
2.
|
6.5 - 7.5
|
|
R.
|
Blood
|
3.
|
4.5 - 5.5
|
|
S.
|
Saliva
|
4.
|
4.0 - 4.4
|
|
T.
|
Coffee
|
5.
|
1.0 - 3.0
|
A)
P-2, Q-1, R-3, S-4, T-5 done
clear
B)
P-1, Q-4, R-3, S-2, T-5 done
clear
C)
P-4, Q-5, R-1, S-2, T-3 done
clear
D)
P-2, Q-3, R-1, S-5, T-4 done
clear
View Solution play_arrow
-
question_answer3)
Certain chemical properties of a substance are given below.
| (i) It turns methyl orange red. |
| (ii) It turns turmeric paper red. |
| (iii) It reacts with zinc and a gas is evolved. |
| (iv) It reacts with solid sodium carbonate to give brisk effervescence. |
Which of the above properties are shown by dilute HCI?
A)
(i) and (ii) only done
clear
B)
(i) and (iii) only done
clear
C)
(i), (iii) and (iv) only done
clear
D)
(iii) and (iv) only done
clear
View Solution play_arrow
-
question_answer4)
|
Sunil set up an experiment as shown in the figure to test a few solutions which contain hydrogen but are not categorized as acids.
|
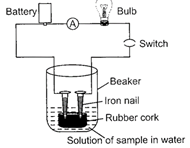
|
Which of the given observations are correct?
| (i) When a solution of glucose is put the bulb does not glow. |
| (ii) When a solution of ethanol is put the bulb does not glow. |
| (iii) When a solution of hydrochloric acid is put the bulb glows. |
| (iv) When a solution of sodium hydroxide is put the bulb does not glow. |
A)
(i), (ii) and (iv) only done
clear
B)
(i), (ii) and (iii) only done
clear
C)
(i) and (ii) only done
clear
D)
(iii) and (iv) only done
clear
View Solution play_arrow
-
question_answer5)
|
Observe the given figure carefully.
|
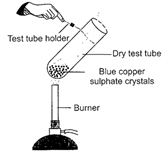
|
Which of the following observations are correct?
| I. A white colored residue is left behind in the test tube. |
| II. Water droplets are observed on the upper cooler part of the test tube. |
| III. On adding water to the residue the colour changes to green. |
A)
I and III only done
clear
B)
II and III only done
clear
C)
I and II only done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer6)
A compound is prepared from gypsum upon heating to a temperature of 373 K and it changes back to gypsum on adding water. Which is the incorrect statement about the compound?
A)
The compound is used for setting fractured bones. done
clear
B)
The compound is called plaster of Paris which is calcium sulphate dehydrate with a formula \[CaS{{O}_{4}}.2HO.\] done
clear
C)
If heated at higher temperature, the compound becomes dehydrated and is called dead burnt plaster. done
clear
D)
Both (a) and (b). done
clear
View Solution play_arrow
-
question_answer7)
Choose the incorrect statement among the following.
A)
Tamarind, tomato and sour milk are the natural sources of tartaric acid, citric acid and lactic acid respectively. done
clear
B)
The atmosphere of Venus is made up of thick white and yellowish clouds of sulphuric acid. done
clear
C)
Bleaching powder is used for disinfecting drinking water. done
clear
D)
During electrolysis of brine,\[C{{l}_{2}}\] gas is given off at the anode and \[{{H}_{2}}\] gas at the cathode. done
clear
View Solution play_arrow
-
question_answer8)
|
Read the given passage and fill in the blanks by choosing an appropriate option.
|
|
Bleaching powder is a ___i___ powder. When exposed to air, it reacts with ___ii___ of the air to liberate \[C{{l}_{2}}\]gas. It is ___iii___ in cold water and milkiness of the solution is due to presence of unreacted ___iv___. It reacts with HCI and \[{{H}_{2}}S{{O}_{4}}\]liberating ___v___ gas.
|
A)
B)
C)
D)
View Solution play_arrow
-
question_answer9)
|
Observe the given experimental set-up carefully.
|
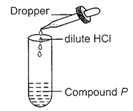
|
|
Which of the following could be the possible observation and inference drawn?
|
A)
The solution becomes blue-green due to formation of copper oxide, P is acidic. done
clear
B)
The solution becomes blue-green due to formation of copper chloride, P is basic. done
clear
C)
The solution becomes white due to formation of calcium hydroxide, P is basic. done
clear
D)
The solution becomes white due to formation of calcium oxide, P is acidic. done
clear
View Solution play_arrow
-
question_answer10)
Few statements regarding the properties of bases are given below. Mark the correct statements?
| (i) Sodium hydroxide and potassium hydroxide are soluble in water. |
| (ii) Calcium hydroxide and magnesium hydroxide are partially soluble in water. |
| (iii) All metallic hydroxides react with acids to form their respective metallic salts. |
| (iv) Metallic oxides are acidic oxides hence they react with acids to form salts. |
A)
(i), (ii) and (iv) only done
clear
B)
(i), (iii) and (iv) only done
clear
C)
(i), (ii) and (iii) only done
clear
D)
(ii) and (iii) only done
clear
View Solution play_arrow
-
question_answer11)
Read the given statements and mark the correct option.
| Statement 1: A solution of pH = 1 has hydrogen ion concentration 3 times than that of solution of pH = 3. |
| Statement 2: \[pH=-\log \frac{1}{[{{H}_{3}}{{O}^{+}}]}\] |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer12)
Baking powder is a mixture of X and a mild edible acid such as Y. When it is heated, Z is produced which makes bread and cake soft and spongy. X, Y and Z are respectively
A)
Sodium hydrogen carbonate, tartaric acid, \[C{{O}_{2}}\] done
clear
B)
Sodium carbonate, acetic add, \[C{{O}_{2}}\] done
clear
C)
Sodium hydroxide, acetic acid, \[{{H}_{2}}\] done
clear
D)
Sodium chloride, oxalic acid, \[{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer13)
A metal carbonate W on reacting with an acid gives a gas X which when passed through a solution Y gives the carbonate back. Solution Y reacts with gas Z to form a compound P used for decolourisation. Identify W, X, Y, Z and P.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer14)
Match column I with column II and mark the correct option from the codes given below.
| Column I | Column II |
| (a)\[NaHC{{O}_{3}}\] | (i) Used for disinfecting water |
| (b)\[N{{a}_{2}}C{{O}_{3}}\] | (ii) Used in soda-acid fire extinguishers |
| (c)\[CaOC{{l}_{2}}\] | (iii) Used for removing Permanent I hardness of water |
| (d)\[CaS{{O}_{4}}.\frac{1}{2}{{H}_{2}}O\] | (iv) Used for making toys, materials for decoration |
A)
(a) - (iii), (b) - (i), (c) - (iv), (d) - (ii) done
clear
B)
(a) - (ii), (b) - (iii), (c) - (i), (d) - (iv) done
clear
C)
(a) - (iii), (b) - (ii), (c) - (i), (d) - (iv) done
clear
D)
(a) - (i), (b) - (ii), (c) - (iv), (d) - (iii) done
clear
View Solution play_arrow
-
question_answer15)
Study the given diagram carefully and identify X and Y respectively.
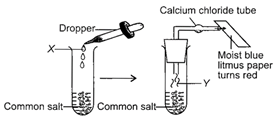
A)
\[Conc.\,HCl,\,C{{O}_{2}}\] done
clear
B)
\[Conc.\,{{H}_{2}}S{{O}_{4}},HCl\,gas\] done
clear
C)
\[Conc.\,HN{{O}_{3}},{{H}_{2}}\] done
clear
D)
\[Conc.\,NaOH,\,C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer16)
Ayaan tested the nature of a few common substances with phenolphthalein indicator and summarised the results in the following table.
| Test tube |
Colour change with phenolphthalein indicator |
| 1 |
Colourless |
| 2 |
Pink |
| 3 |
Colourless |
| 4 |
Pink |
Which of the-following substances could be present in test tubes 1, 2, 3 and 4?
A)
B)
C)
D)
View Solution play_arrow
-
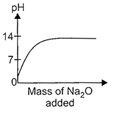
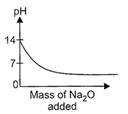
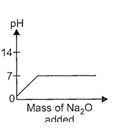
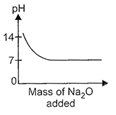
question_answer17)
While demonstrating acid base reactions, Ms. Prabha, a science teacher added sodium oxide to HCI until it was in excess. Which of the following graphs correctly represents the change in pH?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer18)
Aqueous solutions of salts are either acidic, basic or neutral. A few common salts are listed as :
| I. Silver chloride |
| II. Ammonium sulphate |
| III. Sodium nitrate |
| IV. Sodium phosphate |
| V. Sodium acetate |
Which of the following correctly match the given salts with the nature of their aqueous solutions?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer19)
Study the given flow chart carefully and fill in the blanks by choosing an appropriate option.
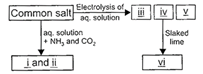
A)
B)
C)
D)
View Solution play_arrow
-
question_answer20)
Observe the given figure carefully, which represents the decomposition of brine solution by passing electricity.
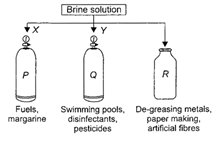
A)
B)
C)
D)
View Solution play_arrow

 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
